Abstract
To determine the contribution of photosynthesis on stomatal conductance, we contrasted the stomatal red light response of wild-type tobacco (Nicotiana tabacum ‘W38’) with that of plants impaired in photosynthesis by antisense reductions in the content of either cytochrome b6f complex (anti-b/f plants) or Rubisco (anti-SSU plants). Both transgenic genotypes showed a lowered content of the antisense target proteins in guard cells as well as in the mesophyll. In the anti-b/f plants, CO2 assimilation rates were proportional to leaf cytochrome b6f content, but there was little effect on stomatal conductance and the rate of stomatal opening. To compare the relationship between photosynthesis and stomatal conductance, wild-type plants and anti-SSU plants were grown at 30 and 300 μmol photon m−2 s−1 irradiance (low light and medium light [ML], respectively). Growth in ML increased CO2 assimilation rates and stomatal conductance in both genotypes. Despite the significantly lower CO2 assimilation rate in the anti-SSU plants, the differences in stomatal conductance between the genotypes were nonsignificant at either growth irradiance. Irrespective of plant genotype, stomatal density in the two leaf surfaces was 2-fold higher in ML-grown plants than in low-light-grown plants and conductance normalized to stomatal density was unaffected by growth irradiance. We conclude that the red light response of stomatal conductance is independent of the concurrent photosynthetic rate of the guard cells or of that of the underlying mesophyll. Furthermore, we suggest that the correlation of photosynthetic capacity and stomatal conductance observed under different light environments is caused by signals largely independent of photosynthesis.
Stomata function as hydraulic valves on the surface of aerial parts of plants, with the guard cells that surround each pore rapidly adjusting their turgor to optimize photosynthetic CO2 uptake and minimize transpirational water loss from leaves. Stomata respond to a variety of signals, either received from the environment or produced within the plant, which lead to changes in the activities of ion or solute channels regulating guard cell turgor. Stomatal opening is induced by low CO2 concentrations, high light intensity, and high humidity, and closing is promoted by high CO2 concentrations, darkness, drought, and the plant hormone abscisic acid (Outlaw, 2003).
In C3 species, stomatal opening in response to light is thought to be induced by distinct mechanisms depending on the wavelength of incident light. Blue light is perceived directly by phototropins (Kinoshita et al., 2001; Doi et al., 2004) and activates a signaling cascade that results in fast stomatal opening under background red light (Shimazaki et al., 2007). The opening response of stomata to red light requires higher irradiance than blue light and shares characteristics of photosynthesis in its action spectra in the red region (Sharkey and Raschke, 1981). Furthermore the red light response can be abolished by 3-(3,4-dichlorophenyl)-1,1-dimethylurea, a PSII inhibitor in whole leaf, epidermal strips, and guard cell protoplasts (Sharkey and Raschke, 1981; Tominaga et al., 2001; Olsen et al., 2002; Messinger et al., 2006). Using isolated guard cell protoplasts, Tominaga et al. (2001) showed that 3-(3,4-dichlorophenyl)-1,1-dimethylurea inhibited proton pumping in red light, suggesting that guard cell chloroplasts provide ATP required for H+ pumping in the guard cell plasma membrane.
It has also been suggested that the guard cell response to red light is in part an indirect response to red-light-driven intercellular CO2 uptake in the mesophyll (Roelfsema et al., 2002). For example, Roelfsema et al. (2006) have shown that chloroplast-containing guard cells in albino sections of variegated leaves do not respond to photosynthetically active radiation, but are sensitive to blue light and CO2, bringing into question a direct role of guard cell photosynthesis on red-light-mediated stomatal opening in intact leaves.
With the exception of the orchid Paphiodedilum, guard cells from all species studied to date contain chloroplasts. Chlorophyll fluorescence measurements (Cardon and Berry, 1992; Goh et al., 1999; Lawson et al., 2002, 2003) and biochemical and immunolocalization experiments (Ueno, 2001; Zeiger et al., 2002) suggest that guard cell chloroplasts have the capacity for electron transport, Rubisco-mediated CO2 assimilation, and photorespiration. Guard cell photophosphorylation has been postulated as a significant energy source driving stomatal opening in red light (Tominaga et al., 2001). These results have suggested a role of guard cell photosynthesis in the red light response of stomata; however, the mechanism underpinning this link has remained elusive.
Across species and under a variety of growth conditions plants regulate their transpiration and photosynthetic rates in parallel, maintaining a balance between the stomata-mediated supply of CO2 to the mesophyll chloroplasts and their photosynthetic demand for CO2. This results in the conservation of the ratio of intercellular (Ci) to ambient (Ca) CO2 partial pressures (pCO2) within the leaf (Wong et al., 1979, 1985; Hetherington and Woodward, 2003). This empirical direct correlation between photosynthesis and stomatal conductance was central to initial models of stomatal control of photosynthesis (Farquhar and Wong, 1984; Ball et al., 1987) and has been carried over to more recent models (Jarvis and Davies, 1998; Dewar, 2002; Buckley et al., 2003). However, the underlying regulatory mechanism is still unclear. It has been proposed that guard cells sense the metabolic status of the mesophyll via a diffusible factor that is a product of photosynthetic activity in the mesophyll (Wong et al., 1979; Lee and Bowling, 1992) and that stomatal aperture would be inversely proportional to the pool size of such metabolites (Farquhar and Wong, 1984). Possible metabolites include ATP, NADPH, or ribulose 1,5-bisphosphate (RuBP), the concentration of which depends strongly on the balance between chloroplast electron transport and the carboxylation reaction catalyzed by Rubisco. However, exogenous ATP added to isolated epidermis did not stimulate opening (Lee and Bowling, 1992). Alternatively, guard cells could respond to photosynthetic demand by direct sensing of Ci (Mott, 1988; Roelfsema et al., 2002).
Contrary to the predictions of the above-mentioned models, transgenic plants with impairments in different steps of the photosynthetic process can maintain normal stomatal conductances, resulting in elevated Ci values casting some doubt on the extent of the control of stomatal movements by Ci (Hudson et al., 1992; Lauerer et al., 1993; Stitt and Schulze, 1994). In antisense plants that accumulate only 10% to 15% of the wild-type levels of Rubisco and show a proportional decrease in CO2 assimilation rate, chlorophyll fluorescence measurements have shown that the antisense reduction of the target gene was effective in guard cells as well as in the mesophyll. However, those plants also maintained wild-type values of conductance under ambient CO2 concentrations and a light source with a mix of red and blue light (von Caemmerer et al., 2004). Most of the gas-exchange measurements made on transgenic plants with impaired photosynthesis have so far been made under white light and this raises the question of whether stomatal conductance would be affected when red light is the only source of illumination.
This report examines the contribution of photosynthetic activity to the stomatal response to red light in intact plants. We contrasted the red light response of stomata of wild-type tobacco (Nicotiana tabacum ‘W38’) with that of antisense plants impaired in photosynthetic CO2 assimilation either by a decrease in chloroplast electron transport rate and ATP synthesis or by a decrease in Rubisco activity and ATP consumption, and we find that these impairments do not affect stomatal conductance. To further explore the relationship between photosynthesis and stomatal conductance, we also examined the stomatal response of wild-type and antisense small subunit of Rubisco (anti-SSU) plants to growth irradiance. Remarkably, despite the large difference in photosynthetic rates, the transpiration machinery of wild-type and anti-SSU plants responded in the same manner to the different light growth conditions.
