Abstract
Stress is a generic term that summarizes how psychosocial and environmental factors influence physical and mental well-being. The interaction between stress and immunity has been widely investigated, involving the neuroendocrine system and several organs. Assays using natural products in stress models deserve further investigation. Propolis immunomodulatory action has been mentioned and it has been the subject of scientific investigation in our laboratory. The aim of this study was to evaluate if and how propolis activated macrophages in BALB/c mice submitted to immobilization stress, as well as the histopathological analysis of the thymus, bone marrow, spleen and adrenal glands. Stressed mice showed a higher hydrogen peroxide (H2O2) generation by peritoneal macrophages, and propolis treatment potentiated H2O2 generation and inhibited nitric oxide (NO) production by these cells. Histopathological analysis showed no alterations in the thymus, bone marrow and adrenal glands, but increased germinal centers in the spleen. Propolis treatment counteracted the alterations found in the spleen of stressed mice. New research is being carried out in order to elucidate propolis immunomodulatory action during stress.
Keywords: lymphoid organs, macrophages, propolis, stress
Introduction
Stress results in the release of neurotransmitters, hormones and cytokines, showing an association of central nervous system with endocrine and immune systems, through the activation of the hypothalamic-pituitary-adrenal (HPA) axis and the autonomous nervous system (1).
Chronic stress alters important behavioral, neurochemical and endocrine parameters, inducing immunosuppression (2), and alterations were found in lymphocyte subsets after chronic immobilization stress, verifying that T cells were more affected than B lymphocytes (3).
Propolis is a resinous substance produced by honeybees and possesses many biological activities, such as antitumor, antioxidant, antimicrobial, antiinflammatory, immunomodulatory, among others (4). Its chemical composition is very complex and is dependent upon the source plant (5). Bud exudates of different poplar species are the main sources of propolis in temperate zones, including Europe, Asia and North America. Samples originating from these regions are characterized by similar chemical composition; the most important constituents appeared to be phenolics: flavonoids, aromatic acids and their esters. The main vegetal source of ‘green’ propolis in Botucatu, São Paulo State, Brazil, is Baccharis dracunculifolia DC., followed by Eucalyptus citriodora Hook and Araucaria angustifolia (Bert.) O. Kuntze (6).
With regards to the immune system, experimental works of our laboratory revealed that murine peritoneal macrophages stimulated with propolis showed an increased fungicidal activity against Paracoccidioides brasiliensis (7) and bactericidal activity against Salmonella typhimurium (8). We also observed that propolis induced hydrogen peroxidase (H2O2) generation by peritoneal macrophages and inhibited NO production by these cells in a dose-dependent way (9), stimulated antibody production in rats (10) and enhanced natural killer activity against tumor cells (11).
Since propolis modulates the immune system and stress leads to immunosuppression, the goal of this work was to evaluate propolis action on macrophage activation, through H2O2 and NO determination, in stressed mice. Morphological alterations in thymus, bone marrow, spleen and adrenal were also evaluated, as well as the seric concentration of glucose and corticosterone.
Methods
Propolis Sample
Propolis was collected in the Beekeeping Section, UNESP. Propolis was ground and 30% ethanolic extracts were prepared (30 g of propolis, completing the volume to 100 ml with 70% ethanol), in the absence of bright light, at room temperature, with moderate shaking. After a week, extracts were filtered and the dry weight of the extracts was calculated (120 mg/ml) (10). Propolis chemical composition was investigated using thin-layer chromatography (TLC), gas-chromatography (GC), and gas chromatography-mass spectrometry (GC-MS) analysis (12–14).
Mice and Stress Procedure
Male BALB/c mice aged between 6 and 8 weeks were used. Mice were stressed by restraint in an immobilization tube (restrainer) of ∼50 ml capacity for 15, 30, 45, 60, 75, 90 and 120 min during seven consecutive days. This procedure is easy to perform and causes no physical pain to the animals (3). This work agrees with Ethical Principles in Animal Research adopted by Brazilian College of Animal Experimentation.
Rats were divided into four groups (G1, G2, G3 and G4) of eight each. G1 was considered control, and received physiological solution (NaCl 0.9%). G2 was submitted to immobilization stress. G3 was daily treated with propolis (200 mg/kg) p.o. by gavage. G4 was submitted to stress and daily treated with propolis, before stress, following the same protocol. All groups had no water and food during stress. After 24 h of the respective treatments, animals were sacrificed by cervical dislocation.
