Abstract
The search for disease-associated T helper 2 (Th2) Leishmania antigens and the induction of a Th1 immune response to them using defined vaccination protocols is a potential strategy to induce protection against Leishmania infection. Leishmania infantum LiP2a and LiP2b acidic ribosomal protein (P proteins) have been described as prominent antigens during human and canine visceral leishmaniasis. In this study we demonstrate that BALB/c mice infected with Leishmania major develop a Th2-like humoral response against Leishmania LiP2a and LiP2b proteins and that the same response is induced in BALB/c mice when the parasite P proteins are immunized as recombinant molecules without adjuvant. The genetic immunization of BALB/c mice with eukaryotic expression plasmids coding for these proteins was unable to redirect the Th2-like response induced by these antigens, and only the co-administration of the recombinant P proteins with CpG oligodeoxynucleotides (CpG ODN) promoted a mixed Th1/Th2 immune response. According to the preponderance of a Th2 or mixed Th1/Th2 responses elicited by the different regimens of immunization tested, no evidence of protection was observed in mice after challenge with L. major. Although alterations of the clinical outcome were not detected in mice presensitized with the P proteins, the enhanced IgG1 and interleukin (IL)-4 response against total Leishmania antigens in these mice may indicate an exacerbation of the disease.
Keywords: adjuvants, antibody responses, CpG DNA, DNA vaccines, Leishmania spp
Introduction
Leishmaniases comprise several diseases caused by intracellular protozoan parasites belonging to the genus Leishmania. The clinical manifestations of leishmaniasis in humans range from cutaneous leishmaniasis, which heal spontaneously, to potentially fatal visceral leishmaniasis [1]. Outcome of infection is determined by interactions between the host immune system and the different parasite species, yet the pathogenesis of leishmaniasis remains unclear. A model of Leishmania virulence has been proposed involving two different groups of parasite molecules [2,3]. One group consists of surface and secreted products that are necessary for the establishment of infection as a prerequisite for virulence but that by themselves do not cause disease. The second group of parasite molecules consists of highly conserved, intracellular molecules referred to as ‘pathoantigens’. The inadequate humoral response induced against these antigens is thought to result in immunopathology, due mainly to the adverse effects of immune complexes, particularly uveitis [4], lesions in the central nervous system [5] or nephritis in dogs suffering visceral leishmaniasis [6–9] and in hamsters and mice infected with L. donovani [10,11]. In addition, immune complexes have been also involved in anaemia in hamsters infected with L. donovani [12]. Finally, it has been shown recently that both in mice infected with L. major and in humans suffering visceral leishmaniasis, the presence of IgG immune complexes correlates with an inability to resolve infections. This effect, that relies on the induction of interleukin (IL)-10, demonstrates that the presence of immune complexes can be detrimental to a host infected with this intracellular pathogen [13].
Effective primary immunity against L. major in mouse requires IL-12-dependent production of interferon (IFN)-γ from CD4+ T cells [T helper 1 (Th1) response] and CD8+ T cells, which mediates nitric oxide (NO)-dependent killing by infected macrophages (reviewed in [14–16]). In contrast, susceptibility correlates with the dominance of an IL-4-driven Th2 response, as it has been observed in certain strains, mainly in BALB/c mice. In recent years, several recombinant leishmanial antigens have been identified and tested as vaccine candidates [16]. Some of them were tested because they elicit primarily a Th1-type response in L. major-infected BALB/c mice, in spite of the strongly biased Th2 responses mounted in these mice [17]. In clear contrast, some other promising vaccine candidates were strong inducers of Th2 responses in BALB/c mice infected with L. major. Thus, it was demonstrated first that BALB/c mice could be protected against L.major infection by simply redirecting the early Th2 response induced against one single antigen, the Leishmania homologue of the receptor for activated C kinase (LACK) towards a Th1 response [18]. The same results were obtained when other Th2-inducing parasite proteins, such as the L. mexicana cysteine protease (CPB2·8) [19] or the parasite nucleosome forming histones [20], were administered with Th1-modulating adjuvants. Thus, redirecting the Th2 responses induced against some Leishmania epitopes towards a Th1 response is a promising strategy to induce protection against L. major infection [21].
The Leishmania P protein family, constituents of the large subunit of ribosomes, comprises three members (namely LiP0, LiP2a and LiP2b) and can be considered as potent immunostimulatory proteins during the leishmaniasis process. They have been described as immunodominant antigens recognized by sera from both human and dogs infected naturally with L. chagasi–L. infantum [22–24]. Also, administration in BALB/c mice of the L. infantum recombinant LiP2a protein (rLiP2a) or LiP0 (rLiP0), in the absence of any added adjuvant, elicited IgG1 humoral responses [25,26]. Remarkably, immunization of the LiP0 in BALB/c mice as a DNA vaccine or as recombinant protein combined with CpG oligodeoxynucleotides (GpG ODN) redirect this response towards a specific Th1 response that correlates with the induction of partial protection after challenge with L. major [26,27].
In this paper we show that BALB/c mice infected with L. major exhibit a Th2 humoral response against the rLiP2a and rLiP2b proteins. Further, we analyse if DNA vaccination or the use of CpG oligodeoxynucleotides (ODN) adjuvant redirect the Th2 responses induced by these antigens. An increased IgG2a–IFN-γ response was observed only by co-administration of CpG ODN with the recombinant proteins in naive mice or in mice primed previously with P proteins genes, suggesting that this adjuvant triggered Th1-specific responses. Notwithstanding, the Th2 response against the parasite LiP2a and LiP2b proteins was not inhibited, as the IgG1 response was also enhanced by this adjuvant. Finally, we demonstrate that these mixed immune responses are not correlated with protection against L. major infection in BALB/c mice.
Materials and methods
Mouse strains and parasites
Female BALB/c mice were 6–8 weeks old (Harlan Interfauna Ibérica SA, Barcelona, Spain). L. major amastigotes (clone WHOM/IR/−173) were obtained from popliteal lymph nodes from infected BALB/c mice and transformed to stationary phase promastigotes by culturing at 26°C in Schneider's medium (Gibco, BRL, Grand Island, NY, USA) plus 20% fetal calf serum (FCS).
