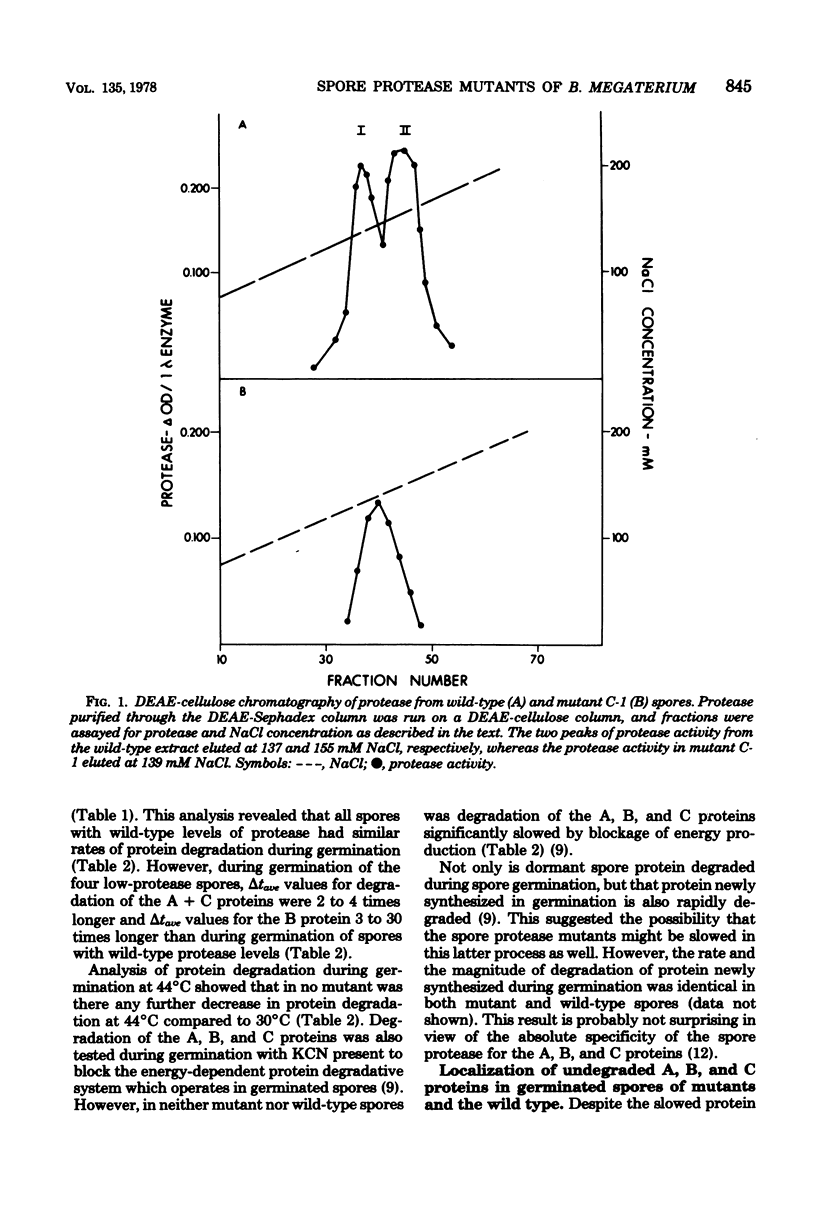
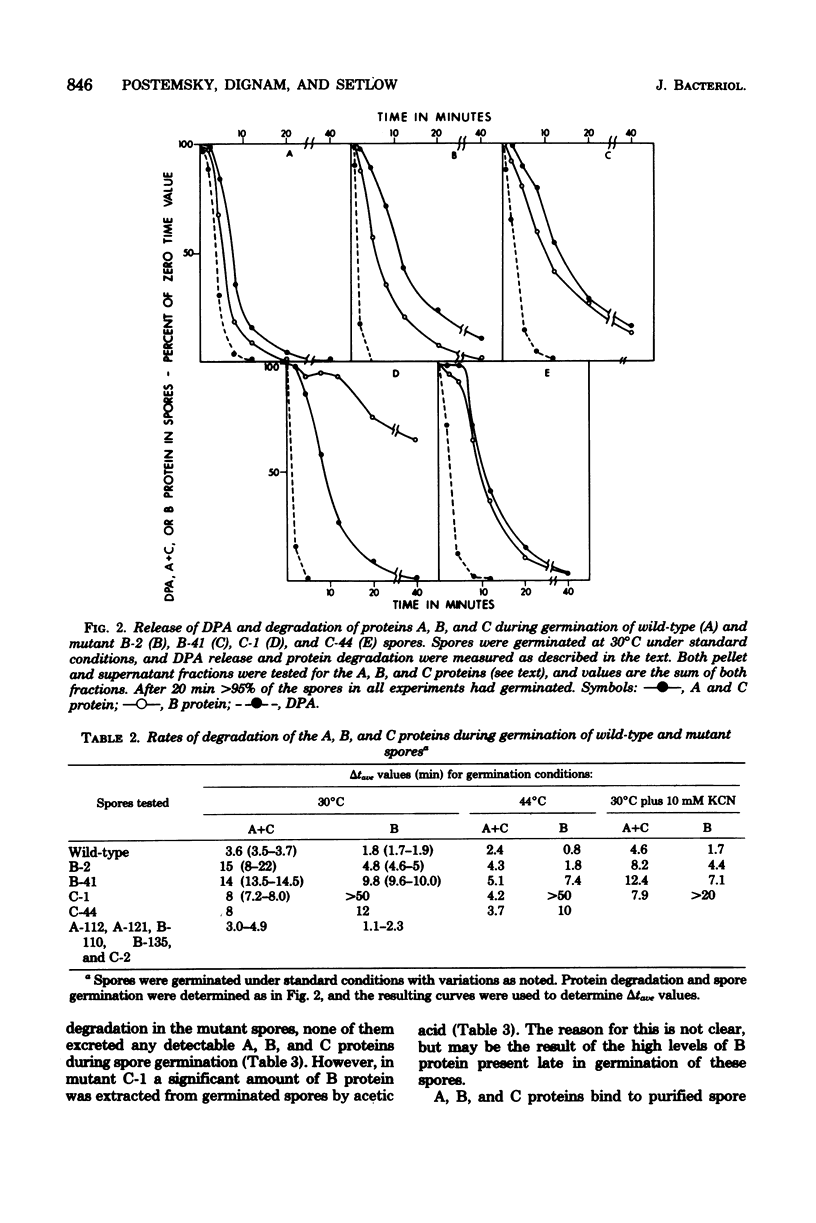
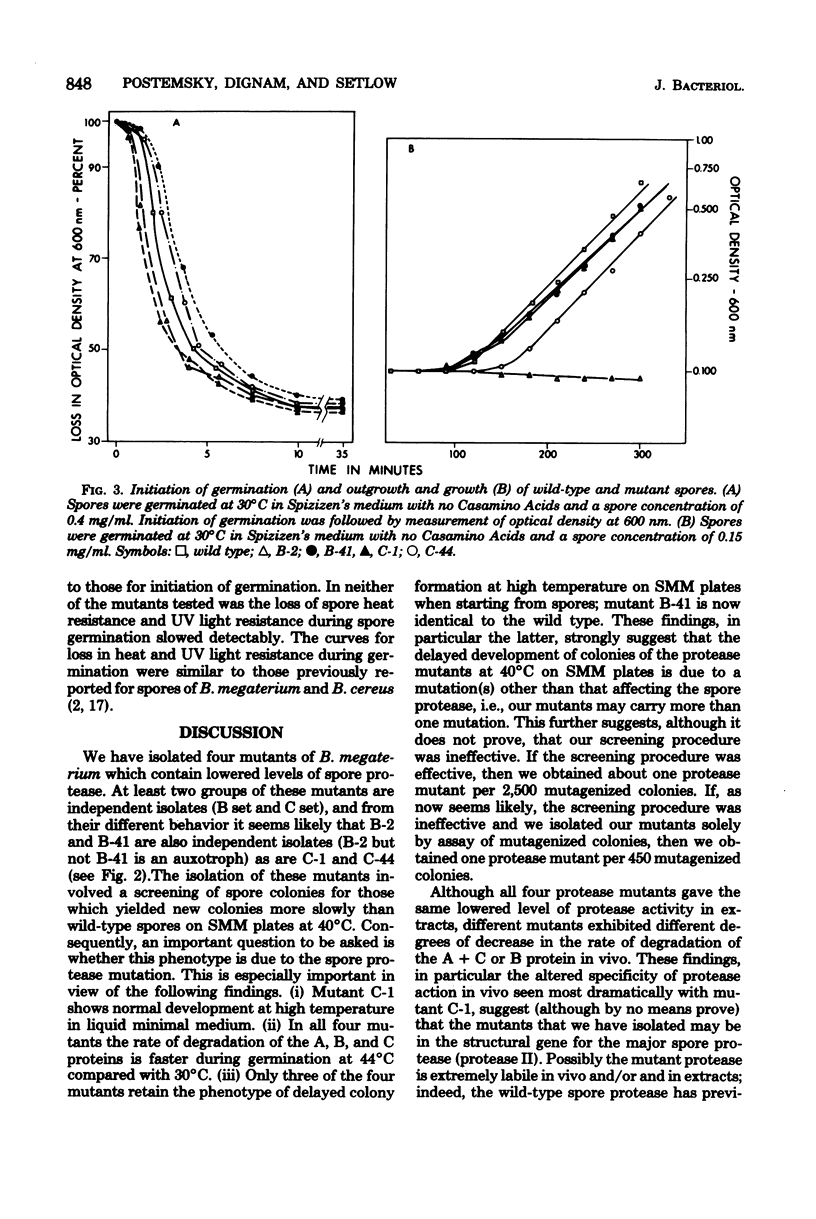
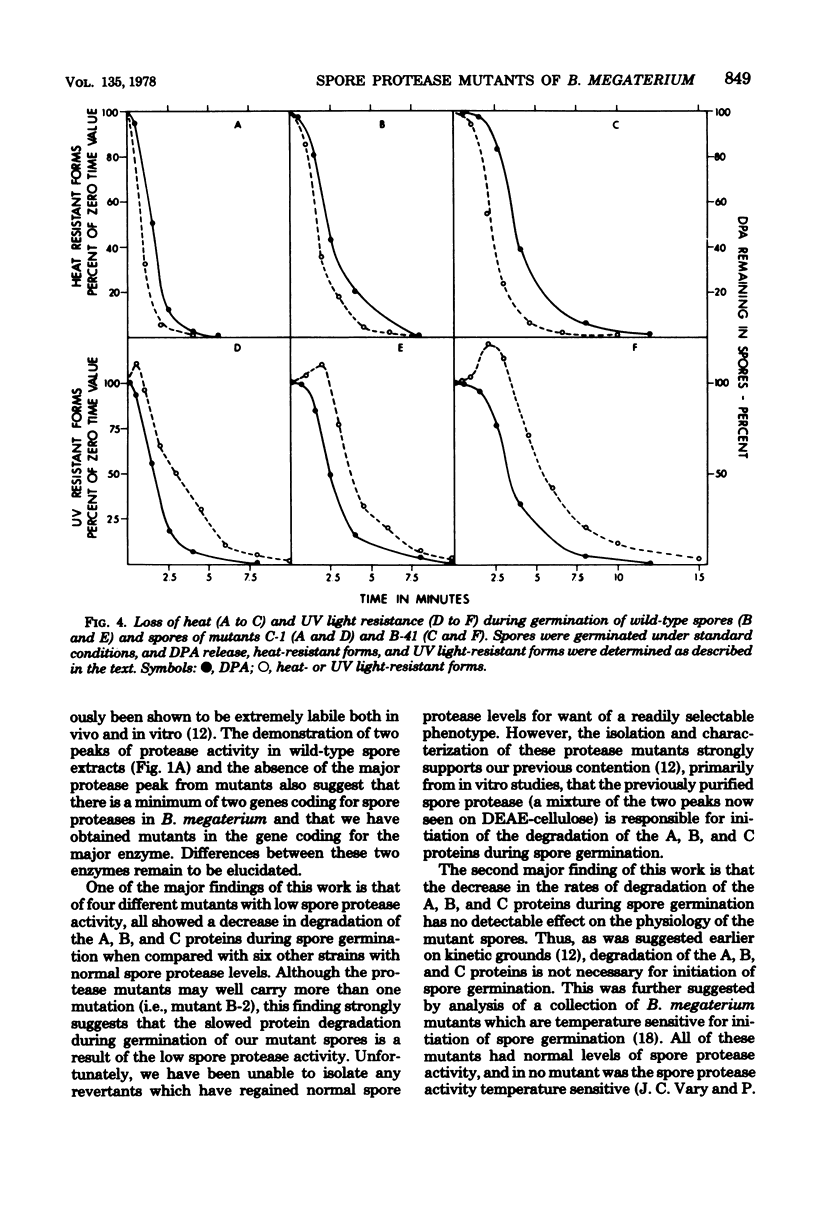
Abstract
A proteolytic activity present in spores of Bacillus megaterium has previously been implicated in the initiation of hydrolysis of the A, B, and C proteins which are degraded during spore germination. Four mutants of B. megaterium containing 20 to 30% of the normal level of spore proteolytic activity have been isolated. Partial purification of the protease from wild-type spores by a reviewed procedure resulted in the resolution of spore protease activity on the A, B, and C proteins into two peaks--a major one (protease II) and a minor one (protease I). The protease mutants tested lacked active protease II. All of the mutants exhibited a decreased rate of degradation of the A, B, and C proteins during spore germination at 30 degrees C, but degradation of the proteins did occur. Degradation of the A, B, and C proteins during germination of the mutant spores was decreased neither by blockade of ATP production nor by germination at 44 degrees C. Initiation of spore germination was normal in all four mutants, and all four mutants went through outgrowth, grew, and sporulated normally in rich medium. Similarly, outgrowth of spores of two of the four mutants was normal in minimal medium at 30 degrees C. In