RESULTS
Cytochrome f and Rubisco Content in the Epidermis of Wild-Type and Transgenic Tobacco
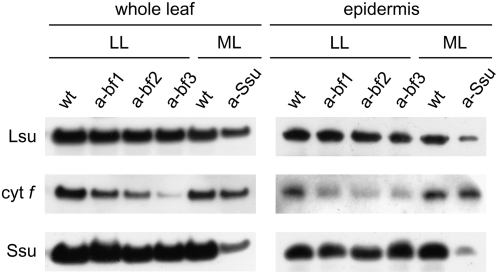
We used three different phenotypes of tobacco, the wild type and two lines with low photosynthetic CO2 assimilation rates, generated by antisense technology: anti-b/f plants, which carry an antisense construct directed against the Rieske iron-sulfur subunit of the chloroplast cytochrome b6f complex (Price et al., 1998), and homozygous anti-SSU plants with 10% to 15% of the wild-type content of Rubisco (Ruuska et al., 1998). To minimize developmental effects of the transgenes, our plants were grown under conditions that provided similar rates of growth for the wild type and transgenic genotypes (see “Materials and Methods”). Figure 1 shows that the known leaf phenotype of the transgenic plants is also expressed in the epidermal tissue. As reported previously (Price et al., 1998), the segregating population of anti-b/f plants exhibited a variety of cytochrome b6f content, as estimated from leaf content of cytochrome f, ranging from 100% to less than 10% of wild-type levels. Three representative examples of plants with leaf cytochrome f content that was 51%, 28%, and 7% of wild type, respectively, are shown in Figure 1. Anti-SSU plants, which are homozygous for a single insertion of the transgene (Hudson et al., 1992), showed the previously observed drastic reduction in Rubisco levels, with only 17% of the wild-type Rubisco SSU content when quantified by immunoblotting (Fig. 1). The antisense transgenes were driven by the CaMV 35S promoter, thus it was expected that both transgenic genotypes would show reduced levels of the target genes in all green cell types, including guard cells. Epidermal tissue prepared from anti-b/f plants with low leaf cytochrome b6f content showed a corresponding decrease in the level of cytochrome f, indicating that the antisense effect of the transgene was also operational in guard cells (Fig. 1). As with the anti-b/f plants, the effect of the anti-SSU transgene was also present in the epidermis, where Rubisco content was 10% of that present in guard cells of the wild type (Fig. 1). Compared to the wild type, anti-b/f plants showed a slight decrease (15% on average) in the amount of Rubisco in whole leaves and the whole leaf cytochrome f content of anti-SSU plants was also decreased (Fig. 1). This is consistent with published data (Jiang and Rodermel, 1995).
Figure 1.
Cytochrome b6f and Rubisco content in leaf and epidermis of wild-type and transgenic tobacco plants determined by immunoblotting. Total protein extracts of leaf discs and epidermal fragments are compared for content of cytochrome f (cyt f in the image) and the large subunit and SSU of Rubisco (Lsu and Ssu in the image, respectively). Plants were grown under LL or ML intensity for comparison of wild-type with anti-b/f and anti-SSU leaves, respectively. Three different anti-b/f plants, labeled a-bf1, a-bf2, and a-bf3, and having CO2 assimilation rates that were 43%, 36%, and 17% of wild-type values, respectively, are shown as representatives of anti-b/f plants with low photosynthetic rates. For whole-leaf samples, gel lanes were loaded on an equal-leaf-area basis, and samples from ML-grown plants were diluted 5-fold relative to samples from LL-grown plants. Equal total protein amounts (20 μg) were loaded when comparing epidermal samples. wt, Wild-type plant; a-Ssu, anti-SSU plant.
Photosynthetic Rates and Stomatal Conductances under Red Measuring Light in Wild-Type and Anti-b/f Plants
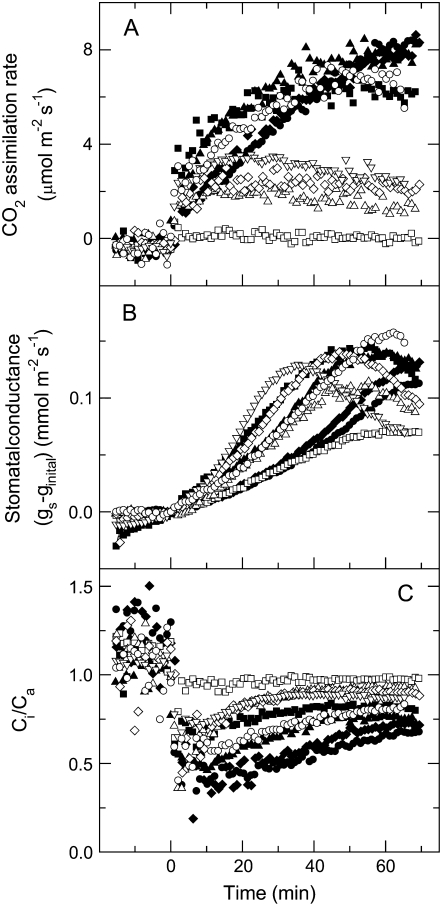
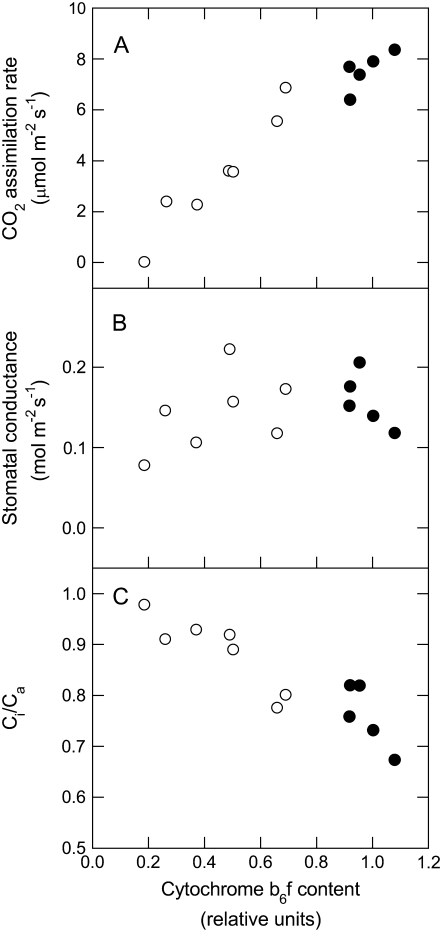
We used red light in our gas-exchange experiments to induce photosynthesis independently of any stimulation of the blue light response of stomata, which is mediated by phototropins (Shimazaki et al., 2007). Attached leaves were equilibrated to ambient pCO2 (362 μbar) in the gas-exchange chamber in the dark for at least 20 min before red light of 1,000 μmol photons m−2 s−1 was turned on. Wild-type plants gradually attained an average steady-state net rate of CO2 assimilation of 7.5 μmol photons m−2 s−1, whereas anti-b/f plants exhibited net rates of CO2 assimilation ranging from wild-type values to only 0.8% of the wild-type rate (Fig. 2A). There was a direct correlation between the steady-state photosynthetic rates under red light and cytochrome b6f content in all plants (Fig. 3A). Both the wild-type and anti-b/f plants showed variability in the rate of stomatal opening and the maximal stomatal conductance in red light (Fig. 2B). We found that stomatal conductance in both wild type and transgenics did not always reach a complete steady state under red light and therefore reported the maximal conductance. There was little effect of low photosynthetic rate on stomatal parameters, and even the anti-b/f plant with the lowest photosynthetic rate in the range (0.8% of wild type) showed a maximal conductance that was 50% of the average wild-type value (Fig. 2, A and B).
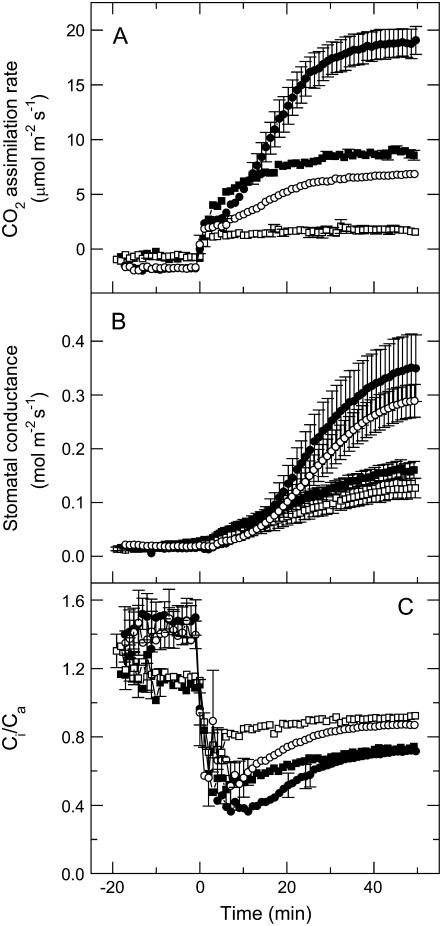
Figure 2.
