Abstract
Quality control of gene expression operates post-transcriptionally at various levels in eukaryotes. Once transcribed, mRNAs associate with a host of proteins throughout their lifetime. These mRNA–protein complexes (mRNPs) undergo a series of remodeling events that are influenced by and/or influence the translation and mRNA decay machinery. In this review we discuss how a decision to translate or to degrade a cytoplasmic mRNA is reached. Nonsense-mediated mRNA decay (NMD) and microRNA (miRNA)-mediated mRNA silencing are provided as examples. NMD is a surveillance mechanism that detects and eliminates aberrant mRNAs whose expression would result in truncated proteins that are often deleterious to the organism. miRNA-mediated mRNA silencing is a mechanism that ensures a given protein is expressed at a proper level to permit normal cellular function. While NMD and miRNA-mediated mRNA silencing use different decision-making processes to determine the fate of their targets, both are greatly influenced by mRNP dynamics. In addition, both are linked to RNA processing bodies. Possible modes involving 3′ untranslated region and its associated factors, which appear to play key roles in both processes, are discussed.
Keywords: microRNA, mRNA decay, NMD, P-bodies, translation
Introduction
Messenger RNA (mRNA) mediates the transfer of genetic information from the cell nucleus to ribosomes in the cytoplasm, where it serves as a template for protein synthesis. Once mRNAs enter the cytoplasm, they are translated, stored for later translation, or degraded. mRNAs that are initially translated may later be temporarily translationally repressed. All mRNAs are ultimately degraded at a defined rate. How are these decisions made? Throughout their lifetime, mRNAs associate with a host of proteins factors, some of which are stably bound while others subject to dynamic exchange (Moore, 2005). Individual mRNA–protein complex (mRNP) components may serve as adaptors that allow mRNAs to interface with the machinery mediating their subcellular localization, translation, and decay. Thus, mRNP remodeling is likely to play a critical role in forming decision as to whether to translate or to degrade an mRNA.
In this review, we use two regulatory mechanisms that control mRNA translation and decay as examples to illustrate how a decision may be reached to translate or to degrade a cytoplasmic mRNA. One is nonsense-mediated mRNA decay (NMD), an RNA surveillance mechanism that rapidly degrades mRNAs harboring premature termination codons (PTCs). The other is microRNA (miRNA)-mediated silencing of gene expression, which involves the base pairing of miRNAs with the 3′ untranslated regions (UTRs) of their target mRNAs. Remodeling events are likely to be crucial for both miRNA-mediated silencing and NMD (Schell et al, 2002; Dreyfuss et al, 2003; Maquat, 2004; Amrani et al, 2006; Chang et al, 2007; Jackson and Standart, 2007; Nilsen, 2007; Pillai et al, 2007). We discuss two distinct models for how NMD distinguishes between normal and aberrant PTC-bearing mRNAs, and suggest ways that they can be reconciled via a ‘unified' model. We describe what is known about how miRNAs target mRNAs for rapid decay and translation repression, and highlight recent studies that have begun to pinpoint how miRNAs inhibit translation initiation. In our discussion of the underlying mechanisms for NMD and miRNA-mediated silencing, we consider the role of RNA-processing bodies (P-bodies), the recently identified cytoplasmic foci that harbor translationally silenced mRNPs and may be the burial grounds for at least some mRNAs (Parker and Sheth, 2007; Eulalio et al, 2007a). We also discuss the role of deadenylation in NMD and miRNA-mediated events, as loss of the poly(A) tail leads to loss of poly(A)-binding protein (PABP), which in turn is known to have profound consequences on both translation and mRNA decay (Jacobson, 1996; Mangus et al, 2003).
NMD: a conserved eukaryotic quality control mechanism
NMD is a conserved pathway found in Saccharomyces cerevisiae (yeast; Losson and Lacroute, 1979), Drosophila melanogaster (Brogna, 1999), Caenorhabditis elegans (Hodgkin et al, 1989), mammals (Maquat et al, 1981), and plants (van Hoof and Green, 1996). Most normal eukaryotic cellular mRNAs are not subject to NMD because they only contain a stop codon at the end of the coding region. In contrast, mutant mRNAs that have an in-frame stop codon upstream of the normal stop codon, are recognized by the NMD machinery, leading to mRNA destabilization. Many human inherited diseases are caused by mutations that trigger NMD (Frischmeyer and Dietz, 1999). Some disease alleles contain a mutation that directly changes a sense codon to a stop codon, and others introduce an in-frame stop codon by more indirect ways such as insertions, deletions, and mutations that disrupt RNA splicing, all of which can result in a shift of the reading frame. It has been estimated that 30% of human disease alleles cause NMD, and in many of these cases, NMD contributes to the disease phenotype (Frischmeyer and Dietz, 1999; Holbrook et al, 2004).
The core factors universally required for NMD (i.e., Upf1p, Upf2p and Upf3p) were originally identified in a genetic screen in yeast (Culbertson et al, 1980). Homologs of these proteins were subsequently identified and shown to function in NMD in humans (Sun et al, 1998), D. melanogaster (Gatfield et al, 2003), C. elegans (Page et al, 1999; Aronoff et al, 2001), and Arabidopsis thaliana (Hori and Watanabe, 2005; Arciga-Reyes et al, 2006). Additional genes are also required for NMD in higher eukaryotes (see below). Despite a large body of work on these three Upf proteins, their mechanism of action in NMD is only beginning to be understood. The only Upf protein with a clearly defined biochemical function is Upf1, which is an ATP-binding protein with RNA helicase activity. Upf1 can catalyze the unwinding of double-stranded RNA (dsRNA), but its substrates have not been identified (Czaplinski et al, 1995; Bhattacharya et al, 2000). It is possible that Upf1 may catalyze some other reactions, such as acting like a motor protein that moves along an RNA or remodeling mRNP for translation termination and/or subsequent mRNA degradation (see below).
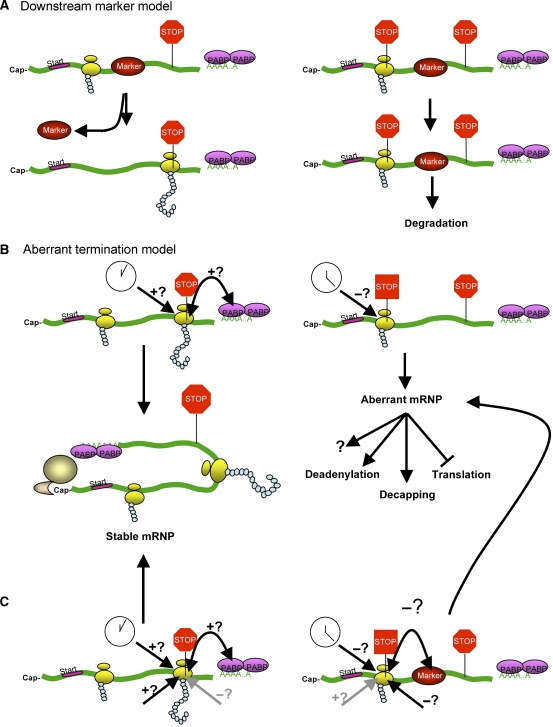
Many models for NMD have been proposed, but they essentially fall into two broad categories. The first group of models we will refer to collectively as the ‘downstream marker model'. This model posits a central role for ‘marker' proteins that are deposited on the mRNA downstream of the PTC and upstream of a normal termination codon (Figure 1). In a normal mRNA, the translating ribosome and/or associated factors displace these marker proteins so that they cannot trigger NMD (Figure 1A). However, in a PTC-containing mRNA, the marker proteins would still be bound when the translational apparatus recognizes the PTC. Interaction of these marker proteins with translation termination factors recruited to the PTC leads to rapid mRNA degradation (Figure 1A). The second group of models will be referred to herein as the ‘aberrant termination model' (Figure 1B). In this model, normal termination induces an mRNP rearrangement, which leads to mRNA stability, whereas aberrant termination induced by a PTC fails to cause this mRNP remodeling or triggers aberrant mRNP remodeling. In the following sections, we will discuss these two groups of models, as well as some important features that we believe could unify them.
Figure 1.
