Abstract
The hematopoietic stem cell (HSC) is defined as a cell that can either self-replicate or generate daughter cells that are destined to commit to mature cells of different specific lineages. Self-replication of the most primitive HSC produces daughter cells that possess a long (possibly unlimited) clonal lifespan, whereas differentiation of HSC produces daughter cells that demonstrate a progressive reduction of their clonal lifespan, a loss of multilineage potential, and lineage commitment. Previous studies indicated that the proliferation of HSC ex vivo favors differentiation at the expense of self-replication, eventually resulting in a complete loss of HSC. In contrast, transplantation studies have shown that a single HSC can repopulate the marrow of a lethally irradiated mouse, demonstrating that self-renewal of HSC occurs in vivo. Thrombopoietin (TPO) has been shown to function both as a proliferative and differentiative factor for megakaryocytes and as a survival and weakly proliferative factor for HSC. Our studies focused on the effects of exogenous TPO on HSC in mouse long-term bone marrow cultures (LTBMC). Previous results indicate that HSC decline in LTBMC in the absence of TPO. In contrast, the continuous presence of TPO resulted in the generation of both long- and short-term repopulating HSC as detected by an in vivo competitive repopulation assay. HSC were generated over a 4-month period at concentrations similar to normal bone marrow. Our results demonstrate that TPO can mediate the self-replication of HSC in LTBMC, and provide proof that HSC can self-replicate ex vivo.
All of the different blood cell types arise from pluripotent hematopoietic stem cells (HSC). These cells can maintain hematopoiesis throughout the lifespan of an individual and can reconstitute and maintain hematopoiesis when transplanted into myeloablated recipients. Transplant experiments in mice using highly purified HSC indicate that complete repopulation of all hematopoietic lineages can be attained with one or very few cells (1, 2). The marrow from transplant recipients then can repopulate secondary (and tertiary) recipients, demonstrating that self-replication of HSC occurs in vivo (1–5).
To better understand the mechanisms regulating hematopoiesis, and their disruption in diseases such as cytopenias, dysplasias, and leukemias, it is necessary to be able to identify and isolate large numbers of HSC. Primitive HSC rarely divide in vivo, complicating attempts to expand these cells in culture. Ex vivo culture of purified or unpurified HSC with stem cell growth factors results in dramatic proliferation coupled with a significant loss of HSC activity, suggesting that proliferation of HSC is coupled with differentiation under these conditions (6–8).
Despite these obstacles, much progress has been made regarding the biology of HSC. Among the different cytokines, combinations of stem cell factor (SCF, or c-kit ligand), thrombopoietin (TPO, c-mpl ligand), and the ligand for the Flt3/Flk2 receptor, have been shown to act directly on HSC (8–14). In addition, TPO as a single growth factor supports survival and modest proliferation of highly purified HSC in vitro (10, 11). TPO also has been shown to be the major effector in the proliferation and differentiation of megakaryocytes, the bone marrow precursor of platelets (15). Several lines of evidence implicate TPO in stem cell proliferation in vivo. Administration of TPO has been shown to speed hematopoietic recovery in myelosuppressed animals (16). Targeted disruptions of TPO or its receptor, c-mpl, result not only in thrombocytopenia and megakaryocytopenia, but also in a decreased number of HSC and their progeny (17, 18). Conversely, constitutive overexpression of TPO, either specifically in hematopoietic tissues by transplant of retrovirally transduced marrow cells, or systemically in transgenic mice, results in myeloproliferation, leading to myelofibrosis, dysplasia, and in some cases leukemic transformation (19–21). Thus, TPO modulates the biological responses of HSC both in vitro and in hematopoietic tissues in vivo (22).
We have added TPO to long-term cultures of whole mouse bone marrow (LTBMC) to analyze its role in hematopoiesis. LTBMC, pioneered by Dexter and colleagues (23), remain one of the best in vitro models of hematopoiesis. These cultures develop a complex adherent stromal layer containing a large variety of cell types and can generate nonadherent (NA) hematopoietic cells for periods of several months. We have explored the effects of exogenous TPO in these cultures. We report here the sustained high-level production of both megakaryocytic and nonmegakaryocytic lineages. Further, we present evidence that these cultures produce significant numbers of cells capable of reconstituting hematopoiesis in lethally irradiated mice.
MATERIALS AND METHODS
Cultures.
LTBMC were initiated by flushing cells from femurs of male B6.SJL-Ptprca Pep3b/BoyJ (Ly5.1) (CD45.1) (B6.SJL) mice (The Jackson Laboratory) with medium consisting of Fischer’s medium (GIBCO/BRL, Life Technologies, Gaithersburg, MD) supplemented with 20% heat-inactivated defined horse serum (HyClone), 100 units/ml penicillin, 10 μg/ml streptomycin, 2 mM l-glutamine, and 1 μM hydrocortisone succinate (Sigma) (23, 24). Different lots of horse serum varied significantly in their ability to support the production of clonogenic (and presumably stem) cells (below). Marrow cells from 10 femurs were flushed with complete medium and pooled, and clumps were broken up by aspiration through an 18-gauge needle. Cells were directly distributed without washing into 25-cm2 tissue culture flasks (B-D Falcon, Lincoln Park, NJ) at a density corresponding to one femur per flask. Medium was added to a final volume of 8 ml per flask. Where indicated, recombinant mouse TPO (R & D Systems) was added to a final concentration of 10 ng per ml. Cultures were incubated in a humidified incubator at 37°C in 5% CO2 in air. LTBMC were fed twice weekly (beginning after week one during which no feeding is done) by removing 4 ml (for the subsequent 4 weeks) or 6 ml (thereafter) medium and NA cells, and replacing with an equivalent volume of medium (and TPO in the appropriate cultures).
