Abstract
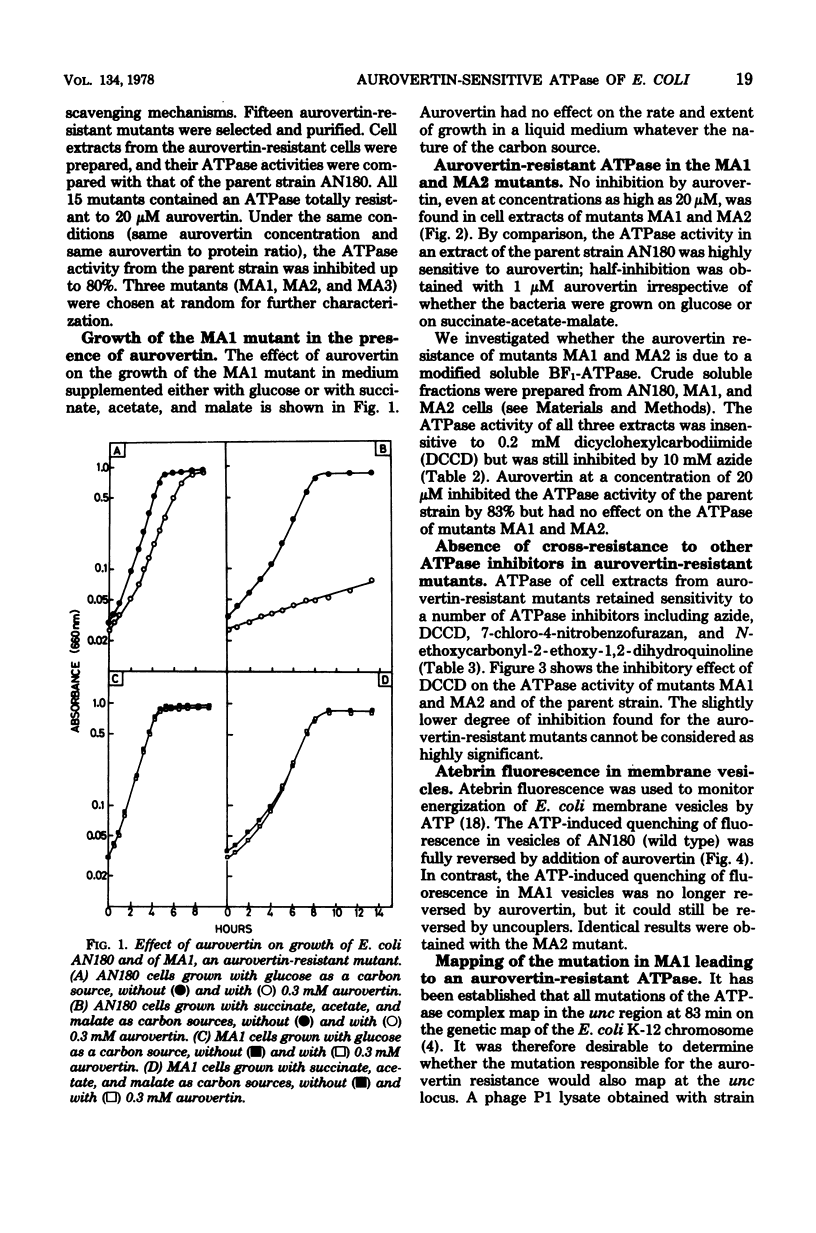
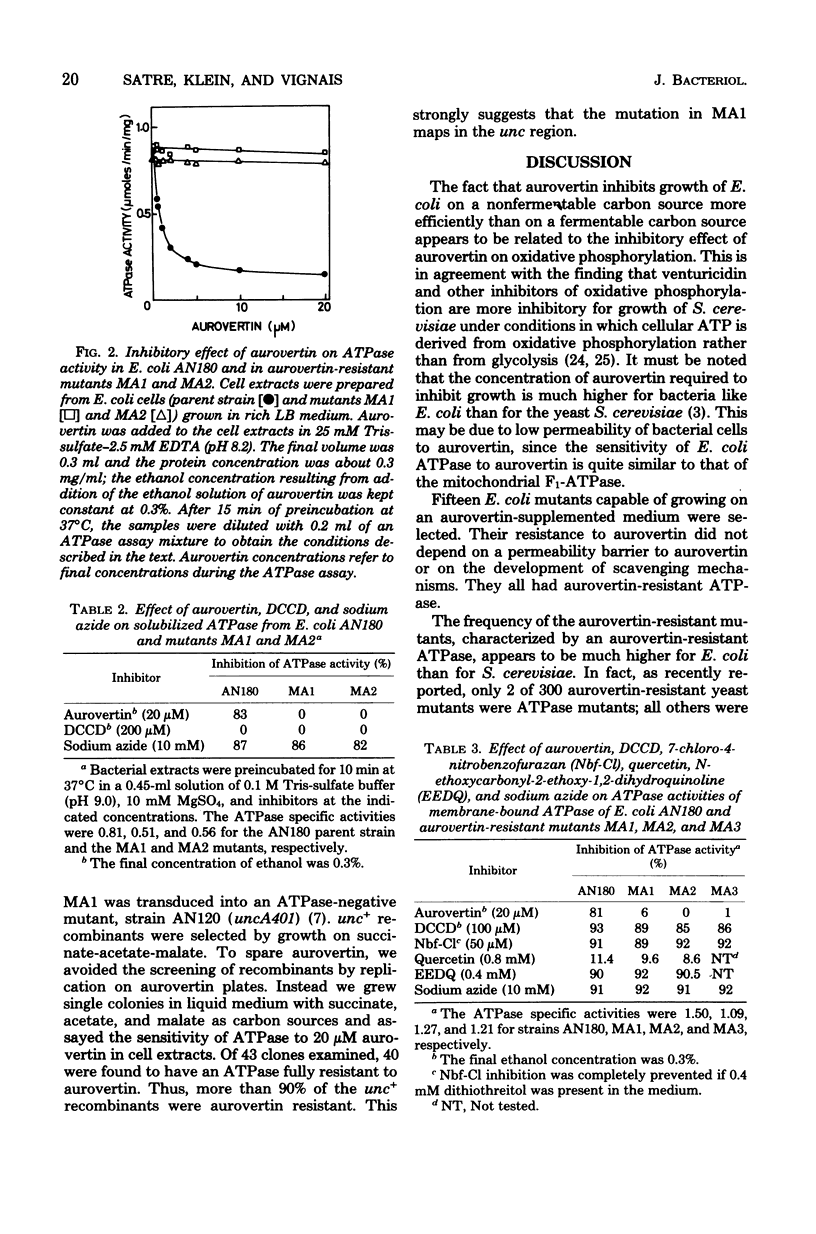
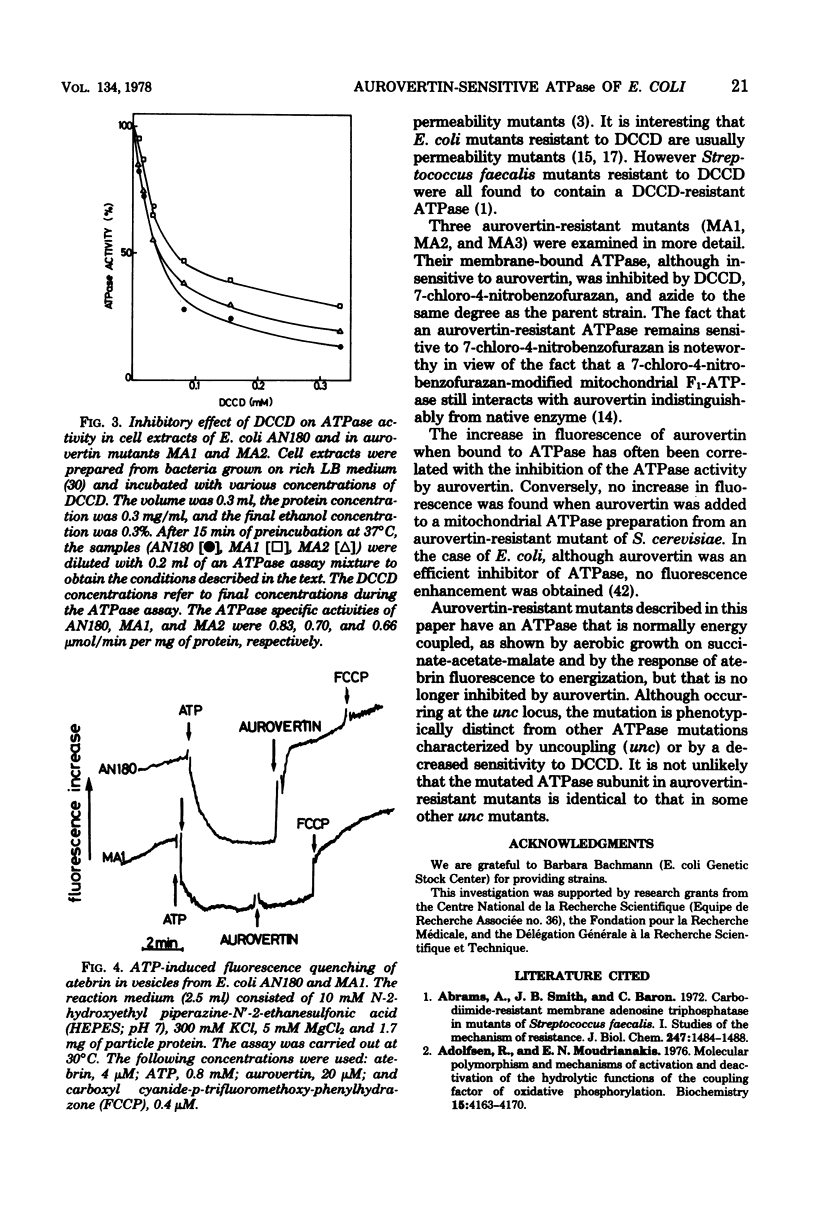
Energy-transducing adenosine triphosphatase (ATPase) from Escherichia coli is inhibited by aurovertin. Aurovertin-resistant mutants were generated by nitrosoguanidine mutagenesis of E. coli AN180, whose growth on a nonfermentable carbon source was blocked by aurovertin. The ATPase activity of cell extracts from 15 different mutants (designated MA1, MA2, MA3, etc.) was found to be at least 20 times less sensitive to aurovertin than that from the parent strain. The aurovertin-resistant mutants did not show cross-resistance towards a number of ATPase inhibitors including azide, dicyclohexylcarbodiimide, quercetin, 7-chloro-4-nitrobenzofurazan, and N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline. Aurovertin inhibited the energization brought about by addition of ATP to E. coli AN180 membrane vesicles; it was without effect on MA1 and MA2 membrane vesicles energized by ATP. The mutation in MA1, like other mutations of the ATPase complex, maps in the unc region of the bacterial chromosome.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abrams A., Smith J. B., Baron C. Carbodiimide-resistant membrane adenosine triphosphatase in mutants of Streptococcus faecalis. I. Studies of the mechanism of resistance. J Biol Chem. 1972 Mar 10;247(5):1484–1488. [PubMed] [Google Scholar]
- Adolfsen R., Moudrianakis E. N. Molecular polymorphism and mechanisms of activation and deactivation of the hydrolytic function of the coupling factor of oxidative phosphorylation. Biochemistry. 1976 Sep 21;15(19):4163–4170. doi: 10.1021/bi00664a005. [DOI] [PubMed] [Google Scholar]
