Abstract
Among 380 Mycoplasma pneumoniae isolates from 3,678 pediatric patients with community-acquired pneumonia, 50 macrolide-resistant strains had an A2063G transition in domain V of the 23S rRNA, whereas 5 had an A2064G transition. These resistant strains increased rapidly from April 2002 to December 2006.
For Mycoplasma pneumoniae, a major etiologic agent of lower respiratory tract infections acquired in the community, 14-membered ring macrolides (ML) generally are recognized as first-choice agents. In Japan, ML-resistant (MLr) M. pneumoniae possessing a 23S rRNA mutation first was isolated from pediatric patients with community-acquired pneumonia (CAP) and bronchitis as reported in 2001 by Okazaki et al. (4). Patient symptoms appeared to be prolonged when isolates showed ML resistance (5).
We subjected M. pneumoniae isolated from pediatric patients with CAP between 2002 and 2006 to susceptibility evaluation for eight agents, including ML. In strains showing ML resistance, the 23S rRNA gene was analyzed.
Between April 2002 and December 2006, 3,678 clinical samples were sent to our laboratory from pediatricians affiliated with 10 institutions participating in the Acute Respiratory Diseases Study Group. All samples originating from pediatric patients diagnosed with pneumonia according to clinical symptoms and chest X-ray images were collected after informed consent was given by the patients and/or their parents or guardians.
Immediately after receipt, the samples were suspended in 1.5 ml of pleuropneumonia-like organism (PPLO) broth (Difco, Detroit, MI). DNA then was extracted by using Extragen II (Tosoh, Tokyo, Japan) according to the manufacturer's protocol. Real-time PCR to detect M. pneumoniae was performed as described previously (2) using the extracted DNA. Culture of M. pneumoniae was carried out for PCR-positive samples using PPLO broth according to previously described methods (6).
The MICs of eight agents for M. pneumoniae isolates were determined with microdilution methods using PPLO broth. These agents were erythromycin, clarithromycin, azithromycin, josamycin, rokitamycin, telithromycin, minocycline, and levofloxacin. M. pneumoniae M129 strain was used as a control.
The full length of the 23S rRNA gene was sequenced by methods described previously (3) in 55 M. pneumoniae strains showing ML resistance.
For patients with adequate clinical information, clinical courses of CAP caused by MLr M. pneumoniae (n = 53) were compared to those of CAP with ML-susceptible (MLs) M. pneumoniae (n = 58). Variables compared included (i) the number of days from initiation of ML treatment until defervescence to 37°C and (ii) whether or not initial treatment with ML was changed later to another agent. Body temperature that exceeded 38°C at least once daily was defined as ongoing fever.
Table 1 shows the numbers of real-time PCR-positive samples for M. pneumoniae among samples tested from April 2002 to December 2006 (approximately 5 years). A total of 3,678 nasopharyngeal samples were collected from pediatric patients with CAP. Culture for M. pneumoniae using PPLO broth was performed in the 521 samples PCR positive for M. pneumoniae; 380 strains were isolated. In 2003 and 2006, M. pneumoniae infection was particularly prevalent in Japan, reflected by the occurrence of more PCR-positive cases and M. pneumoniae isolates than in other years. The percentages of culture positivity for M. pneumoniae in PCR-positive samples ranged from 66.7 to 85.7% during the 5-year period.
TABLE 1.
PCR and culture results for M. pneumoniae from 2002 to 2006
| PCR and culture | No. of strains (%)a in:
|
Total samples (n = 3,678) | ||||
|---|---|---|---|---|---|---|
| 2002 (n = 522) | 2003 (n = 699) | 2004 (n = 533) | 2005 (n = 749) | 2006 (n = 1,175) | ||
| PCR positive | 70 (13.4) | 140 (20.0) | 60 (11.3) | 75 (10.0) | 176 (15.0) | 521 (14.2) |
| Culture positive | 47 (67.1) | 120 (85.7) | 40 (66.7) | 52 (69.3) | 121 (68.8) | 380 (72.9) |
In the first row, the percentage of all samples tested is given in parentheses; in the second row, the percentage of PCR-positive samples is given in parentheses. n, Number of samples tested.
Table 2 shows the MIC range, MIC50, and MIC90 for eight agents according to the presence or absence of a mutation of the 23S rRNA gene in the 380 M. pneumoniae isolates; 50 strains had an A2063G transition in domain V, and 5 strains had an A2064G transition. MLr strains showed high resistance to erythromycin, clarithromycin, azithromycin, telithromycin, and josamycin. Among 16-membered ring ML, only rokitamycin had a effective MIC90 (0.25 μg/ml) for strains with the A2063G transition. However, strains with the A2064G transition showed intermediate resistance to rokitamycin. The MIC90s of minocycline and levofloxacin for MLr strains were equal to the MIC90s for susceptible strains.
