Abstract
The normal expression pattern of the Wnt responsive homeobox gene Siamois is restricted to the dorso-vegetal region of the Xenopus embryo. Because the Wnt signaling pathway (via β-catenin) is active on the entire dorsal side of the early embryo, we have asked why Siamois expression is not seen in the dorsal ectoderm. Only Wnt signaling, via activation of β-catenin, can induce directly Siamois, and signaling via the SMAD1 (BMP2/4) or SMAD2 (activin/Vg-1) pathways cannot. We now directly show that the SMAD2 pathway can cooperate with the Wnt pathway to induce expression of Siamois much more strongly than the Wnt pathway alone, in normal embryos. We demonstrate the significance of this cooperation in normal embryos by blocking the SMAD2 signaling pathway with a dominant negative activin receptor. The activin dominant negative receptor blocks this cooperative effect and reduces the expression of Siamois by threefold in early embryos. Furthermore, we find that this cooperative relationship between the SMAD2 and Wnt pathways is reciprocal. Thus, in normal embryos, the Wnt pathway can enhance induction, by the SMAD 2 pathway, of the organizer genes Gsc and Chd but not the pan-mesodermal marker genes Xbra and Eomes. We conclude that the Wnt and SMAD2 signaling pathways cooperate to induce the expression of Spemann-organizer specific genes and so help to localize their spatial expression.
The Spemann organizer is formed in the dorsal most region of the mesoderm in Xenopus (1). It secretes molecules, such as chordin (2), noggin (3) and Frzb (4), which are involved in mesodermal patterning and are important in the later induction of neural genes in the overlying ectoderm (1, 5). Many of these signaling molecules are expressed specifically in the organizer region of the early gastrula. There are two main regional signaling activities that are thought to be involved in inducing these immediate-early genes (6). First, a transforming growth factor β-like mesoderm-inducing activity (the “Nieuwkoop center”) is thought to be mediated via the activin/Vg-1 pathway and is present in the vegetal cells and in part of the presumptive mesoderm (7, 8). Second, a Wnt-signaling domain is active on the whole dorsal side of the early embryo (9, 10) and acts via nuclear localization of maternal β-catenin, a component of the Wnt-signaling pathway (11). Genes that are expressed in the Spemann organizer fall into two classes: those that are induced by an activin/Vg-1 signal [for example Xgsc (12) and Chordin (2)] and those that are induced by Wnt signaling [for example Siamois (13) and Xnr3 (14)].
Because the maternal β-catenin is active throughout the whole dorsal side of the embryo (11), it might be expected that these Wnt-responsive organizer genes also would be expressed throughout this region. However, the expression pattern of the homeobox gene Siamois is restricted to the dorsal mesendoderm and never is seen in the overlying dorsal ectoderm (13). Therefore, the localization of some other material, in addition to that of β-catenin, is required to explain the localized expression of Siamois (and subsequent organizer genes). We test here the possibility that a spatial overlap between the Wnt and activin pathways may be responsible for the localization of Siamois expression. That Siamois is expressed only in the area of overlap between these two pathways has led us to ask whether there is any cooperation between these pathways in restricting the location of Wnt-responsive gene expression to the Spemann organizer. It is known that ectopic dorsalization of UV ventralized animal cap tissue can be achieved by exposure to a combination of Vg1 and Xwnt8 (15). The presence of a Wnt-responsive element in the Xgsc promoter (8), an activin/Vg-1 responsive gene, has suggested that these two pathways are not entirely independent. Furthermore, there is evidence that Siamois is induced by an activin/Vg-1 signal, albeit at a low level (15, 16), as well as a Wnt signal, although some authors do not see this (17, 18).
Here we show that, although Siamois is not induced directly by activin/Vg-1 signaling, the activin/Vg1 (SMAD2) pathway but not the BMP-4 (SMAD1) pathway cooperates with β-catenin to induce Siamois and Xnr3 much more strongly than β-catenin alone. We show that this cooperation is required for Siamois expression in normal development and that a reciprocal cooperation occurs with those activin/Vg-1 responsive genes that are expressed in the organizer, such as Xgsc and Chd, but not with pan-mesodermal genes like Xbrachyury and Eomesodermin. We conclude that a cooperation between Wnt and activin/Vg-1 signaling is sufficient to explain the localized expression pattern of Spemann organizer genes. This cooperation may illustrate a general mechanism by which the spatial restriction of early gene transcription is localized by the overlapping domains of maternal signaling pathways.
METHODS AND REAGENTS
Synthesis and Microinjection of mRNA.
