Abstract
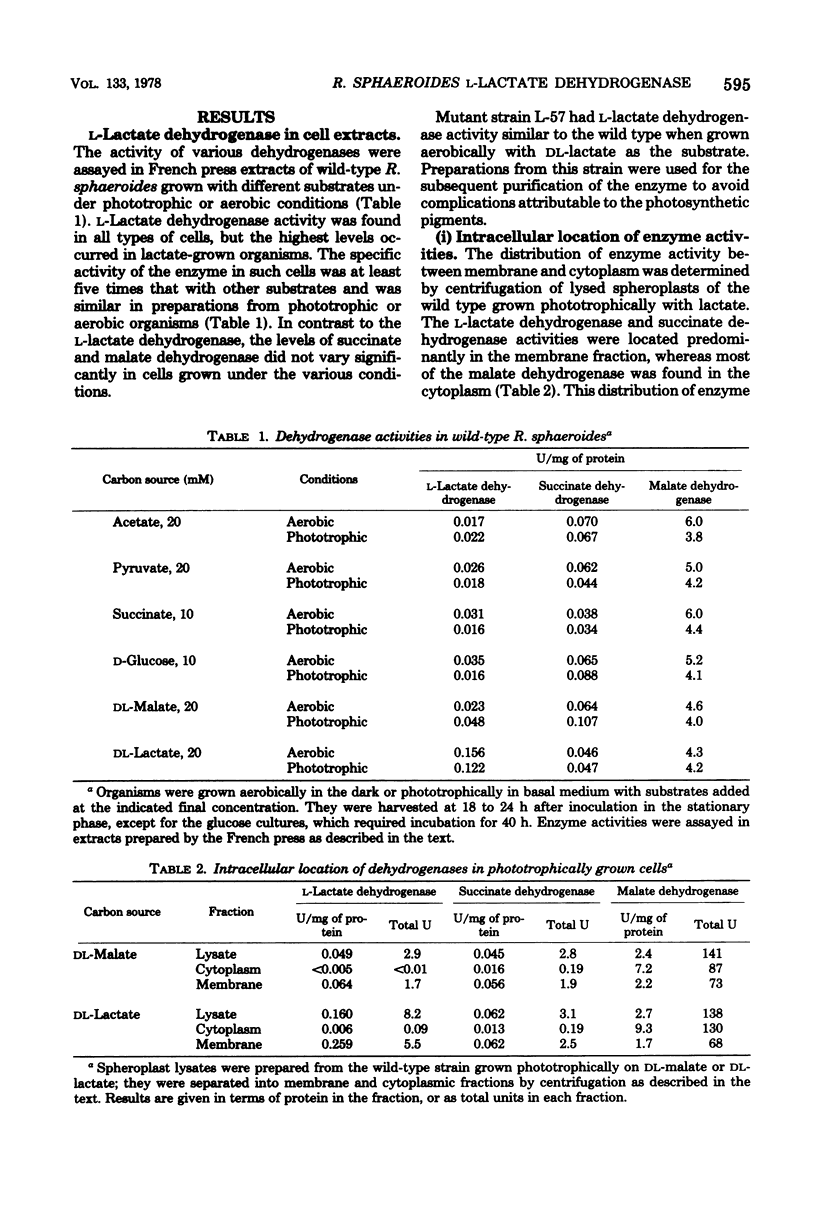
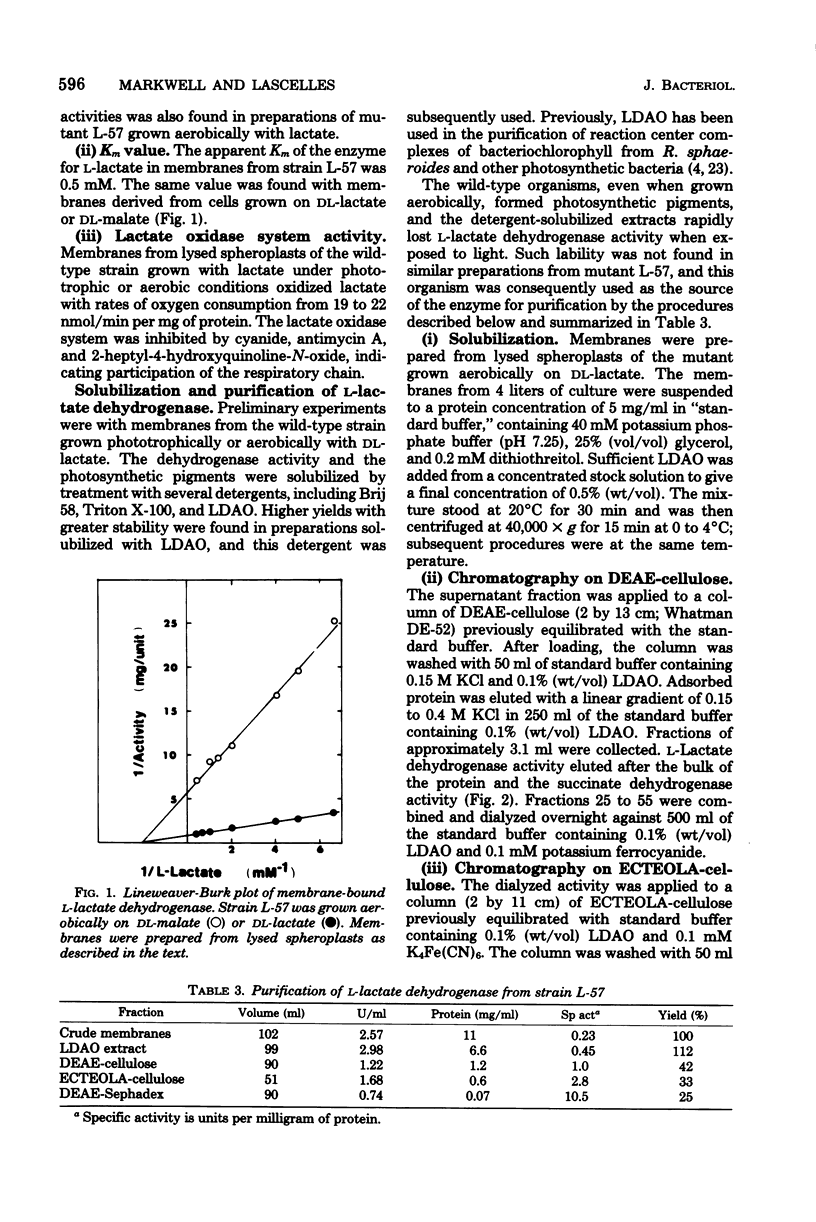
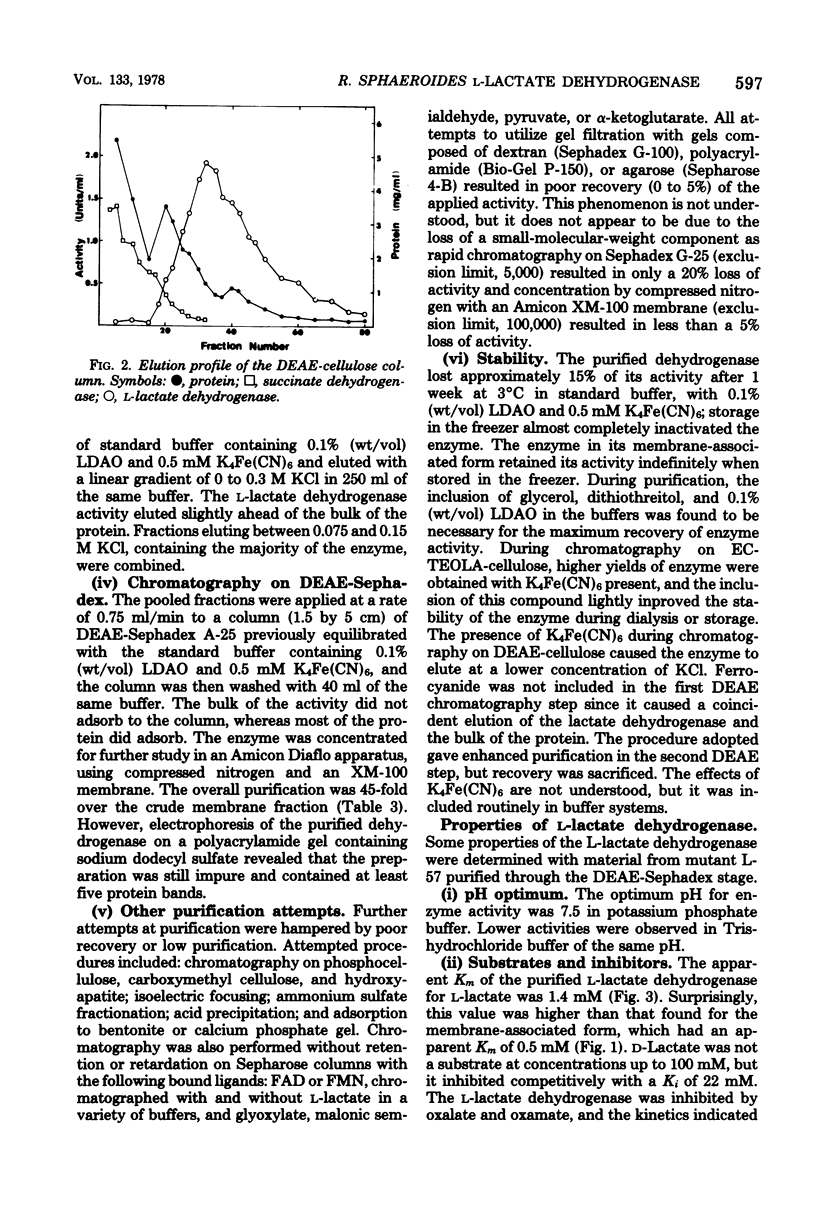
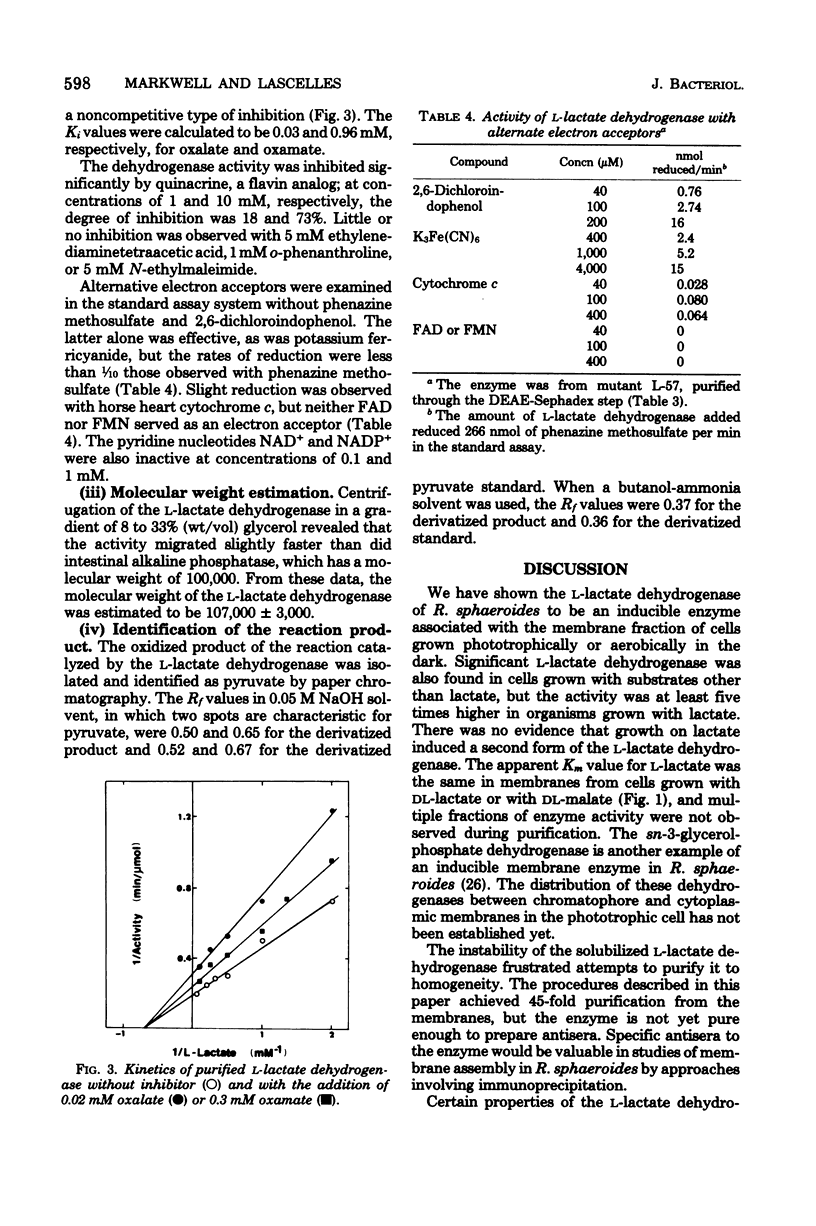
Rhodopseudomonas sphaeroides has a pyridine nucleotide-independent L-lactate dehydrogenase associated with the membrane fraction of cells grown either aerobically or phototrophically. The dehydrogenase is present in cells grown on a variety of carbon sources, but at levels less than 20% of that found in cells grown with DL-lactate. The dehydrogenase has been purified 45-fold from membranes of strain L-57, a non-photosynthetic mutant, by steps involving solubilization with lauryl dimethylamine oxide and three anion-exchange chromatography steps. The purified enzyme was specific for the L-isomer of lactate. The Km of the purified enzyme for L-lactate is 1.4 mM, whereas that of the membrane-associated enzyme is 0.5 mM. The enzyme activity was inhibited competitively by D-lactate and non-competitively by oxalate and oxamate. Quinacrine, a flavin analog, also inhibited the activity. The inducible enzyme may serve as a marker of membrane protein in studies of membrane development.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bensadoun A., Weinstein D. Assay of proteins in the presence of interfering materials. Anal Biochem. 1976 Jan;70(1):241–250. doi: 10.1016/s0003-2697(76)80064-4. [DOI] [PubMed] [Google Scholar]
