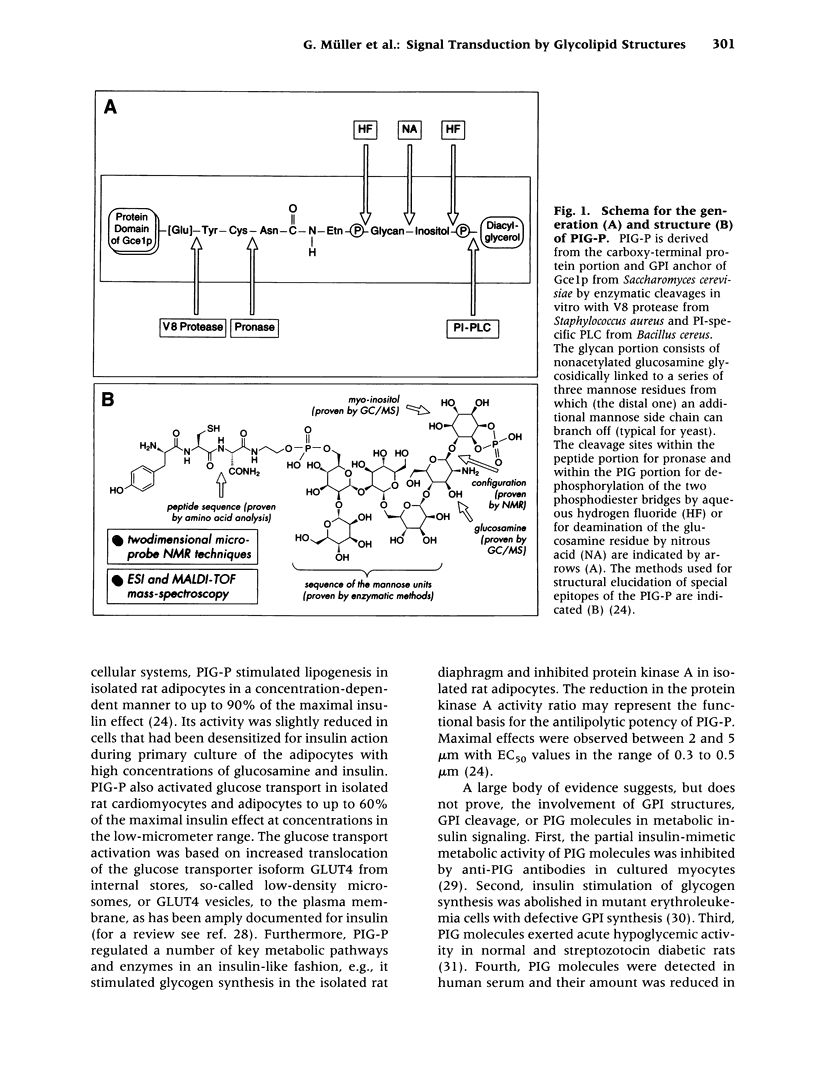
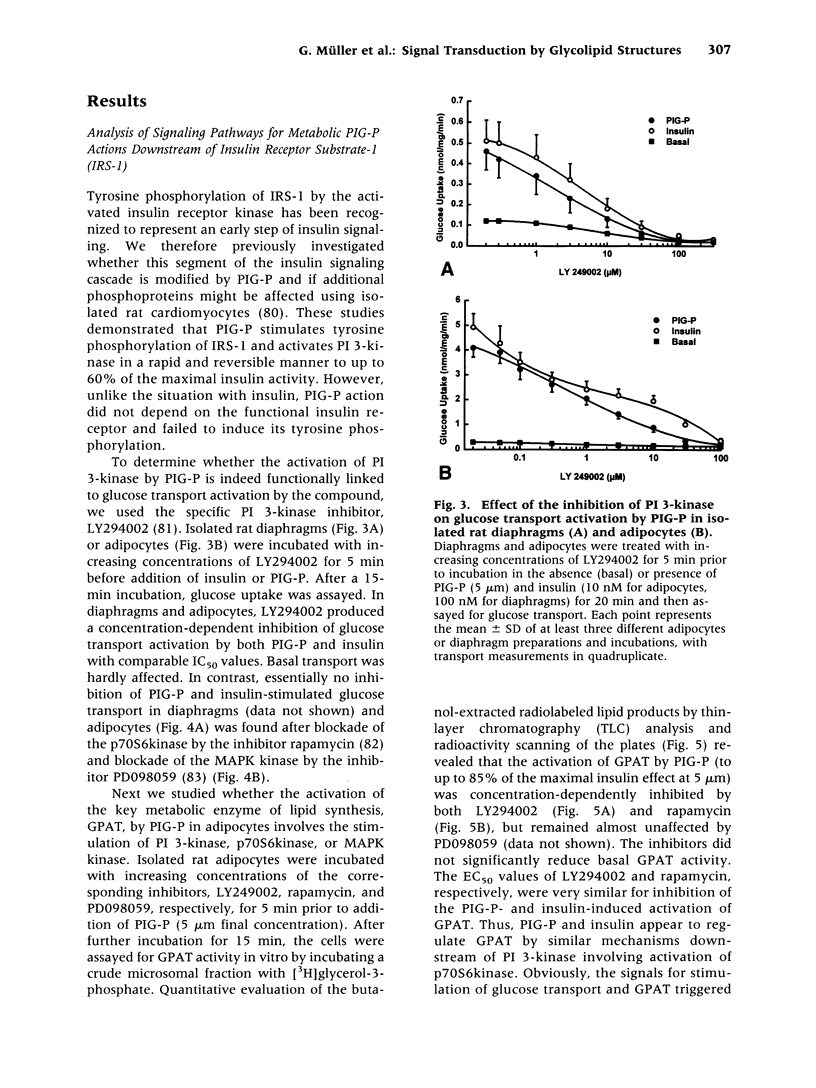
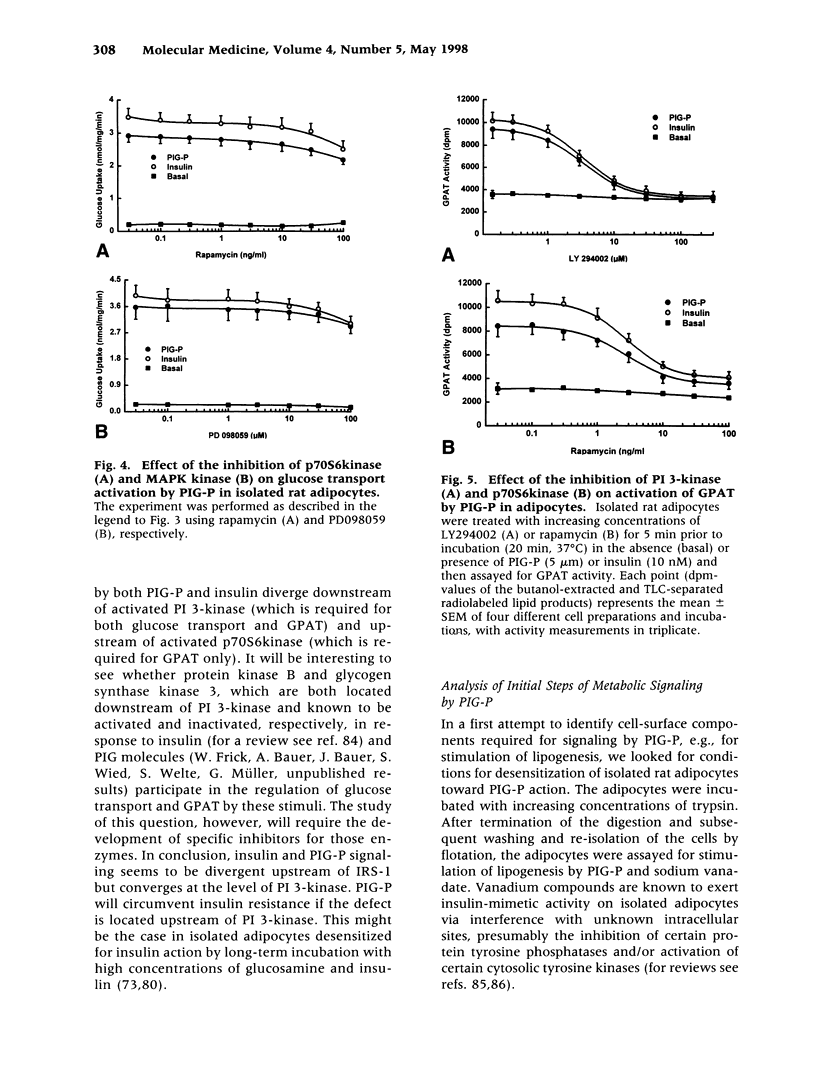
Abstract
Phosphoinositolglycan molecules isolated from insulin-sensitive mammalian tissues have been demonstrated in numerous in vitro studies to exert partial insulin-mimetic activity on glucose and lipid metabolism in insulin-sensitive cells. However, their ill-defined structures, heterogeneous nature, and limited availability have prohibited the analysis of the underlying molecular mechanism. Phosphoinositolglycan-peptide (PIG-P) of defined and homogeneous structure prepared in large scale from the core glycan of a glycosyl-phosphatidylinositol-anchored membrane protein from Saccharomyces cerevisiae has recently been shown to stimulate glucose transport as well as a number of glucose-metabolizing enzymes and pathways to up to 90% (at 2 to 10 microns) of the maximal insulin effect in isolated rat adipocytes, cardiomyocytes, and diaphragms (G. Müller et al., 1997, Endocrinology 138: 3459-3476). Consequently, we used this PIG-P for the present study in which we compare its intracellular signaling with that of insulin. The activation of glucose transport by both PIG-P and insulin in isolated rat adipocytes and diaphragms was found to require stimulation of phosphatidylinositol (PI) 3-kinase but to be independent of functional p70S6kinase and mitogen-activated protein kinase. The increase in glycerol-3-phosphate acyltransferase activity in rat adipocytes in response to PIG-P and insulin was dependent on both PI 3-kinase and p70S6kinase. This suggest that the signaling pathways for PIG-P and insulin to glucose transport and metabolism converage at the level of PI 3-kinase. A component of the PIG-P signaling pathway located up-stream of PI 3-kinase was identified by desensitization of isolated rat adipocytes for PIG-P action by combined treatment with trypsin and NaCl under conditions that preserved