The capped mRNAs were synthesized in vitro (19) by using Ambion Megascript (Austin, TX) SP6 and T7 kits. The clones used were as follows: XE28 β-catenin (a gift of R. T. Moon, University of Washington School of Medicine, Seattle) linearized by NotI and transcribed by SP6; rat glycogen synthase kinase 3β (GSK-3β) (a gift of N. Papalopulu, Wellcome CRC Institute, Cambridge, UK) linearized by SalI and transcribed by T7; activin dominant negative receptor Δ-XAR (a gift of A. Hemmati-Brivanlou, Rockefeller University, New York) linearized by EcoRI and transcribed by SP6; pSP64TNE-Xmad1 and pSP64TNE-Xmad2 (both gifts of D. A. Melton, Howard Hughes Medical Institute, Harvard University) linearized by XbaI and transcribed by SP6.
Embryos were fertilized in vitro, dejellied, and cultured as described (20). Embryos were injected in the animal cap with a known amount of the mRNA solution at the two-cell stage into both blastomeres by using a Drummond Scientific Nanoject system (Broomall, PA). Embryos were injected in the dorsal or ventral equatorial regions at the four-cell stage. Animal caps were dissected at stage 8.5 in 1× strength modified Barth–Hepes saline (20) and cultured to stage 10 or 10.5 and then frozen on dry ice for analysis by RNase protection assays. Whole embryos were cultured to stage 10 or 10.5 and frozen as before.
RNase Protection Assays.
RNase protection assays were carried out as described (21). The fibroblast growth factor receptor (FGF-R), Sia, Xbra, and Xgsc probes were the same as those used previously (21). The Eomes and Chd probes were as described (22). The Xnr3 probe was as described (17). Quantitation of RNase protection assay gels was performed by using a PhosphorImager and the imagequant software package (Molecular Dynamics).
RESULTS
Siamois Is Induced Ectopically in Response to Wnt Signaling but Not Vg-1 Signaling.
To address the question of why the expression of the homeobox gene Siamois is restricted to the dorso-vegetal region when the Wnt pathway also is active in the dorsal ectoderm, we have asked which signaling activities are able to induce Siamois directly in the ectoderm. Two regional and one ubiquitous signaling activities are known to be present before the onset of zygotic transcription, any one of which could be required for Siamois expression and so help to restrict this to the dorsal vegetal region: maternal β-catenin (23), an activin-like transforming growth factor β signal (8), and maternal BMP-2 (24).
Two-cell stage embryos were injected with doses between 5 pg and 1 ng of β-catenin mRNA in the animal pole, a region whose normal fate is to form the ectoderm. Injected embryos were cultured until the early gastrula stage (stage 10.25), when the ectoderm was isolated, and induced gene transcripts then were analyzed by RNase protection. Fig. 1A shows that, as previously reported (16), β-catenin is able to induce strongly Siamois in animal cap tissue and in a dose-dependent manner.
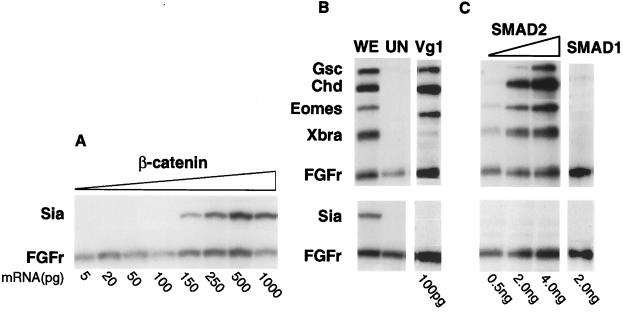
Figure 1.
RNase protection analyses of Siamois expression in animal cap explants. (A) Increasing doses of β-catenin mRNA (in picograms) were injected into two-cell stage eggs. Animal caps were explanted at the mid-blastula stage and cultured to the early gastrula (stage 10.25). Siamois is expressed in response to doses of 150 pg and above of β-catenin mRNA, and the FGF-R is present as a loading control. (B) BVg-1 mRNA (100 pg) was injected into animal caps; explants were taken at the mid-blastula stage and cultured until the very early gastrula (stage 10). The mesodermal marker genes Xgsc, Chd, Eomes, and Xbra are shown as positive controls for Vg-1 activity. (C) Increasing doses of SMAD2 mRNA (0.5–4.0 ng) or 2 ng of SMAD1 mRNA were injected into animal caps, and explants were taken at the mid-blastula stage; these were cultured to the early gastrula (stage 10.25). Expression in one whole embryo equivalent (WE) is shown as a positive control and uninjected animal caps (UN) as negative controls. In all cases, 10 animal cap explants were analyzed per injection.
