Abstract
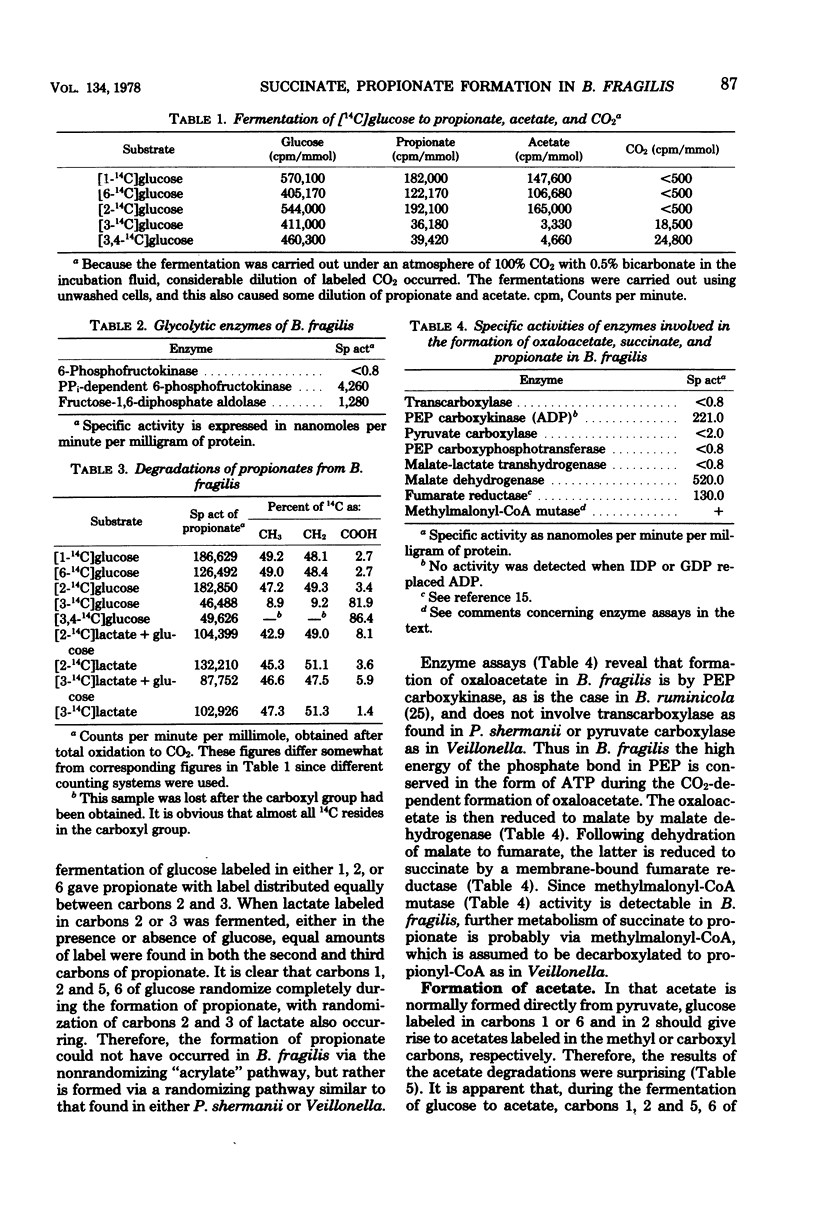
Cell suspensions of Bacteroides fragilis were allowed to ferment glucose and lactate labeled with 14C in different positions. The fermentation products, propionate and acetate, were isolated, and the distribution of radioactivity was determined. An analysis of key enzymes of possible pathways was also made. The results of the labeling experiments showed that: (i) B. fragilis ferments glucose via the Embden-Meyerhof pathway; and (ii) there was a randomization of carbons 1, 2, and 6 of glucose during conversion to propionate, which is in accordance with propionate formation via fumarate and succinate. The enzymes 6-phosphofrucktokinase (pyrophosphate-dependent), fructose-1,6-diphosphate aldolase, phosphoenolpyruvate carboxykinase, malate dehydrogenase, fumarate reductase, and methylmalonyl-coenzyme A mutase could be demonstrated in cell extracts. Their presence supported the labeling results and suggested that propionate is formed from succinate via succinyl-, methylmalonyl-, and propionyl-coenzyme A. From the results it also is clear that CO2 is necessary for growth because it is needed for the formation of C4 acids. There was also a randomization of carbons 1, 2, and 6 of glucose during conversion to acetate, which indicated that pyruvate kinase played a minor role in pyruvate formation from phosphoenolpyruvate. Phosphoenolpyruvate carboxykinase, oxaloacetate decarboxylase, and malic enzyme (nicotinamide adenine dinucleotide phosphate-dependent) were present in cell extracts of B. fragilis, and the results of the labeling experiments agreed with pyruvate synthesis via oxaloacetate and malate if these acids are in equilibrium with fumarate. The conversion of [2-14C]- and [3-14C]lactate to acetate was not associated with a randomization of radioactivity.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Allen S. H. The isolation and characterization of malate-lactate transhydrogenase from Micrococcus lactilyticus. J Biol Chem. 1966 Nov 25;241(22):5266–5275. [PubMed] [Google Scholar]
- Babul J., Robinson J. P., Fraenkel D. G. Are the aerobic and anaerobic phosphofructokinases of Escherichia coli different? Eur J Biochem. 1977 Apr 15;74(3):533–537. doi: 10.1111/j.1432-1033.1977.tb11421.x. [DOI] [PubMed] [Google Scholar]
- Baumann L., Baumann P. Enzymes of glucose catabolism in cell-free extracts of non-fermentative marine eubacteria. Can J Microbiol. 1973 Feb;19(2):302–304. doi: 10.1139/m73-048. [DOI] [PubMed] [Google Scholar]