- Brown A. E., Eiserling F. A., Lascelles J. Bacteriochlorophyll Synthesis and the Ultrastructure of Wild Type and Mutant Strains of Rhodopseudomonas spheroides. Plant Physiol. 1972 Dec;50(6):743–746. doi: 10.1104/pp.50.6.743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clayton R. K., Haselkorn R. Protein components of bacterial photosynthetic membranes. J Mol Biol. 1972 Jul 14;68(1):97–105. doi: 10.1016/0022-2836(72)90265-3. [DOI] [PubMed] [Google Scholar]
- Collins M. L., Niederman R. A. Membranes of Rhodospirillum rubrum: isolation and physicochemical properties of membranes from aerobically grown cells. J Bacteriol. 1976 Jun;126(3):1316–1325. doi: 10.1128/jb.126.3.1316-1325.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collins M. L., Niederman R. A. Membranes of Rhodospirillum rubrum: physicochemical properties of chromatophore fractions isolated from osmotically and mechanically disrupted cells. J Bacteriol. 1976 Jun;126(3):1326–1338. doi: 10.1128/jb.126.3.1326-1338.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ding D. H., Kaplan S. Separation of inner and outer membranes of Rhodopseudomonas spheroides. Prep Biochem. 1976;6(1):61–79. doi: 10.1080/00327487608061599. [DOI] [PubMed] [Google Scholar]
