Abstract
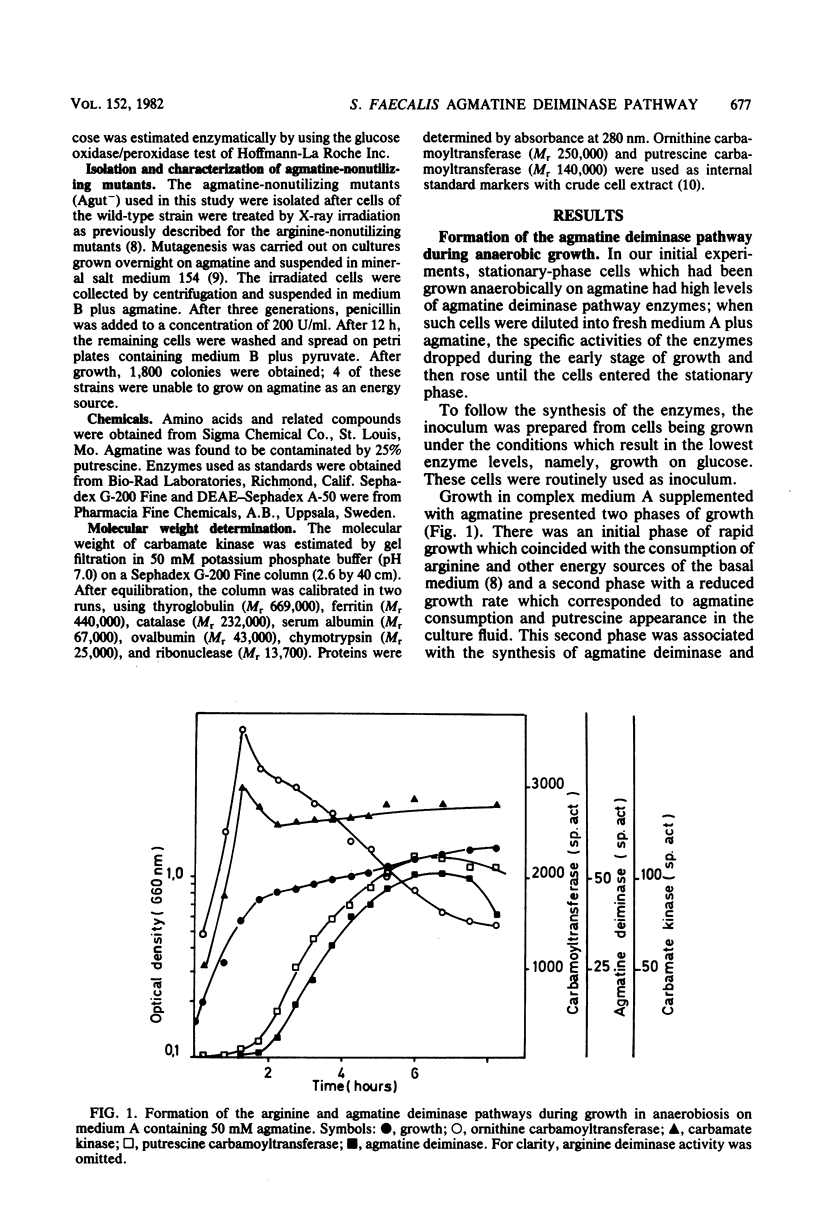
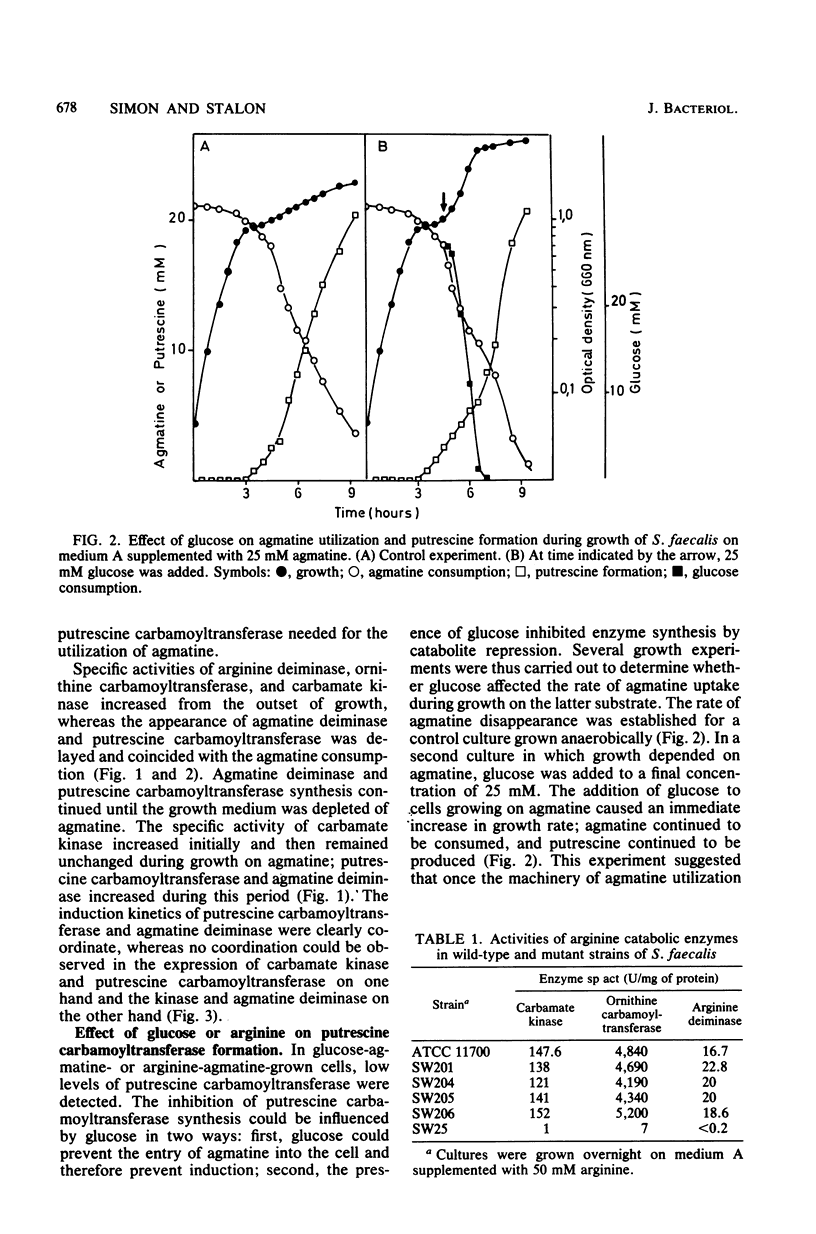
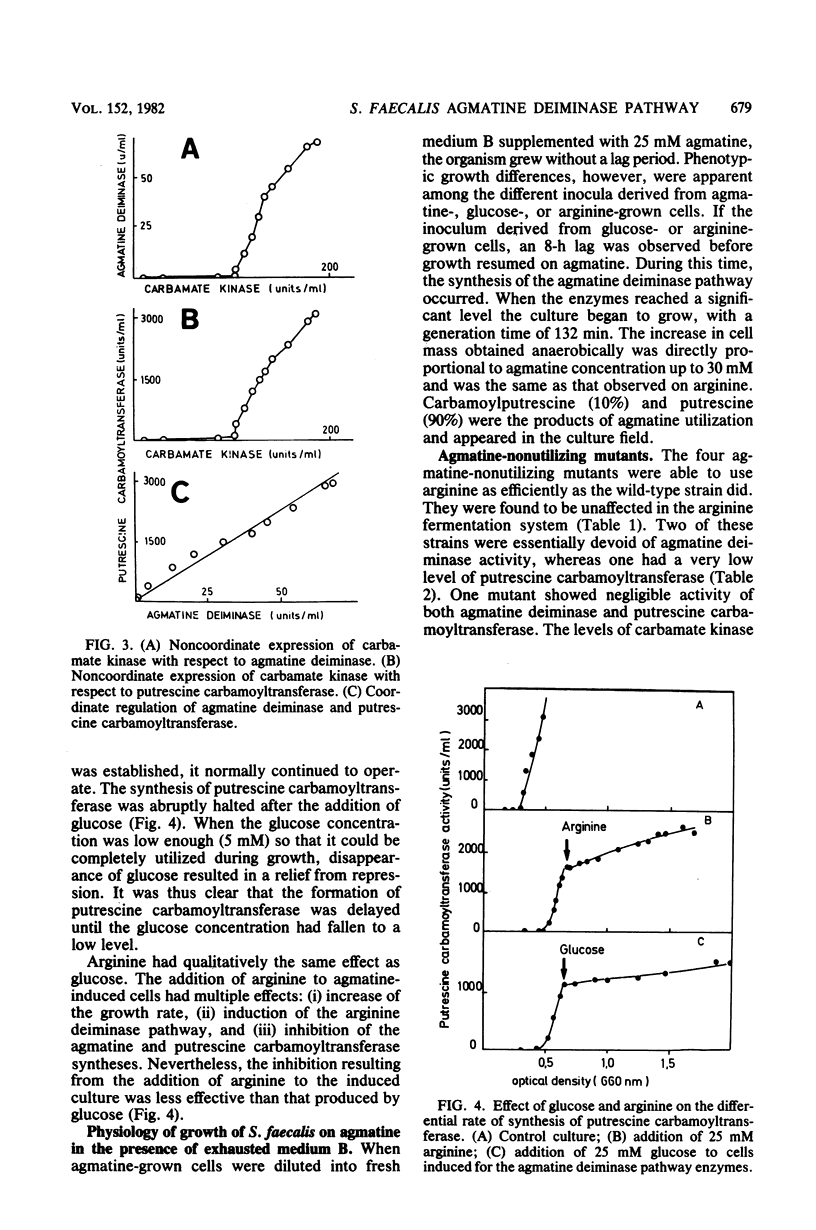
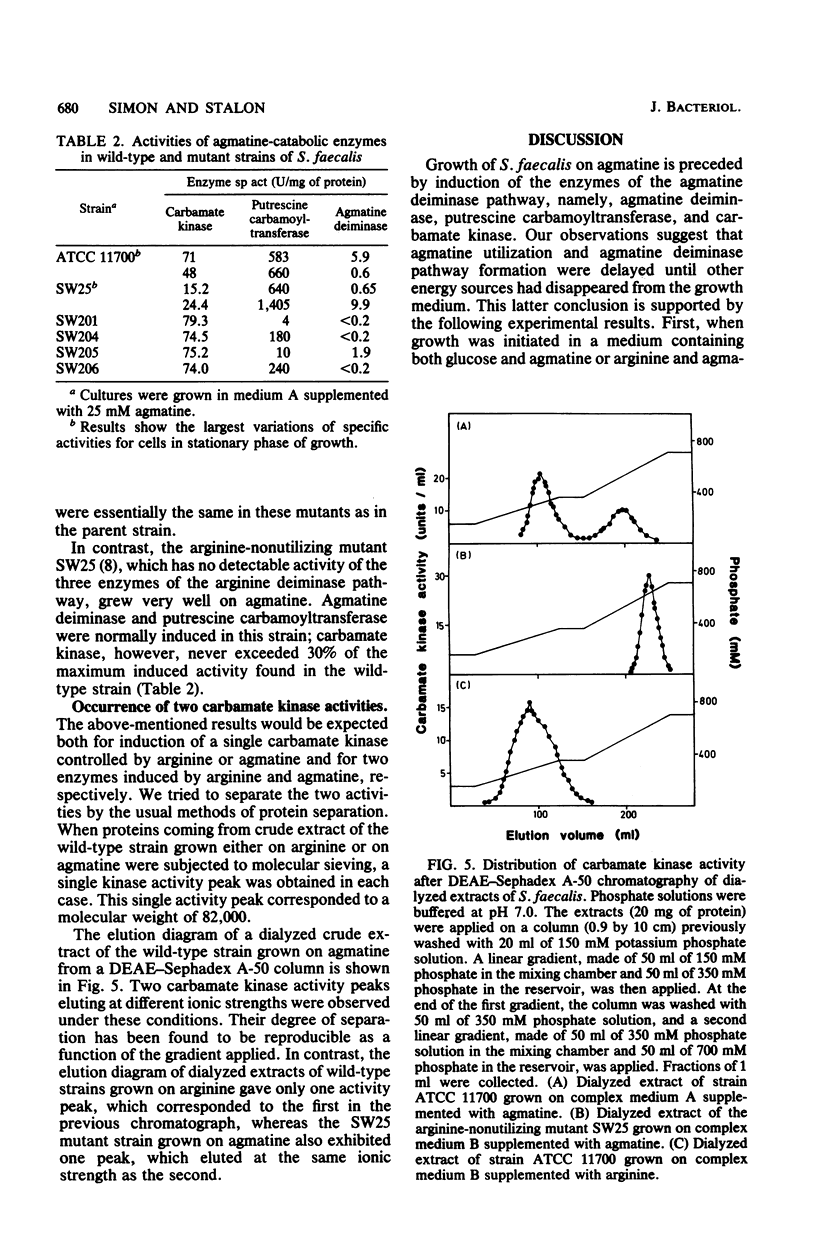
Streptococcus faecalis ATCC 11700 uses agmatine as its sole energy source for growth. Agmatine deiminase and putrescine carbamoyltransferase are coinduced by growth on agmatine. Glucose and arginine were found to exert catabolite repression on the agmatine deiminase pathway. Four mutants unable to utilize agmatine as an energy source, isolated from the wild-type strain, exhibited three distinct phenotypes. Two of these strains showed essentially no agmatine deiminase, one mutant showed negligible activity of putrescine carbamoyltransferase, and one mutant was defective in both activities. Two