Previous reports have provided conflicting evidence as to whether Vg1 (an activin-like transforming growth factor β signal) can induce directly Siamois. Many early genes are known to be inducible by Vg-1/activin in a dose-dependent manner, but the amounts of Vg-1 previously used may have been too low to see induction of Siamois in animal caps. We have repeated this experiment with a stronger, 100-pg injection of BVg-1 mRNA. Animal cap explants were isolated at stage 8.5 and analyzed as in Fig. 1A, except that they were frozen at an earlier stage (stage 10), when Siamois expression is at its peak. We found that Vg-1 strongly induces mesodermal genes such as Eomes (25), Chd, and Xgsc, but no Siamois expression is seen (Fig. 1B). The weaker than expected induction of Xbra is due to the early stage at which these explants were frozen. We have checked the validity of this result by using the intracellular transforming growth factor β signaling component, SMAD2 (25–27). SMAD2 is thought to transduce the signaling of both activin and Vg1 and thus to stimulate the signaling pathway of a putative mesoderm inducer. Two-cell stage embryos were injected with increasing doses of SMAD2 mRNA, from 500 pg to 4 ng, and treated as before. Fig. 1C shows the dose-dependent increase in mesodermal gene transcription in response to injection of SMAD2 mRNA in animal caps. As with Vg-1, mesodermal markers are induced strongly, but no Siamois expression is seen at any dose of SMAD2 (Fig. 1C). We conclude that activin/Vg1 (SMAD2) signaling cannot induce Siamois expression in animal caps at doses up to 30 times stronger than required to induce the activin responsive genes such as Eomes.
Maternal BMP2 signaling, as well as zygotic BMP4 signaling, is believed to be transduced by SMAD1 (25, 28–30). To see whether BMP signaling can activate directly Siamois expression, two-cell stage embryos were injected with 2 ng of SMAD1 and treated as above. Fig. 1C shows that Siamois is not induced by SMAD1 signaling; SMAD1 activity was verified by assaying for induction of β-globin at stage 36 by reverse transcriptase PCR (data not shown) (25). We conclude that only Wnt signaling, via activation of β-catenin, can induce directly Siamois and that signaling through either the SMAD1 or SMAD2 pathways cannot.
Activin/Vg1 Signaling Cooperates with Wnt Signaling to Induce Siamois Strongly.
Siamois expression is seen only within the region of overlap between the SMAD2 (equivalent to Vg-1/activin) and Wnt signaling domains, which led us to ask whether SMAD2 signaling may have a role to play in Siamois expression. Despite not being able to induce directly Siamois in animal caps, SMAD2 might provide a cooperative signal that is required for its in vivo expression in the dorso-vegetal region. To test this, we used combinations of signaling molecules to recreate in animal caps the cooperative conditions that may exist normally in the dorso-vegetal region. We also used Xwnt8 in our assays because it is upstream of β-catenin and thus allows very strong stimulation of the Wnt signaling pathway.
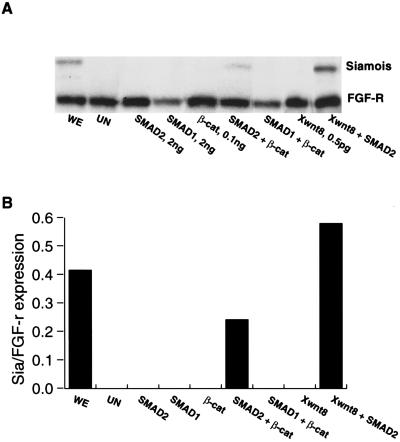
Two-cell stage embryos were injected in the animal pole with 2 ng of SMAD2, 2 ng of SMAD1, 100 pg of β-catenin, or 0.5 pg of Xwnt8 mRNAs either singly or in combination. We chose doses of Xwnt8 (0.5 pg) and β-catenin (100 pg) that are just below the level required to see induction of Siamois (see Fig. 1A). The animal cap explants were analyzed at stage 10.25 for Siamois expression. SMAD2, SMAD1, and the low doses of β-catenin or Xwnt8 failed to induce Siamois expression on their own (Fig. 2). However, combinations of SMAD2 and β-catenin or SMAD2 and Xwnt8 were able to induce Siamois to a level similar to that seen in whole embryos (WE). This cooperative effect is not seen with an equivalent amount of SMAD1 mRNA. We conclude that there is strong cooperation between the Wnt and SMAD2 signals to induce the expression of Siamois in the ectoderm. The effect of this cooperative signaling provides a possible explanation for why Siamois expression is localized to the dorsal vegetal region and is not present in the overlying dorsal animal cap.
