Abstract
We evaluated the effects of sequential therapy with caspofungin (CAS) or amphotericin B (AMB) followed by posaconazole (POS) against Candida glabrata. The susceptibilities to POS of yeast cells pre-exposed to CAS or AMB were identical to those of untreated cells as shown by standard Clinical and Laboratory Standards Institute broth dilution, cell viability, and disk diffusion methods. We then investigated the activity of sequential regimens in an experimental model of disseminated candidiasis. CAS given at 1 mg/kg/day for 2 days followed by POS at either 15 or 30 mg/kg/day significantly reduced the counts compared to the controls, but this treatment was not superior to the use of CAS alone. Also, sequential regimens with AMB given at 1 mg/kg/day for 2 days followed by POS (AMB/POS) were effective at reducing the fungal burden against the controls. In addition, AMB/POS with both doses of the triazole were significantly more effective than AMB alone. Overall, our data showed that there is no therapeutic advantage in using CAS followed by POS, whereas an induction therapy with AMB followed by a maintenance regimen with POS might be a suitable strategy in managing C. glabrata infections.
Systemic fungal infections are a major cause of morbidity and mortality in debilitated patients. The spectrum of invasive fungal infections is changing, with an increased frequency of infections due to non-Candida albicans spp. Candida glabrata has recently emerged as the second most common cause of invasive candidiasis (14, 15). Systemic infections due to C. glabrata are characterized by a high mortality rate, and they are difficult to treat due to a reduced susceptibility of this species to azole drugs, especially to fluconazole (FLU) (14, 15). According to the published guidelines, amphotericin B (AMB) can be used to treat infections due to C. glabrata, especially in profoundly immunocompromised hosts (14). However, recent data suggest that a growing proportion of C. glabrata isolates may be resistant to AMB. This species appears to be susceptible to caspofungin (CAS), making this agent a valuable alternative as first-line therapy against this non-albicans species (10, 14).
Recently, posaconazole (POS) has been developed mainly to combat the development of resistance to azoles in yeasts, in particular to FLU, and to expand the spectrum of susceptible pathogens (1, 3, 5-7, 16-21). Numerous in vitro studies demonstrated that POS has a broad spectrum of activity against the majority of yeasts, filamentous fungi, and azole-resistant Candida species (16, 18). Concurrent and sequential antifungal treatment has been considered for invasive mycoses to improve the results of monotherapy. The new triazole, POS, which is available for oral administration, is expected to be used in different sequences in the course of systemic candidiasis. One possibility is to use CAS or AMB followed by POS. Therefore, in the present study we investigated the effects of sequential therapy with CAS or AMB and POS for the treatment of C. glabrata infections.
MATERIALS AND METHODS
Isolates.
Four clinical isolates of C. glabrata (isolates 4198, 4205, 4293, and 4370) were used. The isolates were recovered from blood. Each strain represented a unique isolate from a patient. Yeast isolates were identified at the species level by conventional morphological and biochemical methods and stored at −70°C in 10% glycerol. Before the initiation of the study, yeast isolates were subcultured on antimicrobial-agent-free medium to ensure viability and purity. Candida parapsilosis ATCC 22019 was used as a quality control (12).
Drugs.
POS (Schering Plough Research Institute, Kenilworth, N.J.) was prepared with polyethylene glycol 200 (PEG-200; Janssen Chimica, Geel, Belgium) for both in vitro and in vivo studies. CAS was used as a commercial preparation (Cancidas; Merck Sharp & Dohme) for both in vitro and in vivo experiments. It was dissolved according to the manufacturer's instructions. AMB was used as pure powder (Sigma) for in vitro studies and as a commercial preparation (Fungizone; Bristol-Myers Squibb) for in vivo studies. It was dissolved in dimethyl sulfoxide and in sterile water according to the manufacturer instructions for in vitro and in vivo studies, respectively.
In vitro studies. (i) Broth dilution.