- Bachmann B. J., Low K. B., Taylor A. L. Recalibrated linkage map of Escherichia coli K-12. Bacteriol Rev. 1976 Mar;40(1):116–167. doi: 10.1128/br.40.1.116-167.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bertina R. M., Schrier P. I., Slater E. C. The binding of aurovertin to mitochondria, and its effect on mitochondrial respiration. Biochim Biophys Acta. 1973 Jun 28;305(3):503–518. doi: 10.1016/0005-2728(73)90072-8. [DOI] [PubMed] [Google Scholar]
- Butlin J. D., Cox G. B., Gibson F. Oxidative phosphorylation in Escherichia coli K-12: the genetic and biochemical characterisations of a strain carrying a mutation in the uncB gene. Biochim Biophys Acta. 1973 Feb 22;292(2):366–375. doi: 10.1016/0005-2728(73)90043-1. [DOI] [PubMed] [Google Scholar]
- Butlin J. D., Cox G. B., Gibson F. Oxidative phosphorylation in Escherichia coli K12. Mutations affecting magnesium ion- or calcium ion-stimulated adenosine triphosphatase. Biochem J. 1971 Aug;124(1):75–81. doi: 10.1042/bj1240075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang T. M., Penefsky H. S. Energy-dependent enhancement of aurovertin fluorescence. An indicator of conformational changes in beef heart mitochondrial adenosine triphosphatase. J Biol Chem. 1974 Feb 25;249(4):1090–1098. [PubMed] [Google Scholar]
- Chang T., Penefsky H. S. Aurovertin, a fluorescent probe of conformational change in beef heart mitochondrial adenosine triphosphatase. J Biol Chem. 1973 Apr 25;248(8):2746–2754. [PubMed] [Google Scholar]
- Cox G. B., Gibson F. Studies on electron transport and energy-linked reactions using mutants of Escherichia coli. Biochim Biophys Acta. 1974 Apr 30;346(1):1–25. doi: 10.1016/0304-4173(74)90010-x. [DOI] [PubMed] [Google Scholar]