Plasmid constructs
The cDNAs coding for the LiP2a and LiP2b proteins were obtained after EcoRI digestion of pUC8-22 and pUC8-26 plasmids [28] and subcloned in pcDNA3 (Invitrogen, San Diego, CA, USA). Endotoxin-free plasmid DNA from pcDNA3-LiP2a and pcDNA3-LiP2b clones was isolated using the EndoFree Plasmid Giga kit (Qiagen, Hilden, Germany).
For expression of rLiP2a and rLiP2b proteins, their coding regions (CR) were polymerase chain reaction (PCR) amplified and subcloned into the pQE30 expression vector (Qiagen). Primers employed were: sense, 5′-CGGGATCCATGCAGTACCTCGCCGCGTA-3′ (positions 1–20 of the P2a-CR); anti-sense, 5′-GCGTCGACTTAGTCAAAGAGACCGAAGC-3′ (reverse and complementary to positions 302–318 of the P2a-CR) and sense, 5′-CGGGATCCATGTCCACCAAGTACCTCGC-3′ (positions 1–20 of the P2b-CR); anti-sense, 5′-CCCAAGCTTAGTCGAACAGACCGAAGC-3′ (reverse and complementary to positions 319–333 of the P2b-CR). BamHI and SalI restriction sites (underlined) were included for cloning purposes.
Leishmania antigens
The rLiP2a and rLiP2b were over-expressed in Escherichia coli transformed with pQE-LiP2a or with pQE-LiP2b and purified under native conditions onto Ni-nitrilotriacetic acid agarose columns (Qiagen). The recombinant proteins were passed through a polymyxin-agarose column (Sigma, St Louis, MO, USA). Residual endotoxin content (< 12 pg/μg of recombinant protein) was measured by the Quantitative Chromogenic Limulus amebocyte assay QCL-1000 (BioWhittaker, Walkersville, MD, USA). Purity of proteins was assessed in a Coomassie blue-stained sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS-PAGE) gel (Fig. 1a). Total soluble Leishmania antigen (SLA) of L. major was prepared as described [27].
Fig. 1.

(a) Purification of Leishmania infantum rLiP2a and rLiP2b after expression in Escherichia coli. Five μg of rLiP2a (lane 1) and rLiP2b (lane 2) were electrophoresed on linear 10–14% gradient sodium dodecyl sulphate-polyacrylamide gel electrophoresis gel. The Coomassie-blue staining of the gel is shown. (b–d) Heterologous cytoplasmic expression of Leishmania P proteins in COS-7 cells. Immunofluorescence of COS-7 cells transfected transiently with pcDNA3-LiP2a (b) and pcDNA3-LiP2b (c); 24 h after transfection cells were fixed and immunostained with a polyclonal anti-LiP2a (b) or anti-LiP2b serum (c). As secondary antibody an Alexa488-conjugated goat anti-mouse IgG was employed. As a control, COS-7 cells transfected with pcDNA3-LiP2a were incubated with the sera from preimmune mice (d). Similar results were obtained when cells transfected with pcDNA-LiP2b were incubated with the corresponding preimmune sera (data not shown).
Expression of pcDNA3-LiP2a and pcDNA3-LiP2b in COS-7 cells
To confirm that the DNA constructs were functional, COS-7 cells were transfected as described [27] and analysed by immunofluorescence. Briefly, cells were fixed 24 h after transfection with 2% paraformaldehyde for 20 min, washed with phosphate-buffered saline (PBS), blocked and permeabilized with 1% bovine serum albumin plus 0·1% saponin in PBS for 1 h at room temperature, and incubated for 1 h with sera from mice vaccinated with rLiP2a and rLiP2b. Afterwards, the coverslips were rinsed with PBS and incubated for 1 h with an Alexa488-conjugated goat anti-mouse IgG secondary antibody (Molecular Probes, Eugene, OR, USA). The coverslips were mounted with Mowiol (Calbiochem, Darmstad, Germany) and visualized on a Zeiss Axiovert 200 microscope (Fig. 1b–d).
Immunizations and parasite challenge
BALB/c mice were immunized following four regimens: recombinant protein inoculation adjuvated or not with CpG ODN, genetic vaccination and a prime-boosting regimen.
Mice were immunized subcutaneously (s.c.) in the right footpad with 2 μg of rLiP2a or rLiP2b alone or plus 50 μg of CpG ODN (25 μg of each phosphorothioate-modified immunostimulatory ODN containing CpG motifs as described previously in [27]). Each group was boosted 2 weeks later using the same regimen. Control mice received PBS or 50 μg CpG-ODN alone.
For genetic vaccinations mice were inoculated s.c. three times at 2-week intervals in the right footpad with either PBS, 100 μg of pcDNA3 (empty vector), 100 μg pcDNA3-LiP2a or 100 μg pcDNA3-LiP2b. Prime-boosted mice were immunized twice, 2 weeks apart, with 100 μg of pcDNA3-LiP2a or 100 μg pcDNA3-LiP2b and boosted 2 weeks later with 2 μg of rLiP2a or rLiP2b adjuvated with 50 μg of CpG ODN. Control mice received two doses of pcDNA3 and were boosted with 50 μg of CpG ODN.
Parasite challenge was carried out by s.c. inoculation with 5 × 104 stationary-phase promastigotes of L. major into the left footpad 4 weeks after the last inoculation. The progress of the infection was followed as described previously [20].
Measurement of cytokines in supernatants
The release of IFN-γ and IL-4 was measured in the supernatants of splenocytes stimulated with rLiP2a or rLiP2b (3 μg/ml) or SLA (12 μg/ml), as described previously [27], using commercial enzyme-linked immunosorbent assay (ELISA) kits (Diaclone, Besançon, France).