Models for nonsense-mediated decay. (A) The downstream marker posits the presence of a marker protein that is bound to the mRNA downstream of the premature stop codon. The presence of this marker triggers degradation of the PTC containing mRNAs (right panel). In a normal mRNA, the translating ribosomes remove the downstream marker from the coding region of the mRNA, thus preventing normal mRNAs from being targeted to the NMD pathway (left panel). (B) The aberrant termination model posits that termination at a normal stop codon (octagonal stop sign) is different from translation termination at a PTC (aberrant square stop sign). The difference in termination may be due to the proximity of PABP to the normal stop codon (double headed arrow), and/or termination at normal stop codons may be faster than termination at a PTC (clock). These two possibilities are not mutually exclusive. Normal termination at a normal stop codon triggers remodeling into a stable mRNP, whereas aberrant termination at a PTC either prevents this remodeling or triggers remodeling into an aberrant mRNP, which in turn triggers mRNA degradation by a variety of mechanisms. (C) The aberrant termination and downstream marker models can be combined into one coherent model. In this model, the difference between normal termination and aberrant termination can be influenced by a number of different signals. For example, proximity to PABP and other features make termination more normal, whereas downstream markers and other features make termination more aberrant. A preponderance of positive signals causes normal termination, which triggers remodeling into a stable mRNP. A preponderance of negative signals either prevents this remodeling, or triggers remodeling into an aberrant mRNP.
The downstream marker model for NMD
One of the best-characterized NMD substrates is yeast PTC-bearing PGK1 mRNA. This mRNA is unstable but can be stabilized by deleting most of the sequence downstream of the PTC. Reinsertion of a small 3′ region of PGK1 mRNA, called the downstream sequence element (DSE), into the deletion mutant restores mRNA instability (Peltz et al, 1993). Further analysis showed that the heterogeneous nuclear RNP protein, Hrp1p, which is able to bind to the DSE in vitro, is required for NMD of PGK1 mRNA (Gonzalez et al, 2000). Thus, Hrp1p is considered as a downstream marker for NMD. However, it is not known whether DSEs and Hrp1p are required for the rapid decay of all PTC-bearing transcripts in yeast.
In mammalian cells, a large exon junction protein complex (EJC) deposited about 20–24 nucleotide (nt) upstream of exon–exon junctions during RNA splicing, is widely considered to be a mark that triggers NMD (Le Hir et al, 2000). Several lines of evidence support this. First, nonsense codons more than 55-nt upstream of the last intron generally trigger NMD, whereas nonsense codons inserted in the last exon do not (Zhang et al, 1998). Second, depletion of EJC components by RNA interference (RNAi) reduces the efficiency of NMD (Mendell et al, 2002; Palacios et al, 2004; Gehring et al, 2005; Kim et al, 2005; Chan et al, 2007). Third, the EJC remains associated with the mRNA while it enters the translating pool of mRNAs (Kim et al, 2001; Le Hir et al, 2001). Lastly, tethering of EJC components downstream of a normal stop codon triggers NMD (Lykke-Andersen et al, 2001; Gehring et al, 2003; Palacios et al, 2004). This model is also consistent with the observation that the normal stop codon in mammalian mRNAs generally occur in the last exon (Nagy and Maquat, 1998).
Although ample evidence supports its role in NMD, the EJC is not universally needed for NMD in mammalian cells (Zhang et al, 1998; Rajavel and Neufeld, 2001; Wang et al, 2002; LeBlanc and Beemon, 2004; Buhler et al, 2006). While in some cases an alternative downstream marker may exist, that does not appear to be so at least in the case of IGμ, (Buhler et al, 2006). Interestingly, although NMD is not conserved in prokaryotes, bacterial genes can undergo NMD when introduced into eukaryotes. For instance, a PTC-containing CAT mRNA can undergo NMD in flies (Gatfield et al, 2003) and a PTC-containing LacZ mRNA can undergo NMD in yeast (Keeling et al, 2004). Moreover, most EJC components are not conserved in S. cerevisiae. Although EJC components are conserved in D. melanogaster and C. elegans, NMD is splicing-independent in these organisms (Gatfield et al, 2003; Longman et al, 2007), suggesting that EJC does not play a role in NMD in these organisms. Thus, it appears that NMD can take place without a known downstream marker.
The aberrant termination model for NMD
The aberrant termination model (Figure 1B) depends on the notion that there is a difference in the translation termination events that occur in normal mRNAs and PTC-containing mRNAs (Amrani et al, 2006). According to this model, normal translation termination occurs at a native stop codon because of the close proximity of a normal 3′UTR, its associated factors, and/or poly(A) tail/PABP. This normal termination is proposed to prevent NMD from occurring. NMD substrates do not have a normal 3′UTR immediately downstream of the stop codon because translation stops in the coding region. The abnormal 3′ end does not permit proper remodeling steps required for normal translation termination. The difference between premature and normal translation termination is unclear. It is possible that translation termination is slower at premature stop codons (Hilleren and Parker, 1999) or termination may be biochemically distinct at normal and premature stop codon (Amrani et al, 2004). This idea is supported by the observations that the frequency of termination (versus translation read through) varies depending on the stop codon identity (UAA, UAG, or UGA) and the nucleotide following the stop codon and other mRNA features (Brown et al, 1990; Bonetti et al, 1995; McCaughan et al, 1995). Consistently, stop codons with low levels of read through caused NMD in yeast, whereas those with higher levels of read through did not (Keeling et al, 2004). A connection between the termination reaction and NMD was also revealed using in vitro translation extracts (Amrani et al, 2004). A toe-printing assay was able to detect a ribosome in the process of terminating at a PTC, but not at several normal stop codons. In addition, ribosomes stalled near PTCs could be detected in extracts made from a wild-type strain, but not from upf1- or upf2-mutant strains. Although these observations suggest that ribosomes associate more tightly with PTCs and/or are released slower from PTCs than from normal stop codons, it is unclear as to how this aberrancy results in NMD and whether it is a conserved feature of NMD.
A central question regarding the aberrant termination model concerns what feature of an mRNA triggers normal or aberrant translation termination. One possibility is that a proper spacing between the stop codon and proteins deposited at 3′UTR of mRNAs during 3′-end formation (e.g., PABP) is important for translation termination (Hilleren and Parker, 1999). This notion is supported by the observation that insertion of extra sequence in the 3′UTR of an mRNA can trigger NMD (Buhler et al, 2006; Behm-Ansmant et al, 2007). Interestingly, NMD can occur when the 3′UTR mRNPs and polyadenylation were generated independent of the normal cleavage and polyadenylation machinery (Baker and Parker, 2006; Behm-Ansmant et al, 2007). Several observations indicate that the protein factors associated with a stop codon, its downstream 3′UTR, and/or the poly(A) tail also play a critical role in determining a translation termination event. For example, tethering of PABP downstream of a PTC recruits the termination factor and rescues the stability of the mRNA (Amrani et al, 2004; Behm-Ansmant et al, 2007). Such stabilization was also observed by tethering PABP downstream of a normal stop codon of an otherwise unstable mRNA (Coller et al, 1998). In addition, when deadenylation in mammalian cells is impaired by knocking down Caf1 poly(A) nuclease or by overexpressing a Caf1 dominant-negative mutant, a PTC-containing mRNA is stabilized, presumably because PABPs remain associated with the unshortened poly(A) tail (N Ezzeddine, D Zheng, C-YA Chen, W Zhu, X He, and A-B Shyu, unpublished observations). Although these findings suggest that PABPs play an inhibitory role to prevent NMD from occurring, proper distinction between a normal stop codon and a PTC can occur in the absence of a poly(A) or PABP. For example, a PTC-containing mRNA harboring the 3′ end of a transcript that does not undergo polyadenylation (histone mRNA) is a substrate for NMD in mammals (Neu-Yilik et al, 2001). Similarly, in yeast, NMD can occur on an unadenylated mRNA or in a mutant that lacks PABP (Meaux et al, 2008). Nevertheless, it is worth noting that these observations are also consistent with the notion that the existence of PABPs prevents NMD from taking place.
While there is considerable support for the aberrant termination model, some observations cannot be explained by this model. For instance, the aberrant termination model does not easily account for the roles of DSEs and EJCs in NMD. Besides, in organisms which have long and heterogeneous 3′UTR length, it is more difficult to conceive of an important role of 3′UTR length.
Important features that may unify the two models for NMD
Neither the ‘downstream marker' model nor the ‘aberrant termination' model appear to apply to all cases of NMD. Nevertheless, both of them explain several critical features of NMD, most of which have to do with signals at or downstream of a PTC. Here, we envision a coherent model that integrates elements of each model to explain how PTCs are recognized by NMD (Figure 1C). It appears that multiple features (e.g., the nature of the stop codon UAA, UAG, or UGA, the nucleotide immediately following the stop codon, and the sequences, length, and associated proteins of 3′UTR) and factors (e.g., DSEs, EJC, PABP) influence the nature of the termination event. These features could work in opposing or dueling fashion (e.g., inhibit or stimulate normal or aberrant termination). It is likely that combination of various features would result in differences in translation termination and/or decay of mRNAs. Depending on the transcript, cell conditions, and/or experimental setup, some of these features may appear to be more important than others.