cell viability and the insulin-mimetic activity of sodium vanadate but completely blunted the insulin response. Incubation of the cells with either trypsin or NaCl alone was ineffective. The desensitized adipocytes were reconstituted for stimulation of lipogenesis by PIG-P by addition of the concentrated trypsin/salt extract. The reconstituted adipocytes exhibited 65-75% of the maximal PIG-P response and similar EC50 values for PIG-P (2 to 5 microns) compared with control cells. A proteinaceous N-ethylmaleimide (NEM)-sensitive component contained in the trypsin/salt extract was demonstrated to bind in a functional manner to the adipocyte plasma membrane of desensitized adipocytes via bipolar interactions. An excess of trypsin/salt extract inhibited PIG-P action in untreated adipocytes in a competitive fashion compatible with a receptor function for PIG-P of this protein. The presence of the putative PIG-P receptor protein in detergent-insoluble complexes prepared from isolated rat adipocytes suggests that caveolae/detergent-insoluble complexes of the plasma membrane may play a role in insulin-mimetic signaling by PIG-P. Furthermore, treatment of isolated rat diaphragms and adipocytes with PIG-P as well as with other agents exerting partially insulin-mimetic activity, such as PI-specific phospholipase C (PLC) and the sulfonylurea glimepiride, triggered tyrosine phosphorylation of the caveolar marker protein caveolin, which was apparently correlated with stimulation of lipogenesis. Strikingly, in adipocytes subjected to combined trypsin/salt treatment, PIG-P, PI-specific PLC, and glimepiride failed completely to provoke insulin-mimetic effects. A working model is presented for a signaling pathway in insulin-sensitive cells used by PIG(-P) molecules which involves GPI structures, the trypsin/salt- and NEM-sensitive receptor protein for PIG-P, and additional proteins located in caveolae/detergent-insoluble complexes.