Immunophenotyping of LTBMC Cells.
NA cells from TPO-containing and control LTBMC were centrifuged and resuspended in 1% (wt/vol) BSA in Dulbecco’s PBS. Fluorochrome-conjugated mAbs to various mouse cluster of differentiation antigens, or biotinylated anti-mouse CD34 and FITC- or phycoerythrin- conjugated strepavidin (PharMingen) were incubated with the cells on ice (1 μg antibody/1–2 × 105 cells). Cells were washed and analyzed by flow cytometry (FACScan, Becton Dickinson) in the presence of propidium iodide to exclude dead cells.
Clonogenic Cell Assays.
Colony formation assays were performed in soft agar cultures in the presence of recombinant cytokines (R & D Systems or PeproTec, Rocky Hill, NJ) (11). Two thousand to 5,000 cells were added per ml of culture and plated in 35-mm dishes. Cultures were incubated for 12 days, and colonies were counted by using an inverted microscope. In some experiments, cells were plucked from colonies, and their morphology was assessed after staining with Giemsa. Cytokines were used at the following concentrations: for colony-forming cells (CFCs), 5 ng/ml mouse granulocyte/macrophage-CSF and 10% (vol/vol) L929 supernatant (mouse macrophage-CSF); for high proliferative potential-CFC (HPP-CFC), 50 ng/ml rat stem cell factor, 20 ng/ml human IL-6, and 10 ng/ml mouse IL-3.
Transplants and Competitive Repopulation Assays.
NA cells from LTBMC established from B6.SJL mice (CD45.1) were harvested, washed, and used unfractionated for transplant. For each test sample, 2–10 recipient C57BL6 mice (CD45.2) were irradiated (950 rad, 137Cesium source) and transplanted by injection via the tail vein with the indicated number of test cells mixed with 4 × 105 fresh unfractionated CD45.2 marrow cells. In some experiments, fresh unfractionated bone marrow cells from CD45.1 animals were transplanted in parallel with the cells from LTBMC. Animals were maintained in microisolator cages in a specific pathogen-free facility. Peripheral blood samples were obtained by retro-orbital bleeding 3, 6, 12, and 24 weeks posttransplant. Expression of the donor CD45.1 allele and lineage-specific antigens was assessed by two-color flow cytometry analysis of peripheral blood leukocytes using directly labeled mAbs as described above for cultured cells. The frequency of long-term repopulating units was estimated by using the maximum-likelihood model that requires limiting dilution cell transplants of the test cells (25).
RESULTS
TPO Alters the Kinetics of Total Cell Production in LTBMC.
The addition of TPO to LTBMC at initiation (TPO-LTBMC) had dramatic effects on both the adherent and NA cells in LTBMC. Although extensive characterization of the adherent cells was not performed, differences in the composition of the stromal layer were apparent between control and TPO-LTBMC, both initially and throughout the life of the cultures. In TPO-LTBMC stroma, adipocytes were rarely observed, and the frequency of macrophages and fibroblasts appeared to be decreased. Early TPO-LTBMC had heavy stromal layers with abundant, adherent small round cells (hematopoietic in morphology), and very large, adherent round cells (presumptive megakaryocytes). At later times in culture (>12 weeks), the stroma in TPO-LTBMC was noticeably more sparse than in control cultures, and megakaryocytes continued to be abundant.
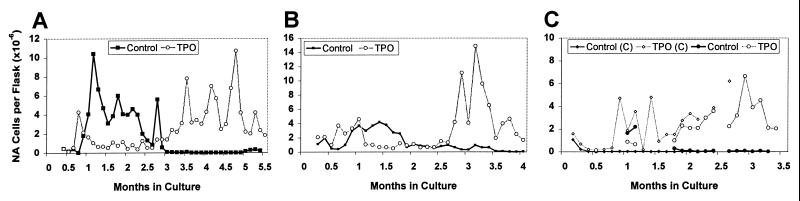
Addition of TPO also affected production of NA hematopoietic cells over the life of the culture. TPO-LTBMC initially produced large cells with a megakaryocytic morphology. As previously reported by Dexter and many other laboratories (6, 23, 24), maintaining LTBMC at 37°C resulted in a decline in NA cell production in control cultures (Fig. 1). In contrast, although NA cell production from the TPO-LTBMC was initially less than that of control LTBMC, it increased after the first 8 weeks and was sustained at high levels throughout the subsequent weeks of culture (Fig. 1). As illustrated, the kinetics of cell production from control cultures varied between different experiments; however, the sustained production of NA cells in TPO-LTBMC was observed in all experiments. These variations may be caused in part by differences between lots of horse serum used in these experiments. The data presented in Fig. 1 A and B are from the cultures used in the experiments described below. In contrast to the predominantly myeloid morphology of the NA cells from untreated LTBMC, the cells from TPO-LTBMC were heterogeneous and included recognizable precursors of all lineages (not shown).
Figure 1.
Production of NA cells in LTBMC. Cells harvested from semiweekly feedings of LTBMC were pooled, counted, averaged, and corrected for the volume of media harvested. (A and B) NA cell production by the cultures used for transplants. Two sets of LTBMC are depicted in C.