Antifungal susceptibility testing was performed by a broth microdilution method in accordance with the Clinical and Laboratory Standards Institute (CLSI; formerly the National Committee for Clinical Laboratory Standards) recommendations (12). The final concentrations of all drugs ranged from 0.0078 to 8 μg/ml. Plates were incubated at 35°C and read at 24 and 48 h. Readings were performed spectrophotometrically (optical density at 490 nm) with an automatic plate reader (ELx800; Biotek). POS and CAS MICs were considered as the first concentration of the antifungal agent at which the turbidity in the well was 50% less than that in the control well (12, 13). AMB MIC was considered the first concentration of the antifungal agent at which the turbidity in the well was 90% less than that in the control well (12). Experiments were performed in quintuplicate.
(ii) Exposure to CAS or AMB.
To study the effects of exposure to either CAS or AMB on the susceptibility patterns of POS, yeast cells were grown overnight in drug-free RPMI 1640 (Sigma) (unexposed cells) or in RPMI 1640 containing CAS or AMB at either one-eighth or one-half the MIC (exposed cells). The cells were then washed twice, counted, and diluted to obtain suitable inocula (0.5 × 103 to 2.5 × 103 CFU/ml for broth dilution and 1 × 106 CFU/ml for halo assay) for POS susceptibility testing. Experiments were performed in quintuplicate.
(iii) CFU determination.
Determination of the numbers of CFU per milliliter was performed only with C. glabrata 4293. After MICs were read, 100-μl samples from the wells corresponding to either 2 or 16 times the POS MICs were withdrawn and plated onto Sabouraud dextrose agar plates. Inoculated plates were incubated at 35°C, and the percentage of viable cells at 48 h was calculated. Experiments were performed in triplicate.
(iv) Halo assay.
Drug-exposed and unexposed yeast cells of C. glabrata 4293 were plated onto Mueller-Hinton agar (Biogenetics srl) plates supplemented with 0.5 μg of methylene blue (4). Six-mm-diameter BBL disks (Becton Dickinson) were embedded with 20 μl of POS at concentrations of 5 or 15 μg. After the disks had dried, they were placed onto inoculated agar plates. The plates were incubated at 35°C, and inhibition zone diameters were measured at 24 and 48 h. Experiments were performed in quintuplicate.
Animal experiments.
CD1 male mice (Charles River, Calco, Lecco, Italy) weighing 25 g were rendered neutropenic by intraperitoneal administration of cyclophosphamide (200 mg/kg of body weight/day) on days −4, +1, and +4 postinfection. The animals were infected intravenously with C. glabrata 4293 given in a 0.2-ml volume. Two studies were performed: in study 1 the mice were challenged with 2.44 × 108 CFU/mouse, and in study 2 the mice were challenged with 1.04 × 108 CFU/mouse. Therapy was initiated 24 h postchallenge: POS was administered at 0.2 ml orally by gavage once daily at doses of 15 mg/kg/day (study 1) and 30 mg/kg/day (study 2). Either CAS or AMB was given intraperitoneally in 0.2 ml at a dose of 1 mg/kg/day in both studies. Drug efficacy was assessed by determining the number of CFU per kidney pair. Briefly, the mice were sacrificed, the kidneys were homogenized and diluted and undiluted aliquots, including the entire organ, were grown on Sabouraud dextrose agar for colony count determination. In both studies, nine groups of animals were considered: (i) placebo treatment (P3; sterile saline solution) from day 1 to day 2 postinfection, (ii) CAS from day 1 to day 2 (CAS), (iii) AMB from day 1 to day 2 (AMB), (iv) placebo treatment from day 1 to day 6 (P7; sterile saline solution from day 1 to day 2 and PEG-200 from day 3 to day 6), (v) P from day 1 to day 2 followed by POS from day 3 to day 6 (P/POS), (vi) CAS from day 1 to day 2 followed by P from day 3 to day 6 (CAS/P), (vii) CAS from day 1 to day 2 followed by POS from day 3 to day 6 (CAS/POS), (viii) AMB from day 1 to day 2 followed by P from day 3 to day 6 (AMB/P), and (ix) AMB from day 1 to day 2 followed by POS from day 3 to day 6 (AMB/POS). Groups 1 to 3 were sacrificed on day 3 postinfection. Groups 4 to 9 were sacrificed on day 7 postinfection. There were seven to eight animals in each group. Animal experiments were conducted with the approval of University of Ancona Ethics Committee.