Determination of antibody titres and isotypes
Serum samples were analysed for specific anti-rLiP2a, anti-rLiP2b or anti-SLA antibodies. ELISA assays were performed as described previously [27]. The reciprocal end-point titre was determined by serial dilution of the sera, and was defined as the inverse of the highest serum dilution factor giving an absorbance > 0·2. The following horseradish peroxidase-conjugated anti-mouse immunoglobulins (Nordic Immunological Laboratories, Tilburg, the Netherlands): anti-IgM (1/2000), anti-IgG (1/2000), anti-IgG1 (1/1000) and anti-IgG2a (1/500). Ortophenylene diamine dihydrochloride-OPD (Dako, A/S, Glostrup, Denmark) was used as peroxidase substrate for ELISA assays. After 15 min, the reaction was stopped by addition of 100 μl of H2SO4 1 M and the absorbance was read at 450 nm.
Statistical analysis
Statistical analysis was performed by a Student's t-test. Differences were considered significant when P < 0·05.
Results
Antigenicity of the Leishmania P proteins: effect of the DNA vaccines and the CpG ODN on the immune responses elicited by the Leishmania P proteins
Using the purified rLiP2a and rLiP2b His-tagged recombinant proteins (Fig. 1a), we detected the presence of anti-Leishmania P proteins antibodies in sera of BALB/c mice infected with L. major. The L. infantum recombinant P proteins were employed as antigens for the ELISA assays because they were 100% conserved with their corresponding L. major orthologues (Accession no. Q4Q6R6 for LmP2a and Q4QF62 for LmP2b). The humoral response was predominantly of the IgG1 isotype (the mean IgG1/IgG2a ratio was 10·2 ± 2·5 for rLiP2a and 8·6 ± 2·5 for rLiP2b), although the intensity of the anti-LiP2b response was somewhat higher than the anti-LiP2a response (Fig. 2a). Because the induction of IgG1 and IgG2a antibodies is used as a marker of Th2-type and Th1-type immune responses, respectively [29], we may conclude that during L. major infection a Th2-like humoral response is induced against these antigens.
Fig. 2.
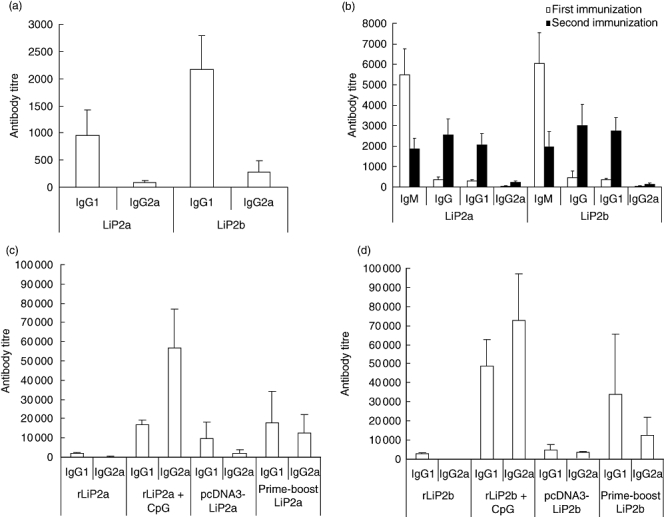
(a) Leishmania P proteins induced a predominant T helper 2-like response in L. major-infected BALB/c mice. Analysis of the anti-LiP2a and anti-LiP2b humoral response in BALB/c mice infected with L. major. Six naive BALB/c mice were inoculated with 5 × 104 stationary-phase L. major promastigotes into the left footpad. Sera were obtained after 8 weeks of infection and tested individually by enzyme-linked immunosorbent assay (ELISA) against the recombinant rLiP2a and rLiP2b P proteins. Reciprocal end-point titres for IgG1 and IgG2a antibodies subclasses were determined as indicated in Materials and methods. (b) Analysis of the specific humoral response induced in BALB/c mice immunized with the LiP2a and LiP2b acidic ribosomal proteins without adjuvants. Six mice were immunized twice in the right footpad with 2 μg of rLiP2a or rLiP2b. Sera were obtained and assayed individually by ELISA 7 days after the first immunization (white bars) or 7 days after the second immunization (solid bars). Reciprocal end-point titre was determined for IgM, IgG, IgG1 and IgG2a antibodies subclasses. No reactivity was detected in the preimmune sera. (c–d) Immune responses elicited by the genetic inoculation of parasite P proteins. BALB/c mice (six mice per group) were immunized with the recombinant rLiP2a or rLiP2b proteins alone and adjuvated with CpG oligodeoxynucleotides (ODN), by genetic vaccination and by prime-boosting regimen, as indicated in Materials and methods. As control, mice were vaccinated in the same manner with phosphate-bufered saline, with CpG ODN alone, with the empty pcDNA3 plasmid and with the empty plasmid and CpG ODN. For measurement of the specific anti-LiP2a (b) or anti-LiP2b (c) humoral responses sera were individually assayed by ELISA 7 days after the last immunization. The reciprocal end-point titre is shown. None of the preimmune or the control sera showed reactivity against the corresponding antigens.
In a previous work we described that the intraperitoneal (i.p.) administration of the rLiP2a protein in the absence of adjuvant elicited a predominant IgG1 humoral response [25]. Here we extend this result to BALB/c mice immunized s.c. with either the rLiP2a or the rLiP2b recombinant proteins alone. A specific antibody response was detected 7 days after the first dose (Fig. 2b). A progressive switch from IgM to IgG antibodies was observed after the protein boosting. Although some IgG2a specific antibodies were detected, the IgG1 response was predominant after the second dose. The IgG1/IgG2a ratio against rLiP2a and rLiP2b was 9·1 ± 1·2 and 20·3 ± 2·1, respectively, demonstrating that BALB/c mice immunized with P proteins develop a specific Th2-like response.
We analysed further if the use of CpG ODN or genetic administration of the Leishmania P proteins counteracts the Th2 response induced by these antigens. For that purpose, eukaryotic expression vectors coding for LiP2a and LiP2b were constructed and their expression was tested in COS-7 cells (Fig. 1b–d). We followed three different inoculation regimens: recombinant protein inoculation adjuvated with CpG ODN, genetic vaccination and a prime-boosting regimen (see Materials and methods for details).