TPO Alters the Cell Types Generated in LTBMC.
The expression of hematopoietic cell markers on the NA cells was assessed by flow cytometry. Many of the NA cells from TPO-LTBMC (45–98%) express the megakaryocytic markers CD41 (platelet GPIIb) and CD61 (GPIIIa) (Table 1). Characterization of several other hematopoietic markers showed a surprisingly high number of NA cells from TPO-LTBMC expressed cell surface antigens characteristic of primitive HSC. At 4 weeks of culture, approximately 0.5% of the NA cells from both control and TPO-LTBMC expressed high levels of c-kit and Sca-1 (Table 1). This phenotype is observed on less than 0.1% of normal unfractionated marrow cells and is associated with HSC (11, 26). In the NA cells from 10-week cultures, 0.02% of cells from control and 0.9% of cells from TPO-LTBMC expressed these two stem cell markers (Table 1). Even after 16 weeks of culture, 0.2% of the NA cells from TPO-LTBMC were c-kithigh/Sca-1high (Table 1). These results indicated that cells with the phenotype of HSC were proliferating in the TPO-LTBMC.
Table 1.
Immunophenotyping of NA cells from LTBMC
| Marker | Fresh marrow | Time in culture
|
|||||
|---|---|---|---|---|---|---|---|
| 4 weeks
|
10 weeks
|
16 weeks
|
|||||
| Control | TPO | Control | TPO | Control | TPO | ||
| c-kithigh/Sca-1high | 0.03 | 0.5 | 0.6 | 0.02 | 0.9 | * | 0.2 |
| CD41/CD61 | 5 | ND | 47.5 | 2.7 | 9.8 | * | 80 |
Numbers in the table indicate the percent positive cells. Percentages for fresh marrow are averages based on multiple determinations. ND, not determined. ∗, not enough cells to analyze.
TPO Increases the Production of Clonogenic Cells, Including HPP-CFC, in LTBMC.
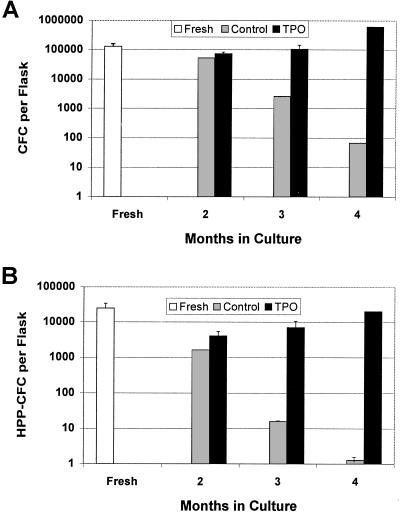
The clonogenic potential of the NA cells from TPO-LTBMC was analyzed by colony formation in soft agar in the presence of various cytokines. As shown in Fig. 2 and Table 2, there was a striking difference in both the frequency and absolute number of CFC in the NA cells from the TPO-LTBMC as compared with the control LTBMC. Both the number and frequency of CFC in control LTBMC decreased with time in culture. In contrast, the frequency of CFC in the NA cells from TPO-LTBMC remained approximately constant up to 4 months in culture. The observed differences in CFC frequency were more striking for the primitive HPP-CFC than for the more mature myeloid CFC (Fig. 2, Table 2). The results of the HPP-CFC assay demonstrated that this class of cells (which includes most HSC, refs. 27 and 28) was being generated throughout the culture period in TPO-LTBMC but not control-LTBMC.
Figure 2.
Production of CFC and HPP-CFC in LTBMC. At various times in culture, 2,000–5,000 NA cells were cultured and colonies were counted as described in Materials and Methods. The frequency of colonies obtained was multiplied by the number of NA cells per flask to obtain the number of CFC per flask. Total number of (A) CFC per flask and (B) HPP-CFC per flask.
Table 2.
In vitro colony-forming cell production in LTBMC
| Culture | Time in culture, months
|
||||
|---|---|---|---|---|---|
| 0 | 2 | 3 | 4 | ||
| Cells per flask (×10−6) | Control | 10 | 6 | 0.08 | 0.006 |
| TPO | " | 0.8 | 1.4 | 4.3 | |
| CFC per flask (×10−3) | Control | 130 ± 30 | 52 ± 4.2 | 2.6 ± 0.1 | 0.068 ± 0.001 |
| TPO | " | 73 ± 1 | 105 ± 10 | 610 ± 40 | |
| HPP-CFC per flask (×10−3) | Control | 25 ± 9 | 1.62 ± 0.36 | 0.016 ± 0.006 | 0.0013 ± 0.0006 |
| TPO | " | 4.03 ± 0.89 | 7.0 ± 1.4 | 20.2 ± 3.55 | |
TPO Mediates the Generation of Short- and Long-Term Repopulating HSC in LTBMC.
The sustained production of HPP-CFC and multilineage CFC, coupled with the primitive cell surface antigen phenotype of the NA cells in TPO-treated LTBMC (c-kit+ and c-kit+/Sca-1+ cells), suggested that HSC were being produced in these cultures. To test this, NA cells from cultures initiated from the CD45.1 strain B6.SJL were transplanted into lethally irradiated C57BL6 (CD45.2) animals along with 4 × 105 competitor cells (normal unfractionated marrow cells from C57BL6 animals). Peripheral blood was assayed for expression of the CD45.1+ donor cells expressing lineage-specific markers at various times posttransplant to assess the contribution of the cultured cells to hematopoiesis. The results are summarized in Table 3 and Fig. 3.