- Daniel J., Roisin M. P., Burstein C., Kepes A. Mutants of Escherichia coli K12 unable to grow on non-fermentable carbon substrates. Biochim Biophys Acta. 1975 Feb 17;376(2):195–209. doi: 10.1016/0005-2728(75)90011-0. [DOI] [PubMed] [Google Scholar]
- Fillingame R. H. Identification of the dicyclohexylcarbodiimide-reactive protein component of the adenosine 5'-triphosphate energy-transducing system of Escherichia coli. J Bacteriol. 1975 Nov;124(2):870–883. doi: 10.1128/jb.124.2.870-883.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friedl P., Schmid B. I., Schairer H. U. A mutant ATP synthetase of Escherichia coli with an altered sensitivity to N,N' -dicyclohexylcarbodiimide: characterization in native membranes and reconstituted proteoliposomes. Eur J Biochem. 1977 Mar 1;73(2):461–468. doi: 10.1111/j.1432-1033.1977.tb11338.x. [DOI] [PubMed] [Google Scholar]
- Gibson F., Cox G. B., Downie J. A., Radik J. Partial diploids of Escherichia coli carrying normal and mutant alleles affecting oxidative phosphorylation. Biochem J. 1977 Mar 15;162(3):665–670. doi: 10.1042/bj1620665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harris D. A., John P., Radda G. K. Tightly bound nucleotides of the energy-transducing ATPase, and their role in oxidative phosphorylation. I. The Paracoccus denitrificans system. Biochim Biophys Acta. 1977 Mar 11;459(3):546–559. doi: 10.1016/0005-2728(77)90053-6. [DOI] [PubMed] [Google Scholar]