Glucose and Corticosterone Determination
Before sacrifice, blood was collected by retro-orbital puncture and immediately used for glucose determination (Accutrend®). Serum was frozen at −20°C and corticosterone was determined by radioimmunoassay, using comercials kits (Coat-A-count®).
Peritoneal Macrophages
Peritoneal macrophages were obtained by inoculation of cold PBS in the abdominal cavity. After a soft abdominal massage, the peritoneal liquid was collected and put in sterile plastic tubes (Falcon) and centrifuged at 200× g for 10 min. Cells were stained with neutral red (0.02%), incubated for 10 min at 37°C and counted in a hematocytometer, in order to obtain a final concentration of 2 × 106 cells/ml. Cells were resuspended in cell culture medium (RPMI 1640 supplemented with 5% fetal calf serum, 2 mM L-glutamine, 20 mM Hepes, 2.5 × 10−5 M 2-mercaptoetanol - Sigma), and cultured in a 96-welled flat-bottomed plate (Corning) at a final concentration of 2 × 105 cells per well. Cells were incubated at 37°C and, after 2 h, non-adherent cells were removed and macrophages were reincubated (37°C) for 24 h (9).
Reactive Oxygen Intermediates (H2O2) Determination
Culture supernatants were collected for further nitric oxide (NO) determination and adherent cells were used for H2O2 determination of Red phenol solution (100 μl), containing 140 mM NaCl, 10 mM K2HPO4, 5.5 mM dextrose and 5.5 mM horseradish peroxidase was added to the adherent cells for H2O2 determination. After 60 min, 10 μl NaOH was added and the absorbances were measured at 620 nm, using an automatic enzyme immunoassay reader (15).
Reactive Nitrogen Intermediates (NO) Determined based on Griess Reaction
NO production was determined based on Griess reaction (16). Supernatants of the 24 h-stimulated cells were collected and 100 μl of Griess reagent was added (N-1-naphthyl-ethyl-enediamine 0.1% + sulfanilamide 1% in H3PO4 5%). After 10 min at room temperature, absorbances were measured at 540 nm, using an automatic enzyme immunoassay reader.
Histological Analysis
Immediately after sacrifice, spleen, thymus, femur and adrenal of all animals were taken and submitted to histological procedures, in order to evaluate these tissues (17,18).
Statistical Analysis
Analysis of variance was used to examine the treatment effect, and comparison between the means was performed by Tukey test, with 0.05 as the significant level (19).
Results
Glucose and Corticosterone Serum Levels were Higher in Stressed Mice
Glucose and corticosterone serum levels were higher in stressed animals, although this difference was not statistically significant (P > 0.05) (Table 1).
Table 1.
Glucose (mg/dl) and corticosterone (ng/ml) seric concentrations of stressed mice
| Control | Stress | |
|---|---|---|
| Glucose | 171.0 ± 17.5 | 190.3 ± 26.4 |
| Corticosterone | 127.2 ± 56.4 | 147.7 ± 36.2 |
Propolis Administration to Stressed Mice Potentiated H2O2 Generation and Inhibited NO Production
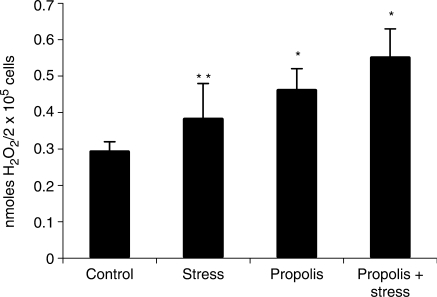
H2O2 generation by peritoneal macrophages of stressed mice was higher than control, although this difference was not statistically significant (P > 0.05). Propolis-treated mice showed an elevation in H2O2 generation. Propolis administration to stressed mice potentiated H2O2 generation (P < 0.05) (Fig. 1).
Figure 1.
Propolis effect on H2O2 generation by peritoneal macrophages of stressed mice. *significantly different from control (P < 0.05), **significantly different from propolis + stress (P < 0.05).
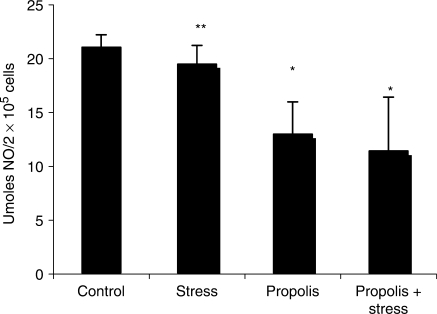
Stressed mice showed a reduced NO liberation as compared with control (P > 0.05) (Fig. 2). Propolis-treated mice showed an inhibition in NO production. Propolis administration to stressed mice also resulted in NO inhibition.