Table 3.
Hematopoietic reconstitution by NA cells from LTBMC
| Cell source | Weeks after transplant
|
|||
|---|---|---|---|---|
| 3 | 6 | 12 | 24 | |
| Two-month LTBMC† | ||||
| Fresh marrow | 86 ± 8.4 (3/3) | 61.7 ± 14.0 (3/3) | 61.6 ± 15.0 (3/3) | 67.2 ± 4.7 (3/3) |
| Control LTBMC | 22.7 ± 7.7 (4/4) | 7.3 ± 2.9 (3/3) | 1.7 ± 1.5 (4/4) | 0.58 ± 0.57 (2/4) |
| TPO-LTBMC | 94.0 ± 4.8 (3/3) | 70.4 ± 15.6 (3/3) | 62.1 ± 20.5 (3/3) | 53.7 ± 32.5 (3/3) |
| Three-month TPO-LTBMC‡ | ||||
| 25,000 | 4.7 ± 5.4 (3/4) | 0.8 ± 1.6 (1/4) | 0.8 ± 1.6 (1/4) | 0.5 ± 1.0 (1/4) |
| 50,000 | 11.5 ± 8.6 (2/2) | 12.3 ± 7.1 (2/2) | 9.0* | 43.6* |
| 100,000 | 39.0 ± 11.0 (3/3) | 34.4 ± 15.0 (3/3) | 26.2 ± 4.1 (3/3) | 17.4 ± 5.4 (3/3) |
| 200,000 | 38.9 ± 9.1 (3/3) | 29.2 ± 14.4 (3/3) | 41.4 ± 36.3 (3/3) | 36.7 ± 41.7 (3/3) |
| Four-month TPO-LTBMC‡ | ||||
| 100,000 | 14.9 ± 3.3 (4/4) | 3.4 ± 2.4 (4/4) | 1.8 ± 2.0 (4/4) | 0.7 ± 1.2 (1/4) |
| 500,000 | 30.0 ± 17.5 (5/5) | 10.3 ± 1.9 (5/5) | 5.6 ± 1.4 (5/5) | 3.4 ± 1.4 (5/5) |
Numbers indicate the percent (±1 SD) CD45.1+ peripheral blood leukocytes. Numbers in parentheses indicate the number of positive animals (>0.5% CD45.1+)/number transplanted.
Only one animal analyzed.
Data are pooled from two experiments, with either 5.5 × 105 or 1.3 × 106 cells transplanted per animal.
Number of NA cells transplanted per animal.
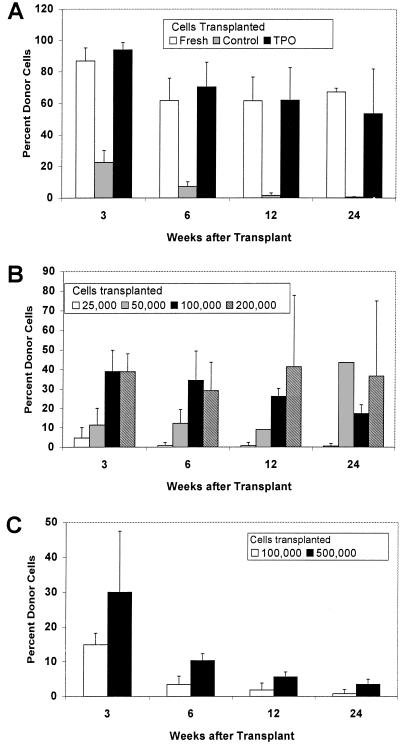
Figure 3.
Competitive repopulation assays of culture-derived HSC. (A) Long-term repopulating HSC in 2-month-old TPO-LTBMC, but not control LTBMC. Lethally irradiated C57BL/6 (CD45.2) mice were transplanted with fresh CD45.1 bone marrow cells only, or with 5 × 105 CD45.2 bone marrow cells plus 5.5 × 105 or 1.34 × 106 CD45.1 NA cells from control or TPO-LTBMC. Bars indicate the percent CD45.1+ cells in peripheral blood at various times after transplant. (B) Limiting dilution analysis of cells from 3-month-old TPO-LTBMC. Lethally irradiated C57BL/6 mice were transplanted with 3 × 105 C57BL/6 bone marrow cells, and 2.5 × 104, 5.0 × 104, 1 × 105, or 2 × 105 NA cells from 3-month TPO-LTBMC. (C) HSC in NA cells from 4-month-old TPO-LTBMC. Lethally irradiated animals were transplanted with syngeneic normal bone marrow and 1 × 105 or 5 × 105 NA cells, and analyzed as described above.
Transplantation of 0.5 or 1.3 × 106 NA cells from 2-month-old TPO-LTBMC resulted in lymphoid and myeloid reconstitution comparable to that of the same number of fresh bone marrow cells (Table 3, Fig. 3A). In both cases, chimeras were generated in which CD45.1+ donor cells comprised 30–80% of mature cells in the peripheral blood up to 6 months after transplant, demonstrating the presence of long-term repopulating HSC in TPO-LTBMC. In contrast, transplant of over 106 NA cells from 2-month-old control LTBMC resulted in only transient repopulation, with few CD45.1+ cells detectable after 1.5 months. Analysis of the circulating CD45.1+ (donor) cells indicated that NA cells from TPO-LTBMC populated both the lymphoid (B220+, B cell; or Thy1+, T cell) and myeloid (Gr-1+, granulocyte; or Mac-1+, monocyte) lineages similarly to unfractionated fresh bone marrow cells (Table 4).