The humoral response elicited by these procedures is depicted in Fig. 2c–d. Only the CpG motifs were able to alter significantly the IgG1/IgG2a ratio driven by the proteins alone. The IgG1/IgG2a ratio after administration of the rLiP2a and rLiP2b proteins in the presence of CpG motifs was 1·42 ± 0·4 and 1·14 ± 0·35, respectively. Notably, the genetic immunization of the proteins modified the IgG1/IgG2a only slightly compared to inoculation of P proteins alone, as the mean IgG1/IgG2a ratio was 4·9 ± 1·4 for rLiP2a and 14·2 ± 2·1 for rLiP2b. Mice primed with P2a or P2b genes and boosted with the corresponding recombinant proteins plus CpG ODN exhibit a variable intensity in the humoral response, but a similar IgG1/IgG2a ratio (1·39 ± 0·45 for LiP2a and 2·3 ± 0·51 for LiP2b).
To determine whether there was a correlation between the switch of the IgG1/IgG2a ratio and the cytokine milieu elicited by these immunizations, we analysed the IFN-γ production in splenocytes cultures established from vaccinated mice and stimulated with the recombinant P proteins. A specific production of IFN-γ was observed in all the groups of vaccinated mice after 48 h of stimulation with the corresponding protein, rLiP2a (Fig. 3a) or rLiP2b (Fig. 3b), although co-administration of CpG ODN (recombinant proteins plus CpG ODN and prime-boost groups) leads to a significant increase in IFN-γ production specific for both P proteins. No IL-4 was detected in these assays.
Fig. 3.

Interferon (IFN)-γ production by splenocytes of BALB/c mice stimulated in vitro with the rLiP2a (a) and rLiP2b (b) P proteins. Groups of mice described in Fig. 2 were killed 4 weeks after the last immunization, and their splenocytes were obtained and cultured in vitro for 48 h in the presence of the corresponding rLiP2a or rLiP2b soluble proteins or medium alone. The supernatants were harvested and assayed individually for IFN-γ production by enzyme-linked immunosorbent assay.
The course of L. major infection in vaccinated mice
We then analysed whether the Th1-like response triggered by CpG ODN adjuvant against P proteins was able to alter the course of a subsequent L. major challenge. The results indicated that the mixed Th1/Th2 immune response induced by the administration of the recombinant proteins plus CpG ODN (Fig. 4a) and the prime-boost regimen (Fig. 4b) did not have any influence in the time–course of the inflammation of the footpad due to L. major infection, nor did the inoculation of these antigens as DNA vaccines alter the footpad swelling due to L. major infection (Fig. 4c). The lack of protection was confirmed by the observation that a similar parasite load was found in the DLN of the control and of the vaccinated mice (data not shown).
Fig. 4.

Course of Leishmania major infection in BALB/c vaccinated mice. Mice (six per group) were immunized twice s. c. with the recombinant rLiP2a or rLiP2b proteins adjuvated with CpG oligodeoxynucleotides (ODN) (a), primed twice with the recombinant plasmid and boosted with the rLiP2a or rLiP2b adjuvated with CpG ODN (b), or three times with the pcDNA3-LiP2a or pcDNA3-LiP2b eukaryotic expression plasmids (c), as indicated in Materials and methods. As control, mice were vaccinated in the same manner with CpG ODN alone (a), with the empty plasmid and CpG ODN (b) and with the empty pcDNA3 plasmid (c). For comparison, unvaccinated groups injected with phosphate-buffered saline were included (a–c). One month after the last immunization, mice were infected in the left hind footpad with 5 × 104 L. major stationary-phase promastigotes. Footpad swelling is given as the difference of thickness between the infected and the uninfected contralateral footpad. No significant differences were found between vaccinated mice and their corresponding controls in any group.
Mice presensitized with P proteins exhibit a mixed Th1/Th2 response against the LiP2a and LiP2b proteins and an enhanced Th2 response against the whole soluble antigens of the parasite.
Humoral and cellular responses against the LiP2a and LiP2b antigens were analysed in mice infected with L. major in order to determine the immunological parameters associated with the lack of protection. After L. major infection, those mice immunized with the P proteins showed an enhanced ability to recognize the recombinant rLiP2a (Fig. 5a) and rLiP2b (Fig. 5b) proteins as the titre for both IgG1 and IgG2a, being higher in the vaccinated mice than in their corresponding controls. No statistical differences between the two isotypes were detected. In order to analyse the antigen-driven cellular responses, splenocytes from different mice groups were stimulated in vitro with either rLiP2a or rLiP2b and the presence of IFN-γ and IL-4 in the supernatants was assessed. The specific production of IFN-γ and IL-4 was higher in the groups of vaccinated mice relative to control mice after 48 h of stimulation with the corresponding protein, rLiP2a (Fig. 5c,e) or rLiP2b (Fig. 5d,f).
Fig. 5.

Analysis of the immune response against the acidic ribosomal proteins in vaccinated and control mice after Leishmania major challenge. Serum samples from different groups of vaccinated and control mice were obtained 7 weeks after challenge and the reciprocal end-point titres for IgG1 and IgG2a antibodies against rLiP2a (a) and rLiP2b (b) were determined by enzyme-linked immunosorbent assay (ELISA). At the same time mice were killed and their splenocytes were obtained and cultured in vitro in complete medium with or without the recombinant proteins. The supernatants were harvested and the specific rLiP2a-driven (c) or rLiP2b-driven (d) production of interferon-γ, and the specific rLiP2a-driven (e) or rLiP2b-driven (f) production of interleukin-4 was assayed by ELISA.