A to C, Kinetics of CO2 assimilation rate (A), leaf conductance (B), and the ratio of intercellular to ambient CO2 (Ci/Ca; C) in wild-type plants (black symbols) and a range of anti-b/f plants with different cytochrome b6f contents (white symbols), during illumination of dark acclimated leaves at an irradiance of 1,000 μmol photons m−2 s−1 of red light. Symbols of different shape (white or black) represent measurements made on different plants. Light was turned on at time 0. Measurements were conducted at 362 μmol mol−1 CO2, a leaf temperature of 25°C, and a leaf chamber humidity of 19 mbar. Leaves were acclimated in the dark for a minimum of 20 min before the measurements. Stomatal conductance (gs) was normalized by subtracting the conductance values at time 0 (ginitial), which ranged between 0.006 and 0.035 mol m−2 s−1.
Figure 3.
Leaf gas-exchange parameters as a function of cytochrome b6f complex content in wild-type (black circles) and anti-b/f (white circles) tobacco. A, CO2 assimilation rate. B, Stomatal conductance. C, The ratio of intercellular to ambient CO2 (Ci/Ca). Leaf cytochrome b6f complex content was determined from immunoblots such as the one shown in Figure 1 and is expressed as a fraction of the maximum wild-type value. Gas-exchange experiments were performed as described in Figure 2. Each point corresponds to a leaf from a different plant.
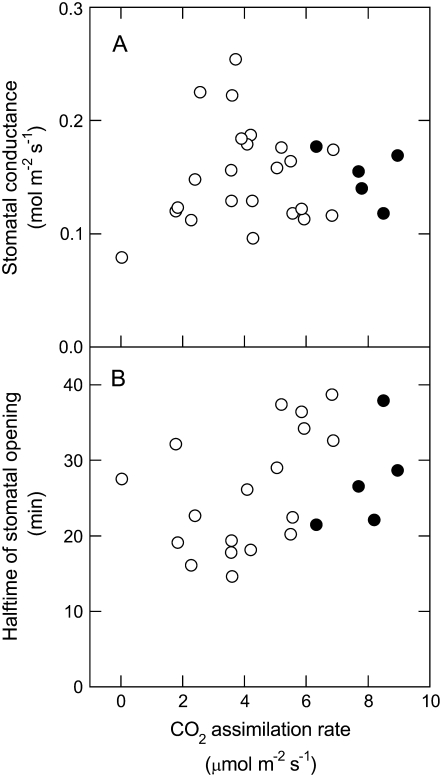
Except for plants with wild-type rates of CO2 assimilation, the anti-b/f plants maintained a ratio of intercellular to ambient CO2 concentration (Ci/Ca) higher than in the wild type (Fig. 2C). There was a marked proportionality of Ci/Ca and cytochrome b6f content, with the anti-b/f plant with the lowest photosynthetic rate showing a Ci/Ca ratio close to 1 (Fig. 3C). Figure 4 shows the maximal stomatal conductance and half-times of stomatal opening of wild type and a large number of individual anti-b/f plants plotted against their steady-state CO2 assimilation rates in red light. Although the presence of the antisense Rieske iron-sulfur protein transgene reduced photosynthetic rates in the anti-b/f plants, their maximal stomatal conductance and rate of stomatal opening remained remarkably similar to that of the wild type when measured with light that stimulates photosynthesis but not the blue light photoreceptors that mediate stomatal opening.
Figure 4.
Relationship between maximal stomatal conductance and CO2 assimilation rate in wild-type and anti-b/f tobacco measured under red light. A, Stomatal conductance. B, Half-times of stomatal opening. Experimental conditions were the same as for Figure 2. Each point corresponds to a different plant. Black circles, wild-type plants; white circles, anti-b/f plants. Half-times were calculated as the time taken to reach half the maximal conductance from the time the light was turned on.
Photosynthetic Rates and Stomatal Conductances under Red Measuring Light in Wild-Type and Anti-SSU Plants and the Effect of Growth Irradiance
To gain further insight on the relationship between stomatal conductance and photosynthesis, we contrasted the red light response of stomata in wild-type and anti-SSU plants that contained between 10% and 15% of wild-type Rubisco content. These transgenic plants do not show the light sensitivity and phenotypic instability of the anti-b/f plants and can be grown under a broader range of irradiances. To compare with our results on anti-b/f plants, we grew the anti-SSU plants at the same low light (LL) intensity (25–35 μmol photons m−2 s−1) and also chose a growth medium light (ML) intensity (300 μmol photons m−2 s−1). This allowed us to contrast the effect of growth irradiance on stomatal conductance in wild-type and photosynthetically impaired plants. Figure 5 shows the kinetics of gas exchange in leaves of wild-type and anti-SSU plants at ambient pCO2 (362 μbar) during a transition from darkness to 1,000 μmol photons m−2 s−1 of red light. Attached leaves were equilibrated to ambient pCO2 (362 μbar) in the gas-exchange chamber in the dark for at least 20 min before red light of 1,000 μmol photons m−2 s−1 was turned on. Wild-type plants grown in LL conditions gradually attained a steady-state rate of net CO2 assimilation of 8.9 ± 0.19 μmol m−2 s−1, whereas anti-SSU plants grown under the same conditions reached a steady-state CO2 assimilation rate of only 1.64 ± 0.3 μmol m−2 s−1 (Fig. 5A). A 10-fold increase in irradiance during growth increased CO2 assimilation rate in the wild type, to 19.1 ± 1.3 μmol m−2 s−1 in ML-grown plants. The anti-SSU plants reached a CO2 assimilation rate of 6.8 ± 0.2 μmol m−2 s−1 when grown in ML. Stomatal conductance in both sets of plants reached steady-state values within 50 min of the onset of red light illumination (Fig. 5B). The maximal stomatal conductance in ML-grown plants was 0.34 ± 0.05 and 0.288 ± 0.03 mol m−2 s−1 for wild type and anti-SSU, respectively, and was higher than that of LL-grown plants, which had conductances of 0.16 ± 0.06 and 0.13 ± 0.02 mol m−2 s−1 for wild type and anti-SSU, respectively. However, the differences in stomatal conductance between wild-type and anti-SSU plants at either light intensity were nonsignificant (P = 0.05). The low CO2 assimilation rates and relatively unchanged stomatal conductances in anti-SSU plants resulted in higher Ci/Ca ratios for the transgenic plants than for the wild type (Fig. 5C). The initial transient lowering of Ci/Ca results from the fact that CO2 assimilation rate increases more rapidly with irradiance than stomatal conductance.
Figure 5.
A to C, Kinetics of CO2 assimilation rate (A), leaf conductance (B), and the ratio of intercellular to ambient CO2 (Ci/Ca; C) in wild-type and anti-SSU plants with 10% to 15% of wild-type Rubisco during illumination of dark acclimated leaves at an irradiance of 1,000 μmol photons m−2 s−1 of red light. Plants were grown under an irradiance of 30 μmol photons m−2 s−1 (LL, squares) or 300 μmol photons m−2 s−1 (ML, circles). During gas-exchange measurements, light was turned on at time 0. Leaves were acclimated in the dark for a minimum of 20 min before the measurements. Experimental conditions were the same as for Figure 2. Data are the means of measurements on four different plants; error bars represent se and are not shown if smaller than the symbols. Black symbols, wild-type plants; white symbols, anti-SSU plants.