Full text
PDF













Images in this article
Selected References
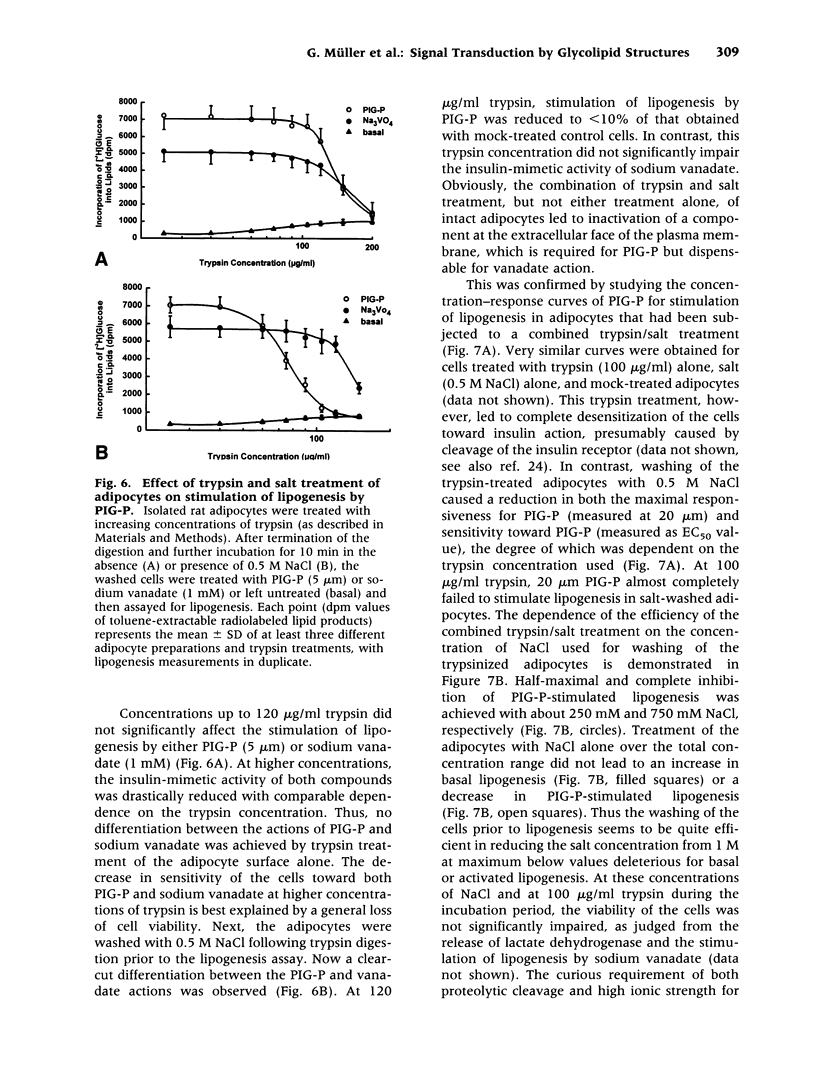
These references are in PubMed. This may not be the complete list of references from this article.
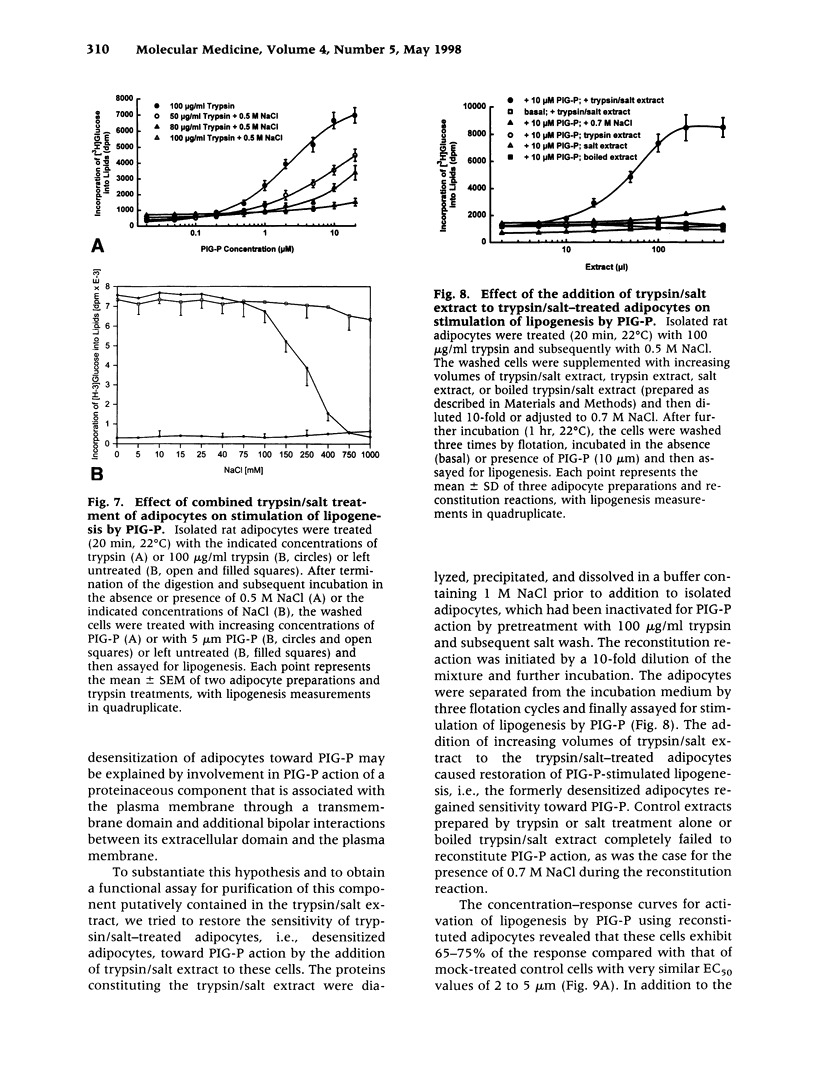
- Anderson R. G. Plasmalemmal caveolae and GPI-anchored membrane proteins. Curr Opin Cell Biol. 1993 Aug;5(4):647–652. doi: 10.1016/0955-0674(93)90135-d. [DOI] [PubMed] [Google Scholar]
- Argetsinger L. S., Hsu G. W., Myers M. G., Jr, Billestrup N., White M. F., Carter-Su C. Growth hormone, interferon-gamma, and leukemia inhibitory factor promoted tyrosyl phosphorylation of insulin receptor substrate-1. J Biol Chem. 1995 Jun 16;270(24):14685–14692. doi: 10.1074/jbc.270.24.14685. [DOI] [PubMed] [Google Scholar]
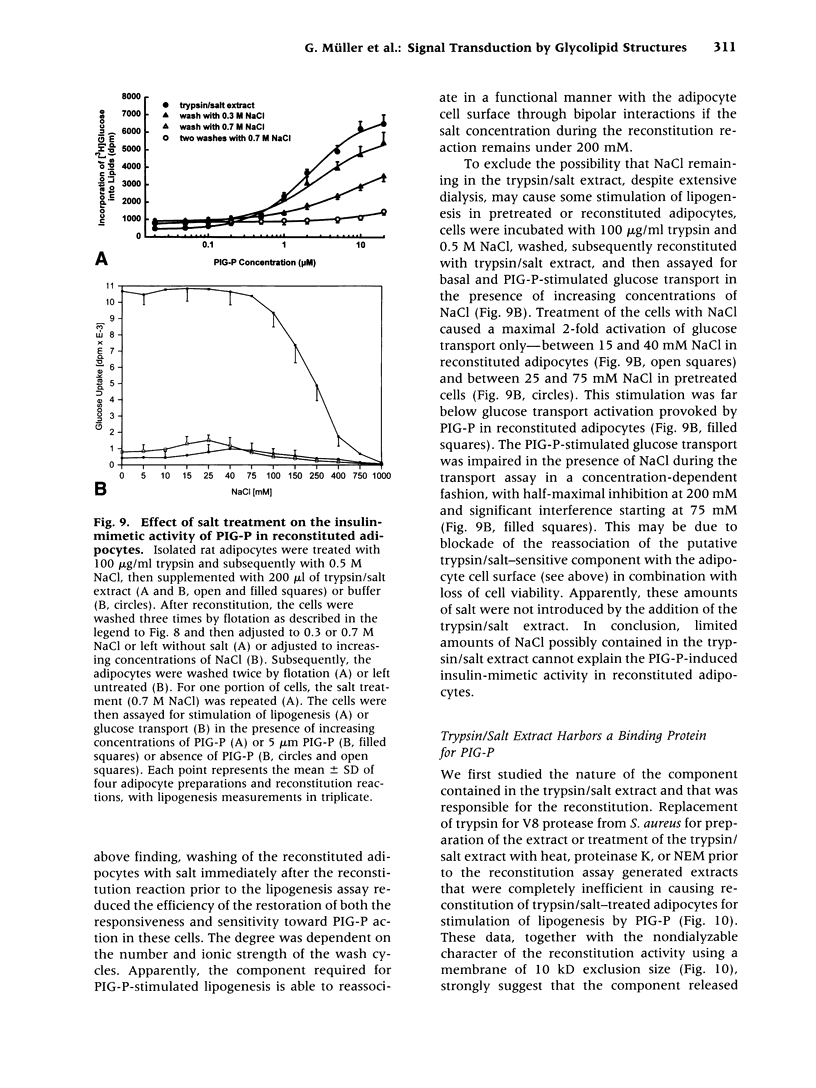
- Asplin I., Galasko G., Larner J. chiro-inositol deficiency and insulin resistance: a comparison of the chiro-inositol- and the myo-inositol-containing insulin mediators isolated from urine, hemodialysate, and muscle of control and type II diabetic subjects. Proc Natl Acad Sci U S A. 1993 Jul 1;90(13):5924–5928. doi: 10.1073/pnas.90.13.5924. [DOI] [PMC free article] [PubMed] [Google Scholar]
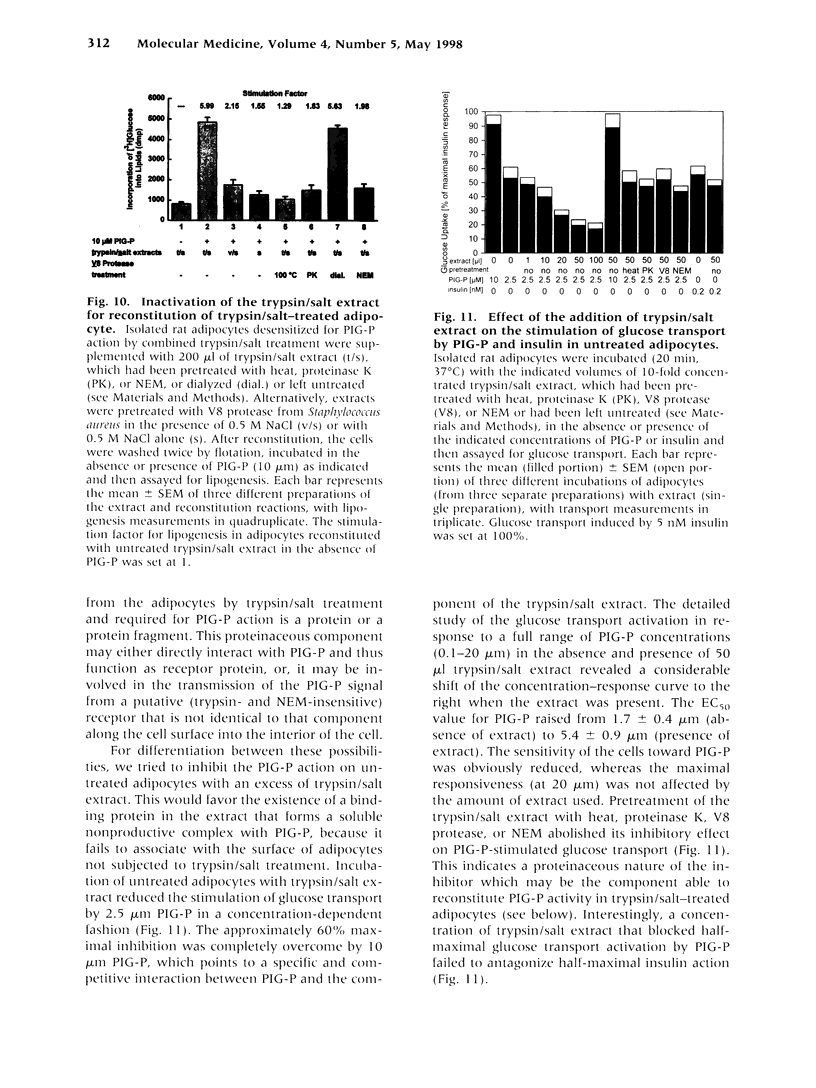
- Bickel P. E., Scherer P. E., Schnitzer J. E., Oh P., Lisanti M. P., Lodish H. F. Flotillin and epidermal surface antigen define a new family of caveolae-associated integral membrane proteins. J Biol Chem. 1997 May 23;272(21):13793–13802. doi: 10.1074/jbc.272.21.13793. [DOI] [PubMed] [Google Scholar]
- Brown D. A., Rose J. K. Sorting of GPI-anchored proteins to glycolipid-enriched membrane subdomains during transport to the apical cell surface. Cell. 1992 Feb 7;68(3):533–544. doi: 10.1016/0092-8674(92)90189-j. [DOI] [PubMed] [Google Scholar]
- Brown D. The tyrosine kinase connection: how GPI-anchored proteins activate T cells. Curr Opin Immunol. 1993 Jun;5(3):349–354. doi: 10.1016/0952-7915(93)90052-t. [DOI] [PubMed] [Google Scholar]
- Cheatham B., Vlahos C. J., Cheatham L., Wang L., Blenis J., Kahn C. R. Phosphatidylinositol 3-kinase activation is required for insulin stimulation of pp70 S6 kinase, DNA synthesis, and glucose transporter translocation. Mol Cell Biol. 1994 Jul;14(7):4902–4911. doi: 10.1128/mcb.14.7.4902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cross G. A. Glycolipid anchoring of plasma membrane proteins. Annu Rev Cell Biol. 1990;6:1–39. doi: 10.1146/annurev.cb.06.110190.000245. [DOI] [PubMed] [Google Scholar]
- Deeg M. A., Brass E. P., Rosenberry T. L. Inositol glycan phosphate derived from human erythrocyte acetylcholinesterase glycolipid anchor and inositol cyclic 1,2-phosphate antagonize glucagon activation of glycogen phosphorylase. Diabetes. 1993 Sep;42(9):1318–1323. doi: 10.2337/diab.42.9.1318. [DOI] [PubMed] [Google Scholar]
- Farese R. V. Lipid-derived mediators in insulin action. Proc Soc Exp Biol Med. 1990 Dec;195(3):312–324. doi: 10.3181/00379727-195-43150c. [DOI] [PubMed] [Google Scholar]
- Farese R. V., Standaert M. L., Yamada K., Huang L. C., Zhang C., Cooper D. R., Wang Z., Yang Y., Suzuki S., Toyota T. Insulin-induced activation of glycerol-3-phosphate acyltransferase by a chiro-inositol-containing insulin mediator is defective in adipocytes of insulin-resistant, type II diabetic, Goto-Kakizaki rats. Proc Natl Acad Sci U S A. 1994 Nov 8;91(23):11040–11044. doi: 10.1073/pnas.91.23.11040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fonteles M. C., Huang L. C., Larner J. Infusion of pH 2.0 D-chiro-inositol glycan insulin putative mediator normalizes plasma glucose in streptozotocin diabetic rats at a dose equivalent to insulin without inducing hypoglycaemia. Diabetologia. 1996 Jun;39(6):731–734. doi: 10.1007/BF00418546. [DOI] [PubMed] [Google Scholar]
