Abstract
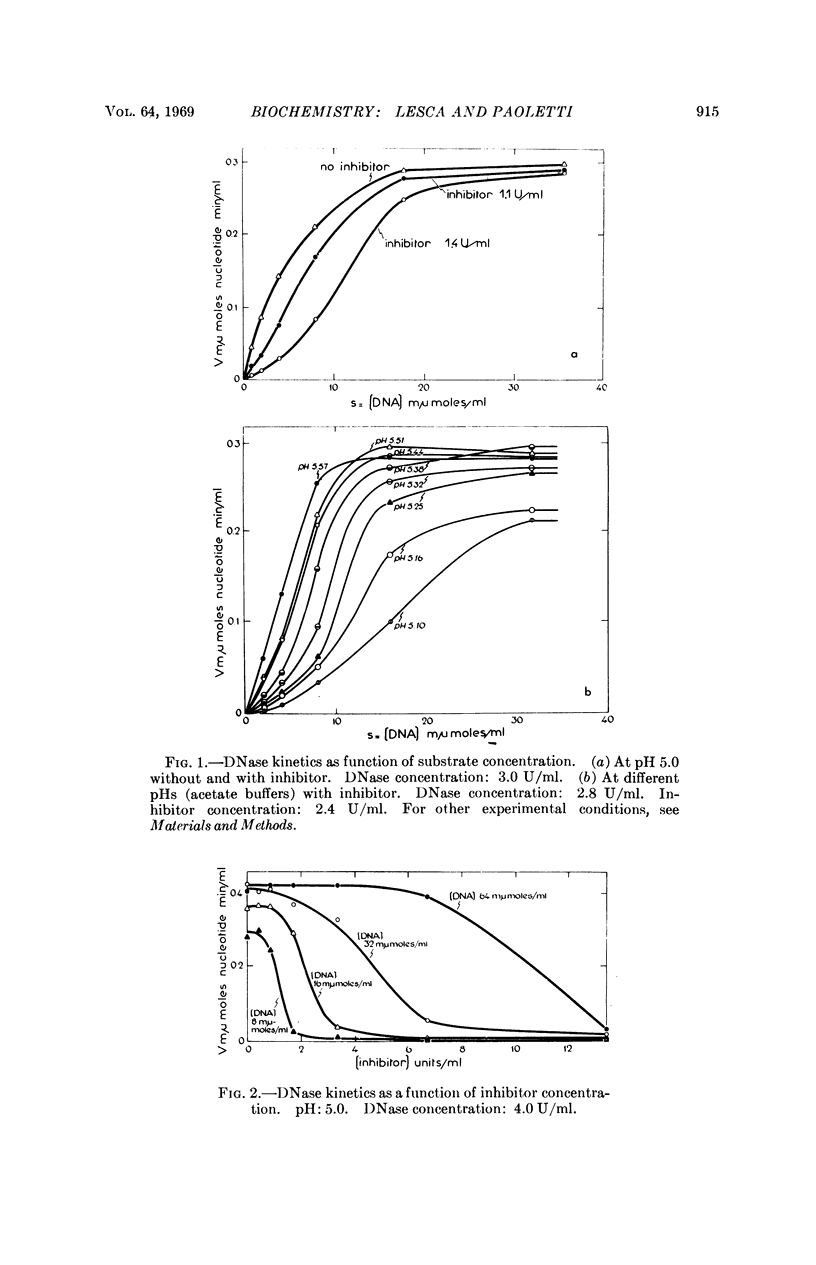
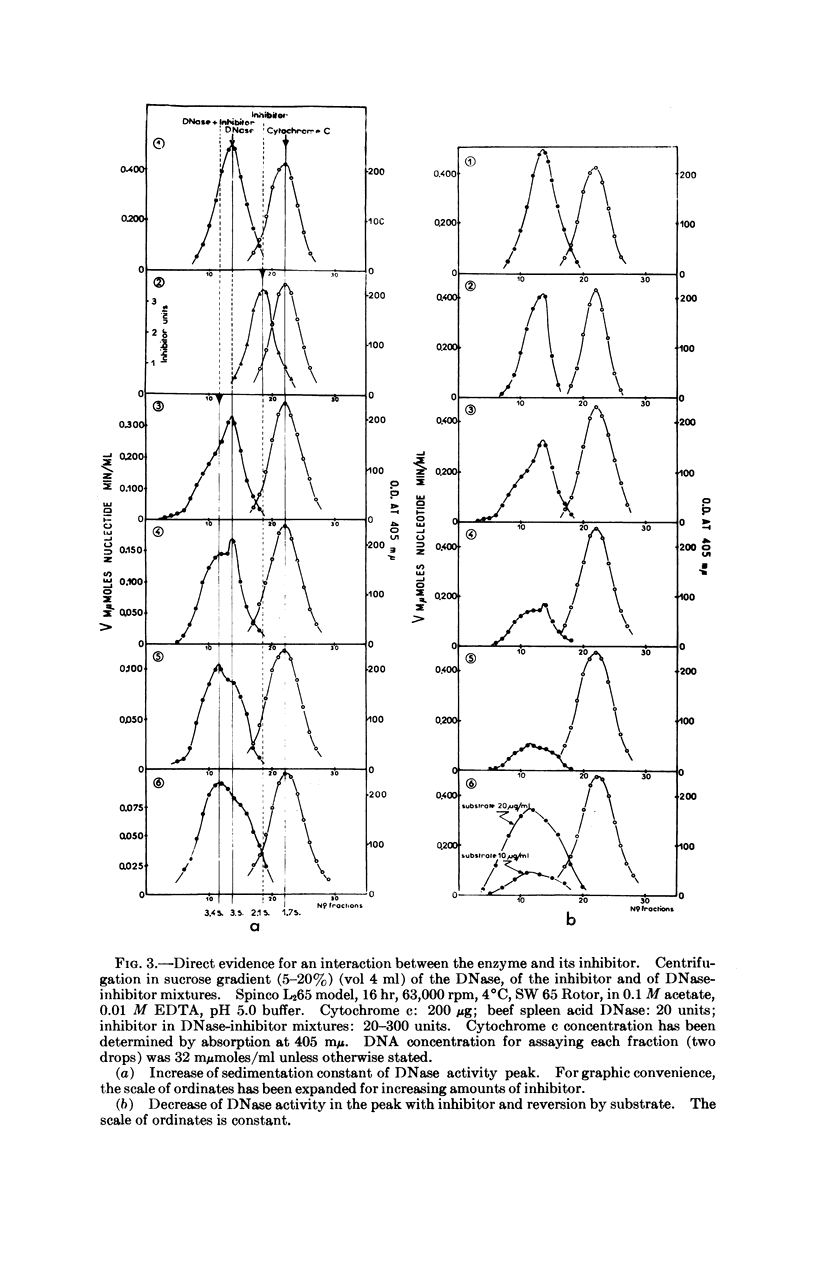
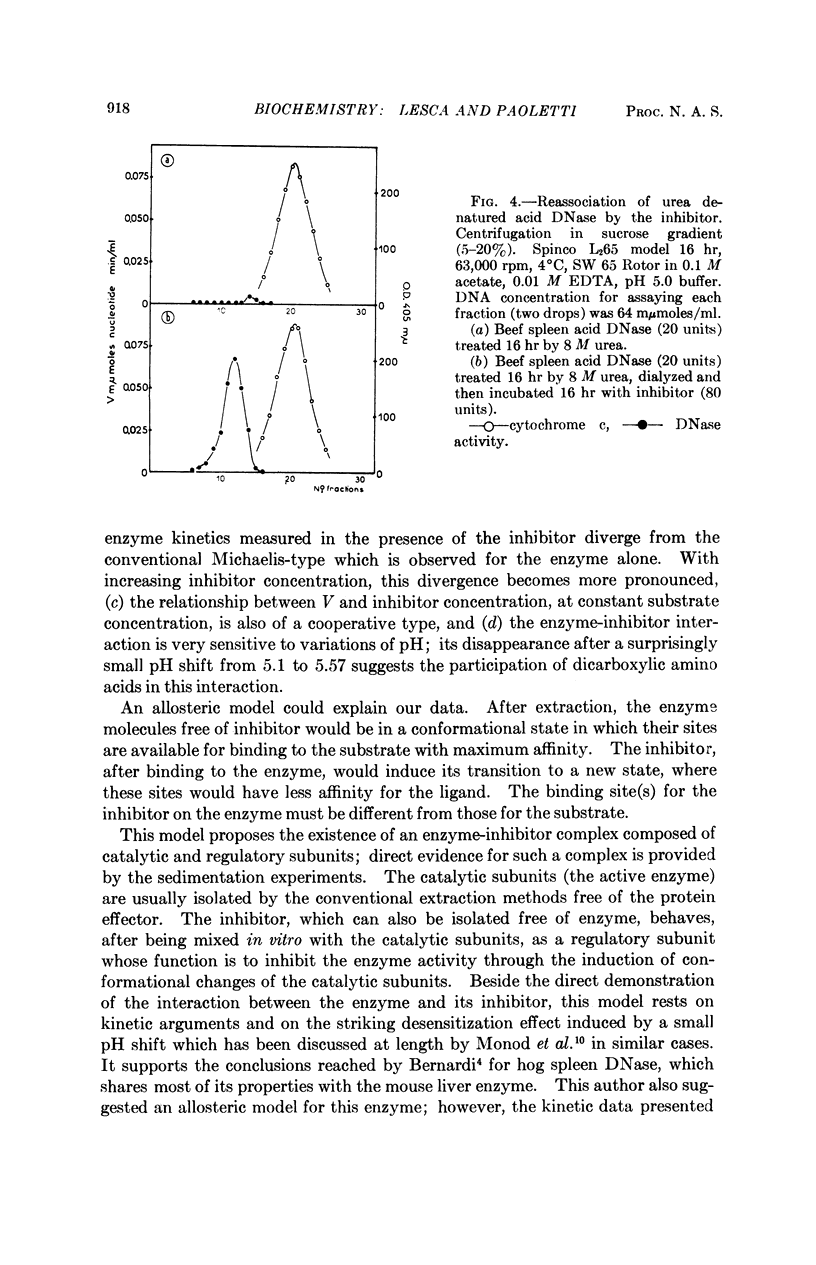
A protein extracted and partially purified (about 100-fold) from mouse liver is able to inhibit the acid DNases from different tissues and species, whereas pancreatic DNase and E. coli endonuclease I are not inhibited. The acid DNase displays typical Michaelis-Menten kinetics in the absence of this inhibitor, but the kinetics become sigmoidal in its presence. The existence of a DNase-inhibitor complex is demonstrated by physicochemical experiments. Moreover, the inhibitor is able to reactivate the DNase treated by urea, probably through a reassociation of the inactive monomers to a dimeric state. An allosteric model in which the DNase-inhibitor complex is composed of catalytic (DNase) and regulatory (inhibitor) subunits could explain these data.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bernardi G. Dimeric structure and allosteric properties of spleen acid deoxyribonuclease. J Mol Biol. 1965 Sep;13(2):603–605. doi: 10.1016/s0022-2836(65)80123-1. [DOI] [PubMed] [Google Scholar]