- Hawrot E., Kennedy E. P. Biogenesis of membrane lipids: mutants of Escherichia coli with temperature-sensitive phosphatidylserine decarboxylase. Proc Natl Acad Sci U S A. 1975 Mar;72(3):1112–1116. doi: 10.1073/pnas.72.3.1112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LARDY H. A., CONNELLY J. L., JOHNSON D. ANTIBIOTIC STUDIES. II. INHIBITION OF PHOSPHORYL TRANSFER IN MITOCHONDRIA BY OLIGOMYCIN AND AUROVERTIN. Biochemistry. 1964 Dec;3:1961–1968. doi: 10.1021/bi00900a030. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lenaz G. Effect of aurovertin on energy-linked processes related to oxidative phosphorylation. Biochem Biophys Res Commun. 1965 Oct 26;21(2):170–175. doi: 10.1016/0006-291x(65)90104-x. [DOI] [PubMed] [Google Scholar]
- Moyle J., Mitchell P. Active/inactive state transitions of mitochondrial ATPase molecules influenced by Mg2+, anions and aurovertin. FEBS Lett. 1975 Aug 1;56(1):55–61. doi: 10.1016/0014-5793(75)80110-4. [DOI] [PubMed] [Google Scholar]
- Ravizzini R. A., Lescano W. I., Vallejos R. H. Effect of aurovertin on energy transfer reactions in Rhodospirillum rubrum chromatophores. FEBS Lett. 1975 Oct 15;58(1):285–288. doi: 10.1016/0014-5793(75)80280-8. [DOI] [PubMed] [Google Scholar]
- Roberton A. M., Beechey R. B., Holloway C. T., Knight I. G. The effect of aurovertin on a soluble mitochondrial adenosine triphosphatase. Biochem J. 1967 Sep;104(3):54C–55C. doi: 10.1042/bj1040054c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roisin M. P., Kepes A. The membrane ATPase of Escherichia coli. I. Ion dependence and ATP-ADP exchange reaction. Biochim Biophys Acta. 1972 Sep 20;275(3):333–346. doi: 10.1016/0005-2728(72)90214-9. [DOI] [PubMed] [Google Scholar]
- Roisin M. P., Kepes A. The membrane ATPase of Escherichia coli. I. Release into solution, allotopic properties and reconstitution of membrane-bound ATPase. Biochim Biophys Acta. 1973 May 30;305(2):249–259. doi: 10.1016/0005-2728(73)90173-4. [DOI] [PubMed] [Google Scholar]