Statistical analysis.
The in vitro susceptibility data were compared by using the Student t test or the Mann-Whitney U test. A P value of <0.05 was considered statistically significant. The Mann-Whitney U test was also used to compare tissue burden counts. Due to multiple comparisons, a P value of <0.016 was considered statistically significant.
RESULTS
In vitro studies. (i) MICs.
MIC data are shown in Table 1. MICs for C. parapsilosis ATCC 22019 were within the expected ranges (12).
TABLE 1.
In vitro susceptibilities of yeast isolates used in this study to FLU, AMB, CAS, and POS
| Isolatesa | Median MIC in μg/ml (range) for ofb:
|
|||
|---|---|---|---|---|
| FLU | AMB | CAS | POSc | |
| C. parapsilosis ATCC 22019 | 4.0 (2.0-4.0) | 1.0 (1.0) | 0.25 (0.25) | ≤0.03 (≤0.03) |
| C. glabrata 4198 | >64 (>64) | 0.5 (0.5-1.0) | 0.25 (0.06-0.25) | 0.5 (0.5-2.0), 0.5 (0.5-1.0)*, 1.0 (0.5-2.0)† |
| C. glabrata 4205 | 1.0 (1.0-4.0) | 0.5 (0.25-0.5) | 0.25 (0.25) | ≤0.03 (≤0.03), ≤0.03 (≤0.03-0.06)*, 0.06 (≤0.03-0.06)† |
| C. glabrata 4293 | 32 (32-64) | 0.5 (0.5) | ≤0.03 (≤0.03-0.06) | 0.25 (0.125-0.25), 0.25 (0.125-0.25)*, 0.125 (0.125-0.25)† |
| C. glabrata 4370 | 16 (8.0-16) | 1.0 (1.0-2.0) | 0.125 (0.125) | 0.06 (0.06-0.125), 0.06 (0.06-0.125)*, 0.125 (0.06-0.25)† |
Each isolate was tested five times.
Readings were performed spectrophotometrically at either 24 or 48 h with an automatic plate reader (ELx800; Biotek) set at 490 nm. The results were similar (within one double dilution). MICs refer to 48-h readings for POS, FLU, and AMB and to 24-h readings for CAS.
*, Susceptibility to POS after overnight exposure of yeast isolates to AMB at their respective one-half MIC; †, susceptibility to POS after overnight exposure of yeast isolates to CAS at their respective one-half MIC.
According to FLU susceptibility definition, only isolate 4205 was susceptible to the triazole (median MIC of 1.0 μg/ml). Isolates 4370 and 4293 were susceptible to FLU in a dose-dependent manner (susceptible dose dependent [SDD], median MICs of 16 and 32 μg/ml, respectively), while isolate 4198 was resistant (median MIC of >64 μg/ml).
The median POS MICs were 0.5, ≤0.03, 0.25, and 0.06 μg/ml for isolates 4198, 4205, 4293, and 4370, respectively. The median CAS MICs were 0.25, 0.25, ≤0.03, and 0.125 μg/ml for isolates 4198, 4205, 4293, and 4370, respectively. The median AMB MICs for all isolates ranged from 0.5 to 1.0 μg/ml.
AMB exposure of yeast cells to one-half of their respective MICs did not have any significant effects on POS susceptibility. In fact, POS MICs of exposed cells were identical or similar (within one double dilution) to those reported for unexposed cells. Similarly, POS MICs of yeasts cells exposed to CAS were similar to those reported for unexposed cells (Table 1).