Figure 2.
SMAD2 but not SMAD1 cooperates with β-catenin and Wnt to induce expression of Siamois in animal caps. (A) RNase protection analysis of Siamois expression in animal cap explants at the early gastrula (stage 10.25). The experimental procedure was as in Fig. 1A; the amounts of mRNA injected were: 2 ng of SMAD2, 2 ng of SMAD1, 100 pg of β-catenin, and 0.5 pg of Xwnt8. The same amounts of these mRNAs were injected in the relevant combinations. (B) Quantitation of the level of Siamois expression; each bar shows the ratio of Siamois to FGF-R (quantitated by PhosphorImager analysis). This experiment was repeated three times and achieved the same result on each occasion.
Blocking the Activin/Vg1 Cooperative Signal Down-Regulates Siamois Expression in Normal Embryos.
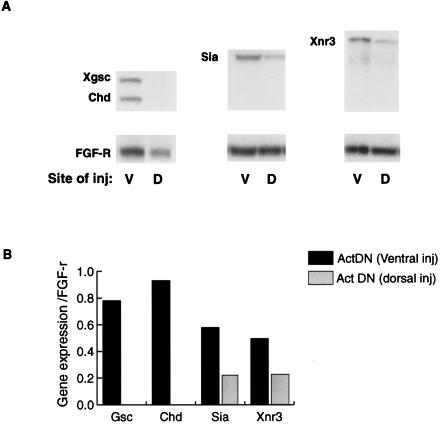
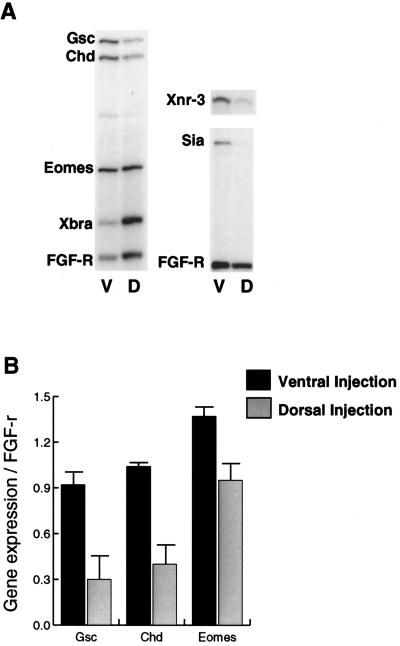
If the cooperation that we have shown in Fig. 2 is of significance in normal development, it would be predicted that an inhibition of the SMAD2 signaling pathway in the dorsal mesendoderm would lead to a down-regulation of normal Siamois expression. To test this, we have overexpressed the Δ-XAR, activin dominant negative receptor (Act DN), which blocks activin, Vg1, and BMP signaling (31, 32), thus reducing the cooperative effect of the SMAD2 pathway (16). We injected 500 pg of mRNA encoding the activin dominant negative receptor into the dorsal subequatorial (presumptive organizer) or ventral subequatorial regions (as a control). Whole embryos were cultured until stage 10, when they were frozen for analysis of gene transcripts, by RNase protection assay. As expected, Xgsc and Chd expression was eliminated by injection on the dorsal side (Fig. 3). However, expression of Siamois and Xnr3 (also a Wnt responsive organizer gene) also was reduced by >50% (Fig. 3A). We conclude that the cooperation we showed for the expression of Siamois in the ectoderm also is required for Siamois expression in its normal dorsal mesendodermal region. This requirement provides further support for the previous observation that, in the absence of a cooperative SMAD2 signal, Siamois is not expressed in the dorsal ectoderm and suggests that organizer gene expression is induced in the region of overlap between Wnt and activin/Vg1 signaling domains.
Figure 3.
The activin dominant negative receptor (Act DN) down-regulates the expression of Siamois in whole embryos. (A) Act DN mRNA (500 pg) was injected into either the ventral (V) or dorsal (D) sub-equatorial region of four-cell stage embryos. Whole embryos were cultured to stage 10, and expression of Siamois as well as the SMAD2 responsive genes Xgsc, and Chd and the β-catenin responsive gene Xnr3 was analyzed by RNase protection assays. (B) The levels of expression of these genes has been quantitated relative to expression of the FGF-R loading control. This experiment was repeated twice and achieved the same result both times.
The Wnt Pathway Enhances the SMAD2 Pathway to Induce Xgsc and Chd Ectopically.