From premature termination to degradation
Once an mRNA is recognized as containing a PTC, how does this lead to its decay? One possibility is that a downstream marker may recruit mRNA decay enzymes to the mRNA by directly interacting with these enzymes (He and Jacobson, 1995). However, to our knowledge, there is no convincing evidence for this possibility. Another possibility for signaling mRNA degradation is that it depends on an mRNP-remodeling step between termination and the actual decay (Hilleren and Parker, 1999; Amrani et al, 2004). For example, a normal translation termination may result in a general remodeling of the mRNP that stabilizes the mRNA. In contrast, aberrant termination would fail to trigger remodeling or trigger an alternative mRNP-remodeling event, either of which could lead to mRNA degradation. One current challenge is to develop assays for mRNP structure that can test this model. Candidates that may mediate these remodeling events are the helicases and GTPase that have been reported to play important roles in mRNP remodeling (Jankowsky and Bowers, 2006; Small et al, 2006; Bleichert and Baserga, 2007). For example, it is possible that the helicase activity of Upf1 and/or the GTPase activity of eRF3 have key roles in the remodeling steps (Kashima et al, 2006). Since eRF3 is a PABP-interacting protein (Uchida et al, 2002), it is possible that the interaction between the Upf1–eRF1–eRF3 trimer and PABP prevents an aberrant mRNP remodeling.
An intermediate mRNP-remodeling step existing between translation termination and mRNA decay allows for versatility in how an mRNA is ultimately degraded by NMD. Thus, while the core of the NMD pathway appears to be conserved in all eukaryotes, the downstream consequences of PTC recognition appear to be different. In yeast, decapping is a major consequence of PTC recognition (i.e., the removal of the 5′-cap structure) (Muhlrad and Parker, 1994), whereas in flies, PTC recognition leads to endonucleolytic cleavage of the mRNA in the vicinity of the aberrant stop codon (Gatfield and Izaurralde, 2004). In other species, including mammals, PTC recognition leads to accelerated deadenylation (Cao and Parker, 2003; Chen and Shyu, 2003). Another feature about the proposed mRNP remodeling step is that the consequence of aberrant or normal termination may not be limited to one specific decay pathway. For instance, PTC recognition in yeast can increase decapping rate (Muhlrad and Parker, 1994), reduce translation (Muhlrad and Parker, 1999), or accelerate deadenylation (Cao and Parker, 2003) and subsequent degradation by the exosome (Cao and Parker, 2003; Mitchell and Tollervey, 2003). We conclude that mRNP remodeling directed by multiple features downstream of the stop codon play an important role in quality control of gene expression. This is a recurring theme in post-transcriptional regulation, including miRNA-mediated mRNA silencing, as described in the next section.
miRNA-mediated downregulation of gene expression
miRNAs are endogenous ∼22-nt non-coding RNAs that control fundamental cellular processes in animals and plants. In vertebrates, miRNA genes are one of the most abundant classes of regulatory genes (∼1% of all the genes) (Lim et al, 2003; Bartel, 2004; Bartel and Chen, 2004; Lim et al, 2005). After incorporation into the RNA-induced silencing complex (RISC), miRNAs guide the RNAi machinery to their target mRNAs by forming RNA duplexes, resulting in sequence-specific repression of productive translation or mRNA decay (Ambros, 2004; Bartel, 2004; Zamore and Haley, 2005). Regulation by miRNAs is typically mediated by the formation of imperfect hybrids with 3′UTR sequences of target mRNAs. A given miRNA targeted mRNA often has multiple miRNA target sites. Computational methods that have been developed to predict miRNA target genes suggest that 20–30% of protein-coding genes are likely targets of miRNAs (Lewis et al, 2003, 2005; Rajewsky, 2006).
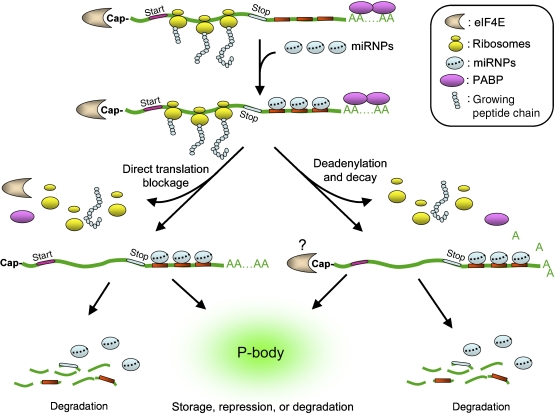
Initially, miRNAs were thought to down-regulate protein expression solely by inhibiting target mRNA translation (Olsen and Ambros, 1999; Seggerson et al, 2002). However, recent studies have indicated that many miRNAs can induce rapid decay of target mRNAs (Bagga et al, 2005; Lim et al, 2005; Behm-Ansmant et al, 2006; Giraldez et al, 2006; Wu et al, 2006; Eulalio et al, 2007c), which then indirectly reduces the amount of protein made. Thus, there are at least two general modes of miRNA-mediated downregulation of targets in metazoan cells: miRNA-mediated translational repression and miRNA-mediated RNA decay (Figure 2; Jackson and Standart, 2007; Nilsen, 2007; Pillai et al, 2007).
Figure 2.
Mechanisms of miRNA-mediated mRNA silencing. After incorporation into the RISC to form miRNPs, miRNAs guide the miRNPs to their target mRNAs by forming imperfect hybrids with 3′UTR sequences of target mRNAs. The interaction between a miRNP and its target mRNA can promote direct inhibition of translation initiation. Alternatively, the miRNP may accelerate deadenylation of the target mRNA, which in turn represses translation initiation or results in mRNA degradation. In P-bodies, miRNA-targeted mRNAs may be sequestered from the translational machinery and degraded or stored for subsequent use.
Mechanisms of miRNA-mediated translational repression
The mechanism of translational repression by miRNAs is still a matter of controversy. Two distinct mechanisms have been proposed to explain how miRNA-mediated translational repression is accomplished without affecting the abundance of target mRNAs. One hypothesizes that miRNAs inhibit translation initiation and the other hypothesizes inhibition of a ‘post-initiation' step in translation, which also elicits co-translational degradation of the nascent peptide. We refer readers to three recent excellent reviews on this controversial issue (Jackson and Standart, 2007; Nilsen, 2007; Pillai et al, 2007). Here, we focus on several new studies, all of which indicate that miRNAs can inhibit translation initiation.
It was found that the miRNA–RISC complex associated with an anti-translation initiation factor, eIF6, which inhibits joining of the 60S to the 40S subunits, thus preventing translation initiation (Chendrimada et al, 2007). Depleting eIF6 in either human cells or C. elegans effectively abolishes miRNA-mediated translational repression. In another study (Thermann and Hentze, 2007), a cell-free system was developed using D. melanogaster embryo extracts, which recapitulated translational repression mediated by the miRNA miR-2 without affecting mRNA stability. The authors found that the translational repression depended on the presence of a physiological cap structure, m7GpppG, at the 5′ of the mRNA substrate, a feature required for cap-dependent translation initiation in eukaryotes (Jacobson, 1996; Gingras et al, 1999). Intriguingly, miR-2 mRNPs co-sedimented with polyribosomes in a sucrose gradient but they did not possess features of a polyribosome. The miRNPs (heavier than 80S monosome) can still form when polyribosome formation and 60S ribosomal subunit joining are blocked, indicating the mRNAs associated with these miRNPs were not translated. In many past studies, it was assumed that the cosedimentation of miRNA-containing complexes with polysomes meant that these complexes contained ribosomes, but the study by Thermann and Hentze (2007) indicates that miRNA-mRNPs co-sedimented with polysomes are not necessarily being translated.
The observation (Kiriakidou et al, 2007) that Argonaute proteins, the catalytic components of RISC (Rand et al, 2005), contain a highly conserved motif binding to the m7G-cap structure also supports that miRNAs inhibit translation initiation. It is possible that Argonaute proteins compete with eIF4E for cap binding, thereby preventing the formation of eIF4F complex on the 5′-cap necessary for cap-dependent translation initiation. This is consistent with the observations that Ago2, but not its variant with mutations in the cap-binding motif, blocks translation when artificially tethered to the 3′UTR of mRNAs (Pillai et al, 2004). In the other two studies, let-7 miRNA-mediated translational repression was recapitulated in two different cell-free systems established with extracts prepared from either mouse Krebs-2 ascitic cells (Mathonnet et al, 2007) or human HEK293F cells over-expressing miRNA pathway components (Wakiyama et al, 2007). In these systems, the poly(A) tail and 5′-cap are both required for the translational repression, suggesting that let-7 represses translation by impairing the synergistic enhancement of translation by the 5′-cap and 3′ poly(A) tail. Collectively, these in vivo and in vitro studies support that inhibition of translation initiation by miRNAs represents one way by which miRNA-mediated translational repression is achieved.
miRNA-mediated RNA decay
Although similar in length, miRNAs are generated by a distinct mechanism from that producing small interfering RNA (siRNA). siRNAs are chopped from long dsRNAs by Dicer (Bernstein et al, 2001). The antisense strand of the siRNA is assembled into RISC, which then degrades RNA molecules with sequences completely complementary to the siRNA by endonucleolytic cleavage (reviewed in Hannon, 2002; Dykxhoorn et al, 2003). On the other hand, miRNAs are derived from native genes and form imperfect matches with target mRNAs that do not elicit endonucleolytic cleavage of target mRNAs (Ambros, 2004; Bartel, 2004). Instead, recent evidence indicates that miRNA-mediated decay can be triggered by deadenylation (see below).