Figure 2.
Propolis effect on NO generation by peritoneal macrophages of stressed mice. *significantly different from control (P < 0.01), **significantly different from propolis and propolis + stress (P < 0.01).
Propolis Administration to Stressed Mice Prevented an Increase in Germinal Centers in the Spleen
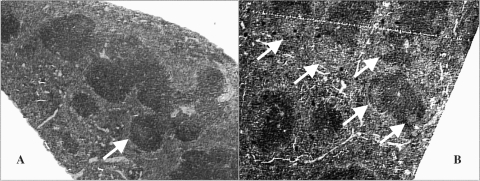
Histological analysis of thymus, bone marrow and adrenal of stressed mice revealed no morphological alterations in such organs. On the other hand, there was an increase in the number of germinal centers in the spleen of stressed mice (Fig. 3). Propolis administration did not lead to such effects in the spleen.
Figure 3.
Histological analysis of the spleen of control (A) and stressed mice (B). Arrows indicate lymphoid follicles with germinal center.
Discussion
Glucose and corticosterone are used as stress indicators, and as observed in Table 1, stressed mice showed glycemic values higher than control, although this difference was not statistically significant. Kioukia-Fougia et al. (1) also found no differences in glucose serum levels using three different stress procedures. In fact, glucose concentration may increase after acute stress, and return to normal levels after prolonged stress. HPA axis has an important role in behavioral and immunological responses during stress. However, no differences were seen in corticosterone serum levels, although it was higher in stressed mice (P > 0.05) (Table 1). Azpiroz et al. (20) also observed no differences in corticosterone of chronically stressed mice, suggesting an adaptive response of HPA axis in the presence of higher concentrations of glucocorticoids.
Our propolis sample, collected in the Beekeeping Section of the University, was analyzed by GC, GC-MS and TLC, revealing that its main components are phenolic compounds (flavonoids, aromatic acids, benzopyranes), di- and triterpenes, essential oils, among others.
The main constituents of our propolis sample were isolated and identified: flavonoids are present in small quantities in Brazilian propolis (kaempferid, 5,6,7-trihydroxy-3,4′-dimethoxyflavone, aromadendrine-4′-methyl ether); a prenylated P-coumaric acid and two benzopyranes: E and Z 2,2-dimethyl-6-carboxyethenyl-8-prenyl-2H-benzopyranes); essential oils (spathulenol, (2Z,6E)-farnesol, benzyl benzoate and prenylated acetophenones); aromatic acids (dihydrocinnamic acid, P-coumaric acid, ferulic acid, caffeic acid, which are common for poplar propolis, 3,5-diprenyl-P-coumaric acid, 2,2-dimethyl-6-carboxy-ethenyl-8-prenyl-2H-1-benzo-pyran); di- and triterpenes, among others. Seasonal variations in propolis composition are not significant and are predominantly quantitative (12–14).
Propolis immunomodulatory action has been widely investigated in our laboratory (7–11,21,22). Thus, in order to verify a possible effect of this bee product during stress, macrophage activation was evaluated after propolis treatment of stressed mice. H2O2 generation by peritoneal macrophages of stressed mice was higher than control, although this difference was not statistically significant (P > 0.05). Propolis-treated mice showed an elevation in H2O2 generation, which is in accordance with previous works of our group (9). Propolis administration to stressed mice potentiated H2O2 generation (P < 0.05), suggesting its important action, stimulating macrophage activation. Palermo-Neto et al. (23), evaluating the foot shock stress during 6 days, also observed an increase in H2O2 liberation by macrophages.
Stressed mice showed a mild decay in NO metabolite liberation as compared with control. Similarly, Palermo-Neto et al. (23), evaluating the effects of maternal stress during pregnancy in the immune response of the offspring, verified no alterations in NO production, as well as a decay in spreading and phagocytosis index of macrophages.
Propolis-treated mice showed an inhibition in NO production. Propolis administration to stressed mice also resulted in NO inhibition. Previous investigations of our laboratory also showed that propolis inhibits NO generation by peritoneal macrophages of BALB/c mice (9).
Although H2O2 and NO production by peritoneal macrophages of mice were evaluated, one may not exclude the possible stress influence in other microbicidal mechanisms.