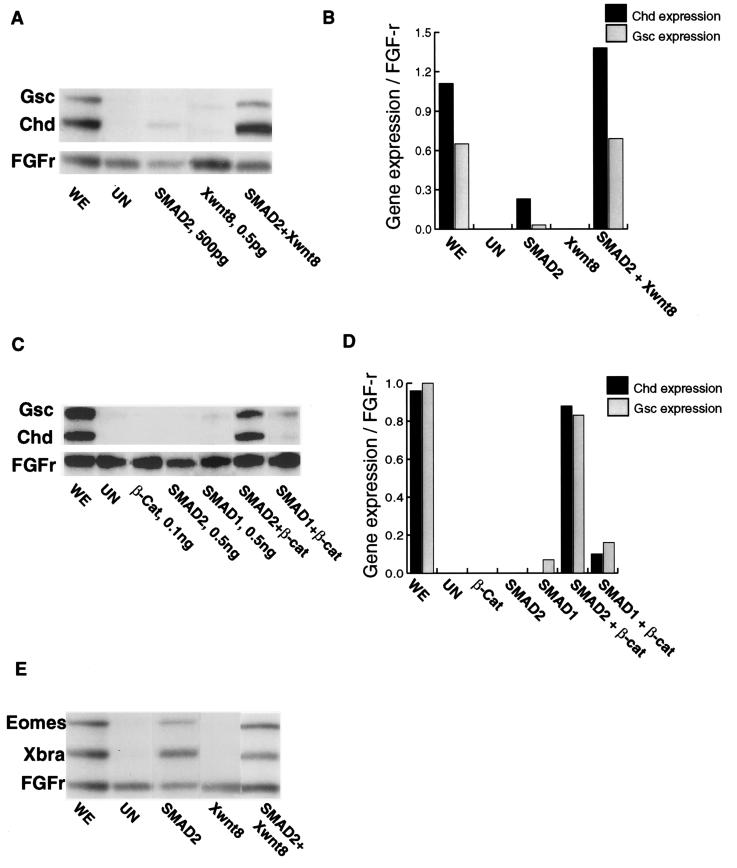
Our finding that the SMAD2 pathway enhances the Wnt pathway led us to ask whether the reciprocal relationship also applies, that is, does the Wnt pathway enhance SMAD2 signaling? There is previous evidence that the Wnt pathway can act as a dorsalizer of already induced mesoderm (33); furthermore, the presence of a Wnt responsive element in the Xgsc promoter (8) suggests that Wnt signals may provide a cooperative stimulus for the induction of genes responsive to the activin/Vg1 signaling pathway. We therefore have looked to see whether this cooperation is required for the expression of dorsal mesodermal genes, e.g., Xgsc and Chd, which are induced by an activin/Vg1 signal. A low concentration of SMAD2 in animal caps is not able to induce appreciable levels of transcription of Chd or Xgsc (Fig. 4A), but it is able to induce expression of the pan-mesodermal genes Xbra and Eomes (Fig. 4E). None of these four genes is induced by the Wnt pathway, as evidenced by the injections of Xwnt8 or β-catenin (Fig. 4 A–E). However, the combination of a low dose (500 pg) of SMAD2 with either Xwnt8 (0.5 pg) or β-catenin (100 pg) strongly induced the expression of Xgsc and Chd (Fig. 4 A–C) to levels at least six times higher than those seen with SMAD2 alone. It is important to note that this cooperative effect is not seen for the pan-mesodermal markers Eomes and Xbra; indeed, the expression of Xbra is reduced slightly (Fig. 4E). This slight but reproducible effect can be explained by the repression of Xbra transcription by increased levels of Xgsc (34). Eomes is not repressed by Xgsc, and consequently, its expression is unaffected (22). It could be argued that cooperation is not seen for the pan-mesodermal genes because maximum levels of transcription of these genes already is reached in the SMAD2 alone condition. However, we have shown previously (Fig. 1) that a dose of 500 pg of SMAD2 mRNA does not induce maximal expression of either Eomes or Xbra. We conclude that, for those genes induced by the activin/Vg-1 pathway, Wnt signaling provides a cooperative effect only for those with organizer-specific expression.
Figure 4.
RNase protection analyses of animal cap explants injected with mRNA, as described in Fig. 1A, and cultured to stage 10.25. Explants were injected with 500 pg of SMAD2 or SMAD1, 0.5 pg of Xwnt8, 100 pg of β-catenin (β-Cat), or their corresponding combinations. (A) The SMAD2 responsive organizer genes Xgsc and Chd are expressed more strongly when SMAD2 mRNA is combined with Xwnt8 mRNA. Whole embryo (WE) expression is shown, and uninjected animal cap explants (UN) are included as a negative control. (B) Quantitation of this gel shows the levels of expression Xgsc and Chd as a proportion of FGF-R loading control. (C) SMAD2 and β-catenin mRNAs induce expression of Xgsc and Chd more strongly than SMAD2 alone. SMAD1 does not cooperate with β-catenin to induce expression of these genes. (D) Quantitation of this gel shows the levels of expression Xgsc and Chd as a proportion of FGF-R loading control. (E) Expression of the SMAD2 responsive pan-mesodermal markers Xbra and Eomes is not enhanced by coinjecting SMAD2 mRNA with Xwnt8 mRNA. These experiments were repeated three times, achieving the same result each time.