A general picture of miRNA-mediated RNA decay emerges from recent studies in D. melanogaster cells (Behm-Ansmant et al, 2006), zebrafish embryos (Giraldez et al, 2006), and human cells (Wu et al, 2006), namely, mRNAs targeted by miRNAs for degradation undergo prior deadenylation. In zebrafish, miRNA miR-430 was shown to target several hundred maternal mRNAs for decay by first triggering rapid deadenylation. This massive destruction of maternal mRNAs is required to silence maternal mRNA expression into proteins so that the embryo development of zebrafish can proceed. This example well illustrates how gene silencing by miRNA is accomplished mainly at the level of mRNA decay triggered by deadenylation. In Drosophila cells, deadenylation is mediated by the Ccr4–Caf1–Not poly(A) nuclease complex (Behm-Ansmant et al, 2006). However, detailed mechanism of miRNA-induced deadenylation and participating poly(A) nucleases and many issues related to miRNA-induced mRNA decay in other organisms remain to be addressed. Given that miRNA-induced deadenylation does not necessarily lead to decay of the RNA body (Behm-Ansmant et al, 2006), it is possible that deadenylation is one way on which different modes of miRNA-mediated mRNA silencing, including miRNA-mediated translational repression and miRNA-mediated RNA decay, can converge. Because the mechanisms of only a few miRNAs have so far been characterized in detail, the generality of any mode of miRNA-mediated mRNA silencing remains to be seen.
The role of deadenylation in miRNA-mediated translational repression
Cytoplasmic PABP proteins interact with both poly(A) tails and the eIF4F complex bound to the 5′ cap, thereby bringing the two ends of the mRNA together (Kahvejian et al, 2001; Mangus et al, 2003). Because this interaction is important for both translation initiation and mRNA stability (Jacobson, 1996), it is not surprising that poly(A) tails are crucial for mRNA stability and in translation initiation. As one major stage at which miRNAs repress translation is the initiation step, it is possible that promoting deadenylation by a miRNP formed on the target mRNA to disrupt 5′–3′ end interaction may represent an effective and immediate way of reducing translation initiation.
Several observations suggest that deadenylation is a cause, but not a consequence, of miRNA-mediated translational repression, particularly at initiation step. Blocking translation initiation by a stem-loop in the 5′UTR of the target mRNA does not abolish its rapid deadenylation and decay induced by miRNA (Wu et al, 2006). Mishima et al (2006) showed that miR-430 directs the deadenylation and translational repression of nanos1 mRNA during zebrafish embryogenesis (Mishima et al, 2006). When the miR-430 target mRNA was provided a non-natural ApppG cap, which significantly impairs normal translation initiation, the rapid deadenylation was unaffected. Using a cell-free system, Wakiyama and co-workers showed that deadenylation triggered by the miRNA let-7 does not require active translation, and can proceed in the presence of cycloheximide, a potent translation inhibitor. Moreover, let-7-mediated deadenylation is independent of the structure of the mRNA 5′-terminus, while the cap and the poly(A) tail are both required for the translational repression by let-7 (Wakiyama et al, 2007). These observations suggest that let-7 miRNAs recruit miRNP complexes to let-7 target mRNAs, resulting in deadenylation, which in turn abolishes the cap-poly(A) synergy, thereby repressing translation initiation. This is reminiscent of the mechanism by which translation repression of maternal mRNAs is accomplished by shortening of the poly(A) tail during Xenopus laevis oocyte maturation (Richter, 1996; Gray and Wickens, 1998).
Although several studies support the idea that accelerated deadenylation induced by miRNAs represents a major way to repress the translation of target mRNAs without affecting mRNA stability, it is unlikely to be the universal mechanism by which this is achieved. For example, in D. melanogaster cells, blocking mRNA deadenylation by knocking down Caf1 poly(A) nuclease complex does not relieve miRNA-mediated translational repression (Behm-Ansmant et al, 2006). In this case, it appears that translational repression and deadenylation are two independent events in miRNA-mediated mRNA silencing. Furthermore, Wu et al (2006) showed that translation of mRNAs lacking a poly(A) tail remains repressed by miRNAs, indicating that deadenylation is not the cause of miRNA-mediated translational silencing in this case. Alternatively, it is possible that when deadenylation is impaired, an alternative fail-safe mechanism that can also effectively block translation initiation (e.g., decapping) is activated to bypass the requirement for deadenylation (Eulalio et al, 2007c).
The role of P-bodies in mRNA quality control
P-bodies are specific cytoplasmic foci that contain proteins known to function in mRNA metabolism (Kedersha and Anderson, 2007; Parker and Sheth, 2007; Eulalio et al, 2007a). These foci are also referred to as GW bodies as they carry GW182 proteins that are required for miRNA-mediated translational repression (Eystathioy et al, 2002; Jakymiw et al, 2005; Meister et al, 2005; Rehwinkel et al, 2005; Liu et al, 2005a; Behm-Ansmant et al, 2006). The function of P-bodies is not yet fully understood, but it is clear that the mRNA in P-bodies can either be degraded or re-enter the translating pool of mRNAs. One important aspect of P-body's protein composition is the presence of enzymes, which promote mRNA decay, including the deadenylase CCR4 (Sheth and Parker, 2003; Andrei et al, 2005) and the DCP1–DCP2 decapping complex (Ingelfinger et al, 2002; Sheth and Parker, 2003). As P-bodies contain the 5′–3′ exonuclease XRN1 (Ingelfinger et al, 2002; Sheth and Parker, 2003) but lack the exosome complex (which contains 3′–5′ exonucleases), it is likely that mRNAs are degraded via 5′ to 3′ decay pathway in P-bodies. P-bodies lack ribosomal components, most translation initiation factors, and PABP, which supports the notion that P-bodies are sites of translational repression. This feature of P-bodies also indicates that ribosomes, PABP, and translation initiation factors must dissociate from mRNPs before or immediately after they enter or aggregate to form P-bodies.
In addition to general decay factors, factors required for NMD (Upf1, Upf2, Upf3, Smg5, and Smg7), as well as PTC-containing mRNAs, are found in P-bodies (Unterholzner and Izaurralde, 2004; Sheth and Parker, 2006). The first NMD factor shown to localize to P-bodies was the human Smg7 protein (Unterholzner and Izaurralde, 2004). As Smg7 is known to bind phosphorylated Upf1 (Kashima et al, 2006), it is possible that after Upf1 detects a PTC-containing mRNA, the interaction between Smg7 and phosphorylated Upf1 targets the NMD substrate to P-bodies for subsequent mRNA degradation. In yeast, Upf1, Upf2, and Upf3 localize to P-bodies, and Upf1 localization is enhanced in upf2 and upf3 mutants (Sheth and Parker, 2006). Collectively, these observations suggest that NMD can occur in P-bodies.
P-bodies also contain protein factors involved in miRNA-mediated translational repression, including the Argonaute proteins, Rck/p54, and GW182 (reviewed in Kedersha and Anderson, 2007; Parker and Sheth, 2007; Eulalio et al, 2007a). Depleting Rck/p54 leads to a loss of P-bodies and a defect in miRNA-mediated translational repression (Chu and Rana, 2006) and miRNA-mediated mRNA decay (Eulalio et al, 2007c), suggesting that P-bodies and miRNA-mediated events are inter-related. However, it is clear that P-bodies are not absolutely required for miRNA function, as depletion of Lsm1 or GW182 in human cells and D. melanogaster cells, which causes a loss of P-bodies and disperses Argonaute proteins throughout the cell, does not affect miRNA function (Chu and Rana, 2006; Stoecklin et al, 2006; Eulalio et al, 2007b). Moreover, it has been reported that miRNAs are associated with polysomes, which seems inconsistent with the notion that P-bodies are required to keep miRNA–mRNPs translationally silenced (Nelson et al, 2004; Maroney et al, 2006; Nottrott et al, 2006). Thus, although there clearly is a close link between P-bodies and miRNA-mediated translation repression, the precise nature of this link remains to be determined.
