Abstract
The baculovirus Autographa californica multiple nuclear polyhedrosis virus (AcMNPV) has been studied as a gene therapy vector. Here, we demonstrated that AcMNPV induces antitumor acquired immunity. These results suggest that AcMNPV has the potential to be an efficient virus or tumor therapy agent which induces innate and acquired immunity.
Autographa californica multiple nuclear polyhedrosis virus (AcMNPV) has a double-stranded circular DNA genome of approximately 130 kb that contains more than 150 open reading frames (4). The ability of AcMNPV to infect insect cells has led to its use in multiple protein expression systems (5, 16) and as plant insecticides (8, 21). AcMNPV also infects a variety of mammalian cell types, with the exception of certain hematopoietic cell lines, although its genome does not replicate or integrate into mammalian chromosomes (9, 11, 13, 17, 23).
Considerable attempts have been made to understand the nature of host immune responses to baculovirus. In vivo studies found that the virus induces antiviral cytokine production, which protects cells from infection with vesicular stomatitis virus and influenza virus (2, 10). Recently, Abe and coworkers demonstrated that AcMNPV is recognized by Toll-like receptor 9 on dendritic cells (DCs) and macrophages (1). More recently, we found that wild-type AcMNPV infects DCs and induces natural killer (NK) cell-dependent antimetastatic effects in mice (12). These results suggest that wild-type AcMNPV induces the activation of NK cells and has the potential for virus or tumor therapy. However, it is not yet known whether such antitumor effects are achieved by the induction of acquired immunity. The purpose of this study, therefore, was to examine wild-type AcMNPV-induced acquired immunity, such as the activation of tumor-specific cytotoxic lymphocytes (CTLs) and antibodies.
To investigate AcMNPV-induced acquired immunity in mice, we used two experimental tumor models: the liver metastasis model and the subcutaneous tumor model. Mice were injected intrasplenically with vehicle, mouse melanoma B16 cells, or hepatocellular carcinoma Hepa1-6 cells (n = 6 to 11) (Fig. 1A). The liver metastasis model was performed as previously described (12). Purified AcMNPV (1 × 108 PFU/mouse) was injected intravenously on day 1. After 2 weeks, mice received a secondary subcutaneous challenge with 2 × 105 B16 cells in 0.1 ml phosphate-buffered saline. Mice were monitored every 2 to 3 days for tumor growth, which was measured with calipers. The tumor volume (mm3) was calculated (18).
FIG. 1.
Acquired immune effects of AcMNPV injection in mouse B16 melanoma tumors. (A) Schematic of the experiment. Mice were injected intrasplenically on day −14 with vehicle or with 1 × 106 B16 or Hepa1-6 cells. AcMNPV (1 × 108 PFU/mouse) was injected intravenously on day −13. After 2 weeks, the mice received a subcutaneous secondary challenge with 2 × 105 B16 cells in 0.1 ml phosphate-buffered saline. Mice were monitored every 2 to 3 days for tumor growth (B). i.v., intravenous; i.s., intrasplenic; s.c., subcutaneous. Error bars indicate standard deviations.
Mice injected with vehicle alone, with AcMNPV alone, or with AcMNPV after intrasplenic delivery of vehicle or Hepa1-6 cells were unable to eradicate the secondary tumor challenge. By contrast, tumor growth was significantly retarded in mice injected with AcMNPV following intrasplenic treatment with B16 cells (Fig. 1B). We demonstrated that no AcMNPV-injected mice in the liver metastasis model survived for 30 days after intrasplenic B16 injection (12). This demonstrates that the AcMNPV-induced antimetastasis effects developed from systematic antitumor acquired immunity toward the first injection of tumor cells.