the two mutants studied, the kinetics of loss of spore heat resistance and spore UV light resistance during germination were identical to those of wild-type spores. This indicates that the A, B, and C proteins alone are not sufficient to account for the heat or UV light resistance of the dormant spore.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Chambon P., DuPraw E. J., Kornberg A. Biochemical studies of bacterial sporulation and germination. IX. Ribonucleic acid and deoxyribonucleic acid polymerases in nuclear fractions of vegetative cells and spores of Bacillus megaterium. J Biol Chem. 1968 Oct 10;243(19):5101–5109. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Levinson H. S., Hyatt M. T. Sequence of events during Bacillus megaterim spore germination. J Bacteriol. 1966 May;91(5):1811–1818. doi: 10.1128/jb.91.5.1811-1818.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rotman Y., Fields M. L. A modified reagent for dipicolinic acid analysis. Anal Biochem. 1968 Jan;22(1):168–168. doi: 10.1016/0003-2697(68)90272-8. [DOI] [PubMed] [Google Scholar]
- SACKS L. E., BAILEY G. F. DRY RUPTURE OF BACTERIAL SPORES. J Bacteriol. 1963 Mar;85:720–721. doi: 10.1128/jb.85.3.720-721.1963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Setlow P. Identification and localization of the major proteins degraded during germination of Bacillus megaterium spores. J Biol Chem. 1975 Oct 25;250(20):8159–8167. [PubMed] [Google Scholar]
