Abstract
We have developed a PCR/electrospray ionization mass spectrometry (PCR/ESI-MS) assay for the rapid detection, identification, and serotyping of human adenoviruses. The assay employs a high-performance mass spectrometer to “weigh” the amplicons obtained from PCR using primers designed to amplify known human adenoviruses. Masses are converted to base compositions and, by comparison against a database of the genetic sequences, the serotype present in a sample is determined. The performance of the assay was demonstrated with quantified viral standards and environmental and human clinical samples collected from a military training facility. Over 500 samples per day can be analyzed with sensitivities greater than 100 genomes per reaction. This approach can be applied to many other families of infectious agents for rapid and sensitive analysis.
Human adenoviruses cause a variety of diseases in humans (13, 14); the types of illness may include gastroenteritis, cystitis, and respiratory disease (pneumonia and respiratory tract infections) (3, 34). In some cases, including closed environments such as schools and military facilities, these diseases can become epidemic (6, 32). The impact on military facilities has been particularly important because the infections have a tremendous negative effect on both the readiness and training schedules of the military personnel. The use of live oral vaccines against serotypes 4 and 7, two of the most common serotypes found in infected military personnel, prevented large epidemic outbreaks until production was discontinued in 1996 (15). Following the cessation of vaccine administration, these outbreaks resumed (16).
Adenoviruses show a high degree of variation. Over 50 serotypes have now been identified based on the use of serotype-specific neutralization assays (5). These serotypes are grouped into six species (A to F) according to a number of criteria, including nucleic acid sequence and agglutination properties (2). Although serotypes 4 and 7 are the most prevalent strains associated with adult respiratory infections, other serotypes are also detected in these populations. Many studies have shown that there is genetic diversity within these serotypes (1, 19-21, 33) and that genome typing of individual serotypes can be used to study the biogeographical origin of outbreak strains and can assist in unraveling the epidemiology of an outbreak (19a).
The ability to rapidly detect and serotype adenovirus is crucial for (i) recognizing that an outbreak is occurring, (ii) prescribing appropriate supportive care, (iii) ruling out other disease etiologies, (iv) making decisions regarding quarantining individuals to reduce the spread of the outbreak, (v) following the epidemiology of the outbreak in real time across multiple sites, and (vi) predicting vaccine effectiveness. The conventional methods used for the detection and serotyping of adenovirus include culture and immunofluorescence to identify adenovirus generically, followed by serotype-discriminating microneutralization assays using serotype-specific antisera (24). Serotyping may also be approximated by sequencing the primary antigenic determinants (30). These assays can take from days to weeks to perform and usually must be performed by highly trained personnel, and results are often difficult to interpret. In recent years, there has been a movement toward more rapid molecular techniques, such as PCR-based assays, for the detection and typing of adenovirus in both clinical and military settings (6-10, 22, 25). Studies have clearly demonstrated the utility of PCR and the high-throughput potential of the assays, but most either (i) are specific for detection of only one or a few serotypes or (ii) are able to detect all serotypes but are incapable of discriminating specific serotypes. Limitations are due either to the inadequate amplification range of the primers or the limited information content provided by probes used to detect the amplicons. Further progress has recently been made by combining the speed of PCR with the power of sequencing. Lin et al. recently demonstrated the use of PCR followed by resequencing microarray technology (23). This technique allows the amplification of a wide variety of serotypes and the detection of point mutations and new serotypes. Although this is a powerful method, it still requires complex reagents and technologies and has not yet reached an optimal level of speed and cost.
This work describes the development of a high-throughput assay capable of both broad detection and high-resolution typing of adenovirus. The approach is equally applicable to pure isolates and mixtures derived from clinical or environmental sources. This work is based on a previously described platform that uses PCR coupled with electrospray ionization mass spectrometry (PCR/ESI-MS) (11, 12, 17, 29). Viral DNA present in a sample is amplified using broad-range PCR primers capable of producing unique amplicons from a wide variety of adenovirus serotypes. The key to the technology is the use of MS to quickly and accurately detect the amplicons and “weigh” them with enough mass accuracy to determine the nucleotide base compositions (i.e., amplicon content in terms of the numbers of A's, G's, C's, and T's). This base composition information is then associated with sequence information to identify and serotype of the adenovirus in the sample. Because it takes approximately 1 minute to analyze each PCR by MS, hundreds of samples can be analyzed in a day in a fully automated mode (12, 17). This level of identification in a rapid-throughput assay format provides an opportunity to change the way in which information about viral pathogens can be used in both clinical diagnosis and public health surveillance activities.
Here we demonstrate the use of this assay in a real-world application with the detailed evaluation of samples from a large human subject population and an environmental sample-based study of a closed, military population known to be exposed to adenovirus infection during recruit training. The PCR/ESI-MS assay is a high-throughput, high-information content assay that can be used with a wide variety of sample types to both identify and serotype adenovirus.
MATERIALS AND METHODS
Primers.
The primers used in this study were originally designed against a human adenovirus E, serotype 4 reference sequence (NC_003266.2). A number of primer pairs were tested. The final primer pairs used were PP769 (forward primer VIR2150F, 5′ TCACCAACACCTACGAGTACATGAA 3′; reverse primer VIR2151R, 5′ TGGTTGAAGGGATTTACGTTGTCCAT 3′), targeting bases 19689 to 19809 of the reference sequence, and PP943 (forward primer VIR2472F, 5′ TTGCAAGATGGCCACCCCATCGAT 3′; reverse primer VIR2473R, 5′ TGTGGCGCGGGCGAACTGCA 3′), targeting bases 18241 to 18352 of the reference sequence. In all cases, a thymidine nucleotide was placed in the first (5′) position of the primer (if not naturally occurring in the target sequence) in order to reduce the addition of nontemplated adenosine nucleotides to the amplicon during the PCR (4).