We suggest that rather than being required for mRNA decay and translational repression, P-bodies increase the efficiency of these events. One possibility is that concentrating repressed mRNPs in P-bodies facilitates additional mRNP-remodeling steps, which reinforce this repression for long-term storage in a repressed form. In other cases, these remodeling events may trigger more efficient mRNA degradation. Sequestration in P-bodies may also provide a rapid means to prevent accidental translation of aberrant mRNAs, such as PTC-containing transcripts, prior to degradation. Moreover, as mRNAs may leave P-bodies and re-enter the translating pool (Brengues et al, 2005; Bhattacharyya et al, 2006), P-bodies could function as temporary storage sites for repressed mRNAs. Thus, P-bodies have the potential to regulate gene expression under various conditions and also provide an additional quality-control point where mRNA that has been mistakenly repressed can be reactivated. In so doing, P-bodies provide an additional layer for fine-tuning gene expression to maintain cellular homeostasis.
Common and distinct features of NMD and miRNA-mediated silencing
NMD and miRNA-mediated silencing have common features, but they clearly differ in many respects. Both occur in the cytoplasm and result in mRNA degradation, but miRNAs have the additional ability to inhibit translation, which provides for the possibility of reversible repression. Mammalian NMD is facilitated by nuclear processing events that deposit the EJC signal (Chang et al, 2007), whereas it is not clear whether miRNA-mediated silencing requires nuclear events other than the Drosha-mediated cleavage that generates miRNA precursors (Lee et al, 2006). Both NMD and miRNA-mediated silencing appear to require sequential mRNP-remodeling steps, raising the possibility that they may use common remodeling events, but this will not be known until these steps are better defined. A clear difference between the two is that NMD absolutely requires translation to define the PTC, whereas miRNA-mediated mRNA decay can occur in the absence of translation. Both NMD and miRNA-mediated silencing appear to be able to take place in P-bodies (Liu et al, 2005b; Sheth and Parker, 2006), but the proportion of these two events that occurs in P-bodies versus other cytoplasmic sites may be quite different, as inhibition of P-body formation down-regulates NMD but has no obvious impact on miRNA-mediated silencing (Chu and Rana, 2006; Eulalio et al, 2007b). Finally, both NMD and miRNA-mediated mRNA silencing can use deadenylation as a crucial step toward mediating their effects, but both can also use deadenylation-independent pathways, possibly as a fail-safe mechanism, to achieve their goals (Yamashita et al, 2005; Behm-Ansmant et al, 2006; Wu et al, 2006).
Future directions
There are many issues in the field that require addressing and clarifying as to how miRNAs determine whether to exert their action through translational repression or mRNA decay as well as how mRNPs are remodeled and what changes in mRNP components occur during NMD and miRNA-mediated mRNA silencing. One key issue is to develop methods to independently examine the many steps required for NMD and miRNA-mediated silencing. It now is apparent that there are multiple separable steps in NMD, including the recognition of the PTC, remodeling of the mRNP, targeting to P-bodies, and mRNA decay. Therefore, simply monitoring steady state level of total mRNA, including both nuclear and cytoplasmic mRNA, is insufficient to address these challenging issues. More attention should be paid to monitoring decay kinetics and studying precursor-production relationship by methods such as the transcriptional pulsing (Yamashita et al, 2005; Chen et al, 2007). While Hrp1p, eRF3, PABP, and EJC factors probably serve to distinguish normal stop codons from PTCs, they could also act on downstream events, including the mRNA degradation event itself. For example, analysis of translation termination in yeast in vitro translation extracts indicates that Upf1p and Upf2p are required in the PTC recognition step (Amrani et al, 2004). Also, it has been shown that Upf1 preferentially associates with NMD substrates in vivo in worms and S. pombe (Rodriguez-Gabriel et al, 2006; Johns et al, 2007). A major challenge for the future will be to clarify the roles of each NMD factor in the various steps of NMD.
A key unanswered question regarding miRNAs is what determines whether they will trigger mRNA decay or translational repression. It is possible that the primary effect of the miRNA machinery is to remodel the mRNP to either avoid forming or disrupt a closed loop structure between 5′-cap and 3′ poly(A) tail that is critical for translation initiation. The subsequent downstream effects of mRNP remodeling may vary depending on physiological conditions, developmental cues, and other factors. In some cases, miRNA-targeted mRNAs may be subjected for rapid degradation, whereas in other cases, they may be simply repressed for translation and stored in P-bodies until needed later; for example, during cellular stress response. Since P-bodies are not absolutely necessary for miRNA-mediated mRNA silencing and normal mRNA decay, a close examination of P-body status during embryogenesis, cell growth and differentiation, and various diseased states may shed new light on the physiological function and significance of P-bodies in regulating gene expression.
The importance of mRNP remodeling may be revealed further by studying when and how PABPs dissociate from an mRNP. This is a particularly critical issue that has not been addressed since PABPs are not present in P-bodies. PABP exhibits a very high binding affinity for its RNA substrate (in the nanomolar range) (Görlach et al, 1994; Kuhn and Pieler, 1996; Deardorff and Sachs, 1997) and thus removal of PABP is particularly challenging if mRNP remodeling exerts its effect on translational repression per se without deadenylation. This raises a question as to what drives PABPs off the P-body entrapped mRNPs so that they can enter existing or form new P-bodies, a key step determining their fate. Future research addressing the key changes in mRNP composition at each critical remodeling step of an mRNA, as it goes on its journey from the nucleus to the cytoplasm, will be crucial for understanding how mRNA decay, translation, and RNA quality-control mechanisms are regulated through an interplay of different mechanisms.
Acknowledgments
We thank Chyi-Ying A Chen for critical reading and valuable comments on the paper, and Nader Ezzeddine for assistance with artwork.
References
- Ambros V (2004) The functions of animal microRNAs. Nature 431: 350–355 [DOI] [PubMed] [Google Scholar]
- Amrani N, Ganesan R, Kervestin S, Mangus DA, Ghosh S, Jacobson A (2004) A faux 3′-UTR promotes aberrant termination and triggers nonsense-mediated mRNA decay. Nature 432: 112–118 [DOI] [PubMed] [Google Scholar]
- Amrani N, Sachs MS, Jacobson A (2006) Early nonsense: mRNA decay solves a translational problem. Nat Rev Mol Cell Biol 7: 415–425 [DOI] [PubMed] [Google Scholar]
- Andrei MA, IIngelfinger D, Heintzmann R, Achsel T, Rivera-Pomar R, Luhrmann R (2005) A role for eIF4E and eIF4E-transporter in targeting mRNPs to mammalian processing bodies. RNA 11: 717–727 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arciga-Reyes L, Wootton L, Kieffer M, Davies B (2006) UPF1 is required for nonsense-mediated mRNA decay (NMD) and RNAi in Arabidopsis. Plant J 47: 480–489 [DOI] [PubMed] [Google Scholar]
- Aronoff R, Baran R, Hodgkin J (2001) Molecular identification of smg-4, required for mRNA surveillance in C. elegans. Gene 268: 153–164 [DOI] [PubMed] [Google Scholar]
- Bagga S, Bracht J, Hunter S, Massirer K, Holtz J, Eachus R, Pasquinelli AE (2005) Regulation by let-7 and lin-4 miRNAs results in target mRNA degradation. Cell 122: 553–563 [DOI] [PubMed] [Google Scholar]
- Baker KE, Parker R (2006) Conventional 3′ end formation is not required for NMD substrate recognition in Saccharomyces cerevisiae. RNA 12: 1441–1445 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bartel DP (2004) MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116: 281–297 [DOI] [PubMed] [Google Scholar]