Although no alterations in the time–course of L. major infection were observed in mice vaccinated with Pproteins, we determined whether the immunization with these antigens alters the IL-4 driven Th2 response that leads to susceptibility in BALB/c mice; the titre of the IgG1 and IgG2a antibodies against the soluble Leishmania antigens (SLA) was determined 7 weeks after challenge in the different groups of mice (Fig. 6a). Notably, the ratio IgG1/IgG2a was higher in mice vaccinated with either rLiP2a or rLiP2b, especially in the prime-boost regimen. This suggests that the cytokine milieu influenced by the administration of the P proteins may have favoured a Th2 response to the whole parasite. In agreement with this finding, it was observed that the in vitro production of IFN-γ specific for SLA was similar in splenocytes from all of the mice groups (Fig. 6b), whereas the level of IL-4 was significantly higher in splenocytes from immunized mice than in controls (Fig. 6c), confirming that presensitization with P proteins enhanced the Th2 response of the BALB/c mice against L. major.
Fig. 6.

Analysis of the immune response against total parasite proteins induced by the Leishmania major challenge in vaccinated and control mice. (a) Analysis of the humoral response against soluble Leishmania antigen (SLA) from the different regimens of vaccination and the corresponding controls. Serum samples were obtained 7 weeks after challenge and the reciprocal end-point titre for IgG1 and IgG2a antibodies against SLA was determined individually by enzyme-linked immunosorbent assay (ELISA). The ratio between IgG1 and IgG2a titres for the different groups has been also included. Seven weeks after infection animals from the different regimens of vaccination and the corresponding controls were killed and their splenocytes were obtained and cultured in vitro in complete medium with or without SLA. The supernatants were harvested and assayed for interferon-γ (b) and interleukin-4 (c) by ELISA. *P < 0·05; **P < 0·02.
Discussion
Upon infection with L. major, mice of the resistant phenotype clearly develop a dominant Th1 immune response to the parasite antigens, whereas BALB/c mice develop a typical Th2 response and succumb to infection. This association of resistance and susceptibility with the emergence of the two phenotypically distinct subsets of CD4+ T cells has been the most striking concept arising from the murine model of cutaneous leishmaniasis (reviewed in [14–16]). Those Leishmania antigens that predominantly stimulate Th1 responses in patient cells or spleen and lymph node cells from mice infected with L. major have been commonly accepted as promising vaccine candidates. In contrast, antigens that stimulate predominantly a Th2 response have been considered as ‘pathoantigens’ because the response induced against them is thought to result in immunopathology [2,3]. Paradoxically, several leishmanial antigens against which a Th1 response is developed during the infection are not necessarily protective, whereas redirecting the Th2 responses induced against some Leishmania epitopes towards a Th1 response may induce a substantial protection against infection (recently reviewed in [21]).
In this study we show that a humoral response is induced against the LiP2a and LiP2b proteins in BALB/c mice infected experimentally with L. major. These proteins have been also described as antigens in dogs [24] and humans [30] infected naturally with L. infantum and in hamster [31] and mouse (N. Parody, laboratory results) infected experimentally with L. infantum. In addition, antibodies against ribosomal P proteins have been found in sera from patients with infectious diseases other than leishmaniasis, such as Chagas' disease [32], patients with fungal allergies [33,34] and patients having autoimmune processes such as systemic lupus erythematosus [35]. Interestingly, it has been reported that these anti-P antibodies are implicated in the immunopathology of these diseases [36–41]. Moreover, our data indicate that the anti-Leishmania P2a and P2b antibodies induced in infected BALB/c mice were predominantly of the IgG1 isotype. Thus, we may conclude that during the murine infection a Th2-like response against these antigens is induced.
The data presented here demonstrate that the parasite P proteins behave as strong immunogens, as they elicit humoral responses when immunized s.c. in the footpads of BALB/c mice in the absence of any adjuvant. The progressive switch from IgM to IgG antibodies after the protein boosting can be taken as an indication that the immunization with these proteins elicited a T cell-dependent immune response. Concomitantly with specific humoral response obtained during infection with L. major, there is a switch of antibody classes to the IgG1 isotype after the second administration of both recombinant proteins. Thus, we conclude that the administration of these proteins induces a Th2-like humoral response in the BALB/c mice.
Strikingly, the genetic administration of these antigens as DNA vaccines, a general strategy employed to promote a Th1 type response, induce only slight changes in the Th2-like response against Leishmania P proteins. As far as we are concerned, only few examples of antigens that generate a dominant Th2-type response in mice when used in genetic immunizations have been described, including the serine repeat antigen (SERA) from Plasmodium falciparum [42] and the 45 W antigen from Taenia ovis [43]. Also, DNA immunization of the Leishmania meta 1 antigen, a protein over-expressed in metacyclic promastigotes, induced a predominant Th2-type of response that correlated with lack of protection after Leishmania infection [44]. Data presented here demonstrate that it is only by co-administration of the CpG ODN adjuvant, both administered with the recombinant rLiP2a and rLiP2b proteins or in a prime-boost regimen, that a balanced IgG2a/IgG1 ratio is observed concomitantly with higher levels of IFN-γ induced in vitro. These data indicate that the use of CpG ODN adjuvant induced Th1 responses against Leishmania P proteins, although these immunization protocols were not able to inhibit completely the Th2-like response induced against these antigens. The potential of CpG ODN to suppress Th2 responses in mouse has been demonstrated widely in parasitic diseases [45–47] and allergy disorders [48], but in some reports it has also been demonstrated that CpG ODN may fail to down-regulate neonatally Th2 triggered responses [49] or ongoing IgE responses [50].