Effect of Growth Irradiance on Stomatal Density and Index in Wild-Type and Anti-SSU Plants
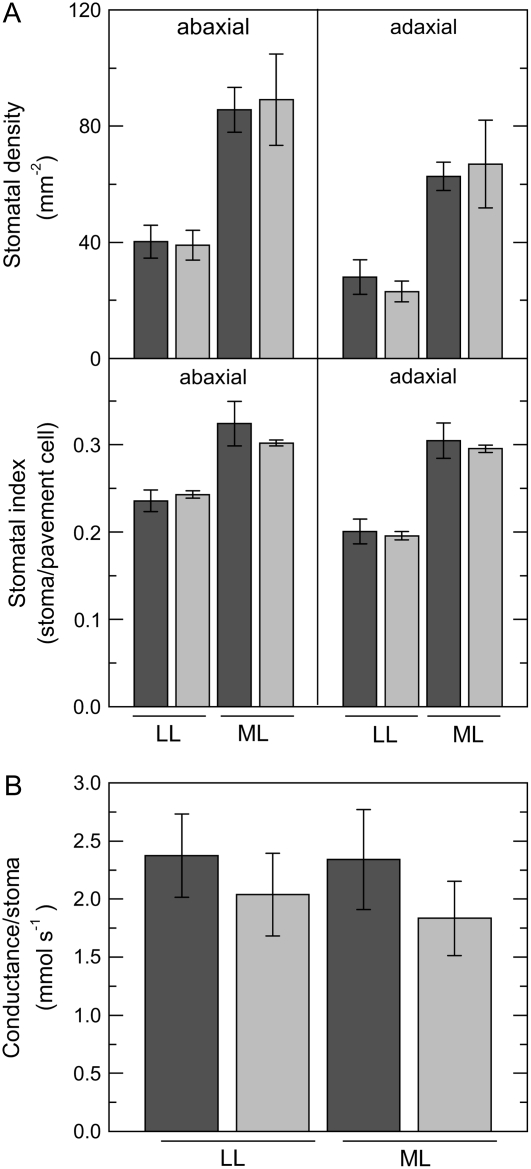
The drastic increase in stomatal conductance in plants grown at ML compared with LL was caused by increases of stomatal density by a factor of approximately 2 in both the abaxial and the adaxial surfaces of the leaf, irrespective of plant genotype (Fig. 6A). In the abaxial leaf surface, the stomatal index rose by 33% and 25% in wild-type and anti-SSU plants, respectively, when grown at ML, whereas the top side of the leaves showed an increase in stomatal index of 50% in both genotypes when grown at ML (Fig. 6A). Compared to LL growth conditions, both types of plants showed a slight decrease in the size of pavement cells when grown in ML. Stomatal dimensions did not vary with different growth light intensity or genotype (data not shown). There was no significant effect of genotype on the stomatal conductance, calculated relative to the combined number of stomata on both leaf surfaces (Fig. 6B).
Figure 6.
Number of stomata on the leaf surface of wild-type (dark gray bars) and anti-SSU (light gray bars) tobacco plants as a function of growth irradiance and stomatal conductance normalized by stomatal numbers. Plants were grown under an irradiance of 30 μmol photons m−2 s−1 (LL) or 300 μmol photons m−2 s−1 (ML). A, Stomatal density. B, Stomatal index. C, Stomatal conductance normalized by stomatal numbers. Data represent mean values ± se from four different plants.
Relationship between Photosynthetic Rate and Stomatal Conductance in Wild-Type and Transgenic Tobacco
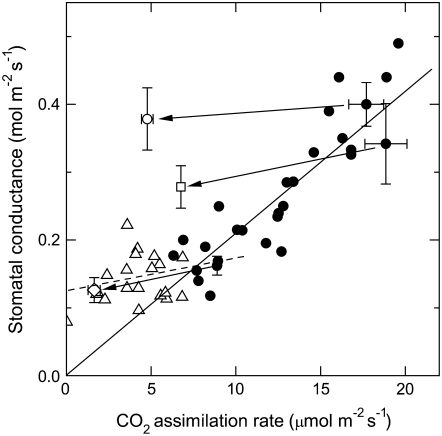
Because it has been shown that there can be a strong correlation between CO2 assimilation rates and stomatal conductance over a range of growth conditions and leaf ages (Wong et al., 1979; for review, see Hetherington and Woodward, 2003), we were interested to see how our data on transgenic tobacco would fit with the expected linear trend. Figure 7 (black circles) shows the covariation of stomatal conductance and CO2 assimilation rates for young wild-type tobacco plants grown in environmental cabinet conditions, at a CO2 concentration of 1,000 μmol mol−1. Differences in growth light intensity and plant-to-plant variation produced a range of wild-type net rates of CO2 assimilation between approximately 7 and 20 μmol m−2 s−1. In these plants stomatal conductance was directly proportional to net CO2 assimilation rates. Transgenic plants with reduced photosynthetic rates, caused either by decreased Rubisco content (Fig. 7, white circle, square, and diamond) or by low cytochrome b6f complex (Fig. 7, triangles), maintain stomatal conductances higher than expected from their low CO2 assimilation rates and thus break the linear relationship of conductance and photosynthetic rate observed for the wild type.
Figure 7.
Relationship between stomatal conductance and CO2 assimilation rate in wild-type and transgenic tobacco plants impaired in photosynthesis either by a decrease in electron transport rates (anti-b/f plants) or in Rubisco function (anti-SSU plants). Plants were grown under elevated CO2 in environmentally controlled chambers and conductance and photosynthesis measurements were performed under ambient CO2. Black circles, wild type; white triangles, anti-b/f plants; white diamond, mean ± se (n = 4) from LL-grown anti-SSU plants; white square, mean ± se (n = 4) of ML-grown anti-SSU plants; white circle, mean ± se (n = 5) from ML-grown anti-SSU plants assayed in red-blue light (von Caemmerer et al., 2004). Arrows link data from anti-SSU plants with the mean ± se of four to five wild-type plants grown and assayed under identical conditions at the same time. The solid and dashed lines represent the linear regression fit of all wild-type data (y = 0.0217 (±0.00069) × x, R = 0.90), and LL-grown wild-type and anti-b/f data as shown in Figure 4A (y = 0.1209 (±0.0159) + 0.00514 (±0.0 29) × x, R = 0.34), respectively. Each data point not showing error bars corresponds to an individual plant. Error bars represent se.
Light Response of CO2 Assimilation Rate and Stomatal Conductance
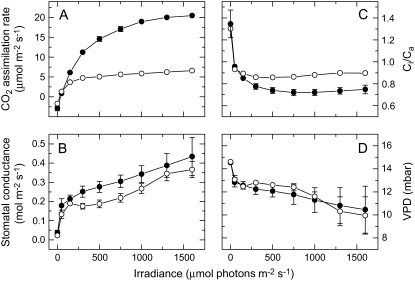
We also examined the fluency response of CO2 assimilation rate and stomatal conductance to varying intensities of red light in wild-type and anti-SSU plants (Fig. 8). Leaves from ML-grown plants were acclimated in the dark for a minimum of 20 min before the red light was turned on for 30 min at each irradiance. Stomatal opening continued even after 50 min in the light and we decided to take measurements at a defined time interval of 30 min. The CO2 assimilation rate was similar for wild-type and anti-SSU plants at LL but saturated for the anti-SSU plants at a low rate around 300 μmol photons m−2 s−1, whereas it continued to increase for wild-type leaves (Fig. 8A). Stomatal conductance on the other hand was similar for wild-type and anti-SSU plants (Fig. 8B). The largest increase in conductance occurred in the first step from dark to 50 μmol photons m−2 s−1; however, conductance continued to increase up to 1,500 μmol photons m−2 s−1 in both genotypes and the response was distinctly biphasic. The different response of CO2 assimilation rate and stomatal conductance to irradiance in the anti-SSU plants resulted in greater ratios of Ci/Ca compared with wild type (Fig. 8C). The humidity of the chamber was not controlled after the initial adjustment and led to a decrease in leaf-to-air vapor pressure difference that was, however, similar in wild-type and anti-SSU plants (Fig. 8D).