- Dulley J. R., Grieve P. A. A simple technique for eliminating interference by detergents in the Lowry method of protein determination. Anal Biochem. 1975 Mar;64(1):136–141. doi: 10.1016/0003-2697(75)90415-7. [DOI] [PubMed] [Google Scholar]
- ENGSTROM L. Studies on calf-intestinal alkaline phosphatase. I. Chromatographic purification, microheterogeneity and some other properties of the purified enzyme. Biochim Biophys Acta. 1961 Sep 2;52:36–48. doi: 10.1016/0006-3002(61)90901-5. [DOI] [PubMed] [Google Scholar]
- Futai M. Membrane D-lactate dehydrogenase from Escherichia coli. Purification and properties. Biochemistry. 1973 Jun 19;12(13):2468–2474. doi: 10.1021/bi00737a016. [DOI] [PubMed] [Google Scholar]
- Hatefi Y., Davis K. A., Baltscheffsky H., Baltscheffsky M., Johansson B. C. Isolation and properties of succinate dehydrogenase from Rhodospirillum rubrum. Arch Biochem Biophys. 1972 Oct;152(2):613–618. doi: 10.1016/0003-9861(72)90257-3. [DOI] [PubMed] [Google Scholar]
- Kohn L. D., Kaback H. R. Mechanisms of active transport in isolated bacterial membrane vesicles. XV. Purification and properties of the membrane-bound D-lactate dehydrogenase from Escherichia coli. J Biol Chem. 1973 Oct 25;248(20):7012–7017. [PubMed] [Google Scholar]