(ii) CFU determinations.
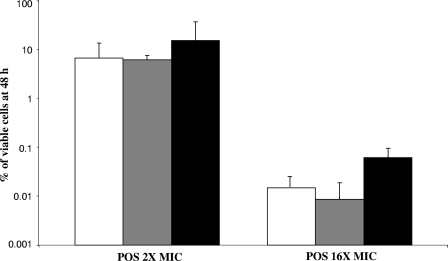
Figure 1 shows the effects of POS on the viability of yeast cells of C. glabrata 4293 grown overnight in drug-free RPMI 1640 medium or medium containing CAS or AMB at one-half MIC. The triazole exerted a dose-dependent activity with percentages of viability ranging from 6.2 to 15.2 and from 0.01 to 0.06 for cells incubated at 2 and 16 times the POS MIC, respectively. In general, exposure to either CAS or AMB did not show any significant variation in the viability of yeast cells with respect to unexposed cells.
FIG. 1.
Effects of posaconazole on the viability of C. glabrata 4293 grown overnight in drug-free RPMI 1640 medium (□) or medium containing caspofungin (░⃞) or amphotericin B (▪) at one-half the MIC (0.015 and 0.25 μg/ml, respectively). Experiments were performed with an initial inoculum that ranged from 0.5 × 103 to 2.5 × 103 CFU/ml. POS MICs were determined according to the CLSI method against either exposed or unexposed cells. The number of CFU per milliliter was determined by plating the wells containing POS at 2× and 16× the MIC (0.5 and 4.0 μg/ml, respectively). The data are averages of the percentage of viable cells at 48 h with respect to the controls, and error bars denote the standard deviations.
(iii) Halo assay.
To further investigate the possible effects of drug exposure on POS susceptibility, we compared the diameters of growth inhibition of exposed and unexposed yeast cells (Table 2). Although there was a tendency toward a slightly reduction of diameters for cells exposed to both CAS and AMB with respect to unexposed cells, the difference was not statistically significant.
TABLE 2.
In vitro activity of POS against C. glabrata 4293 by the halo assaya
| Treatmentb | Mean halo diam (mm) ± SD of POS tested at:
|
|
|---|---|---|
| 5 μg/disk | 15 μg/disk | |
| Unexposed cells | 29.0 ± 2.5 | 30.0 ± 1.9 |
| Exposed to CAS at one-half the MIC | 26.8 ± 0.4 | 29.0 ± 1.2 |
| Exposed to CAS at one-eighth the MIC | 28.0 ± 1.0 | 29.6 ± 0.5 |
| Exposed to AMB at one-half the MIC | 28.4 ± 1.7 | 29.8 ± 0.4 |
| Exposed to AMB at one-half the MIC | 27.0 ± 0.7 | 30.0 ± 0.0 |
C. glabrata 4293 was tested in five independent experiments. Readings were performed at either 24 or 48 h. The results were identical, and 24-h readings are shown in the table.
C. glabrata 4293 was grown overnight in drug-free RPMI 1640 medium (unexposed cells) or medium containing CAS or AMB at one-half the MIC (0.015 and 0.25 mg/ml, respectively) or one-eighth the MIC (0.00375 and 0.0625 mg/ml, respectively).
In vivo studies.