IPC construction.
A plasmid-based internal positive control (IPC) was designed and then constructed by an outside source (Blue Heron Biotechnology, Bothell, WA). The control sequence for each primer pair was identical to the target reference sequence (NC_003266.2) except for a 5-bp deletion in the target region. The deleted sequence for PP769 encompasses bases 19756 to 19760, and the deleted sequence for PP943 is bases 18313 to 18317. An IPC was added to each sample at 300 molecules per well unless otherwise specified and was amplified during PCR.
PCR methods.
PCR for ESI-MS was performed in 50-μl reaction mixtures consisting of 4 U of AmpliTaq Gold (Applied Biosystems, Foster City, CA), 20 mM Tris (pH 8.3), 75 mM KCl, 1.5 mM MgCl2, 0.4 M betaine, 200 μM deoxynucleoside triphosphates (each) (Bioline USA, Randolph, MA), and 250 nM of each primer (primers obtained from Integrated DNA Technologies, Coralville, IA). The reactions were performed in 96-well plates (Bio-Rad, Hercules, CA) using an MJ Dyad thermocycler (Bio-Rad). The following PCR conditions were used to amplify the sequences used for PCR/ESI-MS analysis: 95°C for 10 min, followed by 8 cycles of 95°C for 30 seconds, 48°C for 30 seconds (increasing 0.9°C each cycle), and 72°C for 30 seconds. The PCR was then continued for 37 additional cycles of 95°C for 15 seconds, 56°C for 20 seconds, and 72°C for 20 seconds. The standard BCE multiplex PCR for adenoviruses was performed as previously described (26).
Clinical sample collection.
Within 48 h of arrival for training at the Marine Corps Recruit Depot in San Diego, CA, 271 recruits and 13 support staff were enrolled in the study. Each provided written, informed consent as required by our IRB protocol number CS03. Clinical samples consisted of an oropharynx swab sample as well as a swab sample from the dominant hand of the individual and a blood sample. These samples were collected as described previously (28).
Environmental sample collection.
Environmental samples were collected from a wide variety of sites in the living and working areas of the Marine Corps Recruit Depot in San Diego, CA. Environmental samples included air collection samples obtained using either a dry filter unit (DAAD13-03-P-00021, model 1000; Battelle, Columbus, OH) or electrostatic air-filtration systems (Ionic Breeze Quadra, model SI637; Sharper Image, San Francisco, CA). Samples were collected for 12-h periods (one sample per period) from each type of collector. Environmental samples also included stationary surface samples that were collected using Dacron swabs moistened in viral transport medium (Remel, Lenexa, KS). The swabs were placed in additional media for transport and processing. A detailed description of the protocol for collection of these samples, including sample types and collection methods, was described previously (17).
Study site.
Clinical and environmental samples were acquired at the Marine Corps Recruit Depot in San Diego, CA, over a 2-month study period from October 2004 to November 2004. In all, over 660 environmental samples and samples from 271 enrolled recruits were collected and analyzed. Details on the study design are reported elsewhere (28).
Sample processing.
Viral stock samples consisting of cultured virus or stocks supplied directly from the ATCC were prepared for analysis using the Qiagen (Valencia, CA) QiaAmp virus kit. Both manual (minispin) kits and BioRobot kits were used. Robot-based isolations were done on both the Qiagen MDx and Qiagen BioRobot 8000 platforms. Clinical and environmental samples were also prepared using these same methods. Swab-based samples were prepared by first washing the swabs in 2 ml viral transport medium. Of the resulting solution, 1 ml was passed over a 0.2-μm filter which was then subjected to bead beating in a small amount of lysis buffer (ATL buffer; Qiagen Inc., Valencia, CA). The resulting sample was then prepared using the QiaAmp virus kit (Qiagen Inc., Valencia, CA).
MS and base composition analysis.
Following amplification, 15-μl aliquots of each PCR mixture was desalted and purified using a weak anion-exchange protocol based on the method of Jiang and Hofstadler (18). Accurate-mass (±1 ppm), high-resolution (M/ΔM, >10,000 full width half maximum) mass spectra were acquired for each sample using ESI, time of flight MS, and protocols described previously (17). For each sample, approximately 1.5 μl of analyte solution was consumed during the spectral acquisition. Raw mass spectra were postcalibrated with an internal mass standard and deconvolved to average molecular masses. Unambiguous base compositions were derived from the exact mass measurements of the complementary single-stranded oligonucleotides (27). Quantitative results were obtained by comparing the peak heights with an internal PCR calibration standard present in every PCR well at 300 molecules unless otherwise indicated (17).