TABLE 2.
In vitro antimicrobial activity of eight oral agents against M. pneumoniae strains (n = 380)
| Antimicrobial agent and MLr class | MIC (μg/ml)a
|
||
|---|---|---|---|
| 50% | 90% | Range | |
| Erythromycin | |||
| Susceptible | 0.0078 | 0.0156 | 0.00195-0.0313 |
| Resistant | |||
| A2063G | 64 | >64 | 32->64 |
| A2064G | >64 | >64 | 64->64 |
| Clarithromycin | |||
| Susceptible | 0.0039 | 0.0078 | 0.00049-0.0313 |
| Resistant | |||
| A2063G | 64 | >64 | 32->64 |
| A2064G | 64 | 64 | 16->64 |
| Azithromycin | |||
| Susceptible | 0.00049 | 0.00098 | 0.00024-0.00195 |
| Resistant | |||
| A2063G | 32 | 64 | 16->64 |
| A2064G | 32 | 64 | 16-64 |
| Telithromycin | |||
| Susceptible | 0.00098 | 0.00195 | 0.00024-0.0039 |
| Resistant | |||
| A2063G | 32 | 64 | 16->64 |
| A2064G | 4 | 16 | 1-16 |
| Josamycin | |||
| Susceptible | 0.0313 | 0.0625 | 0.0156-0.0625 |
| Resistant | |||
| A2063G | 8 | 16 | 0.0625-64 |
| A2064G | 64 | >64 | 64->64 |
| Rokitamycin | |||
| Susceptible | 0.0156 | 0.0156 | 0.0039-0.0313 |
| Resistant | |||
| A2063G | 0.125 | 0.25 | 0.0156-16 |
| A2064G | 16 | 16 | 8-16 |
| Minocycline | |||
| Susceptible | 0.5 | 1 | 0.0313-2 |
| Resistant | |||
| A2063G | 0.5 | 1 | 0.0625-1 |
| A2064G | 0.5 | 1 | 0.0313-1 |
| Levofloxacin | |||
| Susceptible | 0.5 | 1 | 0.125-1 |
| Resistant | |||
| A2063G | 1 | 1 | 0.5-1 |
| A2064G | 1 | 1 | 0.5-1 |
MICs were determined by microdilution methods using PPLO broth. Samples (10 μl) from these cultures estimated to contain 105 CFU/ml were inoculated into 96-well microplates filled with 90 μl of PPLO broth containing serially diluted antibiotics. These were incubated aerobically from 10 to 14 days at 37°C until a color change was confirmed in an antibiotic-free growth control. The MIC of each agent was defined as the lowest concentration of each antibiotic preventing the color change.
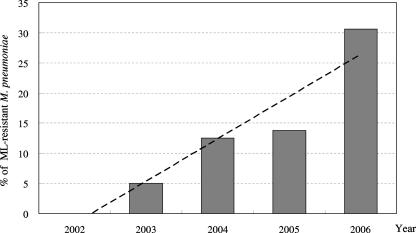
Figure 1 shows year-by-year changes in MLr M. pneumoniae from April 2002 to December 2006. Resistant strains increased rapidly each year: 0% (0/47) in 2002, 5.0% (6/120) in 2003, 12.5% (5/40) in 2004, 13.5% (7/52) in 2005, and 30.6% (37/121) in 2006. In parallel with the overall increased prevalence of M. pneumoniae infections in 2006, the prevalence of MLr strains isolated from widespread regions of Japan clearly also increased.
FIG. 1.
Year-by-year increases in the frequency of MLr M. pneumoniae cases. The dotted line can be expressed by the equation, y = 7x − 8.62 (r = 0.9510).
Treatment was changed from ML to minocycline or levoloxacin in 6.9% (4/58) of patients infected with MLs M. pneumoniae and in 35.8% (19/53) of patients infected with MLr M. pneumoniae, representing a significant difference between groups (P = 0.0023). Only one patient had a change in treatment to levofloxacin, which was done to address coinfection with β-lactamase-nonproducing and ampicillin-resistant Haemophilus influenzae. The mean times from the initiation of macrolide use to defervescence were 1.6 ± 0.8 days for MLs M. pneumoniae and 4.1 ± 2.3 days for MLr M. pneumoniae, a significant difference (P = 0.0020).
In our laboratory, MLr M. pneumoniae first was isolated from a patient with acute bronchitis in 2002 (3) and then began to be isolated from CAP cases in 2003. In parallel with the prevalence of M. pneumoniae infection, the prevalence of MLr isolates have increased rapidly, attaining a 30.6% prevalence in 2006.