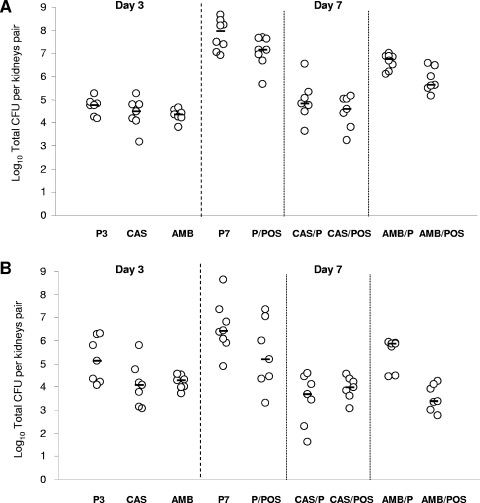
To investigate this interaction in vivo, CD1 mice were infected intravenously with C. glabrata 4293 and treated with several therapeutic regimens, including schemes of sequential therapy. Figure 2A and B show the results of studies 1 and 2, respectively. In both studies, nine groups of animals were considered. Neither CAS nor AMB were effective at reducing the fungal burden on day 3 postinfection in both studies. There was an increase of kidney burdens from day 3 (P3) to day 7 (P7) postinfection in both studies, suggesting a progression of the infection in the untreated animals.
FIG. 2.
Tissue burden of kidneys of CD1 mice infected intravenously with C. glabrata 4239 and treated with P (sterile saline solution) from day 1 to day 2 postinfection (P3), CAS from day 1 to day 2 (CAS), AMB from day 1 to day 2 (AMB), P from day 1 to day 6 (P7; sterile saline solution from day 1 to day 2 and PEG-200 from day 3 to day 6), P (sterile saline solution) from day 1 to day 2 followed by POS from day 3 to day 6 (P/POS), CAS from day 1 to day 2 followed by P (PEG-200) from day 3 to day 6 (CAS/P), CAS from day 1 to day 2 followed by POS from day 3 to day 6 (CAS/POS), AMB from day 1 to day 2 followed by P (PEG-200) from day 3 to day 6 (AMB/P), or AMB from day 1 to day 2 followed by POS from day 3 to day 6 (AMB/POS). The first three groups were sacrificed on day 3 postinfection. The last six groups were sacrificed on day 7 postinfection. In study 1, the animals were infected with 1.04 × 108 CFU/mouse and POS was administered at 15 mg/kg/day (A), while in study 2 the animals were challenged with 1.04 × 108 CFU/mouse and POS was administered at 30 mg/kg/day (B). The bars represent the medians. There were seven to eight animals in each group.
Among the groups sacrificed on day 7, both doses of POS initiated on day 3 postinfection (P/POS) showed a reduction of CFU with respect to their controls, but the difference was not statistically significant. On the other hand, counts for the kidneys of mice treated with CAS for 2 days and sacrificed on day 7 postinfection (CAS/P) were significantly reduced compared to those of untreated controls (P = 0.0002 and 0.0003 in studies 1 and 2, respectively). Similarly, sequential therapy with CAS followed by POS at either 15 or 30 mg/kg/day (CAS/POS) significantly reduced the counts compared to the controls (P = 0.0003). However, neither sequential therapy was more effective than CAS/P.
AMB/P and AMB/POS were all effective at reducing the fungal burden in the controls (P values ranging from 0.0003 to 0.013). In addition, sequential therapies with both doses of the triazole were significantly more effective than AMB/P (P = 0.006 and 0.0006 in studies 1 and 2, respectively).
DISCUSSION
In this study, we evaluated the effects of sequential therapies with AMB or CAS followed by POS for treatment of C. glabrata infections.