- Kung H. F., Henning U. Limiting availability of binding sites for dehydrogenases on the cell membrane of Escherichia coli. Proc Natl Acad Sci U S A. 1972 Apr;69(4):925–929. doi: 10.1073/pnas.69.4.925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lamed R., Levin Y., Wilchek M. Covalent coupling of nucleotides to agarose for affinity chromatography. Biochim Biophys Acta. 1973 Apr 28;304(2):231–235. doi: 10.1016/0304-4165(73)90239-0. [DOI] [PubMed] [Google Scholar]
- Lascelles J. The accumulation of bacteriochlorophyll precursors by mutant and wild-type strains of Rhodopseudomonas spheroides. Biochem J. 1966 Jul;100(1):175–183. doi: 10.1042/bj1000175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lascelles J., Wertlieb D. Mutant strains of Rhodopseudomonas spheroides which form photosynthetic pigments aerobically in the dark. Growth characteristics and enzymic activities. Biochim Biophys Acta. 1971 Mar 2;226(2):328–340. doi: 10.1016/0005-2728(71)90100-9. [DOI] [PubMed] [Google Scholar]
- Niederman R. A., Segen B. J., Gibson K. D. Membranes of Rhodopseudomonas spheroides. I. Isolation and characterization of membrane fractions from extracts of aerobically and anaerobically grown cells. Arch Biochem Biophys. 1972 Oct;152(2):547–560. doi: 10.1016/0003-9861(72)90250-0. [DOI] [PubMed] [Google Scholar]