- Gaulton G. N. Differential regulation of glycosylated phosphatidylinositol subtypes by insulin. Diabetes. 1991 Oct;40(10):1297–1304. doi: 10.2337/diab.40.10.1297. [DOI] [PubMed] [Google Scholar]
- Gaulton G. N., Pratt J. C. Glycosylated phosphatidylinositol molecules as second messengers. Semin Immunol. 1994 Apr;6(2):97–104. doi: 10.1006/smim.1994.1014. [DOI] [PubMed] [Google Scholar]
- Gustavsson J., Parpal S., Strålfors P. Insulin-stimulated glucose uptake involves the transition of glucose transporters to a caveolae-rich fraction within the plasma membrane: implications for type II diabetes. Mol Med. 1996 May;2(3):367–372. [PMC free article] [PubMed] [Google Scholar]
- Häring H. U. The insulin receptor: signalling mechanism and contribution to the pathogenesis of insulin resistance. Diabetologia. 1991 Dec;34(12):848–861. doi: 10.1007/BF00400192. [DOI] [PubMed] [Google Scholar]
- Johnston J. A., Wang L. M., Hanson E. P., Sun X. J., White M. F., Oakes S. A., Pierce J. H., O'Shea J. J. Interleukins 2, 4, 7, and 15 stimulate tyrosine phosphorylation of insulin receptor substrates 1 and 2 in T cells. Potential role of JAK kinases. J Biol Chem. 1995 Dec 1;270(48):28527–28530. doi: 10.1074/jbc.270.48.28527. [DOI] [PubMed] [Google Scholar]
- Kahn B. B. Lilly lecture 1995. Glucose transport: pivotal step in insulin action. Diabetes. 1996 Nov;45(11):1644–1654. doi: 10.2337/diab.45.11.1644. [DOI] [PubMed] [Google Scholar]
- Kahn C. R. Banting Lecture. Insulin action, diabetogenes, and the cause of type II diabetes. Diabetes. 1994 Aug;43(8):1066–1084. doi: 10.2337/diab.43.8.1066. [DOI] [PubMed] [Google Scholar]
- Kandror K. V., Stephens J. M., Pilch P. F. Expression and compartmentalization of caveolin in adipose cells: coordinate regulation with and structural segregation from GLUT4. J Cell Biol. 1995 May;129(4):999–1006. doi: 10.1083/jcb.129.4.999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelly K. L., Merida I., Wong E. H., DiCenzo D., Mato J. M. A phospho-oligosaccharide mimics the effect of insulin to inhibit isoproterenol-dependent phosphorylation of phospholipid methyltransferase in isolated adipocytes. J Biol Chem. 1987 Nov 5;262(31):15285–15290. [PubMed] [Google Scholar]
- Kessler A., Müller G., Wied S., Crecelius A., Eckel J. Signalling pathways of an insulin-mimetic phosphoinositolglycan-peptide in muscle and adipose tissue. Biochem J. 1998 Feb 15;330(Pt 1):277–286. doi: 10.1042/bj3300277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kowalski-Chauvel A., Pradayrol L., Vaysse N., Seva C. Gastrin stimulates tyrosine phosphorylation of insulin receptor substrate 1 and its association with Grb2 and the phosphatidylinositol 3-kinase. J Biol Chem. 1996 Oct 18;271(42):26356–26361. doi: 10.1074/jbc.271.42.26356. [DOI] [PubMed] [Google Scholar]
- Kuo C. J., Chung J., Fiorentino D. F., Flanagan W. M., Blenis J., Crabtree G. R. Rapamycin selectively inhibits interleukin-2 activation of p70 S6 kinase. Nature. 1992 Jul 2;358(6381):70–73. doi: 10.1038/358070a0. [DOI] [PubMed] [Google Scholar]
- Kurzchalia T. V., Dupree P., Monier S. VIP21-Caveolin, a protein of the trans-Golgi network and caveolae. FEBS Lett. 1994 Jun 6;346(1):88–91. doi: 10.1016/0014-5793(94)00466-8. [DOI] [PubMed] [Google Scholar]
- Larner J. Insulin-signaling mechanisms. Lessons from the old testament of glycogen metabolism and the new testament of molecular biology. Diabetes. 1988 Mar;37(3):262–275. doi: 10.2337/diab.37.3.262. [DOI] [PubMed] [Google Scholar]
- Lazar D. F., Knez J. J., Medof M. E., Cuatrecasas P., Saltiel A. R. Stimulation of glycogen synthesis by insulin in human erythroleukemia cells requires the synthesis of glycosyl-phosphatidylinositol. Proc Natl Acad Sci U S A. 1994 Oct 11;91(21):9665–9669. doi: 10.1073/pnas.91.21.9665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lazar D. F., Wiese R. J., Brady M. J., Mastick C. C., Waters S. B., Yamauchi K., Pessin J. E., Cuatrecasas P., Saltiel A. R. Mitogen-activated protein kinase kinase inhibition does not block the stimulation of glucose utilization by insulin. J Biol Chem. 1995 Sep 1;270(35):20801–20807. doi: 10.1074/jbc.270.35.20801. [DOI] [PubMed] [Google Scholar]