Our in vitro data indicate that pre-exposure of C. glabrata to CAS or AMB did not alter the fungal susceptibility to POS. This phenomenon was seen with three different assays. First, CAS or AMB pre-exposure of four C. glabrata isolates to one-half of their respective MICs did not have any significant effect on the POS susceptibility investigated by the CLSI broth dilution method. Similarly, pre-exposure to either CAS or AMB did not cause any significant variations in the viability of yeast cells with respect to the unexposed cells. Finally, POS disk diffusion diameters did not vary significantly when the four C. glabrata isolates were pre-exposed to CAS or AMB at one-half or one-eighth their respective MICs. Previous literature data on sequential therapy of AMB and triazoles have demonstrated that the order of drug administration can play a critical role (11). Although the majority of in vitro data are limited for C. albicans, the sequential exposure to FLU followed by AMB caused the fungus to be transiently resistant to the polyene. It has been proposed that pre-exposure of the fungus to azoles depletes ergosterol, which is the prime target for AMB action (11). Consequently, added AMB will not have the cellular target necessary for its activity, resulting in antagonistic interaction. On the contrary, it has been hypothesized that AMB, by binding to fungal membrane sterols and creating a pore, provides greater access to azoles into the cytoplasm, leading to increased inhibition of ergosterol synthesis. Studies on sequential therapy of CAS followed by triazoles against Candida spp. are limited (2). Our previous in vitro data indicate that exposure to CAS does not alter the initial susceptibility to FLC in C. albicans (2).
The present in vitro data correlate with the outcome of our in vivo murine model of systemic candidiasis. Actually, the switch from CAS or AMB to POS did not yield an antagonistic effect.
We found that sequential therapy CAS/POS was as effective as CAS followed by placebo (CAS/P). This result suggests that there is no therapeutic advantage in using the sequential therapy CAS followed by POS, at least with the doses and regimen schemes investigated here. It is important to note that CAS given for 2 days did not yield a regrowth in fungal burden, as shown by a similar CFU level in the kidneys of mice sacrificed on days 3 and 7 postinfection. Our results can be explained by the findings of Louie et al., who investigated the pharmacodynamic parameters linked with CAS efficacy in a murine model of systemic candidiasis caused by C. albicans (9). These authors demonstrated that therapeutic concentrations of CAS persist at the site of infection in kidney tissue well after serum concentrations fall below the MIC, underscoring the primacy of CAS levels in tissues in determining treatment outcome (9).
In contrast to what we observed with CAS/POS, the sequential scheme of AMB/POS proved more effective than the polyene followed by placebo. Interestingly, this effect was seen at both doses of POS. Also, we observed that AMB given for 2 days yielded a fungal regrowth on day 7 postinfection, as shown by a significant increase in CFU counts between the AMB and AMB/P groups. Recently, Hope et al. applied a mathematical model on the time course of C. albicans kidney burden in neutropenic mice treated with AMB (8). These researchers observed a net growth of C. albicans after the administration of 0.63 mg of AMB/kg, while a dose of 5 mg/kg was required to overcome progressive growth of C. albicans and produce a fungal density that was comparable to that observed at the time of the initiation of therapy (8). Although we utilized a dose of AMB at 1 mg/kg/day for 2 days and a different species of Candida, our results are in agreement with their findings.
To our knowledge, this is the first study in which the efficacy of POS against C. glabrata was investigated after a short exposure to CAS or AMB. Overall, our data showed that there is no therapeutic advantage in using CAS followed by POS, although they seem to indicate that an induction therapy with AMB followed by a maintenance regimen with POS might be a suitable strategy in managing C. glabrata infections. An important point of this approach is the possibility to switch from an intravenous to an oral therapy with both patient and cost advantages.
Additional studies are warranted to further characterize the potential benefits of sequential therapy with AMB and POS against C. glabrata.
Footnotes
Published ahead of print on 3 December 2007.
REFERENCES
- 1.Anstead, G. M., M. Martinez, and J. R. Graybill. 2006. Control of a Candida glabrata prosthetic endovascular infection with posaconazole. Med. Mycol. 44:273-277. [DOI] [PubMed] [Google Scholar]
- 2.Barchiesi, F., E. Spreghini, I. Baldassarri, A. Marigliano, D. Arzeni, D. Giannini, and G. Scalise. 2004. Sequential therapy with caspofungin and fluconazole for Candida albicans infection. Antimicrob. Agents Chemother. 48:4056-4058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cacciapuoti, A., D. Loebenberg, E. Corcoran, F. Menzel, E. L. Moss, C. Norris, M. Michalski, K. Raynor, J. Halpern, C. Mendrick, B. Arnold, B. Antonacci, R. Parmegiani, T. Yarosh-Tomaine, G. H. Miller, and R. S. Hare. 2000. In vitro and in vivo activities of SCH 56592 (posaconazole), a new triazole antifungal agent, against Aspergillus and Candida. Antimicrob. Agents Chemother. 44:2017-2022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Clinical and Laboratory Standards Institute/National Committee for Clinical Laboratory Standards. 2004. Method for antifungal disk diffusion susceptibility testing of yeasts. Approved guideline. Document M44-A. National Committee for Clinical Laboratory Standards, Wayne, PA.