Selye (24) related alterations in thymus, lymph nodes and spleen of stressed mice, as an adaptive response to stress and a consequence of glucocorticoids action, after HPA axis activation. In our work, histological analysis of thymus, bone marrow and adrenal of stressed mice showed no morphological alterations, what could be attributed to the kind and period of stress. On the other hand, there was an increase in the number of germinal centers in the spleen of stressed animals (Fig. 3). Propolis administration did not lead to such effects in the spleen.
Domínguez-Gerpe and Rey-Méndez (3), using a model of immobilization during 14 days, observed a reduction of thymocytes and B cells in lymph nodes, relating that the immune system is compartmentalized and that the response to a stressor may be different in each compartment. These authors also related an increase in T cell number in the spleen and altered percentage of B cells. Conversely, Avitsur et al. (25), in a model of social stress, observed a reduction of T cells in the spleen, but normal numbers of B cells.
Our data showed that propolis potentiated H2O2 generation by stressed mice. Moreover, morphological alterations in the spleen of stressed mice were not seen when mice were treated with propolis, suggesting that this bee product may be useful during stress. Since propolis shows several biological properties (26–28) and it is used as an alternative medicine for health amelioration and disease prevention, new research is being carried out in our laboratory in order to investigate propolis action on cytokine production during stress.
References
- 1.Kioukia-Fougia N, Antoniou K, Bekris S, Liapi C, Christofidis I, Papadopoulou-Daifoti Z. The effects of stress exposure on the hypothalamic-pituitary-adrenal axis, thymus, thyroid hormones and glucose levels. Prog Neuro-Psychoph. 2002;26:823–30. doi: 10.1016/s0278-5846(01)00297-4. [DOI] [PubMed] [Google Scholar]
- 2.Dhabhar FS. Stress-induced augmentation of immune function – the role of stress hormones, leukocyte trafficking, and cytokines. Brain Behav Imm. 2002;16:785–98. doi: 10.1016/s0889-1591(02)00036-3. [DOI] [PubMed] [Google Scholar]
- 3.Domínguez-Gerpe L, Rey-Méndez M. Alterations induced by chronic stress in lymphocyte subsets of blood and primary and secondary immune organs of mice. Immunology. 2001;2:7. doi: 10.1186/1471-2172-2-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Banskota AH, Tezuka V, Kadota S. Recent progress in pharmacological research of propolis. Phytother Res. 2001;15:561–71. doi: 10.1002/ptr.1029. [DOI] [PubMed] [Google Scholar]
- 5.Bankova V. Recent trends and important developments in propolis research. eCAM. 2005;2:29–32. doi: 10.1093/ecam/neh059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bankova V, Boudourova-Krasteva G, Sforcin JM, Frete X, Kujumgiev A, Maimoni-Rodella R, Popov S. Phytochemical evidence for the plant origin of Brazilian propolis from São Paulo State. Z Naturforsch. 1999;54c:401–5. doi: 10.1515/znc-1999-5-616. [DOI] [PubMed] [Google Scholar]
- 7.Murad JM, Calvi SA, Soares AMVC, Bankova V, Sforcin JM. Effects of propolis from Brazil and Bulgaria on fungicidal activity of macrophages against Paracoccidioides brasiliensis. J Ethnopharmacol. 2002;79:331–4. doi: 10.1016/s0378-8741(01)00404-4. [DOI] [PubMed] [Google Scholar]
- 8.Orsi RO, Sforcin JM, Funari SRC, Bankova V. Effects of Brazilian and Bulgarian propolis on bactericidal activity of macrophages against Salmonella Typhimurium. Int Immunopharmacol. 2005;5:359–68. doi: 10.1016/j.intimp.2004.10.003. [DOI] [PubMed] [Google Scholar]
- 9.Orsi RO, Funari SRC, Soares AMVC, Calvi SA, Oliveira SL, Sforcin JM, Bankova V. Immunomodulatory action of propolis on macrophage activation. J Venom Anim Toxins. 2000;6:205–19. [Google Scholar]