Figure 8.
A to D, Red light response of CO2 assimilation rate (A), leaf conductance (B), the ratio of intercellular to ambient CO2 (Ci/Ca; C), and leaf-to-air vapor pressure difference (D) in wild-type and anti-SSU plants. Measurements were conducted at 362 μbar CO2, a leaf temperature of 25°C, and an initial leaf chamber humidity of 19 mbar. Leaves from ML-grown plants were acclimated in the dark for a minimum of 20 min before the red light was turned on and light intensity increased stepwise at 30-min intervals. Data are the means of measurements on three different plants; error bars represent se and are not shown if smaller than the symbols. Black circles, wild-type plants; white circles, anti-SSU plants.
DISCUSSION
The Red Light Response of Stomata in Transgenic Tobacco with Impaired Photosynthesis
We have used transgenic tobacco with low capacity for either chloroplast electron transport (anti-b/f plants) or CO2 fixation capacity (anti-SSU plants) to probe the contribution of photosynthetic capacity to stomatal opening in red light. In both types of transgenic plants, there was an effective decrease in the amount of the proteins targeted by antisense technology in the guard cells, as shown by immunoblotting (Fig. 1). Thus, it is expected that guard cells from these transgenic plants will share at least some of the deficiencies in photosynthetic performance that have been described before for whole leaves. In fact, von Caemmerer et al. (2004) have observed a strong correlation of photosynthetic performance in guard cells and mesophyll cells of anti-SSU plants when comparing measurements of guard cell chlorophyll fluorescence with that of the underlying mesophyll.
The two transgenic genotypes have contrasting phenotypes. The antisense RNA decrease in Rubisco content has been shown to cause an imbalance between the capacity of the photosynthetic carbon reduction cycle to fix CO2 and the chloroplast's capacity for electron transport, resulting in an increase in the pool size of RuBP and ATP (Quick et al., 1991b; Hudson et al., 1992), and zeaxanthin (Ruuska et al., 2000a). Conversely, in the anti-b/f plants, low cytochrome b/f content and hence low electron transport rates cause a decrease in RuBP content and an altered redox state (Price et al., 1998; Ruuska et al., 2000a) together with a lowered capacity for zeaxanthin formation via the xanthophyll cycle (Hurry et al., 1996). Despite the differences in photosynthetic properties and rates, we observed no difference in steady-state stomatal conductance and stomatal opening in red light between wild type and the transgenic plants. Our results clearly indicate that in intact attached leaves, the response of stomata to a dark-to-light transition utilizing red light as irradiance under ambient pCO2 is independent of the concurrent photosynthetic rate of the guard cells or of the underlying mesophyll (Figs. 2–5).
The opening response of stomata to red light has frequently been linked to photosynthesis because the stomatal response saturates at similar irradiance to photosynthesis and can be abolished by PSII inhibitors. Our results confirm that stomatal conductance continues to increase with increasing red irradiance in both wild-type and anti-SSU plants (Fig. 8). The fact that stomatal conductance continued to respond to increasing irradiance in the anti-SSU plants although the CO2 assimilation rate was saturated at low irradiance also suggests that the stomatal response to red light is not linked to the response of the CO2 assimilation rate to red light.
A reduction in the b/f content in our transgenic line leads to a near linear decrease in CO2 assimilation rate (Fig. 3) as has been previously observed (Price et al., 1998). It is thus surprising that this reduction of chloroplast electron transport mediated by the reduction of the cytochrome b/f content does not have a proportional effect on the stomatal red light response and strongly suggests that the red light response of stomata is not quantitatively linked with chloroplast electron transport of guard cells or the mesophyll. All our transgenic plants by necessity have some chloroplast electron transport as they can be grown autotrophically. Thus, we cannot exclude the possibility that a complete inhibition of guard cell chloroplast electron transport is required to decrease the extent of the stomatal red light response.
The lack of a stomatal phenotype in the anti-b/f plants suggests that routes other than photophosphorylation can provide the energy required for stomatal opening. The importance of guard cell respiration as an energy source to drive opening has been pointed out (Parvathi and Raghavendra, 1995). Recent experiments on plants with reduced tricarboxylic acid cycle activity but normal chloroplast electron transport rates support the suggestion that mitochondrial function is necessary to maintain optimal stomatal opening and transpiration rates (Nunes-Nesi et al., 2007).
Is There a Link between Photosynthetic Processes and the Red Light Response of Stomata?
The fact that the transgenic plants used in this study maintain normal conductances but low photosynthetic rates results in higher than wild-type Ci values for a given ambient CO2 partial pressure (Figs. 3 and 5). The lack of sensitivity of guard cells to Ci has been observed in transgenic plants with low Rubisco or cytochrome b6f content before (Quick et al., 1991b; Price et al., 1998; von Caemmerer et al., 2004). However, because those experiments were performed under white light or a red/blue light source, they did not rule out the possibility of an equal, direct blue light stimulation of opening in wild-type and transgenic plants, which could be independent of photosynthesis. To our knowledge, this is the first report to use red light to address this question in intact plants. The nature of the red light response mechanism remains unresolved. The experiments in this article would argue that the effect of red light absorption and utilization by photosynthesis on conductance is not as direct as previously thought. Perhaps another, so far unidentified, photoreceptor is involved. However, the opening response to red light intensity (Fig. 8) clearly shows that no matter how light perception is achieved, stomatal conductance does respond to high light flux levels in a manner not dissimilar to photosynthesis, although there is a distinctly biphasic nature to it with both low and high light response regions. Clearly, more research needs to be done to find alternative mechanisms to explain the red light response of stomata.
Recent mathematical models that attempt to link guard cell photosynthesis with stomatal function hypothesize that the response of stomatal conductance is controlled by the balance between electron transport capacity and Rubisco capacity, and zeaxanthin and ATP have been proposed as possible metabolic links (Zhu et al., 1998; Buckley et al., 2003). In our transgenic lines the balance between electron transport and Rubisco capacity has been perturbed in opposite directions, and our results suggest that the pool size of either metabolite is not the main determinant of stomatal opening under red light. Due to their low electron transport rates relative Rubisco capacity, anti-b/f plants have a substantially decreased zeaxanthin pool (Hurry et al., 1996); however, their maximal stomatal conductance and stomatal opening rate are similar to those of the wild type (Fig. 4). Conversely, anti-SSU plants have a decreased Rubisco relative to electron transport capacity and increased levels of zeaxanthin (Ruuska et al., 2000b); nevertheless, stomatal conductance is similar to wild type. A logical interpretation of our results is that in general they do not support the hypothesis of a direct link between the stomatal response to CO2 and the photosynthetic process.
Suc, either synthesized inside the guard cell or imported from the apoplast, has been proposed to play a key role as an osmoregulatory solute in stomatal movements (Talbott and Zeiger, 1998; Outlaw, 2003). Low leaf sugar content has been reported in anti-SSU plants (Quick et al., 1991a), even under elevated CO2 growth conditions (Masle et al., 1993). We do not know whether these transgenic plants can maintain their apoplastic Suc concentrations despite the lowered bulk leaf concentrations; however, the lack of a stomatal phenotype in anti-SSU plants would suggest that metabolites other than Suc can act as osmoregulators during stomatal opening.