Blocking Wnt Cooperative Signal Down-Regulates Organizer-Specific, but Not Pan-Mesodermal, Gene Expression in Vivo.
To determine whether this cooperative effect we see plays a role in normal development, we have used GSK-3β to block Wnt signaling activity in the presumptive organizer region. GSK-3β is known to function by phosphorylating and targeting for degradation the Wnt signal transduction molecule β-catenin (35). As expected, GSK-3β mRNA injected into the dorsal region inhibits the Wnt responsive genes Siamois and Xnr3 (Fig. 5A). Moreover, it also inhibits Xgsc and Chd, which are primarily under the control of the SMAD2 pathway (Fig. 5A). Eomes also shows a moderate reduction as expected for a gene that is expressed in part within the organizer, but there is an increase in expression of Xbra, which normally is not expressed in the dorsal organizer region at this stage. We believe that this increase in Xbra expression is due to a reduction in repression by the lower levels of endogenous Xgsc.
Figure 5.
The injection of GSK-3β mRNA in the dorsal sub-equatorial region of embryos represses the expression of the SMAD2 responsive organizer genes Xgsc and Chd. (A) RNase protection analysis of whole embryos injected sub-equatorially with 500 pg of GSK-3β mRNA in either the ventral (V) or dorsal (D) blastomeres at the four-cell stage and then cultured to the early gastrula stage (stage 10.25). The reduced level of Sia and Xnr-3 expression on the dorsal side shows that GSK-3β is active. (B) Quantitation of these gel analyses shows the levels of expression, relative to FGF-R loading control, of the genes Xgsc, Chd, and Eomes; error bars are shown for six independent analyses.
DISCUSSION
We have established that Siamois is induced directly by the Wnt pathway but not by Vg-1 through the SMAD2 pathway. There are some previous reports (15, 16) that have given the impression that Siamois is induced weakly by the activin/Vg-1 pathway (see Introduction). It is conceivable that, in these reports, maternal β-catenin activity within a dorsal animal cap may have cooperated with injected Vg-1 mRNA to induce weakly expression of Siamois. This cooperation could have occurred if particularly large animal cap explants were assayed incorporating the most dorsal ectodermal tissue and thereby giving the impression that Siamois was induced directly via the activin/Vg-1 pathway.
We have shown that the SMAD2 and Wnt pathways have a profound influence on the expression of each other’s target genes. We have demonstrated that the activin/Vg-1 and Wnt pathways show clear cooperation with respect to the induction of organizer-specific genes but not for the pan-mesodermal genes responsive to the activin/Vg-1 pathway. It has been the prevailing view (15) that the Wnt pathway does indeed influence the most dorsal responses to an activin/Vg-1 signal, but we believe that our results clearly demonstrate the existence and reciprocal nature of this relationship and its requirement for normal levels of organizer-specific gene expression. Previous studies on the Xgsc promoter (8) have identified the presence of both activin-responsive and Wnt-responsive elements. We suspect that similar elements may be present in the promoters of all organizer-specific genes; in those that are induced by a Wnt signal, the activin responsive element may act as an enhancer of transcription and vice versa. It is important to remember, however, that, of the activin-responsive elements presently known, few conform to a defined consensus and that there is a great deal of complexity within these signaling pathways that is yet to be understood.
In the absence of a definitive in vivo mesoderm inducer, the use of activin and Vg-1 can be questioned. However, these are the strongest mesoderm-inducing molecules so far known. Furthermore, we have tested the effects of the intracellular signal transduction molecules SMAD2 and β-catenin, known to be part of the Vg-1/activin and Wnt signaling pathways, respectively.