RESULTS
The objective in developing this assay was to design a rapid, single test system that could be used to detect the presence of adenovirus in a sample and simultaneously serotype a wide variety of the adenoviruses detected. Based on information from historical outbreaks of adenovirus at the military site of interest, it was important that serotypes 4, 7, and 21 be identified. Initially, a large number of potential primer pairs were designed (data not shown). The design criteria consisted of a number of factors, including melting temperatures of perfect duplexes formed by the primer with the target, GC content, and production of amplicons suitable for MS-based analysis. A number of primer pairs were evaluated against a single adenovirus genome, and from this testing a small subset of primers was selected for further analysis (data not shown). Two primer pairs were selected from this smaller set for development. These pairs were designated PP943 and PP769 (see Materials and Methods). Using both experimental and theoretical data (derived from experimentally based sequence data), we showed that the combination of PP943 and PP769 will detect and discriminate a wide variety of adenovirus serotypes (Table 1). PP943 is designed to produce an amplicon from virtually all known human adenovirus serotypes; however, using this primer pair, many of the resulting base compositions from different adenovirus serotypes were identical. However, PP769 discriminated among a large number of these serotypes, including serotypes 4, 7, and 21.
TABLE 1.
Theoretical and experimental base compositions of amplicons generated when primer pairs PP769 and PP943 were used to amplify adenovirus genetic material
| Serotype | Base composition of amplicon generated with primer paira:
|
|
|---|---|---|
| PP943 | PP769 | |
| 1 | A20 G33 C39 T20 | Does not prime |
| 2 | A20 G33 C38 T21 | A31 G29 C35 T26 |
| 2 | A20 G33 C39 T20 | A31 G29 C34 T27 |
| 2 | A20 G33 C39 T20 | A31 G29 C35 T26 |
| 2 | A20 G33 C39 T20 | A33 G27 C34 T27 |
| 2 | A22 G32 C37 T21 | A31 G30 C34 T26 |
| 3 | A22 G32 C37 T21 | A33 G24 C37 T27 |
| 3 | A22 G33 C36 T21 | A33 G24 C37 T27 |
| 3 | A23 G31 C37 T21 | A33 G24 C37 T27 |
| 3 | A23 G32 C36 T21 | A33 G24 C37 T27 |
| 3 | A23 G32 C36 T21 | A33 G24 C38 T26 |
| 4 | A20 G31 C39 T19 | A30 G31 C38 T22 |
| 4 | A20 G33 C39 T20 | A30 G31 C38 T22 |
| 4 | A20 G34 C38 T20 | A27 G33 C39 T22 |
| 4 | A20 G34 C38 T20 | A27 G34 C38 T22 |
| 4a | A20 G33 C39 T20 | A30 G31 C38 T22 |
| 4a1 | A20 G33 C39 T20 | A30 G31 C38 T22 |
| 4a2 | A20 G33 C39 T20 | A30 G31 C38 T22 |
| 4a3 | A20 G33 C39 T20 | A30 G31 C38 T22 |
| 4p1 | A20 G34 C38 T20 | A27 G33 C39 T22 |
| 4p4 | A20 G34 C38 T20 | A27 G33 C39 T22 |
| 5 | A20 G33 C39 T20 | A33 G27 C34 T27 |
| 5 | A20 G33 C39 T20 | A33 G27 C35 T26 |
| 5 | A23 G32 C36 T21 | A33 G24 C37 T27 |
| 6 | A20 G33 C39 T20 | A31 G29 C34 T27 |
| 7 | A23 G32 C36 T21 | A32 G24 C37 T28 |
| 7 | A23 G32 C36 T21 | A33 G24 C36 T28 |
| 7a | A23 G32 C36 T21 | A32 G24 C37 T28 |
| 7a | A23 G32 C36 T21 | A32 G24 C38 T27 |
| 7d | A23 G31 C38 T20 | A32 G24 C37 T28 |
| 7d | A23 G32 C36 T21 | A32 G24 C37 T28 |
| 8 | A20 G36 C38 T18 | A27 G30 C44 T20 |
| 9 | A20 G36 C38 T18 | A28 G29 C44 T20 |
| 9 | A20 G36 C38 T18 | A29 G28 C44 T20 |
| 10 | A20 G36 C38 T18 | A28 G32 C41 T20 |
| 11p | A22 G32 C37 T21 | A31 G30 C34 T26 |
| 11 | A21 G33 C37 T21 | A31 G30 C34 T26 |
| 12 | A20 G32 C37 T23 | A29 G31 C35 T26 |
| 13 | A20 G36 C38 T18 | A28 G28 C44 T21 |
| 14 | A21 G33 C37 T21 | A31 G30 C34 T26 |
| 15 | A20 G36 C38 T18 | A28 G28 C44 T21 |
| 16 | A23 G32 C36 T21 | A31 G30 C34 T26 |
| 17 | A20 G36 C38 T18 | A30 G28 C43 T20 |
| 18 | A21 G32 C36 T23 | A31 G29 C31 T30 |
| 19 | A20 G36 C38 T18 | A28 G30 C42 T21 |
| 20 | A20 G36 C37 T19 | A30 G26 C45 T20 |
| 21 | A21 G33 C37 T21 | A31 G28 C35 T27 |
| 22 | A20 G36 C38 T18 | A26 G30 C44 T21 |
| 23 | A20 G36 C38 T18 | A29 G29 C42 T21 |
| 24 | A20 G36 C38 T18 | A28 G28 C44 T21 |
| 25 | A20 G36 C38 T18 | A28 G28 C45 T20 |
| 26 | A20 G36 C38 T18 | A28 G28 C44 T21 |
| 26 | A20 G36 C38 T18 | A29 G27 C44 T21 |
| 27 | A20 G36 C38 T18 | A27 G30 C45 T19 |