Correlation between Photosynthetic Capacity and Stomatal Conductance
We used growth light intensity as the environmental variable with which to investigate the effect of low photosynthetic rate in the commonly observed comodulation of stomatal conductance and photosynthesis by environmental conditions (Hetherington and Woodward, 2003). Wild-type and anti-SSU plants responded to an increase in growth irradiance from LL to ML by doubling their CO2 assimilation rates (Fig. 5A) and their stomatal conductance (Fig. 5B) and density (Fig. 6). For wild-type plants this resulted in a strong linear correlation between CO2 assimilation rates and conductance, as expected (Fig. 7). Although the response to growth irradiance was similar in wild-type and anti-SSU plants, the latter maintained a high stomatal conductance relative to their decreased CO2 assimilation rates under the two light conditions, showing that stomatal conductance and photosynthetic rate can be uncoupled by genetic manipulation of Rubisco content (Fig. 7). These results are in agreement with previous findings on transgenic plants with impairments in photosynthesis due to antisense decreases in the levels of Rubisco and other PCR cycle enzymes (Hudson et al., 1992; Lauerer et al., 1993; Haake et al., 1998; Muschak et al., 1999). In contrast to these observations in plants with low photosynthetic rates, the correlation between stomatal conductance and photosynthetic rate was apparently maintained in transgenic plants with decreased mitochondrial respiration (Nunes-Nesi et al., 2007).
Developmental Implications
The density of stomata on the leaf epidermis is controlled by the environmental conditions prevailing during leaf expansion, and once determined it remains unchanged for the lifetime of the leaf. Stomatal densities are higher in plants grown in full sunlight or at high light intensities than in plants grown in shade (Willmer and Fricker, 1996). The stomatal index of dicot plants has also been shown to increase with light intensity (Schoch et al., 1980). Advances have been made recently in elucidating the genetic pathway controlling stomatal development (Bergmann, 2006). It is thought that mature leaves sense the environment and produce a systemic signal that determines stomatal density in expanding leaves (Coupe et al., 2006). Our results suggest that this systemic developmental signal is not directly linked to photosynthetic capacity: We show that when anti-SSU plants are grown under elevated CO2, which prevents a limitation of the carbon fixation reactions, the stomatal developmental program is able to sense light intensities during growths and responds by increasing the stomatal density and index in the same manner as the wild type (Fig. 6). It has been shown previously that a decrease in Rubisco content does not interfere with the acclimation of stomatal conductance to environmental conditions such as growth light intensity under ambient pCO2 (Lauerer et al., 1993), at elevated pCO2 (Masle et al., 1993; Sicher et al., 1994), and under different nitrogen nutrition regimes (Quick et al., 1992), but differences in stomatal densities under different growth conditions have not been reported before in these plants. Taken together, these data suggest that the strong photosynthetic impairment of anti-SSU plants does not significantly affect their ability to acclimate their transpirational machinery to the prevailing growth conditions. Correlation between CO2 assimilation rate and stomatal conductance is also observed throughout the lifespan of a leaf. Jiang and Rodermel (1995) showed that stomatal conductance followed similar developmental changes with leaf age in anti-SSU and wild-type plants despite their different photosynthetic rates. This is an example where the changes in stomatal conductance are not linked to variation in stomatal numbers.
CONCLUSION
Our study illustrates the power of the transgenic approach in unraveling correlative links to reveal mechanistic connections. The results show that the red light response of stomata may not be linked to photosynthesis and that further work is required to discover the nature of the red light receptor. Furthermore, we have shown that the environmentally induced correlation between stomatal conductance and photosynthetic capacity so frequently observed must be caused by signals not directly related to photosynthesis. The results have major implications for our understanding of stomatal function and demonstrate that photosynthetic metabolism can be manipulated with minimal coupling to stomatal function and aperture. This means that if plants can be genetically engineered for improved photosynthesis this should also lead to improved plant water use efficiency.
MATERIALS AND METHODS
Plant Material and Growth Conditions
Wild-type and transgenic tobacco plants (Nicotiana tabacum ‘W38’) were grown in 0.25-L pots in seed-raising soil containing approximately 2 g/L of a slow-release fertilizer (Osmocote; 15:4.8:10.8:1.2 N:P:K:Mg and trace elements, B, Cu, Fe, Mn, Mo, Zn; Scotts Australia) and bottom-watered daily. Two types of transgenic tobacco were used: (1) anti-b/f plants, which exhibit a range of phenotypes with respect to cytochrome b6f content and CO2 assimilation rates (Price et al., 1998), carry an antisense construct directed against the Rieske iron-sulfur protein of the chloroplast cytochrome b6f complex and were raised from seed of selfed T3 plants of the line B6F-2.2-513-16 (Price et al., 1995); and (2) anti-SSU plants, which contain 10% to 15% of the wild-type Rubisco content and carry two copies of an antisense construct directed against the SSU of Rubisco (Ruuska et al., 1998). Plants were grown in environmentally controlled cabinets under a pCO2 of 953 μbar, with a 20-h photoperiod and a constant temperature and relative humidity of 23°C and 65%, respectively. The light intensity at the top of the plant was kept at 25 ± 10 μmol photons m−2 s−1 for the anti-b/f plants, and 30 ± 5 μmol photons m−2 s−1 or 300 ± 20 μmol photons m−2 s−1 for the anti-SSU plants, with their corresponding wild-type controls grown under the same irradiance. The low irradiance and long photoperiod minimized the instability of the anti-b/f phenotype and the high pCO2 ensured that wild-type and transgenic plants had similar growth rates. Plants were assayed 4 to 5 weeks after germination (six-leaf stage), and the uppermost fully expanded leaves were used in the experiments. Transgenic and wild-type plants had similar leaf sizes and number of leaves at the time of measurement.
Preparation of Epidermal Fragments, Protein Extraction, and Immunoblotting
A fraction enriched in epidermal tissue was prepared by adapting the method of Kopka et al. (1997). A young expanding leaf was picked, the major veins were removed and discarded, and the rest was blended with 250 mL of chilled distilled water with a Sorvall Omni Mixer blender at maximum speed, with four pulses of 30 s each and waiting 30 s between pulses. The resulting epidermal fragments were rinsed with 300 mL of chilled distilled water on a 100- to 149-μm Nytal mesh to rid them of contaminating mesophyll cells. The epidermal fragments were drained of excess water, disrupted by grinding with mortar and pestle in liquid nitrogen for 3 min, and stored at −80°C until later use. The resulting fraction was highly enriched in epidermis compared with mesophyll cells (less than one mesophyll cell per 200 stomata was routinely observed under the compound microscope).
Total proteins from 1.28-cm2 leaf discs or 100 mg of epidermal fragments were extracted at room temperature in 0.5 mL of buffer containing 100 mm Tris-HCL, pH 7.8, 20 mm EDTA, 25 mm NaCl, 10 mm dithiothreitol, 2% (w/v) SDS, and 2% (v/v) protease inhibitor cocktail (Sigma) using a 2-mL glass homogenizer. Samples were heated to 65°C in a heat block for 10 min and centrifuged at room temperature in a microcentrifuge at maximum speed for 10 min. Protein concentration in the samples was determined with the bicinchoninic acid method (BCA Protein Assay kit; Pierce). Samples were prepared for gel loading by adding 0.25 volumes of Bio-Rad XT sample buffer (Bio-Rad). Proteins were separated by electrophoresis on NuPAGE Bis-Tris precast gels (4%–12% acrylamide concentration; Novex), using the manufacturer-specified buffer system, and blotted onto nitrocellulose membranes. Blots were probed with polyclonal antibodies raised against the spinach (Spinacia oleracea) Rubisco holoenzyme or spinach cytochrome f. Anti-IgG horseradish peroxidase conjugate (Pierce) was used as secondary antibody. Blots were developed using the enhanced chemiluminescence SuperSignal West Pico substrate system (Pierce). Because of the close correlation of accumulation of cytochrome b6f holocomplex and its cytochrome f subunit (Bruce and Malkin, 1991; Price et al., 1998), anticytochrome f antibody was used instead of a less specific Rieske iron-sulfur protein antibody to quantify content of cytochrome b6f. Protein bands were quantified using Image J software (http://rsb.info.nih.gov/ij/).