- 5.Cornely, O. A., J. Maertens, D. J. Winston, J. Perfect, A. J. Ullmann, T. J. Walsh, D. Helfgott, J. Holowiecki, D. Stockelberg, Y. T. Goh, M. Petrini, C. Hardalo, R. Suresh, and D. Angulo-Gonzalez. 2007. Posaconazole versus fluconazole or itraconazole prophylaxis in patients with neutropenia. N. Engl. J. Med. 356:348-359. [DOI] [PubMed] [Google Scholar]
- 6.Greenberg, R. N., K. Mullane, J. A. van Burik, I. Raad, M. J. Abzug, G. Anstead, R. Herbrecht, A. Langston, K. A. Marr, G. Schiller, M. Schuster, J. R. Wingard, C. E. Gonzalez, S. G. Revankar, G. Corcoran, R. J. Kryscio, and R. Hare. 2006. Posaconazole as salvage therapy for zygomycosis. Antimicrob. Agents Chemother. 50:126-133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gupta, S., N. G. Almyroudis, M. Battiwalla, B. J. Bambach, P. L. McCarthy, A. D. Proefrock, D. Ball, P. Paplham, A. Varma, J. Kwon-Chung, and B. H. Segal. 2007. Successful treatment of disseminated fusariosis with posaconazole during neutropenia and subsequent allogeneic hematopoietic stem cell transplantation. Transpl. Infect. Dis. 9:156-160. [DOI] [PubMed] [Google Scholar]
- 8.Hope, W. W., G. L. Drusano, C. B. Moore, A. Sharp, A. Louie, T. J. Walsh, D. W. Denning, and P. A. Warn. 2007. Effect of neutropenia and treatment delay on the response to antifungal agents in experimental disseminated candidiasis. Antimicrob. Agents Chemother. 51:285-295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Louie, A., M. Deziel, W. Liu, M. F. Drusano, T. Gumbo, and G. L. Drusano. 2005. Pharmacodynamics of caspofungin in a murine model of systemic candidiasis: importance of persistence of caspofungin in tissues to understanding drug activity. Antimicrob. Agents Chemother. 49:5058-5068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mora-Duarte, J., R. Betts, C. Rotstein, A. L. Colombo, L. Thompson-Moya, J. Smietana, R. Lupinacci, C. Sable, N. Kartsonis, and J. Perfect. 2002. Comparison of caspofungin and amphotericin B for invasive candidiasis. N. Engl. J. Med. 347:2020-2029. [DOI] [PubMed] [Google Scholar]
- 11.Mukherjee, P. K., D. J. Sheehan, C. A. Hitchcock, and M. A. Ghannoum. 2005. Combination treatment of invasive fungal infections. Clin. Microbiol. Rev. 18:163-194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.National Committee for Clinical Laboratory Standards. 2002. Reference method for broth dilution antifungal susceptibility testing of yeast, 2nd ed. Approved standard M27-A2. National Committee for Clinical Laboratory Standards, Wayne, PA.