| 28 | A20 G36 C38 T18 | A28 G29 C44 T20 |
| 29 | A20 G36 C38 T18 | A27 G29 C45 T20 |
| 30 | A20 G36 C38 T18 | A28 G29 C43 T21 |
| 31 | A20 G32 C38 T22 | A30 G28 C37 T26 |
| 32 | A20 G36 C38 T18 | A27 G29 C45 T20 |
| 33 | A20 G36 C38 T18 | A27 G29 C45 T20 |
| 34 | A21 G33 C37 T21 | A31 G30 C34 T26 |
| 35 | A22 G32 C37 T21 | A31 G30 C34 T26 |
| 35p | A22 G32 C37 T21 | A31 G30 C34 T26 |
| 36 | A20 G36 C38 T18 | A27 G30 C41 T23 |
| 37 | A20 G36 C38 T18 | A28 G28 C44 T21 |
| 37 | A31 G26 C33 T30 | A28 G28 C44 T21 |
| 38 | A20 G36 C38 T18 | A30 G26 C44 T21 |
| 39 | A20 G36 C38 T18 | A27 G30 C44 T20 |
| 40 | A21 G33 C39 T19 | Does not prime |
| 42 | A20 G36 C38 T18 | A29 G27 C45 T20 |
| 43 | A20 G36 C38 T18 | A27 G29 C45 T20 |
| 44 | A20 G36 C38 T18 | A28 G28 C44 T21 |
| 45 | A20 G36 C38 T18 | A28 G28 C43 T22 |
| 46 | A20 G36 C38 T18 | A27 G29 C44 T21 |
| 47 | A20 G36 C38 T18 | A26 G30 C45 T20 |
| 48 | A20 G36 C38 T18 | A28 G28 C43 T22 |
| 49 | A20 G36 C38 T18 | A29 G27 C44 T21 |
| 50 | A21 G33 C37 T21 | A31 G28 C35 T27 |
| 51 | A20 G36 C38 T18 | A29 G29 C43 T20 |
| 52 | A20 G31 C41 T20 | A31 G29 C38 T23 |
The base compositions are derived from either experimental data (boldface rows) or theoretical data (all other rows) obtained from analysis of the database sequence information. For all tested isolates, an amplicon was generated by primer pair PP943; however, the base compositions of the amplicons for some isolates (for example, serotypes 8 and 9) were identical. Use of primer pair PP769 provided discrimination among virtually all serotypes. “Does not prime” indicates that the primer pair does not produce an amplicon for that particular serotype. When multiple base counts could be assigned to a single serotype, each unique occurrence was noted.
Discrimination between the various adenovirus serotypes in military populations is critical to surveillance efforts in order to guide vaccine development (Kajon et al., submitted). Serotype 4, the serotype often associated with outbreaks in military settings, includes two very divergent genome types, 4a (Ad4a) and 4p (Ad4p), which are distinguished by whole-genome restriction enzyme analysis with the enzyme BamHI. Each of these genome types consists of a number of variants (such as Ad4p1 and Ad4p4), which are distinguished by whole-genome restriction enzyme analysis with additional enzymes. We investigated the ability of the PCR/ESI-MS assay to distinguish between the 4a and 4p genome types and between variants of these types. Samples from U.S. Marine Corps recruits, previously analyzed by traditional genome typing methods (Kajon et al., submitted), were analyzed using the PCR/ESI-MS assay with primer pairs PP943 and PP769. Table 1 also shows the results of the assay for a wide variety of Ad4a and Ad4p samples. In every case, 4a and 4p were clearly distinguished, although the two-primer assay was not able to further distinguish genetic variants within each of these genome types.
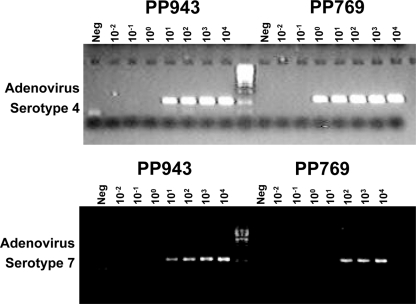
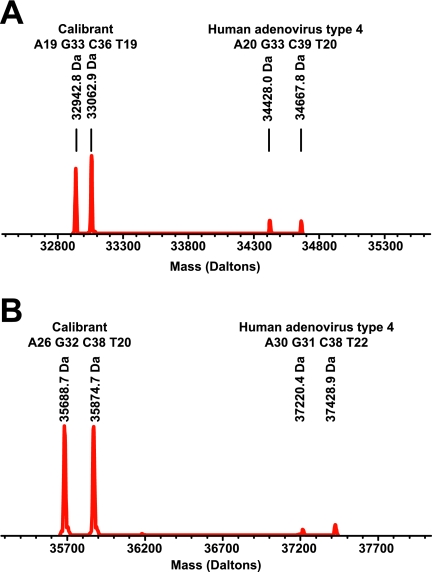
The ability of the system to sensitively detect adenovirus genomic material was tested using adenovirus serotype 4. Genomic material was isolated from adenovirus serotype 4 virus stocks grown in cultured cells. It was not possible to accurately measure the exact quantity of adenovirus genomic material present in the sample by UV spectroscopy since the material contained contaminating DNA from the cell line used for culture. Determination of absolute system sensitivity was therefore performed using the viral titer determined for the stock at the time of collection. Figure 1 shows a dilution-to-extinction experiment. In this experiment, PCR products were detected by gel electrophoresis. The level of detection was less than 100 PFU in all cases, and in most instances, it was 10 or less PFU. Figure 2 shows a similar set of samples analyzed by the PCR/ESI-MS assay. Figure 2A shows that using primer pair PP943, the adenovirus serotype 4 could be detected at a level below 10 PFU. Similar levels of detection were achieved using PP769 (Fig. 2B).