Table 4.
Reconstitution of myeloid and lymphoid lineages by NA cells from TPO-LTBMC
| Cells | Time after transplant
|
|||||
|---|---|---|---|---|---|---|
| Fresh marrow
|
TPO-LTBMC
|
|||||
| 3 weeks | 6 weeks | 24 weeks | 3 weeks | 6 weeks | 24 weeks | |
| Lymphoid | ||||||
| (B220+ or Thy-1+) | 37 | 30 | 61 | 48 | 25 | 64 |
| Myeloid | ||||||
| (Gr-1+ or Mac1+) | 63 | 70 | 39 | 52 | 75 | 36 |
Peripheral blood samples from the animals illustrated in Fig. 3A were analyzed for expression of CD45.1 and B220, Thy-1, Gr-1, or Mac-1 at the times indicated. Numbers indicate the percent of CD45.1+ cells (average of 2–4 animals) that also expressed the markers indicated.
Results of transplanting graded doses of NA cells from 3-month-old TPO-LTBMC are shown in Fig. 3B. Repopulation in most animals was sustained for 24 weeks, again indicating the presence of long-term repopulating HSC in the NA cells from TPO-LTBMC. As few as 5 × 104 cells were capable of persistent (>4 months) repopulation in all of the recipient animals; however, at a dose of 2.5 × 104 cells, 75% of mice were negative (<0.5%) for circulating CD45.1+ donor cells at 4 months. Based on the data from this experiment, the frequency of long-term repopulating HSC in the NA cells of TPO-LTBMC was calculated to be 1 per 3.0 × 104 cells, compared with the frequency of HSC in fresh bone marrow of 1 per 104 (25).
The repopulating potential of the HSC was determined by calculating the absolute number of donor-derived circulating leukocytes generated from a limited number of HSC (Table 5). These numbers were derived by multiplying the percent donor cells by the leukocyte counts per microliter blood. Transplant of 5 × 104 fresh bone marrow cells (containing approximately five long-term repopulating cells) generated 500 leukocytes per μl blood 3 weeks posttransplant, and 2,500 leukocytes/μl at 3 months. In comparison, 1 × 105 TPO-LTBMC-derived cells (containing approximately three long-term repopulating cells) generated 2,700 leukocytes/μl at 3 weeks, and 3,500 leukocytes/μl at 3 months. Because approximately half the donor-derived leukocytes in the circulation are myeloid, with a turnover time of approximately 3 days, these results indicate that the proliferative potential of the culture-derived HSC equals or exceeds that of fresh marrow-derived HSC. In addition, the higher number of donor-derived peripheral blood cells at 3 weeks in animals receiving TPO-LTBMC-derived HSC suggests that the cultured HSC can repopulate the peripheral blood more rapidly than marrow-derived HSC.
Table 5.
Repopulating ability of HSC from marrow and TPO-LTBMC
| Number of cells transplanted | Donor-derived peripheral blood leukocytes/μl
|
|||
|---|---|---|---|---|
| Marrow
|
TPO-LTBMC
|
|||
| 3 weeks* | 3 months* | 3 weeks* | 3 months* | |
| 50,000 | 504 | 2,480 | 360 | 852 |
| 100,000 | 552 | 4,087 | 2,673 | 3,640 |
Peripheral blood samples from the animals illustrated in Fig. 3B were first analyzed by flow cytometry for the percent of peripheral blood leukocytes expressing the donor-derived CD45.1 allele. This percentage was multiplied by the number of leukotyces per microliter of blood (from microscopic counting with a hemocytometer) to obtain the absolute number of donor-derived cells per microliter.
Time after transplant.
The results of transplantation of NA cells from 4-month-old TPO-LTBMC are presented in Fig. 3C. The relatively low percentage of CD45.1+ cells in the recipients, despite the large numbers of cells transplanted, indicates that the frequency of repopulating cells was decreased relative to the previous transplants. Further, the steep decline in the percentage of donor cells detected in the circulation during the first 12 weeks posttransplant suggests that the majority of transplanted cells are short-term repopulating cells. However, the persistence of detectable (average 3.4%) donor-derived cells 24 weeks posttransplant indicates that a low number of long-term repopulating cells are still present after 4 months in culture.
Limiting dilution transplants were performed with a second set of control and TPO-LTBMC to assess HSC frequency after 1 and 2 months of culture (Table 6). At the present time the recipients are only 3 months posttransplant, so the relative contributions of short- and long-term repopulating HSC are not clear; however, the frequency of total HSC can be estimated. After 1 month in culture, HSC frequencies in both control and TPO-LTBMC are approximately one-tenth that of fresh marrow (1.6 and 0.9 per 105 cells, respectively). At 2 months, however, the HSC frequency in TPO-LTBMC reached nearly that of fresh marrow (6.7 per 105 cells), while that of control LTBMC remained low (0.9 per 105 cells). These results are consistent with the results of the earlier transplants. Further, these results strongly suggest that the HSC observed in 2- and 3-month-old TPO-LTBMC are generated by expansion in culture and are not simply the result of prolonged HSC survival.