- 13.Odds, F., M. Motyl, R. Andrade, J. Bille, E. Canton, M. Cuenca-Estrella, A. Davidson, C. Durussel, D. Ellis, E. Foraker, A. W. Fothergill, M. A. Ghannoum, R. A. Giacobbe, M. Gobernado, R. Handke, M. Laverdiere, W. Lee-Yang, W. G. Merz, L. Ostrosky-Zeichner, J. Peman, S. Perea, J. R. Perfect, M. A. Pfaller, L. Proia, J. H. Rex, M. G. Rinaldi, J. L. Rodriguez-Tudela, W. A. Schell, C. Shields, D. A. Sutton, P. E. Verweij, and D. W. Warnock. 2004. Interlaboratory comparison of results of susceptibility testing with caspofungin against Candida and Aspergillus species. J. Clin. Microbiol. 42:3475-3482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pappas, P. G., J. H. Rex, J. D. Sobel, S. G. Filler, W. E. Dismukes, T. J. Walsh, J. E. Edwards, et al. 2004. Guidelines for treatment of candidiasis. Clin. Infect. Dis. 38:161-189. [DOI] [PubMed] [Google Scholar]
- 15.Pfaller, M. A., and D. J. Diekema. 2007. Epidemiology of invasive candidiasis: a persistent public health problem. Clin. Microbiol. Rev. 20:133-163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pfaller, M. A., S. A. Messer, L. Boyken, R. J. Hollis, C. Rice, S. Tendolkar, and D. J. Diekema. 2004. In vitro activities of voriconazole, posaconazole, and fluconazole against 4,169 clinical isolates of Candida spp. and Cryptococcus neoformans collected during 2001 and 2002 in the ARTEMIS global antifungal surveillance program. Diagn. Microbiol. Infect. Dis. 48:201-205. [DOI] [PubMed] [Google Scholar]
- 17.Skiest, D. J., J. A. Vazquez, G. M. Anstead, J. R. Graybill, J. Reynes, D. Ward, R. Hare, N. Boparai, and R. Isaacs. 2007. Posaconazole for the treatment of azole-refractory oropharyngeal and esophageal candidiasis in subjects with HIV infection. Clin. Infect. Dis. 44:607-614. [DOI] [PubMed] [Google Scholar]
- 18.Torres, H. A., R. Y. Hachem, R. F. Chemaly, D. P. Kontoyiannis, and I. I. Raad. 2005. Posaconazole: a broad-spectrum triazole antifungal. Lancet Infect. Dis. 5:775-785. [DOI] [PubMed] [Google Scholar]
- 19.Ullmann, A. J., J. H. Lipton, D. H. Vesole, P. Chandrasekar, A. Langston, S. R. Tarantolo, H. Greinix, W. Morais de Azevedo, V. Reddy, N. Boparai, L. Pedicone, H. Patino, and S. Durrant. 2007. Posaconazole or fluconazole for prophylaxis in severe graft-versus-host disease. N. Engl. J. Med. 356:335-3347. [DOI] [PubMed] [Google Scholar]
- 20.Vazquez, J. A., D. J. Skiest, L. Nieto, R. Northland, I. Sanne, J. Gogate, W. Greaves, and R. Isaacs. 2006. A multicenter randomized trial evaluating posaconazole versus fluconazole for the treatment of oropharyngeal candidiasis in subjects with HIV/AIDS. Clin. Infect. Dis. 42:1179-1186. [DOI] [PubMed] [Google Scholar]
- 21.Walsh, T. J., I. Raad, T. F. Patterson, P. Chandrasekar, G. R. Donowitz, R. Graybill, R. E. Greene, R. Hachem, S. Hadley, R. Herbrecht, A. Langston, A. Louie, P. Ribaud, B. H. Segal, D. A. Stevens, J. A. van Burik, C. S. White, G. Corcoran, J. Gogate, G. Krishna, L. Pedicone, C. Hardalo, and J. R. Perfect. 2007. Treatment of invasive aspergillosis with posaconazole in patients who are refractory to or intolerant of conventional therapy: an externally controlled trial. Clin. Infect. Dis. 44:2-12. [DOI] [PubMed] [Google Scholar]