FIG. 1.
Sensitivity of detection of adenovirus serotype 4 and serotype 7 genomic material by PCR using primer pairs PP769 and PP943 as evaluated by gel-based analysis. Serial dilutions of previously quantified adenovirus serotype 4 and serotype 7 genomic material were tested in PCR assays against PP769 and PP943. The resulting amplicons were analyzed by gel electrophoresis. The concentration of material in each reaction is represented in PFU.
FIG. 2.
Sensitivity of detection of adenovirus serotype 4 genomic material by PCR using primer pairs PP769 and PP943 as evaluated by ESI-MS-based analysis. Adenovirus serotype 4 genomic material was present at 10, 100, or 1,000 PFU per PCR. (A) ESI-MS detection following amplification by PP943. (B) ESI-MS detection following amplification by PP769. The two peaks in each spectrum correspond to sense and antisense strands of the PCR amplicons, which separate under the conditions of ESI.
PCRs used in the PCR/ESI-MS assay normally contain an IPC. The IPC serves two distinct purposes. First, the IPC provides an internal PCR control for the assay that ensures that a successful PCR took place. Additionally, the IPC provides a calibration standard in each well that is present at a known concentration. By comparison of the abundance (intensity of the signal from this peak in the mass spectrum) of the amplicon from the IPC to the abundance of amplicon derived from the target genome(s), an absolute amount of target genome(s) can be accurately determined. IPCs were constructed for both PP769 and PP943 (see Materials and Methods). The region amplified from the IPC is identical to the region from the reference sequence used for primer design, with the exception of a 5-base-pair deletion; the deletion in the IPC allows the IPC amplicon to be distinguished from natural products in the mass spectrum. The sensitivity of both primer pairs PP943 and PP769 for detection of the IPC was evaluated by determining the minimal level of the IPC plasmid detectable in the assay system. Samples containing a series of concentrations of IPC were tested in PCRs containing the PP943 and PP769 primer pairs, and the resulting products of those reactions were analyzed by ESI-MS. For both primer pairs, the PCR/ESI-MS system was able to detect the presence of IPC template at fewer than 100 copies per reaction (data not shown).
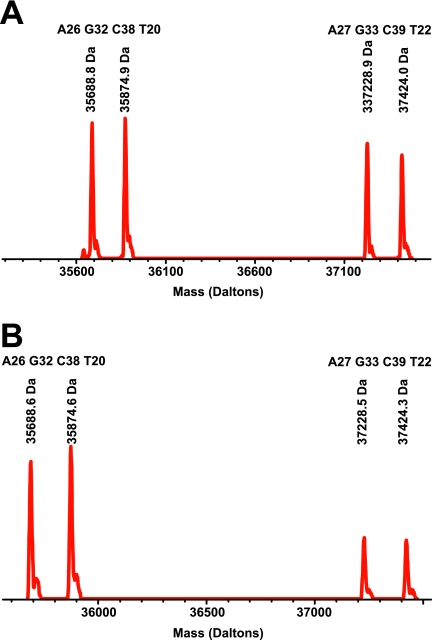
Although the dynamic range of the PCR/ESI-MS system is 1 to 10,000 genome copies per reaction (17), it is ultimately limited by the dynamic range of both the PCR and the mass spectrometer. Therefore, it was important to determine the maximum level of IPC for each primer pair that still allows for sensitive detection of adenovirus in a sample. A measurable amount of IPC amplification product must be present, but the IPC template cannot mask the detection of low levels of target genomic material. To determine the appropriate IPC concentration, a matrix of concentrations of IPC and of adenovirus serotype 4 was evaluated (data not shown). From this series of tests, we determined that the optimal level of IPC was 300 copies/PCR. Figure 3A shows the ESI-MS spectrum of a sample, following PCR using primer pair PP943, of adenovirus serotype 4 genomic material (∼100 genome copies) in the presence of the IPC. Figure 3B shows the spectrum obtained following PCR using PP769. In both cases the mass spectrometer was able to detect ∼100 genomes per reaction in the presence of the IPC. The approximate quantity of genome in the PCR can be calculated by using the ratio of the amplitude of the detected genomic material to that of the known quantity of IPC added to the reaction.
FIG. 3.
Detection of adenovirus serotype 4a in the presence of an IPC. Approximately 100 genomic copies of adenovirus serotype 4a genome were mixed with 300 copies of IPC in a standard RT-PCR using PP943 (A) or PP769 (B). Peaks in each spectrum correspond to sense and antisense strands of IPC and adenovirus. The masses of the individual DNA strands and the amplicon base compositions are indicated.
This assay is intended to detect adenovirus in both environmental and clinical samples; therefore, we determined the assay sensitivity in both backgrounds. For this test, a series of environmental air samples were collected on dry filter units placed in a variety of indoor and outdoor locations at the Marine Corps Recruit Depot in San Diego, CA. A single combined sample was created by using a large number of individual samples, and air from this sample was used as background for evaluation of a dilution series of adenovirus serotype 4 material. The minimum amount of material detected was less than or equal to 100 PFU per well (data not shown). A representative detection is shown in Fig. 4A. In this example, the amount of quantitated genome detected is between 200 and 300 copies using a spike of approximately 100 PFU While the exact ratio of genome to PFU for these samples is not known, Shayakhmetov et al. found that the range of ratios of genome to PFU, in a small number of adenovirus subtypes, was between 5 and several thousand, depending on the subtype (31). Our findings are consistent with that work.