- Lisanti M. P., Scherer P. E., Tang Z., Sargiacomo M. Caveolae, caveolin and caveolin-rich membrane domains: a signalling hypothesis. Trends Cell Biol. 1994 Jul;4(7):231–235. doi: 10.1016/0962-8924(94)90114-7. [DOI] [PubMed] [Google Scholar]
- Liscovitch M., Cantley L. C. Signal transduction and membrane traffic: the PITP/phosphoinositide connection. Cell. 1995 Jun 2;81(5):659–662. doi: 10.1016/0092-8674(95)90525-1. [DOI] [PubMed] [Google Scholar]
- Low M. G., Saltiel A. R. Structural and functional roles of glycosyl-phosphatidylinositol in membranes. Science. 1988 Jan 15;239(4837):268–275. doi: 10.1126/science.3276003. [DOI] [PubMed] [Google Scholar]
- Macaulay S. L., Larkins R. G. Phospholipase C mimics insulin action on pyruvate dehydrogenase and insulin mediator generation but not glucose transport or utilization. Cell Signal. 1990;2(1):9–19. doi: 10.1016/0898-6568(90)90028-9. [DOI] [PubMed] [Google Scholar]
- Marte B. M., Downward J. PKB/Akt: connecting phosphoinositide 3-kinase to cell survival and beyond. Trends Biochem Sci. 1997 Sep;22(9):355–358. doi: 10.1016/s0968-0004(97)01097-9. [DOI] [PubMed] [Google Scholar]
- Mastick C. C., Brady M. J., Saltiel A. R. Insulin stimulates the tyrosine phosphorylation of caveolin. J Cell Biol. 1995 Jun;129(6):1523–1531. doi: 10.1083/jcb.129.6.1523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mato J. M. Insulin mediators revisited. Cell Signal. 1989;1(2):143–146. doi: 10.1016/0898-6568(89)90003-x. [DOI] [PubMed] [Google Scholar]
- Mato J. M., Kelly K. L., Abler A., Jarett L., Corkey B. E., Cashel J. A., Zopf D. Partial structure of an insulin-sensitive glycophospholipid. Biochem Biophys Res Commun. 1987 Jul 31;146(2):764–770. doi: 10.1016/0006-291x(87)90595-x. [DOI] [PubMed] [Google Scholar]
- Mato J. M., Kelly K. L., Abler A., Jarett L. Identification of a novel insulin-sensitive glycophospholipid from H35 hepatoma cells. J Biol Chem. 1987 Feb 15;262(5):2131–2137. [PubMed] [Google Scholar]
- McConville M. J., Ferguson M. A. The structure, biosynthesis and function of glycosylated phosphatidylinositols in the parasitic protozoa and higher eukaryotes. Biochem J. 1993 Sep 1;294(Pt 2):305–324. doi: 10.1042/bj2940305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Misek D. E., Saltiel A. R. An inositol phosphate glycan derived from a Trypanosoma brucei glycosyl-phosphatidylinositol mimics some of the metabolic actions of insulin. J Biol Chem. 1992 Aug 15;267(23):16266–16273. [PubMed] [Google Scholar]
- Movahedi S., Hooper N. M. Insulin stimulates the release of the glycosyl phosphatidylinositol-anchored membrane dipeptidase from 3T3-L1 adipocytes through the action of a phospholipase C. Biochem J. 1997 Sep 1;326(Pt 2):531–537. doi: 10.1042/bj3260531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muñoz P., Mora S., Sevilla L., Kaliman P., Tomàs E., Gumà A., Testar X., Palacín M., Zorzano A. Expression and insulin-regulated distribution of caveolin in skeletal muscle. Caveolin does not colocalize with GLUT4 in intracellular membranes. J Biol Chem. 1996 Apr 5;271(14):8133–8139. doi: 10.1074/jbc.271.14.8133. [DOI] [PubMed] [Google Scholar]