- Bartel DP, Chen CZ (2004) Micromanagers of gene expression: the potentially widespread influence of metazoan microRNAs. Nat Rev Genet 5: 396–400 [DOI] [PubMed] [Google Scholar]
- Behm-Ansmant I, Gatfield D, Rehwinkel J, Hilgers V, Izaurralde E (2007) A conserved role for cytoplasmic poly(A)-binding protein 1 (PABPC1) in nonsense-mediated mRNA decay. EMBO J 26: 1591–1601 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Behm-Ansmant I, Rehwinkel J, Doerks T, Stark A, Bork P, Izaurralde E (2006) mRNA degradation by miRNAs and GW182 requires both CCR4:NOT deadenylase and DCP1:DCP2 decapping complexes. Genes Dev 20: 1885–1898 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bernstein E, Caudy AA, Hammond SM, Hannon GJ (2001) Role for a bidentate ribonuclease in the initiation step of RNA interference. Nature 409: 363–366 [DOI] [PubMed] [Google Scholar]
- Bhattacharya A, Czaplinski K, Trifillis P, He F, Jacobson A, Peltz SW (2000) Characterization of the biochemical properties of the human Upf1 gene product that is involved in nonsense-mediated mRNA decay. RNA 6: 1226–1235 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhattacharyya SN, Habermacher R, Martine U, Closs EI, Filipowicz W (2006) Relief of microRNA-mediated translation repression in human cells subjected to stress. Cell 125: 1111–1124 [DOI] [PubMed] [Google Scholar]
- Bleichert F, Baserga SJ (2007) The long unwinding road of RNA helicases. Mol Cell 27: 339–352 [DOI] [PubMed] [Google Scholar]
- Bonetti B, Fu L, Moon J, Bedwell DM (1995) The efficiency of translation termination is determined by a synergistic interplay between upstream and downstream sequences in Saccharomyces cerevisiae. J Mol Biol 251: 334–345 [DOI] [PubMed] [Google Scholar]
- Brengues M, Teixeira D, Parker R (2005) Movement of eukaryotic mRNAs between polysomes and cytoplasmic processing bodies. Science 310: 486–489 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brogna S (1999) Nonsense mutations in the alcohol dehydrogenase gene of Drosophila melanogaster correlate with an abnormal 3′ end processing of the corresponding pre-mRNA. RNA 5: 562–573 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown CM, Stockwell PA, Trotman CNA, Tate WP (1990) Sequence analysis suggests that tetra-nucleotides signal the termination of protein synthesis in eukaryotes. Nucleic Acids Res 18: 6339–6345 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buhler M, Steiner S, Mohn F, Paillusson A, Muhlemann O (2006) EJC-independent degradation of nonsense immunoglobulin-mu mRNA depends on 3′ UTR length. Nat Struct Mol Biol 13: 462–464 [DOI] [PubMed] [Google Scholar]
- Cao D, Parker R (2003) Computational modeling and experimental analysis of nonsense-mediated decay in yeast. Cell 113: 533–545 [DOI] [PubMed] [Google Scholar]
- Chan WK, Huang L, Gudikote JP, Chang YF, Imam JS, MacLean JA II, Wilkinson MF (2007) An alternative branch of the nonsense-mediated decay pathway. EMBO J 26: 1820–1830 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang Y-F, Imam JS, Wilkinson MF (2007) The nonsense-mediated decay RNA surveillance pathway. Ann Rev Biochem 76: 51–74 [DOI] [PubMed] [Google Scholar]
- Chen C-YA, Shyu A-B (2003) Rapid deadenylation triggered by a nonsense codon precedes decay of the RNA body in a mammalian cytoplasmic nonsense-mediated decay pathway. Mol Cell Biol 23: 4805–4813 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen C-YA, Yamashita Y, Chang T-C, Yamashita A, Zhu W, Zhong Z, Shyu A-B (2007) Versatile applications of transcriptional pulsing to study mRNA turnover in mammalian cells. RNA 13: 1775–1786 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chendrimada TP, Finn KJ, Ji X, Baillat D, Gregory RI, Liebhaber SA, Pasquinelli AE, Shiekhattar R (2007) MicroRNA silencing through RISC recruitment of eIF6. Nature 447: 823–828 [DOI] [PubMed] [Google Scholar]
- Chu CY, Rana TM (2006) Translation repression in human cells by microRNA-induced gene silencing requires RCK/p54. PLoS Biol 4: e210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coller JM, Gray NK, Wickens MP (1998) mRNA stabilization by poly(A) binding protein is independent of poly(A) and requires translation. Genes Dev 12: 3226–3235 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Culbertson MR, Underbrink KM, Fink GR (1980) Frameshift suppression in Saccharomyces cerevisiae. II. Genetic properties of group II suppressors. Genetics 95: 833–853 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Czaplinski K, Weng Y, Hagan KW, Peltz SW (1995) Purification and characterization of the Upf1 protein: a factor involved in translation and mRNA degradation. RNA 1: 610–623 [PMC free article] [PubMed] [Google Scholar]
- Deardorff JA, Sachs AB (1997) Differential effects of aromatic and charged residue substitutions in the RNA binding domains of the yeast poly(A)-binding protein. J Mol Biol 269: 67–81 [DOI] [PubMed] [Google Scholar]
- Dreyfuss G, Kim VN, Kataoka N (2003) Messenger-RNA-binding proteins and the messages they carry. Nat Rev Mol Cell Biol 3: 195–205 [DOI] [PubMed] [Google Scholar]
- Dykxhoorn DM, Novina CD, Sharp PA (2003) Killing the messenger: short RNAs that silence gene expression. Nat Rev Mol Cell Biol 4: 457–467 [DOI] [PubMed] [Google Scholar]
- Eulalio A, Behm-Ansmant I, Izaurralde E (2007a) P bodies: at the crossroads of post-transcriptional pathways. Nat Rev Mol Cell Biol 8: 9–22 [DOI] [PubMed] [Google Scholar]
- Eulalio A, Behm-Ansmant I, Schweizer D, Izaurralde E (2007b) P-body formation is a consequence, not the cause, of RNA-mediated gene silencing. Mol Cell Biol 27: 3970–3981 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eulalio A, Rehwinkel J, Stricker M, Huntzinger E, Yang S-F, Doerks T, Dorner S, Bork P, Boutros M, Izaurralde E (2007c) Target-specific requirements for enhancers of decapping in miRNA-mediated gene silencing. Genes Dev 21: 2558–2570 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eystathioy T, Chan EKL, Tenenbaum SA, Keene JD, Griffith K, Fritzler MJ (2002) A phosphorylated cytoplasmic autoantigen, GW182, associates with a unique population of human mRNAs within novel cytoplasmic speckles. Mol Biol Cell 13: 1338–1351 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frischmeyer PA, Dietz HC (1999) Nonsense-mediated mRNA decay in health and disease. Hum Mol Genet 8: 1893–1900 [DOI] [PubMed] [Google Scholar]
- Gatfield D, Izaurralde E (2004) Nonsense-mediated messenger RNA decay is initiated by endonucleolytic cleavage in Drosophila. Nature 429: 575–578 [DOI] [PubMed] [Google Scholar]
- Gatfield D, Unterholzner L, Ciccarelli FD, Bork P, Izaurralde E (2003) Nonsense-mediated mRNA decay in Drosophila: at the intersection of the yeast and mammalian pathways. EMBO J 22: 3960–3970 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gehring K, Neu-Yilik G, Schell T, Hentze MW, Kulozik AE (2003) Y14 and hUpf3b form an NMD-activating complex. Mol Cell 11: 939–949 [DOI] [PubMed] [Google Scholar]
- Gehring NH, Kunz JB, Neu-Yilik G, Breit S, Viegas MH, Hentze MW, Kulozik AE (2005) Exon-junction complex components specify distinct routes of nonsense-mediated mRNA decay with differential cofactor requirements. Mol Cell 20: 65–75 [DOI] [PubMed] [Google Scholar]
- Gingras A-C, Raught B, Sonenberg N (1999) eIF4 initiation factors: effectors of mRNA recruitment to ribosomes and regulators of translation. Annu Rev Biochem 68: 913–963 [DOI] [PubMed] [Google Scholar]
- Giraldez AJ, Mishima Y, Rihel J, Grocock RJ, Van Dongen S, Inoue K, Enright AJ, Schier AF (2006) Zebrafish MiR-430 promotes deadenylation and clearance of maternal mRNAs. Science 312: 75–79 [DOI] [PubMed] [Google Scholar]
- Gonzalez CI, Ruiz-Echevarria MJ, Vasudevan S, Henry MF, Peltz MF (2000) The yeast hnRNP-like protein Hrp1/Nab4 marks a transcript for nonsense-mediated mRNA decay. Mol Cell 5: 489–499 [DOI] [PubMed] [Google Scholar]
- Görlach M, Burd CG, Dreyfuss G (1994) The mRNA poly(A)-binding protein: localization, abundance, and RNA-binding specificity. Exp Cell Res 211: 400–407 [DOI] [PubMed] [Google Scholar]
- Gray NK, Wickens M (1998) Control of translation initiation in animals. Annu Rev Cell Dev Biol 14: 399–458 [DOI] [PubMed] [Google Scholar]