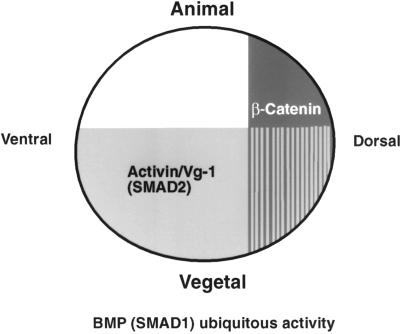
Our primary aim in pursuing this work was to seek an explanation for how the major activin/Vg-1-like and Wnt-like signaling processes present at mid blastula transition could define the position of the Spemann organizer. We believe that our results support the hypothesis that the overlapping domains of the Wnt and activin/Vg-1 signaling pathways are capable of defining the location of the organizer in the dorsal equatorial region of the embryo, as indicated in Fig. 6.
Figure 6.
A diagram to show how the SMAD2 and β-catenin signaling domains overlap in the dorsal mesendoderm and may cooperate to induce expression of organizer-specific genes in this region.
An important further question to ask is how these signaling domains lay down the different patterns of expression of particular genes within the organizer itself. Xnr3 is restricted to the dorsal epithelial layer, whereas Siamois and Xgsc are widely expressed throughout the organizer, with Siamois also extending into the dorsal vegetal tissue. The discrete expression domains of these genes within the organizer region could be due to a gradient of activin/Vg-1 across the tissue. Perhaps each of these organizer genes is induced optimally at different levels of SMAD2 and β-catenin activity. Such a system could impart a fine level of patterning information across the early organizer domain.
Acknowledgments
We thank P. Lemaire and S. Darras for critical reading of the manuscript. We are grateful to R. T. Moon, N. Papalopulu, A. Hemmati-Brivanlou, and D. A. Melton for useful reagents. We also thank K. Ryan and K. Butler for help and advice. This work was supported by the Cancer Research Campaign. D.J.C and S.D. are also supported by the Medical Research Council.
ABBREVIATIONS
- Xgsc, Xenopusgoosecoid
Chd, Chordin
- Xbra
Xbrachyury
- Eomes
Eomesodermin
- Xnr3
Xenopus nodal related 3
- BMP2/4
bone morphogenetic protein 2/4
- GSK-3β
glycogen synthase kinase 3β
- FGF-R
fibroblast growth factor receptor
References
- 1.Lemaire P, Kodjabachian L. Trends Genet. 1996;12:525–531. doi: 10.1016/s0168-9525(97)81401-1. [DOI] [PubMed] [Google Scholar]
- 2.Sasai Y, Lu B, Steinbeisser H, Geissert D, Gont L K, De Robertis E M. Cell. 1994;79:779–790. doi: 10.1016/0092-8674(94)90068-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Smith W C, Harland R M. Cell. 1992;70:829–840. doi: 10.1016/0092-8674(92)90316-5. [DOI] [PubMed] [Google Scholar]
- 4.Leyns L, Bouwmeester T, Kim S H, Piccolo S, De Robertis E M. Cell. 1997;88:747–756. doi: 10.1016/s0092-8674(00)81921-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lamb T M, Knecht A K, Smith W C, Stachel S E, Economides A N, Stahl N, Yancopolous G D, Harland R M. Science. 1993;262:713–718. doi: 10.1126/science.8235591. [DOI] [PubMed] [Google Scholar]
- 6.Heasman J. Development. 1997;124:4179–4191. doi: 10.1242/dev.124.21.4179. [DOI] [PubMed] [Google Scholar]
- 7.Weeks D L, Melton D A. Cell. 1987;51:861–867. doi: 10.1016/0092-8674(87)90109-7. [DOI] [PubMed] [Google Scholar]
- 8.Watabe T, Kim S, Candia A, Rothbacher U, Hashimoto C, Inoue K, Cho K W. Genes Dev. 1995;9:3038–3050. doi: 10.1101/gad.9.24.3038. [DOI] [PubMed] [Google Scholar]
- 9.Rowning B A, Wells J, Wu M, Gerhart J C, Moon R T, Larabell C A. Proc Natl Acad Sci USA. 1997;94:1224–1229. doi: 10.1073/pnas.94.4.1224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wylie C, Kofron M, Payne C, Anderson R, Hosobuchi M, Joseph E, Heasman J. Development. 1996;122:2987–2996. doi: 10.1242/dev.122.10.2987. [DOI] [PubMed] [Google Scholar]