In acquired immunity, CTLs have a very important role (3, 14), and many studies have investigated their antitumor effects. Here, we examined whether tumor-specific CTL cytotoxicity was induced by AcMNPV treatment in B16 metastasis-inhibited mice. Mice were injected intrasplenically with vehicle, B16 cells, or Hepa1-6 cells and then with AcMNPV 1 day later (n = 8 to 9). After 14 days, lymphocytes from mouse intestinal lymph nodes were pooled and stimulated in vitro with B16 cells as previously described (18). Cytotoxic activity was assayed after 5 days of incubation. Target cells (0.5 to 1 × 104/well) were incubated with effector cells in RPMI 1640 containing 10% fetal calf serum (total volume, 100 μl) in 96-well round-bottom microtiter plates. Cytotoxicity was detected with the CytoTox 96 nonradioactive cytotoxicity assay kit (Promega Corp., Madison, WI). Spontaneous release from target cells was less than 15% of maximum release.
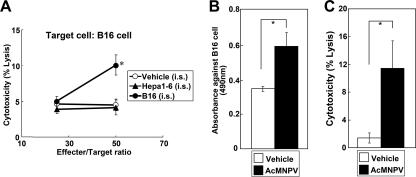
CTL cytotoxicity against B16 target cells was significantly increased in B16 cell-challenged mice (10.1% ± 1.41%) compared with mice challenged with vehicle or Hepa1-6 cells (4.13% ± 0.81% and 4.49% ± 1.05%, respectively) at an effector/target cell ratio of 50 (Fig. 2A), indicating that AcMNPV induces B16 cell-specific CTL activity. Data were analyzed using a two-tailed Student t test.
FIG. 2.
Effect of AcMNPV injection on CTL activity and specific antibody titer. (A) CTL activity against B16 melanoma cells in mice intrasplenically challenged with vehicle, Hepa1-6 cells, or B16 cells challenged and treated with AcMNPV. (B) B16-specific antibody titer in serum. (C) Antibody-mediated complement-dependent cytotoxicity in serum. Data represent means ± standard deviations. *, P < 0.05.
Mice were injected intrasplenically with vehicle or 1 × 106 B16 cells and then with AcMNPV 1 day later. After 14 days, serum was harvested and stored at −80°C. Immunodetection using an antibody against B16 cells was performed as previously described (19). The measured B16 cell-specific absorbance of serum from AcMNPV-injected mice was significantly increased compared with that of serum from vehicle-injected mice (AcMNPV, 11.5% ± 3.9%; vehicle, 1.41% ± 0.74%) (Fig. 2B), indicating that AcMNPV increased the antibody titer against challenged tumor cells. Next, we determined whether the serum antibodies could mediate cytotoxicity against tumor cells. B16 cell-specific antibody-mediated complement-dependent cytotoxicity was determined as previously described (6). Cytotoxicity was detected with the CytoTox 96 nonradioactive cytotoxicity assay kit. B16 cell-specific cytotoxicity from the sera of AcMNPV-treated mice was significantly increased compared with that from sera of vehicle-injected mice (AcMNPV, 11.5% ± 3.9%; vehicle, 1.41% ± 0.74%) (Fig. 2C), suggesting that AcMNPV induced effective antitumor antibodies. Data were analyzed using a two-tailed Student t test.
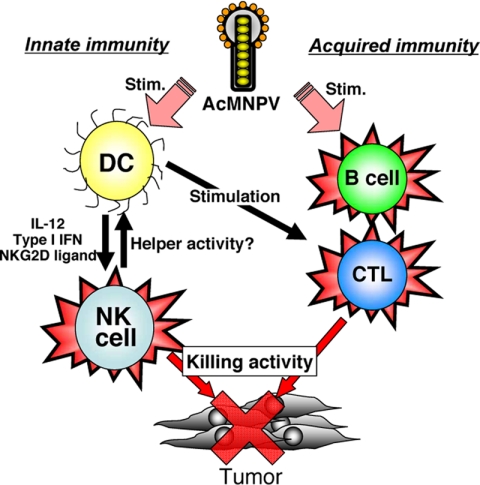
Figure 3 shows a hypothetical model of AcMNPV-induced antitumor mechanisms in mice. Initially, AcMNPV induces NK cell-dependent killing activity against tumor cells through the activation of cytokines and NKG2D ligands (12). In acquired immunity, tumor-specific CTLs and antibodies are induced. Thus, these results suggest that AcMNPV induces strong antitumor effects in both innate and acquired immunity.
FIG. 3.