FIG. 4.
Detection of adenovirus serotype 4p, using PP769, in the presence of an IPC and a relevant background. (A) Spectrum obtained following PCR/ESI-MS of approximately 100 PFU of adenovirus serotype 4p in the presence of an air background. (B) Spectrum obtained following PCR/ESI-MS of approximately 100 PFU of adenovirus serotype 4p in the presence of a throat background from healthy volunteers.
As a control background for the clinical samples, we used a mixture of a number of throat swabs from healthy volunteers. These samples were pooled and tested using primer pair PP769 to ensure the lack of any background adenovirus material in the samples. As with the environmental samples, a range of concentrations of adenovirus serotype 4 material was tested in the presence of this clinical background to determine the minimal detectable number of PFU per well. The minimal detectable level of adenovirus in this background was higher than in the case of no background but was still less than or equal to 100 PFU per well (data not shown). A representative detection is shown in Fig. 4B.
A comparison test of the PCR/ESI-MS assay with a conventional PCR assay designed to detect and serotype adenovirus was performed using a number of previously characterized samples (Table 2). These samples consisted of both cultured virus stocks and archived clinical samples. For each sample tested, both the PCR/ESI-MS assay and the standard PCR BCE-2 assay were performed. Table 2 shows the detections from each assay type. Generally, the PCR/ESI-MS assay was able to detect the adenovirus present and identify the serotype. In the case of serotype 1 (species C), the PCR/ESI-MS assay detects the presence of adenovirus but is unable to identify the exact serotype (although it does identify the correct species). In the case of serotype 40 (species F), the PCR/ESI-MS assay is similarly unable to identify the correct serotype. Both of these cases result from the inability of PP769 to prime these specific serotypes. This can be corrected by the use of a more comprehensive assay setup with more primer pairs (data not shown).
TABLE 2.
Comparison of PCR/ESI-MS detections with those of a standard BCE-2 PCR assay
| Identification of previously characterized sample | Identification by:
|
Base count for T5000 detection with primer pair:
|
||
|---|---|---|---|---|
| T5000 | Standard BCE-2 PCR | PP943 | PP769 | |
| Cultured isolates | ||||
| Ad C1 | Ad C | C | A20 G33 C39 T20 | Does not prime |
| Ad C5 | Ad C5 | C | A20 G33 C39 T20 | A33 G27 C35 T26 |
| Ad C6 | Ad C6 | C | A20 G33 C39 T20 | A31 G29 C34 T27 |
| Ad B3 | Ad B3 | C | A23 G31 C37 T21 | A33 G24 C37 T27 |
| Ad B7 | Ad B7 | B | A23 G32 C36 T21 | A33 G24 C36 T28 |
| Ad B21 | Ad B21 | B | A21 G33 C37 T21 | A31 G28 C35 T27 |
| Ad B14 | Ad B14/B34 | B | A21 G33 C37 T21 | A31 G30 C34 T26 |
| Ad B16 | Ad B16 | B | A23 G32 C36 T21 | A31 G30 C34 T26 |
| Ad F40 | Ad F40 | C | A21 G33 C39 T19 | Does not prime |
| Clinical specimens | ||||
| Ad B7 | Ad B7 | B | A23 G32 C36 T21 | A32 G24 C37 T28 |
| Ad B14 | Ad B14/B34 | B | A21 G33 C37 T21 | A31 G30 C34 T26 |
| Ad E4a | Ad E4a | E4a | A20 G33 C39 T20 | A30 G31 C38 T22 |
| Ad E4p | Ad E4p | E4p | A21 G33 C37 T21 | A27 G33 C39 T22 |
As previously described, the detection methodology used here was tested in a clinical study and was able to identify 134 of the 135 clinical samples that were identified as positive using classic culture techniques (28). In addition, 78 throat samples that were negative with classic culture were positive by PCR/ESI-MS. We also tested a large number of environmental samples using the PCR/ESI-MS system. A smaller number of these samples were also tested using classical culture/immunofluorescence. The culture techniques provide information about the presence or absence of virus in the sample; in addition to this information, the PCR/ESI-MS system measures of the quantity of virus present (Fig. 3 and 4).
Table 3 shows the correlation of the detections by both culture and PCR/ESI-MS for each of three distinct cutoff values as well as the specificity and sensitivity values at these same cutoff levels. The cutoff values represent the minimum level of virus (in number of molecules) detected in a sample that would then lead to a declaration of a positive detection by the analysis software. Levels below a particular cutoff value were declared negative. For the majority of the samples and cutoff values, there was an excellent correlation between PCR/ESI-MS and culture. Higher cutoff values led to an apparent increase in false-negative detections (Table 3, Vir+/MS−) and a decrease in false-positive detections (Table 3, Vir−/MS+). However, the strong correlation between the estimated load of genetic material as determined by PCR/ESI-MS and the ability to culture with classic techniques implies that the PCR/ESI-MS detection was actually more sensitive rather than less specific and that the “false” positives were actually true positives that were unculturable (Table 3).
TABLE 3.