Taking into account the antigenicity of the LiP2a and LiP2b proteins, and the correlation of Th2 and Th1 responses with susceptibility and resistance to L. major infection in BALB/c mice, we analysed the effects of the different inoculation regimens in the course of L. major infection. As indicated above, genetic immunization of some defined Leishmania antigens that induce Th2 responses during infection protects BALB/c mice against cutaneous leishmaniasis. It has also been described that immunization with a quimeric protein (Q protein) containing the LiP2a and LiP2b, engineered to have deletions of the carboxyl-terminal regions and the antigenic regions of the H2A and the LiP0 Leishmania proteins [51] mixed with live bacille Calmette–Guérin (BCG), confers protection to dogs challenged with L. infantum [52]. In addition, co-administration of Q protein mixed with CpG ODN resulted in a reduction of the parasite burden of BALB/c mice infected with L. infantum [53]. However, no differences in the clinical outcomes of infection after challenge with L. major were observed in any of the groups of mice employed in this work. No protection was observed in mice vaccinated with the rLiP2a or rLiP2b antigens mixed with CpG (recombinant proteins plus CpG ODN and prime-boost), in spite of the specific Th1-like response elicited against the P proteins. This lack of protection correlates with the induction of mixed Th1/Th2 responses against the P proteins in all the vaccinated mice groups after infection. As it has been suggested that the induction of a Th2 response simultaneous to a Th1 could abrogate the protective function of this response in this particular model of infection [26,54], this may explain the lack of protection of the mixed Th1/Th2 induced by the inoculation of the P proteins with CpG ODN. In fact, we have described that the IgG1 and the IL-4 response against the whole parasite is enhanced in those mice presensitized with the acidic ribosomal proteins. A slight exacerbation of the disease may be expected due to this enhancement and, probably, due to the pre-existing immune response against Leishmania P proteins induced by the vaccination. However, this enhancement seems not to be enough to change the clinical features of the disease in the BALB/c, otherwise a highly susceptible strain of mice.
Given that immunomodulation of the Th2 response induced against Leishmania antigens towards a Th1-type has been demonstrated to be a potential strategy for the development of vaccines against leishmaniasis, the existence of certain parasite antigens inducing Th2 responses which cannot be redirected fully by the usual Th1 inducers should be taken into account for the design of a molecularly defined vaccine against this disease.
Acknowledgments
This work was supported by grant PI050973 from FIS. An institutional grant from Fundación Ramón Areces is also acknowledged.
References
- 1.Herwaldt BL. Leishmaniasis. Lancet. 1999;354:1191–9. doi: 10.1016/S0140-6736(98)10178-2. [DOI] [PubMed] [Google Scholar]
- 2.Chang KP, McGwire BS. Molecular determinants and regulation of Leishmania virulence. Kinetoplastid Biol Dis. 2002;1:1. doi: 10.1186/1475-9292-1-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chang KP, Reed SG, McGwire BS, Soong L. Leishmania model for microbial virulence: the relevance of parasite multiplication and pathoantigenicity. Acta Trop. 2003;85:375–90. doi: 10.1016/s0001-706x(02)00238-3. [DOI] [PubMed] [Google Scholar]
- 4.Garcia-Alonso M, Blanco A, Reina D, Serrano FJ, Alonso C, Nieto CG. Immunopathology of the uveitis in canine leishmaniasis. Parasite Immunol. 1996;18:617–23. doi: 10.1046/j.1365-3024.1996.d01-39.x. [DOI] [PubMed] [Google Scholar]
- 5.Garcia-Alonso M, Nieto CG, Blanco A, Requena JM, Alonso C, Navarrete I. Presence of antibodies in the aqueous humour and cerebrospinal fluid during Leishmania infections in dogs. Pathological features at the central nervous system. Parasite Immunol. 1996;18:539–46. doi: 10.1046/j.1365-3024.1996.d01-28.x. [DOI] [PubMed] [Google Scholar]
- 6.Mancianti F, Poli A, Bionda A. Analysis of renal immune-deposits in canine leishmaniasis. Preliminary results. Parasitologia. 1989;31:213–30. [PubMed] [Google Scholar]
- 7.Lopez R, Lucena R, Novales M, Ginel PJ, Martin E, Molleda JM. Circulating immune complexes and renal function in canine leishmaniasis. Zentralbl Veterinarmed B. 1996;43:469–74. doi: 10.1111/j.1439-0450.1996.tb00342.x. [DOI] [PubMed] [Google Scholar]
- 8.Nieto CG, Navarrete I, Habela MA, Serrano F, Redondo E. Pathological changes in kidneys of dogs with natural Leishmania infection. Vet Parasitol. 1992;45:33–47. doi: 10.1016/0304-4017(92)90025-5. [DOI] [PubMed] [Google Scholar]
- 9.Nieto CG, Barrera R, Habela MA, et al. Changes in the plasma concentrations of lipids and lipoprotein fractions in dogs infected with Leishmania infantum. Vet Parasitol. 1992;44:175–82. doi: 10.1016/0304-4017(92)90115-p. [DOI] [PubMed] [Google Scholar]
- 10.Sartori A, Roque-Barreira MC, Coe J, Campos-Neto A. Immune complex glomerulonephritis in experimental kala-azar. II. Detection and characterization of parasite antigens and antibodies eluted from kidneys of Leishmania donovani-infected hamsters. Clin Exp Immunol. 1992;87:386–92. doi: 10.1111/j.1365-2249.1992.tb03007.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Jain A, Berthwal M, Tiwari V, Maitra SC. Immune complex mediated lesions in experimental Kala azar: an ultrastructural study. Indian J Pathol Microbiol. 2000;43:13–16. [PubMed] [Google Scholar]
- 12.Agu WE, Farrell JP, Soulsby EJ. Pathogenesis of anaemia in hamsters infected with Leishmania donovani. Z Parasitenkd. 1982;68:27–32. doi: 10.1007/BF00926654. [DOI] [PubMed] [Google Scholar]