- Myers M. G., Jr, Wang L. M., Sun X. J., Zhang Y., Yenush L., Schlessinger J., Pierce J. H., White M. F. Role of IRS-1-GRB-2 complexes in insulin signaling. Mol Cell Biol. 1994 Jun;14(6):3577–3587. doi: 10.1128/mcb.14.6.3577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Müller G., Dearey E. A., Korndörfer A., Bandlow W. Stimulation of a glycosyl-phosphatidylinositol-specific phospholipase by insulin and the sulfonylurea, glimepiride, in rat adipocytes depends on increased glucose transport. J Cell Biol. 1994 Sep;126(5):1267–1276. doi: 10.1083/jcb.126.5.1267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Müller G., Dearey E. A., Pünter J. The sulphonylurea drug, glimepiride, stimulates release of glycosylphosphatidylinositol-anchored plasma-membrane proteins from 3T3 adipocytes. Biochem J. 1993 Jan 15;289(Pt 2):509–521. doi: 10.1042/bj2890509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Müller G., Ertl J., Gerl M., Preibisch G. Leptin impairs metabolic actions of insulin in isolated rat adipocytes. J Biol Chem. 1997 Apr 18;272(16):10585–10593. doi: 10.1074/jbc.272.16.10585. [DOI] [PubMed] [Google Scholar]
- Müller G., Geisen K. Characterization of the molecular mode of action of the sulfonylurea, glimepiride, at adipocytes. Horm Metab Res. 1996 Sep;28(9):469–487. doi: 10.1055/s-2007-979839. [DOI] [PubMed] [Google Scholar]
- Müller G., Jordan H., Petry S., Wetekam E. M., Schindler P. Analysis of lipid metabolism in adipocytes using a fluorescent fatty acid derivative. I. Insulin stimulation of lipogenesis. Biochim Biophys Acta. 1997 Jul 12;1347(1):23–39. doi: 10.1016/s0005-2760(97)00044-1. [DOI] [PubMed] [Google Scholar]
- Müller G., Schubert K., Fiedler F., Bandlow W. The cAMP-binding ectoprotein from Saccharomyces cerevisiae is membrane-anchored by glycosyl-phosphatidylinositol. J Biol Chem. 1992 Dec 15;267(35):25337–25346. [PubMed] [Google Scholar]
- Müller G., Wied S., Crecelius A., Kessler A., Eckel J. Phosphoinositolglycan-peptides from yeast potently induce metabolic insulin actions in isolated rat adipocytes, cardiomyocytes, and diaphragms. Endocrinology. 1997 Aug;138(8):3459–3475. doi: 10.1210/endo.138.8.5308. [DOI] [PubMed] [Google Scholar]
- Müller G., Wied S. The sulfonylurea drug, glimepiride, stimulates glucose transport, glucose transporter translocation, and dephosphorylation in insulin-resistant rat adipocytes in vitro. Diabetes. 1993 Dec;42(12):1852–1867. doi: 10.2337/diab.42.12.1852. [DOI] [PubMed] [Google Scholar]
- Müller G., Wied S., Wetekam E. M., Crecelius A., Unkelbach A., Pünter J. Stimulation of glucose utilization in 3T3 adipocytes and rat diaphragm in vitro by the sulphonylureas, glimepiride and glibenclamide, is correlated with modulations of the cAMP regulatory cascade. Biochem Pharmacol. 1994 Aug 30;48(5):985–996. doi: 10.1016/0006-2952(94)90369-7. [DOI] [PubMed] [Google Scholar]
- Müller G., Zimmermann R. Import of honeybee prepromelittin into the endoplasmic reticulum: structural basis for independence of SRP and docking protein. EMBO J. 1987 Jul;6(7):2099–2107. doi: 10.1002/j.1460-2075.1987.tb02476.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nosjean O., Briolay A., Roux B. Mammalian GPI proteins: sorting, membrane residence and functions. Biochim Biophys Acta. 1997 Sep 8;1331(2):153–186. doi: 10.1016/s0304-4157(97)00005-1. [DOI] [PubMed] [Google Scholar]
- Okada T., Kawano Y., Sakakibara T., Hazeki O., Ui M. Essential role of phosphatidylinositol 3-kinase in insulin-induced glucose transport and antilipolysis in rat adipocytes. Studies with a selective inhibitor wortmannin. J Biol Chem. 1994 Feb 4;269(5):3568–3573. [PubMed] [Google Scholar]
- Parpal S., Gustavsson J., Strålfors P. Isolation of phosphooligosaccharide/phosphoinositol glycan from caveolae and cytosol of insulin-stimulated cells. J Cell Biol. 1995 Oct;131(1):125–135. doi: 10.1083/jcb.131.1.125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parton R. G. Caveolae and caveolins. Curr Opin Cell Biol. 1996 Aug;8(4):542–548. doi: 10.1016/s0955-0674(96)80033-0. [DOI] [PubMed] [Google Scholar]