- Brockman H. L., Wood W. A. D-Lactate dehydrogenase of Peptostreptococcus elsdenii. J Bacteriol. 1975 Dec;124(3):1454–1461. doi: 10.1128/jb.124.3.1454-1461.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brockman H. L., Wood W. A. Electron-transferring flavoprotein of Peptostreptococcus elsdenii that functions in the reduction of acrylyl-coenzyme A. J Bacteriol. 1975 Dec;124(3):1447–1453. doi: 10.1128/jb.124.3.1447-1453.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dehority B. A. Characterization of several bovine rumen bacteria isolated with a xylan medium. J Bacteriol. 1966 May;91(5):1724–1729. doi: 10.1128/jb.91.5.1724-1729.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galivan J. H., Allen S. H. Methylmalonyl coenzyme A decarboxylase. Its role in succinate decarboxylation by Micrococcus lactilyticus. J Biol Chem. 1968 Mar 25;243(6):1253–1261. [PubMed] [Google Scholar]
- Kornberg H. L., Malcovati M. Control in situ of the pyruvate kinase activity of Escherichia coli. FEBS Lett. 1973 Jun 1;32(2):257–259. doi: 10.1016/0014-5793(73)80846-4. [DOI] [PubMed] [Google Scholar]
- LEAVER F. W., WOOD H. G., STJERNHOLM R. The fermentation of three carbon substrates by Clostridium propionicum and Propionibacterium. J Bacteriol. 1955 Nov;70(5):521–530. doi: 10.1128/jb.70.5.521-530.1955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Macy J., Probst I., Gottschalk G. Evidence for cytochrome involvement in fumarate reduction and adenosine 5'-triphosphate synthesis by Bacteroides fragilis grown in the presence of hemin. J Bacteriol. 1975 Aug;123(2):436–442. doi: 10.1128/jb.123.2.436-442.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ng S. K., Hamilton I. R. Carbon dioxide fixation by Veillonella parvula M 4 and its relation to propionic acid formation. Can J Microbiol. 1973 Jun;19(6):715–723. doi: 10.1139/m73-116. [DOI] [PubMed] [Google Scholar]
- O'Brien W. E., Bowien S., Wood H. G. Isolation and characterization of a pyrophosphate-dependent phosphofructokinase from Propionibacterium shermanii. J Biol Chem. 1975 Nov 25;250(22):8690–8695. [PubMed] [Google Scholar]
- PHARES E. F. Degradation of labeled propionic and acetic acids. Arch Biochem Biophys. 1951 Sep;33(2):173–178. doi: 10.1016/0003-9861(51)90094-x. [DOI] [PubMed] [Google Scholar]
- Reeves R. E., South D. J., Blytt H. J., Warren L. G. Pyrophosphate:D-fructose 6-phosphate 1-phosphotransferase. A new enzyme with the glycolytic function of 6-phosphofructokinase. J Biol Chem. 1974 Dec 25;249(24):7737–7741. [PubMed] [Google Scholar]
- SWIM H. E., KRAMPITZ L. O. Acetic acid oxidation by Escherichia coli; evidence for the occurrence of a tricarboxylic acid cycle. J Bacteriol. 1954 Apr;67(4):419–425. doi: 10.1128/jb.67.4.419-425.1954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanwal B. D. Regulatory characteristics of the diphosphopyridine nucleotide-specific malic enzyme of Escherichia coli. J Biol Chem. 1970 Mar 10;245(5):1212–1216. [PubMed] [Google Scholar]