Measurement of Leaf Gas Exchange
Gas-exchange measurements were made with a LI-6400 portable gas-exchange system (LI-COR), equipped with a red light-emitting diode light source with a maximum emission peak centered at 670 nm (LI-6400-02; LI-COR). This light source was fitted on the standard 6-cm2 clamp-on leaf chamber. Sample pCO2, flow rate, and temperature were kept constant at 362 μbar, 500 μmol s−1, and 25°C, respectively. Leaves were equilibrated in the gas-exchange leaf chamber in darkness, at an initial humidity of 19 mbar for a minimum of 20 min before measurements. For light response curves, the conditions were the same, and after the initial 20 min of dark adaptation the intensity of red light was increased in steps of 30-min duration. The average atmospheric pressure was 950 mbar. Gas-exchange parameters were calculated using the equation derived by von Caemmerer and Farquhar (1981).
Determination of Stomatal Numbers
Stomatal numbers were determined from the same or similar leaves as used for gas-exchange measurements, from impressions taken from both sides of the leaves with dental silicone (Optosil-Xantopren; Heraeus Kulzer). Stomata and epidermal cells were counted from positives made from the impressions with nail polish, in 10 different fields of view per leaf, with a compound microscope using a magnification of 200-fold. Digital photographs of each field were taken and cells were counted and measured with Image J software (http://rsb.info.nih.gov/ij/).
Acknowledgments
The authors thank Drs. John Andrews and Graham Hudson for the use of anti-SSU plants, and Drs. Spencer Whitney and Richard Malkin for the Rubisco and cytochrome f antibodies, respectively. We thank Dr. Asaph Cousins for the careful reading of the manuscript.
This work was supported by the Australian Research Council (Discovery Grant no. DP0343270).
The author responsible for distribution of materials integral to the findings presented in this article in accordance with the policy described in the Instructions for Authors (www.plantphysiol.org) is: Susanne von Caemmerer (susanne.caemmerer@anu.edu.au).
Open Access articles can be viewed online without a subscription.
References
- Ball J, Woodrow I, Berry J (1987) A model predicting stomatal conductance and its contribution to the control of photosynthesis under different environmental conditions. In J Biggins, ed, Progress in Photosynthesis Research, Vol IV. Martinus Nijhoff, Leiden, The Netherlands, pp 221–224
- Bergmann D (2006) Stomatal development: from neighbourly to global communication. Curr Opin Plant Biol 9 478–483 [DOI] [PubMed] [Google Scholar]
- Bruce B, Malkin R (1991) Biosynthesis of the chloroplast b6f complex: studies in a photosynthetic mutant of Lemna. Plant Cell 3 203–212 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buckley TN, Mott KA, Farquhar GD (2003) A hydromechanical and biochemical model of stomatal conductance. Plant Cell Environ 26 1767–1785 [Google Scholar]
- Cardon ZG, Berry J (1992) Effects of O2 and CO2 concentration on the steady-state fluorescence yield of single guard cell pairs in intact leaf discs of Tradescantia albiflora: evidence for Rubisco-mediated CO2 fixation and photorespiration in guard cells. Plant Physiol 99 1238–1244 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coupe SA, Palmer BG, Lake JA, Overy SA, Oxborough K, Woodward FI, Gray JE, Quick WP (2006) Systemic signalling of environmental cues in Arabidopsis leaves. J Exp Bot 57 329–341 [DOI] [PubMed] [Google Scholar]
- Dewar RC (2002) The Ball-Berry-Leuning and Tardieu-Davies stomatal models: synthesis and extension within a spatially aggregated picture of guard cell function. Plant Cell Environ 25 1383–1398 [Google Scholar]
- Doi M, Shigenaga A, Emi T, Kinoshita T, Shimazaki K (2004) A transgene encoding a blue-light receptor, phot1, restores blue light responses in the Arabidopsis phot1 phot2 double mutant. J Exp Bot 55 517–523 [DOI] [PubMed] [Google Scholar]
- Farquhar G, Wong S (1984) An empirical model of stomatal conductance. Funct Plant Biol 11 191–210 [Google Scholar]
- Goh CH, Schreiber U, Hedrich R (1999) New approach of monitoring changes in chlorophyll a fluorescence of single guard cells and protoplasts in response to physiological stimuli. Plant Cell Environ 22 1057–1070 [Google Scholar]
- Haake V, Zrenner R, Sonnewald U, Stitt M (1998) A moderate decrease of plastid aldolase activity inhibits photosynthesis, alters the levels of sugars and starch, and inhibits growth of potato plants. Plant J 14 147–157 [DOI] [PubMed] [Google Scholar]
- Hetherington A, Woodward FI (2003) The role of stomata in sensing and driving environmental change. Nature 424 901–908 [DOI] [PubMed] [Google Scholar]
- Hudson GS, Evans JR, von Caemmerer S, Arvidsson YBC, Andrews TJ (1992) Reduction of ribulose-1,5-bisphosphate carboxylase/oxygenase content by antisense RNA reduces photosynthesis in transgenic tobacco plants. Plant Physiol 98 294–302 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hurry V, Anderson JM, Badger MR, Price GD (1996) Reduced levels of cytochrome b6f in transgenic tobacco increases the excitation pressure on photosystem II without increasing sensitivity to photoinhibition in vivo. Photosynth Res 50 159–169 [DOI] [PubMed] [Google Scholar]
- Jarvis A, Davies W (1998) The coupled response of stomatal conductance to photosynthesis and transpiration. J Exp Bot 49 399–406 [Google Scholar]
- Jiang CZ, Rodermel SR (1995) Regulation of photosynthesis during leaf development in RbcS antisense DNA mutants of tobacco. Plant Physiol 107 215–224 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kinoshita T, Doi M, Suetsugu N, Kagawa T, Wada M, Shimazaki K (2001) phot1 and phot2 mediate blue light regulation of stomatal opening. Nature 414 656–660 [DOI] [PubMed] [Google Scholar]
- Kopka J, Provart NJ, Müller Röber B (1997) Potato guard cells respond to drying soil by a complex change in the expression of genes related to carbon metabolism and turgor regulation. Plant J 11 871–882 [DOI] [PubMed] [Google Scholar]
- Lauerer M, Saftic D, Quick WP, Labate C, Fichtner K, Schulze ED, Rodermel SR, Bogorad L, Stitt M (1993) Decreased ribulose-1,5-bisphosphate carboxylase-oxygenase in transgenic tobacco transformed with antisense rbcS. VI. Effect on photosynthesis in plants grown at different irradiance. Planta 190 332–345 [Google Scholar]
- Lawson T, Oxborough K, Morison JIL, Baker NR (2002) Responses of photosynthetic electron transport in stomatal guard cells and mesophyll cells in intact leaves to light, CO2, and humidity. Plant Physiol 128 52–62 [PMC free article] [PubMed] [Google Scholar]
- Lawson T, Oxborough K, Morison JIL, Baker NR (2003) The responses of guard and mesophyll cell photosynthesis to CO2, O-2, light, and water stress in a range of species are similar. J Exp Bot 54 1743–1752 [DOI] [PubMed] [Google Scholar]
- Lee J, Bowling DJF (1992) Effect of the mesophyll on stomatal opening in Commelina communis. J Exp Bot 43 951–957 [Google Scholar]