- Robbins S. M., Quintrell N. A., Bishop J. M. Myristoylation and differential palmitoylation of the HCK protein-tyrosine kinases govern their attachment to membranes and association with caveolae. Mol Cell Biol. 1995 Jul;15(7):3507–3515. doi: 10.1128/mcb.15.7.3507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodgers W., Crise B., Rose J. K. Signals determining protein tyrosine kinase and glycosyl-phosphatidylinositol-anchored protein targeting to a glycolipid-enriched membrane fraction. Mol Cell Biol. 1994 Aug;14(8):5384–5391. doi: 10.1128/mcb.14.8.5384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Romero G., Gámez G., Huang L. C., Lilley K., Luttrell L. Anti-inositolglycan antibodies selectively block some of the actions of insulin in intact BC3H1 cells. Proc Natl Acad Sci U S A. 1990 Feb;87(4):1476–1480. doi: 10.1073/pnas.87.4.1476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Romero G., Larner J. Insulin mediators and the mechanism of insulin action. Adv Pharmacol. 1993;24:21–50. doi: 10.1016/s1054-3589(08)60932-1. [DOI] [PubMed] [Google Scholar]
- Romero G., Luttrell L., Rogol A., Zeller K., Hewlett E., Larner J. Phosphatidylinositol-glycan anchors of membrane proteins: potential precursors of insulin mediators. Science. 1988 Apr 22;240(4851):509–511. doi: 10.1126/science.3282305. [DOI] [PubMed] [Google Scholar]
- Rothberg K. G., Heuser J. E., Donzell W. C., Ying Y. S., Glenney J. R., Anderson R. G. Caveolin, a protein component of caveolae membrane coats. Cell. 1992 Feb 21;68(4):673–682. doi: 10.1016/0092-8674(92)90143-z. [DOI] [PubMed] [Google Scholar]
- Saad M. J., Velloso L. A., Carvalho C. R. Angiotensin II induces tyrosine phosphorylation of insulin receptor substrate 1 and its association with phosphatidylinositol 3-kinase in rat heart. Biochem J. 1995 Sep 15;310(Pt 3):741–744. doi: 10.1042/bj3100741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saltiel A. R., Cuatrecasas P. Insulin stimulates the generation from hepatic plasma membranes of modulators derived from an inositol glycolipid. Proc Natl Acad Sci U S A. 1986 Aug;83(16):5793–5797. doi: 10.1073/pnas.83.16.5793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saltiel A. R., Fox J. A., Sherline P., Cuatrecasas P. Insulin-stimulated hydrolysis of a novel glycolipid generates modulators of cAMP phosphodiesterase. Science. 1986 Aug 29;233(4767):967–972. doi: 10.1126/science.3016898. [DOI] [PubMed] [Google Scholar]
- Saltiel A. R. Insulin generates an enzyme modulator from hepatic plasma membranes: regulation of adenosine 3',5'-monophosphate phosphodiesterase, pyruvate dehydrogenase, and adenylate cyclase. Endocrinology. 1987 Mar;120(3):967–972. doi: 10.1210/endo-120-3-967. [DOI] [PubMed] [Google Scholar]
- Saltiel A. R., Osterman D. G., Darnell J. C., Sorbara-Cazan L. R., Chan B. L., Low M. G., Cuatrecasas P. The function of glycosyl phosphoinositides in hormone action. Philos Trans R Soc Lond B Biol Sci. 1988 Jul 26;320(1199):345–358. doi: 10.1098/rstb.1988.0081. [DOI] [PubMed] [Google Scholar]
- Saltiel A. R. Second messengers of insulin action. Diabetes Care. 1990 Mar;13(3):244–256. doi: 10.2337/diacare.13.3.244. [DOI] [PubMed] [Google Scholar]
- Saltiel A. R., Sorbara-Cazan L. R. Inositol glycan mimics the action of insulin on glucose utilization in rat adipocytes. Biochem Biophys Res Commun. 1987 Dec 31;149(3):1084–1092. doi: 10.1016/0006-291x(87)90519-5. [DOI] [PubMed] [Google Scholar]
- Sargiacomo M., Sudol M., Tang Z., Lisanti M. P. Signal transducing molecules and glycosyl-phosphatidylinositol-linked proteins form a caveolin-rich insoluble complex in MDCK cells. J Cell Biol. 1993 Aug;122(4):789–807. doi: 10.1083/jcb.122.4.789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scherer P. E., Lisanti M. P., Baldini G., Sargiacomo M., Mastick C. C., Lodish H. F. Induction of caveolin during adipogenesis and association of GLUT4 with caveolin-rich vesicles. J Cell Biol. 1994 Dec;127(5):1233–1243. doi: 10.1083/jcb.127.5.1233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sekar N., Li J., Shechter Y. Vanadium salts as insulin substitutes: mechanisms of action, a scientific and therapeutic tool in diabetes mellitus research. Crit Rev Biochem Mol Biol. 1996 Dec;31(5-6):339–359. doi: 10.3109/10409239609108721. [DOI] [PubMed] [Google Scholar]