Table 6.
HSC production early in LTBMC
| Culture
|
Percent positive animals (no. positive/transplanted)
|
Repopulating units per 105 cells (95% confidence interval) | |||||
|---|---|---|---|---|---|---|---|
| Type | Time | 5,000 cells* | 10,000 cells* | 25,000 cells* | 50,000 cells* | 100,000 cells* | |
| TPO | 1 month | 0 (0/6) | 0 (0/7) | 33 (2/6) | 75 (3/4) | 0.9 (0.1–1.7) | |
| 2 months | 13 (1/8) | 57 (5/7) | 100 (5/5) | 100 (5/5) | 6.7 (2.8–11.5) | ||
| Control | 1 month | 13 (1/8) | 60 (3/5) | 1.6 (0.1–3.0) | |||
| 2 months | 0 (0/5) | 14 (1/7) | 33 (2/6) | 60 (3/5) | 0.9 (0.2–1.6) | ||
Animals were assayed 3 months posttransplant. Transplanted animals were considered positive if >0.5% of the peripheral leukocytes expressed CD45.1. Repopulating units were calculated by using the maximum-likelihood model (25).
Number of NA cells transplanted.
DISCUSSION
The results presented here demonstrate that the addition of TPO alone to Dexter LTBMC supports the expansion of both short- and long-term repopulating HSC. We report that HSC can be significantly expanded, rather than maintained, ex vivo. Our results indicate that HSC can be generated de novo in TPO-LTBMC. In addition, the culture-derived HSC are functionally normal, and transplant recipients with chimeras up to 85–90% donor cells are normal and healthy >9 months after transplant.
Previous experiments indicated that, although NA cells from LTBMC retained colony-forming potential, stem cell activity was rapidly lost from culture (29). Certain stromal cell lines maintain HSC activity for up to 5 weeks in vitro, but the expansion of HSC reported was modest (30). Our results indicate that the addition of TPO alone is sufficient to modify LTBMC conditions such that HSC are produced without significant loss of differentiative or repopulating potential for at least 4 months in culture. After 2 months of culture in the presence of TPO, the frequency of transplantable cells appears to be nearly that of normal bone marrow. Transplant of NA cells from 3-month-old TPO-LTBMC indicates that, although the frequency of stem cells is approximately one-third that of fresh bone marrow, the ability of these cells to mediate short- and long-term reconstitution equals or exceeds that of fresh cells. Long-term repopulating cells also are found in NA cells from 4-month-old TPO-LTBMC, albeit at a lower frequency. These results indicate that HSC are produced in these cultures at a rate only slightly less than that of total NA cell production. Finally, the function of the culture-derived HSC, as assessed by the kinetics of repopulation, the lineages repopulated, and the number of progeny generated, is similar to that of fresh marrow-derived HSC.
The kinetics of HSC production in TPO-LTBMC are complex. The effect of TPO on HSC can be influenced in part by unknown factors present in different lots of horse serum. The delay in production of NA cells in TPO-LTBMC as compared with control LTBMC, as well as the sustained production at later culture times (Fig. 1), suggests that TPO suppresses differentiation and/or promotes survival of HSC. Our preliminary data indicate that HSC levels decline to low numbers after 1 month in both control and TPO-LTBMC but rebound in TPO-LTBMC to levels comparable to fresh marrow by 2 months. This result suggests that the HSC observed at later times in TPO-LTBMC arise de novo in culture.
The effect of TPO on hematopoiesis in LTBMC extends to more mature progenitor cells. Previous studies demonstrated that CFC can be generated in LTBMC incubated at 33°C for extended periods, and that culture at 37°C reduces the generation of CFC (23). Our results show that the addition of TPO to LTBMC at 37°C supports the generation not only of CFC, but also of the more primitive HPP-CFC. In fact, our results show that, over 4 months of culture at 37°C, the total number of HPP-CFC increased despite the semiweekly removal of 2/3 of the NA cells during feeding. Thus, HPP-CFC are continually produced in TPO-LTBMC at a rate higher than that of overall cell production. HPP-CFC have been shown to reflect HSC, CFU-S, and other primitive cells, some of which are not transplantable (27, 28).
Our results are in contrast to those of several laboratories that indicate that proliferation of HSC is associated with the loss of repopulating capacity (6–8). Many of these studies used highly purified cell populations cultured in the presence of recombinant cytokines. Previous work from several laboratories indicates that combinations including TPO with other cytokines support the survival and expansion of primitive progenitor cells (10–13, 16, 22). In combination with Flt3/Flk2 ligand, TPO has been reported to support the expansion of human umbilical cord blood HSC up to 2 × 105-fold (31). In addition, TPO modifies the expression of adhesion receptors on HSC and cell lines, inducing adhesion to stroma and extracellular matrix (32, 33). Finally, long-term repopulating HSC express c-mpl (34). Thus, it is possible that TPO is acting directly on the HSC in these cultures. Recent results from our laboratory indicate that TPO can act on individual purified HSC to increase the generation of HPP-CFC, as opposed to more mature myeloid CFC (35).