- 11.Larabell C A, Torres M, Rowning B A, Yost C, Miller J R, Wu M, Kimelman D, Moon R T. J Cell Biol. 1997;136:1123–1136. doi: 10.1083/jcb.136.5.1123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Cho K H Y, Blumberg B, Steinbeisser H, De Robertis E M. Cell. 1991;67:1111–1120. doi: 10.1016/0092-8674(91)90288-a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lemaire P, Garrett N, Gurdon J B. Cell. 1995;81:85–94. doi: 10.1016/0092-8674(95)90373-9. [DOI] [PubMed] [Google Scholar]
- 14.Smith W C, KcKendry R, Ribisi S J, Harland R M. Cell. 1995;82:37–46. doi: 10.1016/0092-8674(95)90050-0. [DOI] [PubMed] [Google Scholar]
- 15.Cui Y, Tian Q, Christian J L. Dev Biol. 1996;180:22–34. doi: 10.1006/dbio.1996.0281. [DOI] [PubMed] [Google Scholar]
- 16.Brannon M, Kimelman D. Dev Biol. 1996;180:344–347. doi: 10.1006/dbio.1996.0306. [DOI] [PubMed] [Google Scholar]
- 17.Carnac G, Kodjabachian L, Gurdon J B, Lemaire P. Development. 1996;122:3055–3065. doi: 10.1242/dev.122.10.3055. [DOI] [PubMed] [Google Scholar]
- 18.Fagotto F, Guger K, Gumbiner B M. Development. 1997;124:453–460. doi: 10.1242/dev.124.2.453. [DOI] [PubMed] [Google Scholar]
- 19.Krieg P A, Melton D A. Methods Enzymol. 1987;155:397–415. doi: 10.1016/0076-6879(87)55027-3. [DOI] [PubMed] [Google Scholar]
- 20.Gurdon J B, Tiller E, Roberts J, Kato K. Curr Biol. 1993;3:1–11. doi: 10.1016/0960-9822(93)90139-f. [DOI] [PubMed] [Google Scholar]
- 21.Lemaire P, Gurdon J B. Development. 1994;120:1191–1199. doi: 10.1242/dev.120.5.1191. [DOI] [PubMed] [Google Scholar]
- 22.Ryan K, Garrett N, Mitchell A, Gurdon J B. Cell. 1996;87:989–1000. doi: 10.1016/s0092-8674(00)81794-8. [DOI] [PubMed] [Google Scholar]
- 23.Heasman J, Crawford A, Goldstone K, Garner-Hamrick P, Gumbiner B, McCrea P, Kintner C, Yoshida Noro C, Wylie C. Cell. 1994;79:791–803. doi: 10.1016/0092-8674(94)90069-8. [DOI] [PubMed] [Google Scholar]
- 24.Hemmati-Brivanlou A, Thomsen G H. Dev Genet. 1995;17:78–89. doi: 10.1002/dvg.1020170109. [DOI] [PubMed] [Google Scholar]
- 25.Graff J M, Bansal A, Melton D A. Cell. 1996;85:479–487. doi: 10.1016/s0092-8674(00)81249-0. [DOI] [PubMed] [Google Scholar]
- 26.Baker J C, Harland R M. Genes Dev. 1996;10:1880–1889. doi: 10.1101/gad.10.15.1880. [DOI] [PubMed] [Google Scholar]
- 27.Sekelsky J J, Newfeld S J, Raftery L A, Chartoff E H, Gelbart W M. Genetics. 1995;139:1347–1358. doi: 10.1093/genetics/139.3.1347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Liu F, Hata A, Baker J C, Doody J, Carcamo J, Harland R M, Massague J. Nature (London) 1996;381:620–623. doi: 10.1038/381620a0. [DOI] [PubMed] [Google Scholar]
- 29.Wilson P A, Lagna G, Suzuki A, Hemmati-Brivanlou A. Development. 1997;124:3177–3184. doi: 10.1242/dev.124.16.3177. [DOI] [PubMed] [Google Scholar]
- 30.Thomsen G H. Development. 1996;122:2359–2366. doi: 10.1242/dev.122.8.2359. [DOI] [PubMed] [Google Scholar]
- 31.Hemmati-Brivanlou A, Melton D A. Nature (London) 1992;359:609–614. doi: 10.1038/359609a0. [DOI] [PubMed] [Google Scholar]
- 32.Schulte-Merker S, Smith J C, Dale L. EMBO J. 1994;13:3533–3541. doi: 10.1002/j.1460-2075.1994.tb06660.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sokol S Y, Melton D A. Dev Biol. 1992;154:348–355. doi: 10.1016/0012-1606(92)90073-p. [DOI] [PubMed] [Google Scholar]
- 34.Artinger M, Blitz I, Inoue K, Tran U, Cho K W. Mech Dev. 1997;65:187–196. doi: 10.1016/s0925-4773(97)00073-7. [DOI] [PubMed] [Google Scholar]
- 35.He X, Saint-Jeannet J P, Woodgett J R, Varmus H E, Dawid I B. Nature (London) 1995;374:617–622. doi: 10.1038/374617a0. [DOI] [PubMed] [Google Scholar]