- Sanwal B. D., Smando R. Malic enzyme of Escherichia coli. Diversity of the effectors controlling enzyme activity. J Biol Chem. 1969 Apr 10;244(7):1817–1823. [PubMed] [Google Scholar]
- Sawyer M. H., Baumann P., Baumann L. Pathways of D-fructose and D-glucose catabolism in marine species of Alcaligenes, Pseudomonas marina, and Alteromonas communis. Arch Microbiol. 1977 Mar 1;112(2):169–172. doi: 10.1007/BF00429331. [DOI] [PubMed] [Google Scholar]
- South D. J., Reeves R. E. Pyruvate, orthophosphate dikinase from Bacteroides symbiosus. Methods Enzymol. 1975;42:187–181. doi: 10.1016/0076-6879(75)42114-0. [DOI] [PubMed] [Google Scholar]
- Tung K. K., Wood W. A. Purification, new assay, and properties of coenzyme A transferase from Peptostreptococcus elsdenii. J Bacteriol. 1975 Dec;124(3):1462–1474. doi: 10.1128/jb.124.3.1462-1474.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WOOD H. G., KULKA R. G., EDSON N. L. The metabolism of [1-14C] glucose in an enzyme system from Propionibacterium. Biochem J. 1956 Jun;63(2):177–182. doi: 10.1042/bj0630177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WOOD H. G., STJERNHOLM R., LEAVER F. W. The metabolism of labeled glucose by the propionic acid bacteria. J Bacteriol. 1955 Nov;70(5):510–520. doi: 10.1128/jb.70.5.510-520.1955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wallnöfer P., Baldwin R. L. Pathway of propionate formation in Bacteroides ruminicola. J Bacteriol. 1967 Jan;93(1):504–505. doi: 10.1128/jb.93.1.504-505.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whiteley H. R. The Mechanism of Propionic Acid Formation by Succinate Decarboxylation: I. The Activation of Succinate. Proc Natl Acad Sci U S A. 1953 Aug;39(8):772–779. doi: 10.1073/pnas.39.8.772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wittenberger C. L., Fulco J. G. Purification and allosteric properties of a nicotinamide adenine dinucleotide-linked D(-)-specific lactate dehydrogenase from Butyribacterium rettgeri. J Biol Chem. 1967 Jun 25;242(12):2917–2924. [PubMed] [Google Scholar]
- Wood H. G., O'brien W. E., Micheales G. Properties of carboxytransphosphorylase; pyruvate, phosphate dikinase; pyrophosphate-phosphofructikinase and pyrophosphate-acetate kinase and their roles in the metabolism of inorganic pyrophosphate. Adv Enzymol Relat Areas Mol Biol. 1977;45:85–155. doi: 10.1002/9780470122907.ch2. [DOI] [PubMed] [Google Scholar]
- von Hugo H., Schoberth S., Madan V. K., Gottschalk G. Coenzyme specificity of dehydrogenases and fermentation of pyruvate by clostridia. Arch Mikrobiol. 1972;87(3):189–202. doi: 10.1007/BF00424880. [DOI] [PubMed] [Google Scholar]