- Setlow P. Identification of several unique low molecular weight basic proteins in dormant spores of Bacillus megaterium and their degradation during spore germination. Biochem Biophys Res Commun. 1974 Dec 23;61(4):1110–1117. doi: 10.1016/s0006-291x(74)80398-0. [DOI] [PubMed] [Google Scholar]
- Setlow P., Kornberg A. Biochemical studies of bacterial sporulation and germination. XVII. Sulfhydryl and disulfide levels in dormancy and germination. J Bacteriol. 1969 Dec;100(3):1155–1160. doi: 10.1128/jb.100.3.1155-1160.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Setlow P. Polyamine levels during growth, sporulation, and spore germination of Bacillus megaterium. J Bacteriol. 1974 Mar;117(3):1171–1177. doi: 10.1128/jb.117.3.1171-1177.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Setlow P., Primus G. Protein metabolism during germination of Bacillus megaterium spores. I. Protein synthesis and amino acid metabolism. J Biol Chem. 1975 Jan 25;250(2):623–630. [PubMed] [Google Scholar]
- Setlow P. Protein metabolism during germination of Bacillus megaterium spores. II. Degradation of pre-existing and newly synthesized protein. J Biol Chem. 1975 Jan 25;250(2):631–637. [PubMed] [Google Scholar]
- Setlow P. Purification and properties of a specific proteolytic enzyme present in spores of Bacillus magaterium. J Biol Chem. 1976 Dec 25;251(24):7853–7862. [PubMed] [Google Scholar]