Results of culture versus PCR/ESI-MS assay at various detection amplitude cutoffs for 786 clinical and environmental samples
| Amplitude cutoff for detection (no. of molecules)a | No. of samplesb:
|
Sensitivity (%) | Specificity (%) | ||||
|---|---|---|---|---|---|---|---|
| Vir+/MS+ | Vir+/MS− | Vir−/MS+ | Vir−/MS− | Total | |||
| 50 | 131 | 4 | 40 | 611 | 786 | 0.97 | 0.94 |
| 100 | 129 | 6 | 35 | 616 | 786 | 0.96 | 0.95 |
| 300 | 124 | 11 | 19 | 632 | 786 | 0.92 | 0.97 |
Minimum level of virus in a sample that would lead to a declaration of a positive detection by the analysis software.
Vir, culture result; MS, PCR/ESI-MS result.
DISCUSSION
Traditional methods for the detection and characterization of pathogenic organisms in both environmental and clinical samples are both cumbersome and time-consuming. For the most part, these methods depend upon the ability to culture the organism(s) of concern. Typing beyond the species level is generally accomplished by the use of specific immunoassays performed on cultured material. Although decisions regarding clinical treatment do not always require typing, the ability to obtain such detailed information is critical to any epidemiological study and can play a significant role in understanding the level and form of pathogenicity that any one strain may possess.
The PCR/ESI-MS technology described in this work does not require culture and uses molecular-based methods both to detect adenovirus and to identify the serotype of the virus or viruses present. For the assay described here, two primer pairs (PP943 and PP769) were used to amplify adenovirus present in clinical or environmental samples. The base compositions of the resulting amplicons were determined following MS using bioinformatic analysis (12). From the base compositions, the serotype can be assigned. The sensitivity of the primers for both the internal calibrant and a prototypic set of viruses (serotypes 4 and 7) was better than 100 copies/reaction, and the effect of adding a background obtained from either air or throat swabs was minimal.
Evaluation of samples of various serotypes showed that the assay was capable of detection of all tested adenovirus strains. The ability to assign an absolute serotype to the detected adenovirus in each sample provides a level of information currently available only with additional, time-consuming steps. The ability of the assay to detect adenovirus in both clinical and environmental samples was tested in a study comparing the PCR/ESI-MS methodology to traditional detection methods (28). The correlation of the PCR/ESI-MS to the gold standard culture methods was very good (Table 2). Overall, this assay produces more positive detections than does gold standard culture. This is, at least in part, due to the high sensitivity of the PCR-based assay and the ability to detect genomic material rather than PFU. Since the ratio of genome present to PFU is normally much greater than 1.0, one would expect a higher sensitivity in assays directed against nucleic acid. The PCR/ESI-MS assay requires no culture and provides both general detection of adenovirus and serotype determination in a single, fast, easy test. Critically, the PCR/ESI-MS assay runs on a fully automated bench top instrument that can analyze >1,500 PCRs per day, and results from a sample are obtained within 24 h (12). Overall, the PCR/ESI-MS technology will provide a significant improvement relative to current methods for adenovirus detection and serotyping. The quantitative nature of the assay allows for the evaluation of the data at multiple detection cutoff levels to provide enhanced sensitivity and specificity compared to the culture-based system.
Acknowledgments
We acknowledge the administrative support of the Henry M. Jackson Foundation for Military Medicine and the efforts of the entire Naval Health Research Center team, especially the technicians and collection personnel who made this work possible.
The views expressed in this work are those of the authors and do not reflect the official policy or position of the Department of the Navy, Department of the Army, Department of Defense, or U.S. government. This research has been conducted in compliance with all applicable federal and international regulations governing the protection of human subjects in research (DoD protocol NHRC.1999.0002).
L.B.B., T.A.H., B.L., R.R., R.S., S.A.H., and D.J.E. are employed by Ibis Biosciences, the provider of the PCR/ESI-MS technology, and are fully compensated by the company for their contributions.
Footnotes
Published ahead of print on 19 December 2007.
REFERENCES
- 1.Adrian, T., et al. 1986. DNA restriction analysis of adenovirus prototypes 1 to 41. Arch. Virol. 91277-290. [DOI] [PubMed] [Google Scholar]
- 2.Benko, M., et al. 2004. Family Adenoviridae, p. 1162. In C. M. Fauquet, et al. (ed.), Virus taxonomy. VIIIth report of the International Committee on Taxonomy of Viruses. Academic Press, New York, NY.