TPO additionally may mediate its effects in LTBMC by acting through megakaryocytes or other responsive cells. Studies indicate that the presence of stromal cells preserve the repopulating potential of cultured HSC (30, 36). Bone marrow stromal cells and cell lines express TPO, along with many other cytokines (37–41). An endothelial cell line that supports survival of HSC expresses both TPO and c-mpl, the TPO receptor (42). The role of megakaryocytes in the regulation of early hematopoiesis is unknown; however, megakaryocytes produce a variety of cytokines that act on HSC, including granulocyte/macrophage-CSF and transforming growth factor β (43, 44). TPO also can stimulate release of cytokines from c-mpl-expressing cells (45). Thus, the addition of TPO may significantly alter the spectrum of cytokines produced by megakaryocytes and other cells in LTBMC stroma.
The availability of large numbers of cycling but still pluripotent cells with significant reconstituting ability may be useful for autologous stem cell transplant and retroviral gene therapy. The delayed production of HSC in TPO-LTBMC would not necessarily preclude their use in stem cell expansion; patients may undergo chemotherapy and radiation treatments of several weeks duration before transplant. Attempts to attain high levels of introduction of exogenous genes by using retroviral vectors have been hampered both by the relative rarity of HSC and their quiescence (46). The rarity of HSC in total marrow makes infection inefficient, and retroviral vectors require at least one round of cellular DNA synthesis to stably integrate in the genome. Furthermore, stimulation of highly purified HSC to undergo cell division generally is accompanied by a loss in repopulating potential. Therefore, most attempts to achieve high levels of retrovirally transduced transplantable stem cells have met with limited success. Thus, the NA cells from TPO-treated LTBMC, which appear to be cycling cells with significant repopulating ability, are an ideal target for transduction by viral vectors.
Acknowledgments
We thank Doris Damian, Department of Biostatistics, University of Washington, for assistance with statistical modeling, and Susan Danner for excellent technical help. This work was supported in part by National Institutes of Health Awards HL02959 (K.A.R.), HL39947 (G.J.R.), and DK48708 (S.B.), and a Merit Review Award from the Department of Veterans Affairs (G.J.R.).
ABBREVIATIONS
- CFC
colony-forming cell
- HPP-CFC
high proliferative potential CFC
- HSC
hematopoietic stem cell
- LTBMC
long-term bone marrow culture
- NA
nonadherent
- TPO
thrombopoietin
References
- 1.Smith L G, Weissman I L, Heimfeld S. Proc Natl Acad Sci USA. 1991;88:2788–2792. doi: 10.1073/pnas.88.7.2788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Osawa M, Hanada K, Hamada H, Nakauchi H. Science. 1996;273:242–245. doi: 10.1126/science.273.5272.242. [DOI] [PubMed] [Google Scholar]
- 3.Dick J E, Magli M C, Huszar D, Phillips R A, Bernstein A. Cell. 1985;42:71–79. doi: 10.1016/s0092-8674(85)80102-1. [DOI] [PubMed] [Google Scholar]
- 4.Jordan C T, Lemischka I R. Genes Dev. 1990;4:220–232. doi: 10.1101/gad.4.2.220. [DOI] [PubMed] [Google Scholar]
- 5.Keller G, Snodgrass R. J Exp Med. 1990;171:1407–1418. doi: 10.1084/jem.171.5.1407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.van der Loo J C M, Ploemacher R E. Blood. 1995;85:2598–2606. [PubMed] [Google Scholar]
- 7.Traycoff C M, Cornetta K, Yoder M C, Davidson A, Srour E F. Exp Hematol. 1996;24:299–306. [PubMed] [Google Scholar]
- 8.Ogawa M, Yonemura Y, Ku H. Stem Cells. 1997;15, Suppl. 1:7–12. doi: 10.1002/stem.5530150803. [DOI] [PubMed] [Google Scholar]
- 9.Ku H, Yonemura Y, Kaushansky K, Ogawa M. Blood. 1996;87:4544–4551. [PubMed] [Google Scholar]
- 10.Ramsfjell V, Borge O J, Veiby O P, Cardier J, Murphy M J, Jr, Lyman S D, Lok S, Jacobsen S E. Blood. 1996;88:4481–4492. [PubMed] [Google Scholar]
- 11.Sitnicka E, Lin N, Priestley G V, Fox N, Broudy V C, Wolf N S, Kaushansky K. Blood. 1996;87:4998–5005. [PubMed] [Google Scholar]
- 12.Young J C, Bruno E, Luens K M, Wu S, Backer M, Murray L J. Blood. 1996;88:1619–1631. [PubMed] [Google Scholar]