- Masle J, Hudson G, Badger MR (1993) Effects of ambient CO2 concentration on growth and nitrogen use in tobacco (Nicotiana tabacum) plants transformed with an antisense gene to the small subunit of ribulose-1,5-bisphosphate carboxylase/oxygenase. Plant Physiol 103 1075–1088 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Messinger SM, Buckley TN, Mott KA (2006) Evidence for involvement of photosynthetic processes in the stomatal response to CO2. Plant Physiol 140 771–778 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mott KA (1988) Do stomata respond to CO2 concentrations other than intercellular? Plant Physiol 86 200–203 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muschak M, Willmitzer L, Fisahn J (1999) Gas-exchange analysis of chloroplastic fructose-1,6-bisphosphatase antisense potatoes at different air humidities and at elevated CO2. Planta 209 104–111 [DOI] [PubMed] [Google Scholar]
- Nunes-Nesi A, Carrari F, Gibon Y, Sulpice R, Lytovchenko A, Fisahn J, Graham J, Ratcliffe RG, Sweetlove LJ, Fernie AR (2007) Deficiency of mitochondrial fumarase activity in tomato plants impairs photosynthesis via an effect on stomatal function. Plant J 50 1093–1106 [DOI] [PubMed] [Google Scholar]
- Olsen RL, Pratt RB, Gump P, Kemper A, Tallman G (2002) Red light activates a chloroplast-dependent ion uptake mechanism for stomatal opening under reduced CO2 concentrations in Vicia spp. New Phytol 153 497–508 [DOI] [PubMed] [Google Scholar]
- Outlaw WH Jr (2003) Integration of cellular and physiological functions of guard cells. CRC Crit Rev Plant Sci 22 503–529 [Google Scholar]
- Parvathi K, Raghavendra AS (1995) Bioenergetic processes in guard cells related to stomatal function. Physiol Plant 93 146–154 [Google Scholar]
- Price G, von Caemmerer S, Evans J, Siebke K, Anderson J, Badger M (1998) Photosynthesis is strongly reduced by antisense suppression of chloroplastic cytochrome bf complex in transgenic tobacco. Aust J Plant Physiol 25 445–452 [Google Scholar]
- Price G, Yu J, von Caemmerer S, Evans J, Chow W, Anderson J, Hurry V, Badger M (1995) Chloroplast cytochrome b6f and ATP synthase complexes in tobacco: transformation with antisense RNA against nuclear-encoded transcripts for the Rieske FeS and ATPδ polypeptides. Funct Plant Biol 22 285–297 [Google Scholar]
- Quick WP, Fichtner K, Schulze ED, Wendler R, Leegood RC, Mooney H, Rodermel SR, Bogorad L, Stitt M (1992) Decreased ribulose-1,5-bisphosphate carboxylase-oxygenase in transgenic tobacco transformed with “antisense” rbcS. VI. Impact on photosynthesis in conditions of altered nitrogen supply. Planta 188 522–531 [DOI] [PubMed] [Google Scholar]
- Quick WP, Schurr U, Fichtner K, Schulze ED, Rodermel SR, Bogorad L, Stitt M (1991. a) The impact of decreased Rubisco on photosynthesis, growth, allocation and storage in tobacco plants which have been transformed with antisense rbcS. Plant Journal 1 51–58 [Google Scholar]
- Quick WP, Schurr U, Scheibe R, Schulze ED, Rodermel SR, Bogorad L, Stitt M (1991. b) Decreased ribulose-1,5-bisphosphate carboxylase-oxygenase in transgenic tobacco transformed with “antisense” rbcS. I. Impact on photosynthesis in ambient growth conditions. Planta 183 542–554 [DOI] [PubMed] [Google Scholar]
- Roelfsema MRG, Hanstein S, Felle HH, Hedrich R (2002) CO2 provides an intermediate link in the red light response of guard cells. Plant J 32 65–75 [DOI] [PubMed] [Google Scholar]
- Roelfsema MRG, Konrad KR, Marten H, Psaras GK, Hartung W, Hedrich R (2006) Guard cells in albino leaf patches do not respond to photosynthetically active radiation, but are sensitive to blue light, CO2 and abscisic acid. Plant Cell Environ 29 1595–1605 [DOI] [PubMed] [Google Scholar]
- Ruuska SA, Andrews TJ, Badger MR, Hudson GS, Laisk A, Price GD, von Caemmerer S (1998) The interplay between limiting processes in C3 photosynthesis studied by rapid-response gas exchange using transgenic tobacco impaired in photosynthesis. Funct Plant Biol 25 859–870 [Google Scholar]
- Ruuska SA, Andrews TJ, Badger MR, Price GD, von Caemmerer S (2000. a) The role of chloroplast electron transport and metabolites in modulating Rubisco activity in tobacco. Insights from transgenic plants with reduced amounts of cytochrome b/f complex or glyceraldehyde 3-phosphate dehydrogenase. Plant Physiol 122 491–504 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruuska SA, von Caemmerer S, Badger MR, Andrews TJ, Price GD, Robinson SA (2000. b) Xanthophyll cycle, light energy dissipation and electron transport in transgenic tobacco with reduced carbon assimilation capacity. Funct Plant Biol 27 289–300 [Google Scholar]
- Schoch PG, Zinsou C, Sibi M (1980) Dependence of the stomatal index on environmental factors during stomatal differentiation in leaves of Vigna sinensis L.: 1. Effect of light intensity. J Exp Bot 31 1211–1216 [Google Scholar]
- Sharkey TD, Raschke K (1981) Effect of light quality on stomatal opening in leaves of Xanthium strumarium L. Plant Physiol 68 1170–1174 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimazaki K, Doy M, Assmann SM, Kinoshita T (2007) Light regulation of stomatal movement. Annu Rev Plant Biol 58 219–247 [DOI] [PubMed] [Google Scholar]
- Sicher RC, Kremer DF, Rodermel SR (1994) Photosynthetic acclimation to elevated CO2 occurs in transformed tobacco with decreased ribulose-1,5-bisphosphate carboxylase/oxygenase content. Plant Physiol 104 409–415 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stitt M, Schulze D (1994) Does Rubisco control the rate of photosynthesis and plant growth? An exercise in molecular ecophysiology. Plant Cell Environ 17 465–487 [Google Scholar]
- Talbott LD, Zeiger E (1998) The role of sucrose in guard cell osmoregulation. J Exp Bot 49 329–337 [Google Scholar]
- Tominaga M, Kinoshita T, Shimazaki K (2001) Guard-cell chloroplasts provide ATP required for H+ pumping in the plasma membrane and stomatal opening. Plant Cell Physiol 42 795–802 [DOI] [PubMed] [Google Scholar]
- Ueno O (2001) Ultrastructural localization of photosynthetic and photorespiratory enzymes in epidermal, mesophyll, bundle sheath, and vascular bundle cells of the C4 dicot Amaranthus viridis. J Exp Bot 52 1003–1013 [DOI] [PubMed] [Google Scholar]
- von Caemmerer S, Farquhar GD (1981) Some relationships between the biochemistry of photosynthesis and the gas exchange of leaves. Planta 153 376–387 [DOI] [PubMed] [Google Scholar]
- von Caemmerer S, Lawson T, Oxborough K, Baker NR, Andrews TJ, Raines CA (2004) Stomatal conductance does not correlate with photosynthetic capacity in transgenic tobacco with reduced amounts of Rubisco. J Exp Bot 55 1157–1166 [DOI] [PubMed] [Google Scholar]
- Willmer C, Fricker M (1996) Stomata, Ed 2. Chapman & Hall, London
- Wong SC, Cowan IR, Farquhar GD (1979) Stomatal conductance correlates with photosynthetic capacity. Nature 282 424–426 [Google Scholar]
- Wong SC, Cowan IR, Farquhar GD (1985) Leaf conductance in relation to the rate of CO2 assimilation. I. Influence of nitrogen nutrition, phosphorous nutrition, photon flux density, and ambient partial pressure of CO2 during ontogeny. Plant Physiol 78 821–825 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeiger E, Talbott LD, Frechilla S, Srivastava A, Zhu JX (2002) The guard cell chloroplast: a perspective for the twenty-first century. Review. New Phytol 153 415–424 [DOI] [PubMed] [Google Scholar]
- Zhu J, Talbott LD, Jin X, Zeiger E (1998) The stomatal response to CO2 is linked to changes in guard cell zeaxanthin. Plant Cell Environ 21 813–820 [Google Scholar]