- Shashkin P. N., Shashkina E. F., Fernqvist-Forbes E., Zhou Y. P., Grill V., Katz A. Insulin mediators in man: effects of glucose ingestion and insulin resistance. Diabetologia. 1997 May;40(5):557–563. doi: 10.1007/s001250050715. [DOI] [PubMed] [Google Scholar]
- Shenoy-Scaria A. M., Dietzen D. J., Kwong J., Link D. C., Lublin D. M. Cysteine3 of Src family protein tyrosine kinase determines palmitoylation and localization in caveolae. J Cell Biol. 1994 Jul;126(2):353–363. doi: 10.1083/jcb.126.2.353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song K. S., Scherer P. E., Tang Z., Okamoto T., Li S., Chafel M., Chu C., Kohtz D. S., Lisanti M. P. Expression of caveolin-3 in skeletal, cardiac, and smooth muscle cells. Caveolin-3 is a component of the sarcolemma and co-fractionates with dystrophin and dystrophin-associated glycoproteins. J Biol Chem. 1996 Jun 21;271(25):15160–15165. doi: 10.1074/jbc.271.25.15160. [DOI] [PubMed] [Google Scholar]
- Stefanová I., Horejsí V., Ansotegui I. J., Knapp W., Stockinger H. GPI-anchored cell-surface molecules complexed to protein tyrosine kinases. Science. 1991 Nov 15;254(5034):1016–1019. doi: 10.1126/science.1719635. [DOI] [PubMed] [Google Scholar]
- Su B., Waneck G. L., Flavell R. A., Bothwell A. L. The glycosyl phosphatidylinositol anchor is critical for Ly-6A/E-mediated T cell activation. J Cell Biol. 1991 Feb;112(3):377–384. doi: 10.1083/jcb.112.3.377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki S., Sugawara K., Satoh Y., Toyota T. Insulin stimulates the generation of two putative insulin mediators, inositol-glycan and diacylglycerol in BC3H-1 myocytes. J Biol Chem. 1991 May 5;266(13):8115–8121. [PubMed] [Google Scholar]
- Tachado S. D., Schofield L. Glycosylphosphatidylinositol toxin of Trypanosoma brucei regulates IL-1 alpha and TNF-alpha expression in macrophages by protein tyrosine kinase mediated signal transduction. Biochem Biophys Res Commun. 1994 Dec 15;205(2):984–991. doi: 10.1006/bbrc.1994.2763. [DOI] [PubMed] [Google Scholar]
- Thomas P. M., Samelson L. E. The glycophosphatidylinositol-anchored Thy-1 molecule interacts with the p60fyn protein tyrosine kinase in T cells. J Biol Chem. 1992 Jun 15;267(17):12317–12322. [PubMed] [Google Scholar]
- Uddin S., Yenush L., Sun X. J., Sweet M. E., White M. F., Platanias L. C. Interferon-alpha engages the insulin receptor substrate-1 to associate with the phosphatidylinositol 3'-kinase. J Biol Chem. 1995 Jul 7;270(27):15938–15941. doi: 10.1074/jbc.270.27.15938. [DOI] [PubMed] [Google Scholar]
- Varela-Nieto I., León Y., Caro H. N. Cell signalling by inositol phosphoglycans from different species. Comp Biochem Physiol B Biochem Mol Biol. 1996 Oct;115(2):223–241. doi: 10.1016/0305-0491(96)00087-9. [DOI] [PubMed] [Google Scholar]
- Velloso L. A., Folli F., Sun X. J., White M. F., Saad M. J., Kahn C. R. Cross-talk between the insulin and angiotensin signaling systems. Proc Natl Acad Sci U S A. 1996 Oct 29;93(22):12490–12495. doi: 10.1073/pnas.93.22.12490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vila M. C., Milligan G., Standaert M. L., Farese R. V. Insulin activates glycerol-3-phosphate acyltransferase (de novo phosphatidic acid synthesis) through a phospholipid-derived mediator. Apparent involvement of Gi alpha and activation of a phospholipase C. Biochemistry. 1990 Sep 18;29(37):8735–8740. doi: 10.1021/bi00489a033. [DOI] [PubMed] [Google Scholar]
- Ying Y. S., Anderson R. G., Rothberg K. G. Each caveola contains multiple glycosyl-phosphatidylinositol-anchored membrane proteins. Cold Spring Harb Symp Quant Biol. 1992;57:593–604. doi: 10.1101/sqb.1992.057.01.065. [DOI] [PubMed] [Google Scholar]