- 13.Yoshida M, Tsuji K, Ebihara Y, Muraoka K, Tanaka R, Miyazaki H, Nakahata T. Br J Haematol. 1997;98:254–264. doi: 10.1046/j.1365-2141.1997.2283045.x. [DOI] [PubMed] [Google Scholar]
- 14.Matsunaga T, Kato T, Miyazaki H, Ogawa M. Blood. 1998;92:452–461. [PubMed] [Google Scholar]
- 15.Kaushansky K. Blood. 1995;86:419–431. [PubMed] [Google Scholar]
- 16.Grossmann A, Lenox J, Ren H P, Humes J M, Forstrom J W, Kaushansky K, Sprugel K H. Exp Hematol. 1996;24:1238–1246. [PubMed] [Google Scholar]
- 17.Kimura S, Roberts A W, Metcalf D, Alexander W S. Proc Natl Acad Sci USA. 1998;95:1195–1200. doi: 10.1073/pnas.95.3.1195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Murone M, Carpenter D A, de Sauvage F J. Stem Cells. 1998;16:1–6. doi: 10.1002/stem.160001. [DOI] [PubMed] [Google Scholar]
- 19.Yan X Q, Lacey D, Fletcher F, Hartley C, McElroy P, Sun Y, Xia M, Mu S, Saris C, Hill D, et al. Blood. 1995;86:4025–4033. [PubMed] [Google Scholar]
- 20.Villeval J L, Cohen S-K, Tulliez M, Giraudier S, Guichard J, Burstein S A, Cramer E M, Vainchenker W, Wendling F. Blood. 1997;90:4369–4383. [PubMed] [Google Scholar]
- 21.Zhou W, Toombs C F, Zou T, Guo J, Robinson M O. Blood. 1997;89:1551–1559. [PubMed] [Google Scholar]
- 22.Kaushansky K. Blood. 1998;92:1–3. [PubMed] [Google Scholar]
- 23.Dexter T M, Simmons P, Purnell R A, Spooncer E, Schofield R. Prog Clin Biol Res. 1984;148:13–33. [PubMed] [Google Scholar]
- 24.Pollack S B, Tsuji J, Rosse C. Cell Immunol. 1992;139:352–362. doi: 10.1016/0008-8749(92)90077-3. [DOI] [PubMed] [Google Scholar]
- 25.Taswell C. J Immunol. 1981;126:1614–1619. [PubMed] [Google Scholar]
- 26.Okada S, Nakauchi H, Nagayoshi K, Nishikawa S, Miura Y, Suda T. Blood. 1992;80:3044–3050. [PubMed] [Google Scholar]
- 27.Wolf N S, Kone A, Priestly G V, Bartelmez S H. Exp Hematol. 1993;21:614–622. [PubMed] [Google Scholar]
- 28.Sitnicka E, Ruscetti F W, Priestly G V, Wolf N S, Bartelmez S H. Blood. 1996;88:82–88. [PubMed] [Google Scholar]
- 29.van der Sluijs J P, van den Bos C, Baert M R, van Beurden C A, Ploemacher R E. Leukemia. 1993;7:725–732. [PubMed] [Google Scholar]
- 30.Moore K A, Ema H, Lemischka I R. Blood. 1997;89:4337–4347. [PubMed] [Google Scholar]
- 31.Piacibello W, Sanavio F, Garetto L, Severino A, Bergandi D, Ferrario J, Fagioli F, Berger M, Aglietta M. Blood. 1997;89:2644–2653. [PubMed] [Google Scholar]
- 32.Cui L, Ramsfjell V, Borge O J, Veiby O P, Lok S, Jacobsen S E. J Immunol. 1997;159:1961–1969. [PubMed] [Google Scholar]
- 33.Gotoh A, Ritchie A, Takahira H, Broxmeyer H E. Ann Hematol. 1997;75:207–213. doi: 10.1007/s002770050344. [DOI] [PubMed] [Google Scholar]
- 34.Solar G P, Kerr W G, Zeigler F C, Hess D, Donahue C, de Sauvage F J, Eaton D L. Blood. 1998;92:4–10. [PubMed] [Google Scholar]
- 35.Sitnicka E, Storey C, Bartelmez S H. Blood. 1998;92, Suppl. 1:59a. (abstr.). [Google Scholar]
- 36.Breems D A, Blokland E A W, Siebel K E, Mayen A E M, Engels L J A, Ploemacher R E. Blood. 1998;91:111–117. [PubMed] [Google Scholar]
- 37.Guerriero A, Worford L, Holland H K, Guo G R, Sheehan K, Waller E K. Blood. 1997;90:3444–3455. [PubMed] [Google Scholar]
- 38.Hirayama Y, Sakamaki S, Matsunaga T, Kuga T, Kuroda H, Kusakabe T, Sasaki K, Fujikawa K, Kato J, Kogawa K, et al. Blood. 1998;92:46–52. [PubMed] [Google Scholar]
- 39.Mitjavila M T, Filippi M D, Cohen S-K, Le P-F, Vainchenker W, Sainteny F. Exp Hematol. 1998;26:124–134. [PubMed] [Google Scholar]
- 40.Sensebe L, Deschaseaux M, Li J, Herve P, Charbord P. Stem Cells. 1997;15:133–143. doi: 10.1002/stem.150133. [DOI] [PubMed] [Google Scholar]
- 41.Sungaran R, Markovic B, Chong B H. Blood. 1997;89:101–107. [PubMed] [Google Scholar]
- 42.Cardier J E, Dempsey J. Blood. 1998;91:923–929. [PubMed] [Google Scholar]
- 43.Jiang S, Levine J D, Fu Y, Deng B, London R, Groopman J E, Avraham H. Blood. 1994;84:4151–4156. [PubMed] [Google Scholar]
- 44.Wickenhauser C, Hillienhof A, Jungheim K, Lorenzen J, Ruskowski H, Hansmann M L, Thiele J, Fischer R. Leukemia. 1995;9:310–315. [PubMed] [Google Scholar]
- 45.Bobik R, Hong Y, Breier G, Martin J F, Erusalinsky J D. FEBS Lett. 1998;423:10–14. doi: 10.1016/s0014-5793(98)00056-8. [DOI] [PubMed] [Google Scholar]
- 46.Tisdale J F, Hanazono Y, Sellers S E, Agricola B A, Metzger M E, Donahue R E, Dunbar C E. Blood. 1998;92:1131–1141. [PubMed] [Google Scholar]