- Hannon GJ (2002) RNA interference. Nature 418: 244–251 [DOI] [PubMed] [Google Scholar]
- He F, Jacobson A (1995) Identification of a novel component of the nonsense-mediated mRNA decay pathway by use of an interacting protein screen. Genes Dev 9: 437–454 [DOI] [PubMed] [Google Scholar]
- Hilleren P, Parker R (1999) mRNA surveillance in eukaryotes: kinetic proofreading of proper translation termination as assessed by mRNP domain organization? RNA 5: 711–719 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hodgkin J, Papp A, Pulak R, Ambros V, Anderson P (1989) A new kind of informational suppression in the nematode Caenorhabditis elegans. Genetics 123: 301–313 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holbrook JA, Neu-Yilik G, Hentze MW, Kulozik AE (2004) Nonsense-mediated decay approaches the clinic. Nat Genet 36: 801–808 [DOI] [PubMed] [Google Scholar]
- Hori K, Watanabe Y (2005) UPF3 suppresses aberrant spliced mRNA in Arabidopsis. Plant J 43: 530–540 [DOI] [PubMed] [Google Scholar]
- Ingelfinger D, Arndt-Jovin DJ, Luhrmann R, Achsel T (2002) The human LSm1-7 proteins colocalize with the mRNA-degrading enzymes Dcp1/2 and Xrnl in distinct cytoplasmic foci. RNA 8: 1489–1501 [PMC free article] [PubMed] [Google Scholar]
- Jackson RJ, Standart N (2007) How do microRNAs regulate gene expression? Sci STKE 2007: re1. [DOI] [PubMed] [Google Scholar]
- Jacobson A (1996) Poly(A) metabolism and translation: the colsed-loop model. In Tanslational Control, Hershey JWB, Mathews MB, Sonenberg N (eds), pp 451–480. Plainview: Cold Spring Harbor Laboratory Press [Google Scholar]
- Jakymiw A, Lian S, Eystathioy T, Li S, Satoh M, Hamel JC, Fritzler MJ, Chan EK (2005) Disruption of GW bodies impairs mammalian RNA interference. Nat Cell Biol 8: 1267–1274 [DOI] [PubMed] [Google Scholar]
- Jankowsky E, Bowers H (2006) Remodeling of ribonucleoprotein complexes with DExH/D RNA helicases. Nucleic Acids Res 34: 4181–4188 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johns L, Grimson A, Kuchma SL, Newman CL, Anderson P (2007) Caenorhabditis elegans SMG-2 selectively marks mRNAs containing premature translation termination codons. Mol Cell Biol 27: 5630–5638 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kahvejian A, Roy G, Sonenberg N (2001) The mRNA closed-loop model: the function of PABP and PABP-interacting proteins in mRNA translation. Cold Spring Harb Symp Quant Biol 66: 293–300 [DOI] [PubMed] [Google Scholar]
- Kashima I, Yamashita A, Izumi N, Kataoka N, Morishita R, Hoshino S, Ohno M, Dreyfuss G, Ohno S (2006) Binding of a novel SMG-1–Upf1–eRF1–eRF3 complex (SURF) to the exon junction complex triggers Upf1 phosphorylation and nonsense-mediated mRNA decay. Genes Dev 20: 355–367 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kedersha N, Anderson P (2007) Mammalian stress granules and processing bodies. Methods Enzymol 431: 61–81 [DOI] [PubMed] [Google Scholar]
- Keeling KM, Lanier J, Du M, Salas-Marco JOE, Gao LIN, Kaenjak-Angeletti A, Bedwell DM (2004) Leaky termination at premature stop codons antagonizes nonsense-mediated mRNA decay in S. cerevisiae. RNA 10: 691–703 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim VN, Yong J, Kataoka N, Abel L, Diem MD, Dreyfuss G (2001) The Y14 protein communicates to the cytoplasm the position of exon–exon junctions. EMBO J 20: 2062–2068 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim YK, Furic L, Desgroseillers L, Maquat LE (2005) Mammalian Staufen1 recruits Upf1 to specific mRNA 3′UTRs so as to elicit mRNA decay. Cell 120: 195–208 [DOI] [PubMed] [Google Scholar]
- Kiriakidou M, Tan GS, Lamprinaki S, De Planell-Saguer M, Nelson PT, Mourelatos Z (2007) An mRNA m7G cap binding-like motif within human Ago2 represses translation. Cell 129: 1141–1151 [DOI] [PubMed] [Google Scholar]
- Kuhn U, Pieler T (1996) Xenopus poly(A) binding protein: functional domains in RNA binding and protein–protein interaction. J Mol Biol 256: 20–30 [DOI] [PubMed] [Google Scholar]
- Le Hir H, Gatfield D, Izaurralde E, Moore MJ (2001) The exon–exon junction complex provides a binding platform for factors involved in mRNA export and nonsense-mediated mRNA decay. EMBO J 20: 4987–4997 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Le Hir H, Moore MJ, Maquat LE (2000) Pre-mRNA splicing alters mRNP composition: evidence for stable association of proteins at exon–exon junctions. Genes Dev 14: 1098–1108 [PMC free article] [PubMed] [Google Scholar]
- LeBlanc JJ, Beemon KL (2004) Unspliced rous sarcoma virus genomic RNAs are translated and subjected to nonsense-mediated mRNA decay before packaging. J Virol 78: 5139–5146 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee Y, Han J, Yeom KH, Jin H, Kim VN (2006) Drosha in primary MicroRNA processing. Cold Spring Harb Symp Quant Biol 71: 51–57 [DOI] [PubMed] [Google Scholar]
- Lewis BP, Burge CB, Bartel DP (2005) Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell 120: 15–20 [DOI] [PubMed] [Google Scholar]
- Lewis BP, Shih IH, Jones-Rhoades MW, Bartel DP, Burge CB (2003) Prediction of mammalian microRNA targets. Cell 115: 787–798 [DOI] [PubMed] [Google Scholar]
- Lim LP, Glasner ME, Yekta S, Burge CB, Bartel DP (2003) Vertebrate miRNA genes. Science 299: 1540. [DOI] [PubMed] [Google Scholar]
- Lim LP, Lau NC, Garrett-Engele P, Grimson A, Schelter JM, Castle J, Bartel DP, Linsley PS, Johnson JM (2005) Microarray analysis shows that some microRNAs downregulate large numbers of target mRNAs. Nature 433: 769–773 [DOI] [PubMed] [Google Scholar]
- Liu J, Rivas FV, Wohlschlegel J, Yates JR III, Parker R, Hannon GJ (2005a) A role for the P-body component GW182 in microRNA function. Nat Cell Biol 7: 1261–1266 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu J, Valencia-Sanchez MA, Hannon GJ, Parker R (2005b) MicroRNA-dependent localization of targeted mRNAs to mammalian P-bodies. Nat Cell Biol 7: 719–723 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Longman D, Plasterk RHA, Johnstone IL, Caceres JF (2007) Mechanistic insights and identification of two novel factors in the C. elegans NMD pathway. Genes Dev 21: 1075–1085 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Losson R, Lacroute F (1979) Interference of nonsense mutations with eukaryotic messenger RNA stability. Proc Natl Acad Sci USA 76: 5134–5137 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lykke-Andersen J, Shu MD, Steitz JA (2001) Communication of the position of exon–exon junctions to the mRNA surveillance machinery by the protein RNPS1. Science 293: 1836–1839 [DOI] [PubMed] [Google Scholar]
- Mangus DA, Evans MC, Jacobson A (2003) Poly(A)-binding proteins: multifunctional scaffolds for the post-transcriptional control of gene expression. Genome Biol 4: 233 http://genomebiology.com/2003/2004/ 2007/2223 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maquat LE (2004) Nonsense mediated mRNA decay: splicing, translation and mRNP dynamics. Nat Rev Mol Cell Biol 5: 89–99 [DOI] [PubMed] [Google Scholar]
- Maquat LE, Kinniburgh AJ, Rachmilewitz EA, Ross J (1981) Unstable beta-globin mRNA in mRNA-deficient beta o thalassemia. Cell 27: 543–553 [DOI] [PubMed] [Google Scholar]
- Maroney PA, Yu Y, Fisher J, Nilsen TW (2006) Evidence that microRNAs are associated with translating messenger RNAs in human cells. Nat Struct Mol Biol 13: 1102–1107 [DOI] [PubMed] [Google Scholar]
- Mathonnet G, Fabian MR, Svitkin YV, Parsyan A, Huck L, Murata T, Biffo S, Merrick WC, Darzynkiewicz E, Pillai RS, Filipowicz W, Duchaine TF, Sonenberg N (2007) MicroRNA inhibition of translation initiation in vitro by targeting the cap-binding complex eIF4F. Science 317: 1764–1767 [DOI] [PubMed] [Google Scholar]
- McCaughan KK, Brown CM, Dalphin ME, Berry MJ, Tate WP (1995) Translational termination efficiency in mammals is influenced by the base following the stop codon. Proc Natl Acad Sci USA 92: 5431–5435 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meaux S, van Hoof A, Baker KE (2008) Nonsense-mediated mRNA decay in yeast does not require PAB1 or a poly(A) tail. Mol Cell (in press) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meister G, Landthaler M, Peters L, Chen PY, Urlaub H, Luhrmann R, Tuschl T (2005) Identificiation of novel argonaute-associated proteins. Curr Biol 15: 2149–2155 [DOI] [PubMed] [Google Scholar]