- 13.Miles SA, Conrad SM, Alves RG, Jeronimo SM, Mosser DM. A role for IgG immune complexes during infection with the intracellular pathogen Leishmania. J Exp Med. 2005;201:747–54. doi: 10.1084/jem.20041470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gumy A, Louis JA, Launois P. The murine model of infection with Leishmania major and its importance for the deciphering of mechanisms underlying differences in Th cell differentiation in mice from different genetic backgrounds. Int J Parasitol. 2004;34:433–44. doi: 10.1016/j.ijpara.2003.11.021. [DOI] [PubMed] [Google Scholar]
- 15.Scott P, Artis D, Uzonna J, Zaph C. The development of effector and memory T cells in cutaneous leishmaniasis: the implications for vaccine development. Immunol Rev. 2004;201:318–38. doi: 10.1111/j.0105-2896.2004.00198.x. [DOI] [PubMed] [Google Scholar]
- 16.Sacks D, Noben-Trauth N. The immunology of susceptibility and resistance to Leishmania major in mice. Nat Rev Immunol. 2002;2:845–58. doi: 10.1038/nri933. [DOI] [PubMed] [Google Scholar]
- 17.Campos-Neto A, Porrozzi R, Greeson K, et al. Protection against cutaneous leishmaniasis induced by recombinant antigens in murine and nonhuman primate models of the human disease. Infect Immun. 2001;69:4103–8. doi: 10.1128/IAI.69.6.4103-4108.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mougneau E, Altare F, Wakil AE, et al. Expression cloning of a protective Leishmania antigen. Science. 1995;268:563–6. doi: 10.1126/science.7725103. [DOI] [PubMed] [Google Scholar]
- 19.Pollock KG, McNeil KS, Mottram JC, et al. The Leishmania mexicana cysteine protease, CPB2.8, induces potent Th2 responses. J Immunol. 2003;170:1746–53. doi: 10.4049/jimmunol.170.4.1746. [DOI] [PubMed] [Google Scholar]
- 20.Iborra S, Soto M, Carrion J, Alonso C, Requena JM. Vaccination with a plasmid DNA cocktail encoding the nucleosomal histones of Leishmania confers protection against murine cutaneous leishmaniosis. Vaccine. 2004;22:3865–76. doi: 10.1016/j.vaccine.2004.04.015. [DOI] [PubMed] [Google Scholar]
- 21.Campos-Neto A. What about Th1/Th2 in cutaneous leishmaniasis vaccine discovery? Braz J Med Biol Res. 2005;38:979–84. doi: 10.1590/s0100-879x2005000700001. [DOI] [PubMed] [Google Scholar]
- 22.Skeiky YA, Benson DR, Elwasila M, Badaro R, Burns JM, Jr, Reed SG. Antigens shared by Leishmania species and Trypanosoma cruzi: immunological comparison of the acidic ribosomal P0 proteins. Infect Immun. 1994;62:1643–51. doi: 10.1128/iai.62.5.1643-1651.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Soto M, Requena JM, Quijada L, Guzman F, Patarroyo ME, Alonso C. Identification of the Leishmania infantum P0 ribosomal protein epitope in canine visceral leishmaniasis. Immunol Lett. 1995;48:23–8. doi: 10.1016/0165-2478(95)02436-0. [DOI] [PubMed] [Google Scholar]
- 24.Soto M, Requena JM, Quijada L, et al. During active viscerocutaneous leishmaniasis the anti-P2 humoral response is specifically triggered by the parasite P proteins. Clin Exp Immunol. 1995;100:246–52. doi: 10.1111/j.1365-2249.1995.tb03661.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Soto M, Alonso C, Requena JM. The Leishmania infantum acidic ribosomal protein LiP2a induces a prominent humoral response in vivo and stimulates cell proliferation in vitro and interferon-gamma (IFN-gamma) production by murine splenocytes. Clin Exp Immunol. 2000;122:212–18. doi: 10.1046/j.1365-2249.2000.01372.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Iborra S, Soto M, Carrion J, et al. The Leishmania infantum acidic ribosomal protein P0 administered as a DNA vaccine confers protective immunity to Leishmania major infection in BALB/c mice. Infect Immun. 2003;71:6562–72. doi: 10.1128/IAI.71.11.6562-6572.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Iborra S, Carrion J, Anderson C, Alonso C, Sacks D, Soto M. Vaccination with the Leishmania infantum acidic ribosomal P0 protein plus CpG oligodeoxynucleotides induces protection against cutaneous leishmaniasis in C57BL/6 mice but does not prevent progressive disease in BALB/c mice. Infect Immun. 2005;73:5842–52. doi: 10.1128/IAI.73.9.5842-5852.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Soto M, Requena JM, Garcia M, Gomez LC, Navarrete I, Alonso C. Genomic organization and expression of two independent gene arrays coding for two antigenic acidic ribosomal proteins of Leishmania. J Biol Chem. 1993;268:21835–43. [PubMed] [Google Scholar]
- 29.Coffman RL. Mechanisms of helper T-cell regulation of B-cell activity. Ann NY Acad Sci. 1993;681:25–8. doi: 10.1111/j.1749-6632.1993.tb22865.x. [DOI] [PubMed] [Google Scholar]
- 30.Soto M, Requena JM, Quijada L, Alonso C. Specific serodiagnosis of human leishmaniasis with recombinant Leishmania P2 acidic ribosomal proteins. Clin Diagn Lab Immunol. 1996;3:387–91. doi: 10.1128/cdli.3.4.387-391.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Requena JM, Soto M, Doria MD, Alonso C. Immune and clinical parameters associated with Leishmania infantum infection in the golden hamster model. Vet Immunol Immunopathol. 2000;76:269–81. doi: 10.1016/s0165-2427(00)00221-x. [DOI] [PubMed] [Google Scholar]
- 32.Levin MJ, Vazquez M, Kaplan D, Schijman AG. The Trypanosoma cruzi ribosomal P protein family: classification and antigenicity. Parasitol Today. 1993;9:381–4. doi: 10.1016/0169-4758(93)90088-w. [DOI] [PubMed] [Google Scholar]
- 33.Kurup VP, Banerjee B. Fungal allergens and peptide epitopes. Peptides. 2000;21:589–99. doi: 10.1016/s0196-9781(00)00181-9. [DOI] [PubMed] [Google Scholar]