- Schairer H. U., Friedl P., Schmid B. I., Vogel G. The use of several energy-coupling reactions in characterizing mutants of Escherichia coli K12 defective in oxidative phosphorylation. Eur J Biochem. 1976 Jul 1;66(2):257–268. doi: 10.1111/j.1432-1033.1976.tb10515.x. [DOI] [PubMed] [Google Scholar]
- Senior A. E. The structure of mitochondrial ATPase. Biochim Biophys Acta. 1973 Dec 31;301(3):249–277. doi: 10.1016/0304-4173(73)90006-2. [DOI] [PubMed] [Google Scholar]
- Simoni R. D., Postma P. W. The energetics of bacterial active transport. Annu Rev Biochem. 1975;44:523–554. doi: 10.1146/annurev.bi.44.070175.002515. [DOI] [PubMed] [Google Scholar]
- Simoni R. D., Shandell A. Energy transduction in Escherichia coli. Genetic alteration of a membrane polypeptide of the (Ca2+,Mg2+)-ATPase. J Biol Chem. 1975 Dec 25;250(24):9421–9427. [PubMed] [Google Scholar]
- van Thienen G., Postma P. W. Coupling between energy conservation and active transport of serine in Escherichia coli. Biochim Biophys Acta. 1973 Oct 25;323(3):429–440. doi: 10.1016/0005-2736(73)90188-0. [DOI] [PubMed] [Google Scholar]
- van de Stadt R. J., van Dam K. Binding of aurovertin to phosphorylating submitochondrial particles. Biochim Biophys Acta. 1974 May 22;347(2):253–263. doi: 10.1016/0005-2728(74)90049-8. [DOI] [PubMed] [Google Scholar]
- van de Stadt R. J., van Dam K., Slater E. C. Interaction of aurovertin with submitochondrial particles, deficient in ATPase inhibitor. Biochim Biophys Acta. 1974 May 22;347(2):224–239. doi: 10.1016/0005-2728(74)90047-4. [DOI] [PubMed] [Google Scholar]