- Mendell JT, ap Rhys CM, Dietz HC (2002) Separable roles for rent1/hUpf1 in altered splicing and decay of nonsense transcripts. Science 298: 419–422 [DOI] [PubMed] [Google Scholar]
- Mishima Y, Giraldez AJ, Takeda Y, Fujiwara T, Sakamoto H, Schier AF, Inoue K (2006) Differential regulation of germline mRNAs in soma and germ cells by zebrafish miR-430. Curr Biol 16: 2135–2142 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitchell P, Tollervey D (2003) An NMD pathway in yeast involving accelerated deadenylation and exosome-mediated 3′ → 5′ degradation. Mol Cell 11: 1405–1413 [DOI] [PubMed] [Google Scholar]
- Moore MJ (2005) From birth to death: the complex lives of eukaryotic mRNAs. Science 309: 1514–1518 [DOI] [PubMed] [Google Scholar]
- Muhlrad D, Parker R (1994) Premature translational termination triggers mRNA decapping. Nature 370: 578–581 [DOI] [PubMed] [Google Scholar]
- Muhlrad D, Parker R (1999) Recognition of yeast mRNAs as ‘nonsense containing' leads to both inhibition of mRNA translation and mRNA degradation: implications for the control of mRNA decapping. Mol Biol Cell 10: 3971–3978 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagy E, Maquat LE (1998) A rule for termination-codon position within intron-containing genes: when nonsense affects RNA abundance. Trends Biochem Sci 23: 198–199 [DOI] [PubMed] [Google Scholar]
- Nelson PT, Hatzigeorgiou AG, Mourelatos Z (2004) miRNP:mRNA association in polyribosomes in a human neuronal cell line. RNA 10: 387–394 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neu-Yilik G, Gehring NH, Thermann R, Frede U, Hentze MW, Kulozik AE (2001) Splicing and 3′ end formation in the definition of nonsense-mediated decay-competent human {beta}-globin mRNPs. EMBO J 20: 532–540 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nilsen TW (2007) Mechanisms of miroRNA-mediated gene regulation in animal cells. Trends Genet 23: 243–249 [DOI] [PubMed] [Google Scholar]
- Nottrott S, Simard MJ, Richter JD (2006) Human let-7a miRNA blocks protein production on actively translating polyribosomes. Nat Struct Mol Biol 13: 1108–1114 [DOI] [PubMed] [Google Scholar]
- Olsen PH, Ambros V (1999) The lin-4 regulatory RNA controls developmental timing in Caenorhabditis elegans by blocking LIN-14 protein synthesis after the initiation of translation. Dev Biol 216: 671–680 [DOI] [PubMed] [Google Scholar]
- Page MF, Carr B, Anders KR, Grimson A, Anderson P (1999) SMG-2 is a phosphorylated protein required for mRNA surveillance in Caenorhabditis elegans and related to Upf1p of yeast. Mol Cell Biol 19: 5943–5951 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palacios IM, Gatfield D, St Johnston D, Izaurralde E (2004) An eIF4AIII-containing complex required for mRNA localization and nonsense-mediated mRNA decay. Nature 427: 753–757 [DOI] [PubMed] [Google Scholar]
- Parker R, Sheth U (2007) P bodies and the control of mRNA translation and degradation. Mol Cell 25: 635–646 [DOI] [PubMed] [Google Scholar]
- Peltz SW, Brown AH, Jacobson A (1993) mRNA destabilization triggered by premature translational termination depends on at least three cis-acting sequence elements and one trans-acting factor. Genes Dev 7: 1737–1754 [DOI] [PubMed] [Google Scholar]
- Pillai RS, Artus CG, Filipowicz W (2004) Tethering of human Ago proteins to mRNA mimics the miRNA-mediated repression of protein synthesis. RNA 10: 1518–1525 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pillai RS, Bhattacharyya SN, Filipowicz W (2007) Repression of protein sysnthesis by miRNAs: how many mechanisms? Trends Cell Biol 17: 118–126 [DOI] [PubMed] [Google Scholar]
- Rajavel KS, Neufeld EF (2001) Nonsense-mediated decay of human HEXA mRNA. Mol Cell Biol 21: 5512–5519 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rajewsky N (2006) microRNA target predictions in animals. Nat Genet 38 (Suppl): S8–S13 [DOI] [PubMed] [Google Scholar]
- Rand TA, Petersen S, Du F, Wang X (2005) Argonaute2 cleaves the anti-guide strand of siRNA during RISC activation. Cell 123: 621–629 [DOI] [PubMed] [Google Scholar]
- Rehwinkel JAN, Behm-Ansmant I, Gatfield D, Izaurralde E (2005) A crucial role for GW182 and the DCP1:DCP2 decapping complex in miRNA-mediated gene silencing. RNA 11: 1640–1647 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richter JD (1996) Dynamics of poly(A) addition and removal during development. In Translational Control, Hershey JWB, Mathews MB, Sonenberg N (eds), pp 481–503. Plainview: Cold Spring Harbor Laboratory Press [Google Scholar]
- Rodriguez-Gabriel MA, Watt S, Bahler J, Russell P (2006) Upf1, an RNA helicase required for nonsense-mediated mRNA decay, modulates the transcriptional response to oxidative stress in fission yeast. Mol Cell Biol 26: 6347–6356 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schell T, Kulozik A, Hentze M (2002) Integration of splicing, transport and translation to achieve mRNA quality control by the nonsense-mediated decay pathway. Genome Biol 3: reviews1006.1–1006.6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seggerson K, Tang L, Moss EG (2002) Two genetic circuits repress the Caenorhabditis elegans heterochronic gene lin-28 after translation initiation. Dev Biol 243: 215–225 [DOI] [PubMed] [Google Scholar]
- Sheth U, Parker R (2003) Decapping and decay of messenger RNA occur in cytoplasmic processing bodies. Science 300: 805–808 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sheth U, Parker R (2006) Targeting of aberrant mRNAs to cytoplasmic processing bodies. Cell 125: 1095–1109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Small EC, Leggett SR, Winans AA, Staley JP (2006) The EF-G-like GTPase Snu114p regulates spliceosome dynamics mediated by Brr2p, a DExD/H box ATPase. Mol Cell 23: 389–399 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stoecklin G, Mayo T, Anderson P (2006) ARE-mRNA degradation requires the 5′–3′ decay pathway. EMBO Rep 7: 72–77 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun X, Perlick HA, Dietz HC, Maquat LE (1998) A mutated human homologue to yeast Upf1 protein has a dominant-negative effect on the decay of nonsensecontaining mRNAs in mammalian cells. Proc Natl Acad Sci USA 95: 10009–10014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thermann R, Hentze MW (2007) Drosophila miR2 induces pseudo-polysomes and inhibits translation initiation. Nature 447: 875–878 [DOI] [PubMed] [Google Scholar]
- Uchida N, Hoshino S-I, Imataka H, Sonenberg N, Katada T (2002) A novel role of the mammalian GSPT/eRF3 associating with poly(A)-binding protein in Cap/poly(A)-dependent translation. J Biol Chem 277: 50286–50292 [DOI] [PubMed] [Google Scholar]
- Unterholzner L, Izaurralde E (2004) SMG7 acts as a molecular link between mRNA surveillance and mRNA decay. Mol Cell 16: 587–596 [DOI] [PubMed] [Google Scholar]
- van Hoof A, Green PJ (1996) Premature nonsense codons decrease the stability of phytohemagglutinin mRNA in a position-dependent manner. Plant J 10: 415–424 [DOI] [PubMed] [Google Scholar]
- Wakiyama M, Takimoto K, Ohara O, Yokoyama S (2007) Let-7 microRNA-mediated mRNA deadenylation and translational repression in a mammalian cell-free system. Genes Dev 21: 1857–1862 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J, Gudikote JP, Olivas OR, Wilkinson MF (2002) Boundary-independent polar nonsense-mediated decay. EMBO Rep 3: 274–279 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu L, Fan J, Belasco JG (2006) MicroRNAs direct rapid deadenylation of mRNA. Proc Natl Acad Sci USA 103: 4034–4039 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamashita A, Chang TC, Yamashita Y, Zhu W, Zhong Z, Chen CY, Shyu AB (2005) Concerted action of poly(A) nucleases and decapping enzyme in mammalian mRNA turnover. Nat Struct Mol Biol 12: 1054–1063 [DOI] [PubMed] [Google Scholar]
- Zamore PD, Haley B (2005) Ribo-genome: the big world of small RNAs. Science 309: 1519–1524 [DOI] [PubMed] [Google Scholar]
- Zhang J, Sun X, Qian Y, LaDuca JP, Maquat LE (1998) At least one intron is required for the nonsense-mediated decay of triosephosphate isomerase mRNA: a possible link between nuclear splicing and cytoplasmic translation. Mol Cell Biol 18: 5272–5283 [DOI] [PMC free article] [PubMed] [Google Scholar]