- 34.Tchorzewski M. The acidic ribosomal P proteins. Int J Biochem Cell Biol. 2002;34:911–15. doi: 10.1016/s1357-2725(02)00012-2. [DOI] [PubMed] [Google Scholar]
- 35.Elkon K, Skelly S, Parnassa A, et al. Identification and chemical synthesis of a ribosomal protein antigenic determinant in systemic lupus erythematosus. Proc Natl Acad Sci USA. 1986;83:7419–23. doi: 10.1073/pnas.83.19.7419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hoff M, Trueb RM, Ballmer-Weber BK, Vieths S, Wuethrich B. Immediate-type hypersensitivity reaction to ingestion of mycoprotein (Quorn) in a patient allergic to molds caused by acidic ribosomal protein P2. J Allergy Clin Immunol. 2003;111:1106–10. doi: 10.1067/mai.2003.1339. [DOI] [PubMed] [Google Scholar]
- 37.Mayer C, Appenzeller U, Seelbach H, et al. Humoral and cell-mediated autoimmune reactions to human acidic ribosomal P2 protein in individuals sensitized to Aspergillus fumigatus P2 protein. J Exp Med. 1999;189:1507–12. doi: 10.1084/jem.189.9.1507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ersvaer E, Bertelsen LT, Espenes LC, et al. Characterization of ribosomal P autoantibodies in relation to cell destruction and autoimmune disease. Scand J Immunol. 2004;60:189–98. doi: 10.1111/j.0300-9475.2004.01450.x. [DOI] [PubMed] [Google Scholar]
- 39.Hirohata S, Nakanishi K. Antiribosomal P protein antibody in human systemic lupus erythematosus reacts specifically with activated T cells. Lupus. 2001;10:612–21. doi: 10.1191/096120301682430195. [DOI] [PubMed] [Google Scholar]
- 40.Sun KH, Tang SJ, Lin ML, Wang YS, Sun GH, Liu WT. Monoclonal antibodies against human ribosomal P proteins penetrate into living cells and cause apoptosis of Jurkat T cells in culture. Rheumatology (Oxf) 2001;40:750–6. doi: 10.1093/rheumatology/40.7.750. [DOI] [PubMed] [Google Scholar]
- 41.Lopez Bergami P, Scaglione J, Levin MJ. Antibodies against the carboxyl-terminal end of the Trypanosoma cruzi ribosomal P proteins are pathogenic. FASEB J. 2001;15:2602–12. doi: 10.1096/fj.01-0132com. [DOI] [PubMed] [Google Scholar]
- 42.Belperron AA, Feltquate D, Fox BA, Horii T, Bzik DJ. Immune responses induced by gene gun or intramuscular injection of DNA vaccines that express immunogenic regions of the serine repeat antigen from Plasmodium falciparum. Infect Immun. 1999;67:5163–9. doi: 10.1128/iai.67.10.5163-5169.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Drew DR, Lightowlers M, Strugnell RA. Humoral immune responses to DNA vaccines expressing secreted, membrane bound and non-secreted forms of the Taenia ovis 45W antigen. Vaccine. 2000;18:2522–32. doi: 10.1016/s0264-410x(00)00020-7. [DOI] [PubMed] [Google Scholar]
- 44.Serezani CH, Franco AR, Wajc M, et al. Evaluation of the murine immune response to Leishmania meta 1 antigen delivered as recombinant protein or DNA vaccine. Vaccine. 2002;20:3755–63. doi: 10.1016/s0264-410x(02)00354-7. [DOI] [PubMed] [Google Scholar]
- 45.Rhee EG, Mendez S, Shah JA, et al. Vaccination with heat-killed Leishmania antigen or recombinant leishmanial protein and CpG oligodeoxynucleotides induces long-term memory CD4+ and CD8+ T cell responses and protection against Leishmania major infection. J Exp Med. 2002;195:1565–73. doi: 10.1084/jem.20020147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Zimmermann S, Egeter O, Hausmann S, et al. CpG oligodeoxynucleotides trigger protective and curative Th1 responses in lethal murine leishmaniasis. J Immunol. 1998;160:3627–30. [PubMed] [Google Scholar]
- 47.Chiaramonte MG, Hesse M, Cheever AW, Wynn TA. CpG oligonucleotides can prophylactically immunize against Th2-mediated schistosome egg-induced pathology by an IL-12-independent mechanism. J Immunol. 2000;164:973–85. doi: 10.4049/jimmunol.164.2.973. [DOI] [PubMed] [Google Scholar]
- 48.Serebrisky D, Teper AA, Huang CK, et al. CpG oligodeoxynucleotides can reverse Th2-associated allergic airway responses and alter the B7.1/B7.2 expression in a murine model of asthma. J Immunol. 2000;165:5906–12. doi: 10.4049/jimmunol.165.10.5906. [DOI] [PubMed] [Google Scholar]
- 49.Kovarik J, Bozzotti P, Love-Homan L, et al. CpG oligodeoxynucleotides can circumvent the Th2 polarization of neonatal responses to vaccines but may fail to fully redirect Th2 responses established by neonatal priming. J Immunol. 1999;162:1611–17. [PubMed] [Google Scholar]
- 50.Peng Z, Wang H, Mao X, HayGlass KT, Simons FE. CpG oligodeoxynucleotide vaccination suppresses IgE induction but may fail to down-regulate ongoing IgE responses in mice. Int Immunol. 2001;13:3–11. doi: 10.1093/intimm/13.1.3. [DOI] [PubMed] [Google Scholar]
- 51.Soto M, Requena JM, Quijada L, Alonso C. Multicomponent chimeric antigen for serodiagnosis of canine visceral leishmaniasis. J Clin Microbiol. 1998;36:58–63. doi: 10.1128/jcm.36.1.58-63.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Molano I, Alonso MG, Miron C, et al. A Leishmania infantum multi-component antigenic protein mixed with live BCG confers protection to dogs experimentally infected with L. infantum. Vet Immunol Immunopathol. 2003;92:1–13. doi: 10.1016/s0165-2427(02)00315-x. [DOI] [PubMed] [Google Scholar]
- 53.Parody N, Soto M, Requena JM, Alonso C. Adjuvant guided polarization of the immune humoral response against a protective multicomponent antigenic protein (Q) from Leishmania infantum. A CpG + Q mix protects Balb/c mice from infection. Parasite Immunol. 2004;26:283–93. doi: 10.1111/j.0141-9838.2004.00711.x. [DOI] [PubMed] [Google Scholar]
- 54.Sjolander A, Baldwin TM, Curtis JM, Handman E. Induction of a Th1 immune response and simultaneous lack of activation of a Th2 response are required for generation of immunity to leishmaniasis. J Immunol. 1998;160:3949–57. [PubMed] [Google Scholar]