- Brew K., Vanaman T. C., Hill R. L. The role of alpha-lactalbumin and the A protein in lactose synthetase: a unique mechanism for the control of a biological reaction. Proc Natl Acad Sci U S A. 1968 Feb;59(2):491–497. doi: 10.1073/pnas.59.2.491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Changeux J. P., Gerhart J. C., Schachman H. K. Allosteric interactions in aspartate transcarbamylase. I. Binding of specific ligands to the native enzyme and its isolated subunits. Biochemistry. 1968 Feb;7(2):531–538. doi: 10.1021/bi00842a007. [DOI] [PubMed] [Google Scholar]
- Changeux J. P., Rubin M. M. Allosteric interactions in aspartate transcarbamylase. 3. Interpretation of experimental data in terms of the model of Monod, Wyman, and Changeux. Biochemistry. 1968 Feb;7(2):553–561. doi: 10.1021/bi00842a601. [DOI] [PubMed] [Google Scholar]
- FESTY B., PAOLETTI C. MISE EN 'EVIDENCE ET PROPRI'ET'ES D'UN (OU PLUSIEURS) INHIBITEUR(S) NATUREL(S) DE LA D'ESOXYRIBONUCL'EASE NEUTRE PANCR'EATIQUE. C R Hebd Seances Acad Sci. 1963 Dec 4;257:3682–3685. [PubMed] [Google Scholar]
- LEHMAN I. R., ROUSSOS G. G., PRATT E. A. The deoxyribonucleases of Escherichia coli. II. Purification and properties of a ribonucleic acid-inhibitable endonuclease. J Biol Chem. 1962 Mar;237:819–828. [PubMed] [Google Scholar]
- Laval J., Laval F., Paoletti C. Purification et propriétés d'une désoxyribonucléase acide d'hépato-pancréas d'escargot (Helix pomatia L.) C R Acad Sci Hebd Seances Acad Sci D. 1966 May 9;262(19):2093–2095. [PubMed] [Google Scholar]
- Lindberg M. U. Crystallization from calf spleen of two inhibitors of deoxyribonuclease I. J Biol Chem. 1966 Mar 10;241(5):1246–1249. [PubMed] [Google Scholar]
- Swenson M. K., Hodes M. E. The separation of the phosphodiesterase and deoxyribonuclease II activities of bovine spleen. J Biol Chem. 1969 Apr 10;244(7):1803–1807. [PubMed] [Google Scholar]