A question might be posed as to whether MLr M. pneumoniae represents a serious clinical therapeutic issue, since M. pneumoniae infection typically produces mild symptoms that spontaneously diminish, with ultimate recovery. However, in CAP patients infected with MLr strains, ML frequently was changed to minocycline or levofloxacin because of persistent fever and cough or nonresolution or worsening of chest X-ray abnormalities. Levofloxacin and minocycline are not ordinarily recommended for children. Only in cases where other antibiotics cannot be used or are ineffective against organisms is the use of these antibiotics approved by the Japanese Ministry of Health, Labor, and Welfare. If ML are ineffective against M. pneumoniae infection, pediatricians have little choice but to use minocycline. In the future, improvement or augmentation of treatment for patients with MLr M. pneumoniae infections should be considered promptly, including symptomatic measures such as steroid therapy.
In addition to affecting children, M. pneumoniae is a common pathogen among young adults with CAP (1, 6). No MLr M. pneumoniae was observed among 30 isolates from adult patients with CAP. Although the absence of resistant isolates is unexplained, the wide use of new quinolone agents for adult patients with acute respiratory tract infections may have contributed to this finding. Nonetheless, oral ML treatment still is a common choice for adult patients, amounting to 25% or more compared to ca. 24% for quinolones. In this population, MLr M. pneumoniae strains may also ultimately emerge and become more prevalent.
Based upon our results, we recommend increased worldwide surveillance for MLr M. pneumoniae and stress the need to establish the most appropriate chemotherapy against those infections.
Acknowledgments
We thank the members of the Acute Respiratory Diseases Study Group: Shinobu Ishizawa, Mika Numata, and Michiyo Kojima (Tohoku Rosai Hospital), Takeshi Tajima (Hakujikai Memorial Hospital), Haruo Kuroki (Sotobo Kodomo Clinic), Kouichi Shimizu (Saiseikai Ibaraki Hospital), Naohisa Kawamura (Osaka Rosai Hospital), Ritsuko Sakai (Sakai Clinic), Masaaki Kobayashi (Kobayashi Pediatric Clinic), Kota Saito (Saito Pediatric Clinic), Masahiko Nitta (Yaotokushukai Hospital and Osaka Medical College), Shigeru Ohnari (Nakafukawa Pediatric Clinic), Takao Morinobu (Seikeikai Hospital), Masato Nonoyama (Ebina General Hospital), and Tomohiro Oishi (Niigata Kenritsu Shibata Hospital).
This study was supported by a grant from Kawano Masanori Memorial Foundation for the Promotion of Pediatrics.
Footnotes
Published ahead of print on 22 October 2007.
REFERENCES
- 1.Hammerschlag, M. R. 2001. Mycoplasma pneumoniae infections. Curr. Opin. Infect. Dis. 14:181-186. [DOI] [PubMed] [Google Scholar]
- 2.Morozumi, M., E. Nakayama, S. Iwata, Y. Aoki, K. Hasegawa, R. Kobayashi, N. Chiba, T. Tajima, and K. Ubukata. 2006. Simultaneous detection of pathogens in community-acquired pneumonia by real-time PCR with pathogen-specific molecular beacon probes. J. Clin. Microbiol. 44:1440-1446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Morozumi, M., K. Hasegawa, R. Kobayashi, N. Inoue, S. Iwata, H. Kuroki, N. Kawamura, E. Nakayama, T. Tajima, K. Shimizu, and K. Ubukata. 2005. Emergence of macrolides-resistant Mycoplasma pneumoniae with a 23S rRNA gene mutation. Antimicrob. Agents Chemother. 49:2302-2306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Okazaki, N., M. Narita, S. Yamada, K. Izumikawa, M. Umetsu, T. Kenri, Y. Sasaki, Y. Arakawa, and T. Sasaki. 2001. Characteristics of macrolide-resistant Mycoplasma pneumoniae strains isolated from patients and induced with erythromycin in vitro. Microbiol. Immunol. 45:617-620. [DOI] [PubMed] [Google Scholar]
- 5.Suzuki, S., T. Yamazaki, M. Narita, N. Okazaki, I. Suzuki, T. Andoh, M. Matsuoka, T. Kenri, Y. Arakawa, and T. Sasaki. 2006. Clinical evaluation of macrolide-resistant Mycoplasma pneumoniae. Antimicrob. Agents Chemother. 50:709-712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Waites, K. B., Y. Rikihisa, and D. Taylor-Robinson. 2003. Mycoplasma and ureaplasma, p. 972-990. In P. R. Murray, E. J. Baron, J. H. Jorgensen, M. A. Pfaller, and R. H. Yolken (ed.), Manual of clinical microbiology, 8th ed. ASM Press, Washington, DC.