- 10.Sforcin JM, Orsi RO, Bankova V. Effects of propolis, some isolated compounds and its source plant on antibody production. J Ethnopharmacol. 2005;98:301–5. doi: 10.1016/j.jep.2005.01.042. [DOI] [PubMed] [Google Scholar]
- 11.Sforcin JM, Kaneno R, Funari SRC. Absence of seasonal effect on the immunomodulatory action of Brazilian propolis on natural killer activity. J Venom Anim Toxins. 2002;8:19–29. [Google Scholar]
- 12.Boudourova-Krasteva G, Bankova V, Sforcin JM, Nikolova N, Popov S. Phenolics from Brazilian propolis. Z Naturforsch. 1997;52c:676–9. [Google Scholar]
- 13.Bankova V, Boudourova-Krasteva G, Popov S, Sforcin JM, Funari SRC. Seasonal variations in essential oil from Brazilian propolis. J Essent Oil Res. 1998a;10:693–6. [Google Scholar]
- 14.Bankova V, Boudourova-Krasteva G, Popov S, Sforcin JM, Funari SRC. Seasonal variations of the chemical composition of Brazilian propolis. Apidologie. 1998b;29:361–7. [Google Scholar]
- 15.Pick E, Mizel D. Rapid microassay for the measured of hydrogen peroxide produced by macrophages in culture using an automatic enzyme immunoassay reader. J Immunol Met. 1981;46:216–26. doi: 10.1016/0022-1759(81)90138-1. [DOI] [PubMed] [Google Scholar]
- 16.Green LC, Ruiz De Luzuriaga K, Wagner DA, Rand W, Istfand N, Young VR, et al. Nitrate biosyntesis in man. Proc Natl Acad Sci. 1989;78:7764–8. doi: 10.1073/pnas.78.12.7764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kuper CF, Harleman JH, Richter-Reichelm HB, Vos JG. Histopathologic approaches to detect changes indicative of immunotoxicity. Toxicol Pathol. 2000;28:454–66. doi: 10.1177/019262330002800317. [DOI] [PubMed] [Google Scholar]
- 18.Jones TC, Carpen CC, Mohr U. Monographs on Pathology of Laboratory Animals. International Life Sciences Institute. New York: Springer; 1996. Endocrine system; p. 521. [Google Scholar]
- 19.Zar JH. Biostatistical Analysis. 2nd. Englewood Cliffs: Prentice-Hall; 1996. p. 718. [Google Scholar]
- 20.Azpiroz A, Fano E, Garmendia L, Arregi A, Cacho R, Beitia G, et al. Effects of chronic mild stress (CMS) and imipramine administration, on spleen mononuclear cell proliferative response, serum corticosterone level and brain norepinephrine content in male mice. Psychoneuroendocrino. 1999;24:345–61. doi: 10.1016/s0306-4530(98)00084-5. [DOI] [PubMed] [Google Scholar]
- 21.Lopes FC, Bankova V, Sforcin JM. Effect of three vegetal sources of propolis on macrophages activation. Phytomedicine. 2003;10:343. doi: 10.1078/094471103322004848. [DOI] [PubMed] [Google Scholar]
- 22.Sá-Nunes A, Faccioli LH, Sforcin JM. Propolis: lymphocyte proliferation and IFN-gamma production. J Ethnopharmacol. 2003;87:93–7. doi: 10.1016/s0378-8741(03)00121-1. [DOI] [PubMed] [Google Scholar]
- 23.Palermo-Neto J, Massoco CO, Fávare RC. Effects of maternal stress on anxiety levels, macrophage activity, and Ehrlich tumor growth. Neurotoxicol Teratol. 2001;23:497–507. doi: 10.1016/s0892-0362(01)00164-7. [DOI] [PubMed] [Google Scholar]
- 24.Selye H. The Stress of Life. New York: McGraw-Hill; 1978. [Google Scholar]
- 25.Avitsur R, Stark JL, Dhabhar FS, Sheridan JF. Social stress alters splenocyte phenotype and function. J Neuroimmunol. 2002;132:66–71. doi: 10.1016/s0165-5728(02)00310-7. [DOI] [PubMed] [Google Scholar]
- 26.Inokuchi Y, Shimazawa M, Nakajima Y, Suemori S, Mishima S, Hara H. Brazilian green propolis protects against retinal damage in vitro and in vivo. eCAM. 2006;3:71–7. doi: 10.1093/ecam/nek005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Shimazawa M, Chikamatsu S, Morimoto N, Mishima S, Nagai H, Hara H. Neuroprotection by Brazilian green propolis against in vitro and in vivo ischemic neuronal damage. eCAM. 2005;2:201–7. doi: 10.1093/ecam/neh078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Trusheva B, Popova M, Bankova V, Simova S, Marcucci MC, Miorin PL, et al. Bioactive constituents of Brazilian red propolis. eCAM. 2006;3:249–54. doi: 10.1093/ecam/nel006. [DOI] [PMC free article] [PubMed] [Google Scholar]