- Noël H., Van der Rest M., Gingras G. Isolation and partial characterization of P870 reaction center complex from wild type Rhodospirillum rubrum. Biochim Biophys Acta. 1972 Aug 17;275(2):219–230. doi: 10.1016/0005-2728(72)90043-6. [DOI] [PubMed] [Google Scholar]
- Oelze J., Drews G. Membranes of photosynthetic bacteria. Biochim Biophys Acta. 1972 Apr 18;265(2):209–239. doi: 10.1016/0304-4157(72)90003-2. [DOI] [PubMed] [Google Scholar]
- Pike L., Sojka G. A. Glycerol dissimilation in Rhodopseudomonas sphaeroides. J Bacteriol. 1975 Dec;124(3):1101–1105. doi: 10.1128/jb.124.3.1101-1105.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takemoto J., Lascelles J. Function of membrane proteins coupled to bacteriochlorophyll synthesis. Studies with wild type and mutant strains of Rhodopseudomonas spheroides. Arch Biochem Biophys. 1974 Aug;163(2):507–514. doi: 10.1016/0003-9861(74)90508-6. [DOI] [PubMed] [Google Scholar]
- Weiner J. H., Heppel L. A. Purification of the membrane-bound and pyridine nucleotide-independent L-glycerol 3-phosphate dehydrogenase from Escherichia coli. Biochem Biophys Res Commun. 1972 Jun 28;47(6):1360–1365. doi: 10.1016/0006-291x(72)90222-7. [DOI] [PubMed] [Google Scholar]
- van Niel C. B. THE CULTURE, GENERAL PHYSIOLOGY, MORPHOLOGY, AND CLASSIFICATION OF THE NON-SULFUR PURPLE AND BROWN BACTERIA. Bacteriol Rev. 1944 Mar;8(1):1–118. doi: 10.1128/br.8.1.1-118.1944. [DOI] [PMC free article] [PubMed] [Google Scholar]