- 3.Brandt, C. D., et al. 1969. Infections in 18,000 infants and children in a controlled study of respiratory tract disease. I. Adenovirus pathogenicity in relation to serologic type and illness syndrome. Am. J. Epidemiol. 90484-500. [DOI] [PubMed] [Google Scholar]
- 4.Brownstein, M. J., J. D. Carpten, and J. R. Smith. 1996. Modulation of non-templated nucleotide addition by Taq DNA polymerase: primer modifications that facilitate genotyping. BioTechniques 201004-1006, 1008-1110. [DOI] [PubMed] [Google Scholar]
- 5.De Jong, J. C., et al. 1999. Adenoviruses from human immunodeficiency virus-infected individuals, including two strains that represent new candidate serotypes Ad50 and Ad51 of species B1 and D, respectively. J. Clin. Microbiol. 373940-3945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Echavarria, M., et al. 2003. Rapid detection of adenovirus in throat swab specimens by PCR during respiratory disease outbreaks among military recruits. J. Clin. Microbiol. 41810-812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Echavarria, M., et al. 1998. PCR method for detection of adenovirus in urine of healthy and human immunodeficiency virus-infected individuals. J. Clin. Microbiol. 363323-3326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Echavarria, M., et al. 2000. Detection of adenoviruses (AdV) in culture-negative environmental samples by PCR during an AdV-associated respiratory disease outbreak. J. Clin. Microbiol. 382982-2984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Echavarria, M., et al. 2001. Prediction of severe disseminated adenovirus infection by serum PCR. Lancet 358384-385. [DOI] [PubMed] [Google Scholar]
- 10.Echavarria, M., et al. 2006. Use of PCR to demonstrate presence of adenovirus species B, C, or F as well as coinfection with two adenovirus species in children with flu-like symptoms. J. Clin. Microbiol. 44625-627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ecker, D. J., et al. 2005. Rapid identification and strain-typing of respiratory pathogens for epidemic surveillance. Proc. Natl. Acad. Sci. USA 1028012-8017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ecker, D. J., et al. 2006. The Ibis T5000 universal biosensor—an automated platform for pathogen identification and strain typing. J. Assoc. Lab. Automat. 11341-351. [Google Scholar]
- 13.Edwards, K. M., et al. 1985. Adenovirus infections in young children. Pediatrics 76420-424. [PubMed] [Google Scholar]
- 14.Fox, J. P., et al. 1969. The virus watch program: a continuing surveillance of viral infections in metropolitan New York families. VI. Observations of adenovirus infections: virus excretion patterns, antibody response, efficiency of surveillance, patterns of infections, and relation to illness. Am. J. Epidemiol. 8925-50. [DOI] [PubMed] [Google Scholar]
- 15.Gaydos, C. A., and J. C. Gaydos. 1995. Adenovirus vaccines in the U.S. military. Mil. Med. 160300-304. [PubMed] [Google Scholar]
- 16.Gray, G. C., et al. 2000. Adult adenovirus infections: loss of orphaned vaccines precipitates military respiratory disease epidemics. Clin. Infect. Dis. 31663-670. [DOI] [PubMed] [Google Scholar]
- 17.Hofstadler, S. A., et al. 2005. TIGER: the universal biosensor. Int. J. Mass Spectrom. 24223-41. [Google Scholar]
- 18.Jiang, Y., and S. A. Hofstadler. 2003. A highly efficient and automated method of purifying and desalting PCR products for analysis by electrospray ionization mass spectrometry. Anal. Biochem. 31650-57. [DOI] [PubMed] [Google Scholar]
- 19.Kajon, A. E., et al. 1999. Genome type analysis of Brazilian adenovirus strains of serotypes 1,2,3,5, and 7 collected between 1976 and 1995. J. Med. Virol. 58408-412. [DOI] [PubMed] [Google Scholar]
- 19a.Kajon, A. E., et al. 2007. Molecular epidemiology of adenovirus type 4 infections in the post-vaccination era (1997-2003). J. Infect. Dis. 19667-75. [DOI] [PubMed] [Google Scholar]
- 20.Li, Q. G., and G. Wadell. 1988. Comparison of 17 genome types of adenovirus type 3 identified among strains recovered from six continents. J. Clin. Microbiol. 261009-1015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Li, Q. G., and G. Wadell. 1986. Analysis of 15 different genome types of adenovirus type 7 isolated on five continents. J. Virol. 60331-335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lin, B., et al. 2004. Use of oligonucleotide microarrays for rapid detection and serotyping of acute respiratory disease-associated adenoviruses. J. Clin. Microbiol. 423232-3239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lin, B., et al. 2007. Using a resequencing microarray as a multiple respiratory pathogen detection assay. J. Clin. Microbiol. 45443-452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Malasig, M. D., et al. 2001. Simplified microneutralization test for serotyping adenovirus isolates. J. Clin. Microbiol. 392984-2986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Metzgar, D., et al. 2005. PCR analysis of Egyptian respiratory adenovirus isolates, including identification of species, serotypes, and coinfections. J. Clin. Microbiol. 435743-5752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Metzgar, D., et al. 2007. Abrupt emergence of diverse species B adenoviruses at US military recruit training centers. J. Infect. Dis. 1961465-1473. [DOI] [PubMed] [Google Scholar]
- 27.Muddiman, D. C., et al. 1997. Length and base composition of PCR-amplified nucleic acids using mass measurements from electrospray ionization mass spectrometry. Anal. Chem. 691543-1549. [DOI] [PubMed] [Google Scholar]
- 28.Russell, K. L., et al. 2006. Transmission dynamics and prospective environmental sampling of adenovirus in a military recruit setting. J. Infect. Dis. 194877-885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sampath, R., et al. 2005. Rapid identification of emerging pathogens: coronavirus. Emerg. Infect. Dis. 11373-379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sarantis, H., et al. 2004. Comprehensive detection and serotyping of human adenoviruses by PCR and sequencing. J. Clin. Microbiol. 423963-3969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Shayakhmetov, D. M., et al. 2000. Efficient gene transfer into human CD34+ cells by a retargeted adenovirus vector. J. Virol. 742567-2583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Top, F. H., Jr. 1975. Control of adenovirus acute respiratory disease in U.S. Army trainees. Yale J. Biol. Med. 48185-195. [PMC free article] [PubMed] [Google Scholar]
- 33.Videla, C., G. Carballal, and A. Kajon. 1999. Genomic analysis of adenovirus isolated from Argentinian children with acute lower respiratory infections. J. Clin. Virol. 1467-71. [DOI] [PubMed] [Google Scholar]
- 34.Weissenbacher, M., et al. 1990. Etiologic and clinical evaluation of acute lower respiratory tract infections in young Argentinian children: an overview. Rev. Infect. Dis. 12(Suppl. 8)S889-S898. [DOI] [PubMed] [Google Scholar]