Hypothetical AcMNPV-induced antitumor mechanisms in mice. AcMNPV stimulates antigen-presenting cells such as DCs and B cells. In innate immunity, cytokines and NKG2D ligands provide NK cell killing activity against tumor cells. In acquired immunity, tumor-specific CTLs and antibodies are induced.
Most viruses currently employed for gene therapy can utilize humans as a host, and their replication can lead to pathogenesis and toxicity. By contrast, baculoviruses do not replicate in humans (23), instead using insects as hosts. AcMNPV has been registered as a type of insecticide with the U.S. Environmental Protection Agency (8, 22) and is used clinically in multiple protein expression systems (4, 7). Recently, it was shown that injection of B6 mice with AcMNPV did not lead to activation of hepatic enzymes, such as plasma alanine aminotransferase or aspartate aminotransferase (12). Although these observations suggest that baculovirus is potentially a safer option for applications in humans, this remains to be confirmed, and toxicity in animals must be examined prior to its use in clinical trials.
In conclusion, we have shown that wild-type AcMNPV induces antitumor acquired immunity against challenged tumor cells in the form of increased CTL activity and tumor-specific antibody production. This suggests that it could be a useful and efficient antitumor agent which induces antitumor innate and acquired immunity.
Acknowledgments
We thank Y. Shoji and Y. Tsuyama for secretarial work.
This work was supported, in part, by a Grant-in-Aid for High Technology Research (no. 09309011) from the Ministry of Education, Science, Sports, and Culture, Japan, and by a grant from the Research and Development Program for New Bio-industry Initiatives.
Footnotes
Published ahead of print on 5 December 2007.
REFERENCES
- 1.Abe, T., H. Hemmi, H. Miyamoto, K. Moriishi, S. Tamura, H. Takaku, S. Akira, and Y. Matsuura. 2005. Involvement of the Toll-like receptor 9 signaling pathway in the induction of innate immunity by baculovirus. J. Virol. 79:2847-2858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Abe, T., H. Takahashi, H. Hamazaki, N. Miyano-Kurosaki, Y. Matsuura, and H. Takaku. 2003. Baculovirus induces an innate immune response and confers protection from lethal influenza virus infection in mice. J. Immunol. 171:1133-1139. [DOI] [PubMed] [Google Scholar]
- 3.Andersen, M. H., D. Schrama, P. Thor Straten, and J. C. Becker. 2006. Cytotoxic T cells. J. Investig. Dermatol. 126:32-41. [DOI] [PubMed] [Google Scholar]
- 4.Ayres, M. D., S. C. Howard, J. Kuzio, M. Lopez-Ferber, and R. D. Possee. 1994. The complete DNA sequence of Autographa californica nuclear polyhedrosis virus. Virology 202:586-605. [DOI] [PubMed] [Google Scholar]
- 5.Berger, I., D. J. Fitzgerald, and T. J. Richmond. 2004. Baculovirus expression system for heterologous multiprotein complexes. Nat. Biotechnol. 22:1583-1587. [DOI] [PubMed] [Google Scholar]
- 6.Chen, H. W., Y. P. Lee, Y. F. Chung, Y. C. Shih, J. P. Tsai, M. H. Tao, and C. C. Ting. 2003. Inducing long-term survival with lasting anti-tumor immunity in treating B cell lymphoma by a combined dendritic cell-based and hydrodynamic plasmid-encoding IL-12 gene therapy. Int. Immunol. 15:427-435. [DOI] [PubMed] [Google Scholar]
- 7.Cheng, T., C. Y. Xu, Y. B. Wang, M. Chen, T. Wu, J. Zhang, and N. S. Xia. 2004. A rapid and efficient method to express target genes in mammalian cells by baculovirus. World J. Gastroenterol. 10:1612-1618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cory, J. S., M. L. Hirst, T. Williams, R. S. Hails, D. Goulson, B. M. Green, T. M. Carty, R. D. Possee, P. J. Cayley, and D. H. L. Bishop. 1994. Field trial of a genetically improved baculovirus insecticide. Nature 370:138-140. [Google Scholar]