carbamate kinases are present in S. faecalis, one belonging to the arginine deiminase pathway, the other being induced by growth on agmatine. These two enzymes have the same molecular weight, 82,000, and seem quite different in size from the kinases isolated from other streptococci.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Broman K., Lauwers N., Stalon V., Wiame J. M. Oxygen and nitrate in utilization by Bacillus licheniformis of the arginase and arginine deiminase routes of arginine catabolism and other factors affecting their syntheses. J Bacteriol. 1978 Sep;135(3):920–927. doi: 10.1128/jb.135.3.920-927.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Marshall M., Cohen P. P. A kinetic study of the mechanism of crystalline carbamate kinase. J Biol Chem. 1966 Sep 25;241(18):4197–4208. [PubMed] [Google Scholar]
- Mercenier A., Simon J. P., Haas D., Stalon V. Catabolism of L-arginine by Pseudomonas aeruginosa. J Gen Microbiol. 1980 Feb;116(2):381–389. doi: 10.1099/00221287-116-2-381. [DOI] [PubMed] [Google Scholar]
- Mercenier A., Simon J. P., Vander Wauven C., Haas D., Stalon V. Regulation of enzyme synthesis in the arginine deiminase pathway of Pseudomonas aeruginosa. J Bacteriol. 1980 Oct;144(1):159–163. doi: 10.1128/jb.144.1.159-163.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mercenier A., Stalon V., Simon J. P., Haas D. Mapping of the arginine deiminase gene in Pseudomonas aeruginosa. J Bacteriol. 1982 Feb;149(2):787–788. doi: 10.1128/jb.149.2.787-788.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simon J. P., Wargnies B., Stalon V. Control of enzyme synthesis in the arginine deiminase pathway of Streptococcus faecalis. J Bacteriol. 1982 Jun;150(3):1085–1090. doi: 10.1128/jb.150.3.1085-1090.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stalon V., Ramos F., Piérard A., Wiame J. M. The occurrence of a catabolic and an anabolic ornithine carbamoyltransferase in Pseudomonas. Biochim Biophys Acta. 1967 May 16;139(1):91–97. doi: 10.1016/0005-2744(67)90115-5. [DOI] [PubMed] [Google Scholar]
- Wargnies B., Lauwers N., Stalon V. Structure and properties of the putrescine carbamoyltransferase of Streptococcus faecalis. Eur J Biochem. 1979 Nov 1;101(1):143–152. doi: 10.1111/j.1432-1033.1979.tb04226.x. [DOI] [PubMed] [Google Scholar]