- Setlow P. Purification and properties of some unique low molecular weight basic proteins degraded during germination of Bacillus megaterium spores. J Biol Chem. 1975 Oct 25;250(20):8168–8173. [PubMed] [Google Scholar]
- Setlow P., Waites W. M. Identification of several unique, low-molecular-weight basic proteins in dormant spores of clastridium bifermentans and their degradation during spore germination. J Bacteriol. 1976 Aug;127(2):1015–1017. doi: 10.1128/jb.127.2.1015-1017.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spizizen J. TRANSFORMATION OF BIOCHEMICALLY DEFICIENT STRAINS OF BACILLUS SUBTILIS BY DEOXYRIBONUCLEATE. Proc Natl Acad Sci U S A. 1958 Oct 15;44(10):1072–1078. doi: 10.1073/pnas.44.10.1072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stafford R. S., Donnellan J. E., Jr Photochemical evidence for conformation changes in DNA during germination of bacterial spores. Proc Natl Acad Sci U S A. 1968 Mar;59(3):822–828. doi: 10.1073/pnas.59.3.822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vary J. C., Kornberg A. Biochemical studies of bacterial sporulation and germination. XXI. Temperature-sensitive mutants for initiation of germination. J Bacteriol. 1970 Jan;101(1):327–329. doi: 10.1128/jb.101.1.327-329.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]