- 9.Fipaldini, C., B. Bellei, and N. La Monica. 1999. Expression of hepatitis C virus cDNA in human hepatoma cell line mediated by a hybrid baculovirus-HCV vector. Virology 255:302-311. [DOI] [PubMed] [Google Scholar]
- 10.Gronowski, A. M., D. M. Hilbert, K. C. Sheehan, G. Garotta, and R. D. Schreiber. 1999. Baculovirus stimulates antiviral effects in mammalian cells. J. Virol. 73:9944-9951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hofmann, C., V. Sandig, G. Jennings, M. Rudolph, P. Schlag, and M. Strauss. 1995. Efficient gene transfer into human hepatocytes by baculovirus vectors. Proc. Natl. Acad. Sci. USA 92:10099-10103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kitajima, M., T. Abe, N. Miyano-Kurosaki, M. Taniguchi, T. Nakayama, and H. Takaku. Baculovirus Autographa caliphornica multiple nuclear polyhedrosis virus-induced natural killer-dependent antitumor immunity. Mol. Ther., in press. [DOI] [PubMed]
- 13.Kitajima, M., H. Hamazaki, N. Miyano-Kurosaki, and H. Takaku. 2006. Characterization of baculovirus Autographa californica multiple nuclear polyhedrosis virus infection in mammalian cells. Biochem. Biophys. Res. Commun. 343:378-384. [DOI] [PubMed] [Google Scholar]
- 14.Liu, S. H., M. Zhang, and W. G. Zhang. 2005. Strategies of antigen-specific T-cell-based immunotherapy for cancer. Cancer Biother. Radiopharm. 20:491-501. [DOI] [PubMed] [Google Scholar]
- 15.Reference deleted.
- 16.Matsuura, Y., R. D. Possee, H. A. Overton, and D. H. Bishop. 1987. Baculovirus expression vectors: the requirements for high level expression of proteins, including glycoproteins. J. Gen. Virol. 68:1233-1250. [DOI] [PubMed] [Google Scholar]
- 17.McCormick, C. J., D. J. Rowlands, and M. Harris. 2002. Efficient delivery and regulable expression of hepatitis C virus full-length and minigenome constructs in hepatocyte-derived cell lines using baculovirus vectors. J. Gen. Virol. 83:383-394. [DOI] [PubMed] [Google Scholar]
- 18.Okada, N., Y. Masunaga, Y. Okada, H. Mizuguchi, S. Iiyama, N. Mori, A. Sasaki, S. Nakagawa, T. Mayumi, T. Hayakawa, T. Fujita, and A. Yamamoto. 2003. Dendritic cells transduced with gp100 gene by RGD fiber-mutant adenovirus vectors are highly efficacious in generating anti-B16BL6 melanoma immunity in mice. Gene Ther. 10:1891-1902. [DOI] [PubMed] [Google Scholar]
- 19.Overwijk, W. W., D. S. Lee, D. R. Surman, K. R. Irvine, C. E. Touloukian, C. C. Chan, M. W. Carroll, B. Moss, S. A. Rosenberg, and N. P. Restifo. 1999. Vaccination with a recombinant vaccinia virus encoding a “self” antigen induces autoimmune vitiligo and tumor cell destruction in mice: requirement for CD4(+) T lymphocytes. Proc. Natl. Acad. Sci. USA 96:2982-2987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Reference deleted.
- 21.Song, S. U., and F. M. Boyce. 2001. Combination treatment for osteosarcoma with baculoviral vector mediated gene therapy (p53) and chemotherapy (adriamycin). Exp. Mol. Med. 33:46-53. [DOI] [PubMed] [Google Scholar]
- 22.Stewart, L. M., M. Hirst, M. Lopez Ferber, A. T. Merryweather, P. J. Cayley, and R. D. Possee. 1991. Construction of an improved baculovirus insecticide containing an insect-specific toxin gene. Nature 352:85-88. [DOI] [PubMed] [Google Scholar]
- 23.Tjia, S. T., G. M. zu Altenschildesche, and W. Doerfler. 1983. Autographa californica nuclear polyhedrosis virus (AcNPV) DNA does not persist in mass cultures of mammalian cells. Virology 125:107-117. [DOI] [PubMed] [Google Scholar]