Abstract
Broth microdilution was used to determine the MICs of AR-709 and comparator antimicrobial agents for 224 invasive multidrug-resistant isolates of Streptococcus pneumoniae. AR-709 was highly active, with a MIC50 of 0.25 μg/ml, a MIC90 of 0.5 μg/ml, and a range of ≤0.008 μg/ml to 1 μg/ml.
Streptococcus pneumoniae is a significant cause of meningitis, pneumonia, bacteremia, sinusitis, and otitis media, with a U.S. national estimate of over 40,000 cases of invasive disease per year and 1.6 deaths per 100,000 population (1). Of concern is the global emergence of drug-resistant S. pneumoniae, especially strains resistant to beta-lactams and macrolides which are commonly prescribed for community-acquired pneumonia (6, 10, 16). High rates of resistance to trimethoprim-sulfamethoxazole (SXT) have limited the utility of that inexpensive oral agent, and resistance to fluoroquinolones might become more common in the future (4). Important risk factors for antimicrobial resistance among invasive pneumococcal infections include nosocomial or nursing home acquisition and antimicrobial use in the preceding three months before the onset of infection (15, 17). Discordant empirical antimicrobial therapy has led to treatment failure, especially in cases of meningitis, otitis media, and community-acquired pneumonia, although increased mortality with discordant therapy in community-acquired pneumonia is less clear (9, 16, 18).
The emergence of multidrug-resistant (MDR) S. pneumoniae has prompted a need for newer antimicrobial agents. AR-709 is an investigational diaminopyrimidine that inhibits microbial dihydrofolate reductase and is under development by Arpida AG (Reinach, Switzerland) for the therapy of community-acquired upper and lower respiratory tract infections. In vitro studies have shown the drug to have excellent bactericidal activity against pneumococci (8). In addition, microdosing studies in humans have revealed that AR-709 achieves very high levels in different key lung compartments (12). The goal of this study was to assess the in vitro activity of AR-709 against invasive, highly drug-resistant strains of S. pneumoniae in the United States.
The S. pneumoniae isolates selected for testing in this study were recovered from blood and sterile body fluid sources collected from the CDC's Active Bacterial Core surveillance (ABCs) program during 2002 to 2006. The ABCs program surveys a total population of almost 27 million persons in ten states (California, Colorado, Connecticut, Georgia, Maryland, Minnesota, New Mexico, New York, Oregon, and Tennessee) and is a core component of the CDC's Emerging Infections Programs Network. These strains were previously tested with a battery of 15 antimicrobial agents, and isolates from eight of the different geographic sites (excluding GA and MN) that were found to be MDR (i.e., exhibiting resistance to agents from at least three different classes) or were fluoroquinolone resistant were selected for inclusion in this study.
A total of 224 isolates were tested, and MICs of AR-709 were determined for each isolate in addition to MICs of penicillin, amoxicillin, cefotaxime, meropenem, erythromycin, clindamycin, SXT, doxycycline, and levofloxacin. Isolates were obtained from patients of all ages (mean of 54 years), most of whom (138 [62%]) had bacteremic pneumonia. The most-common serotypes represented were 19A (54 isolates), 14 (54 isolates), 19F (23 isolates), 9V (23 isolates), and 6A (14 isolates).
Microdilution susceptibility testing was performed using the Clinical and Laboratory Standards Institute (CLSI)-recommended procedure incorporating cation-adjusted Mueller-Hinton lysed horse blood broth (2). Colonies grown for 20 to 24 h on sheep blood agar and grown at 35°C in 5% CO2 were suspended in 0.9% saline to obtain a suspension equivalent to the 0.5 McFarland standard. The inoculum was further diluted to provide a final inoculum density of approximately 5 × 105 CFU/ml in the wells of the broth microdilution panels. Following incubation at 35°C in ambient air for 20 to 24 h, MICs were read and interpreted using CLSI breakpoint criteria (3). S. pneumoniae ATCC 49619 was used for quality control and was tested on each day of testing.
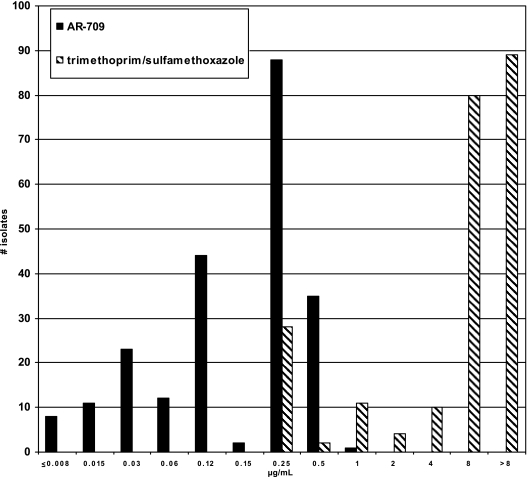
Of the 224 isolates tested, 202 isolates were MDR (82%, 71%, and 43% of the isolates were resistant to four, five, and six drug classes, respectively) and 22 isolates were selected because of fluoroquinolone resistance. Overall rates of resistance and the MICs of the isolate collection for the antimicrobial agents tested can be seen in Table 1. Despite a large percentage of isolates having SXT resistance, AR-709 was highly active against this group of MDR strains with a MIC50 of 0.25 μg/ml, a MIC90 of 0.5 μg/ml, and a range of ≤0.008 μg/ml to 1 μg/ml. Figure 1 illustrates the MIC distribution of AR-709 compared to that of SXT. AR-709 MICs were highest with high-level SXT-resistant strains but did not exceed 1 μg/ml with any strain. All isolates with AR-709 MICs of 0.5 μg/ml or 1 μg/ml had SXT MICs of ≥8 μg/ml; however, some isolates with high-level SXT resistance had very low MICs of AR-709.
TABLE 1.
S. pneumoniae MIC data for AR-709 and comparator antimicrobial agents
| Drug | MIC (μg/ml)
|
% Resistance | ||
|---|---|---|---|---|
| Range | 50% | 90% | ||
| AR-709 | ≤0.008-1 | 0.25 | 0.5 | NAb |
| Erythromycin | ≤0.03->32 | 8 | >32 | 83.5 |
| Penicillin | ≤0.03-8 | 4 | 4 | 85.7 |
| Cefotaxime | ≤0.03-16 | 1 | 2 | 41.1 |
| Amoxicillin | ≤0.03->8 | 2 | 8 | 48.2 |
| Meropenem | ≤0.03-2 | 0.5 | 1 | 75.9 |
| Clindamycin | ≤0.03->8 | 0.06 | >8 | 34.8 |
| Doxycycline | ≤0.06->8 | 0.12 | 8 | 40.6 |
| SXTa | ≤0.25->8 | 8 | >8 | 86.6 |
| Levofloxacin | ≤0.5->8 | 1 | >8 | 21.0 |
Tested in a 1:19 ratio of trimethoprim to sulfamethoxazole.
NA, not applicable.
FIG. 1.
MIC distribution ranges for AR-709 and SXT.
Drug-resistant S. pneumoniae has been an increasing problem worldwide. One factor that seems to have changed the current epidemiology of S. pneumoniae is the introduction of the 7-valent pneumococcal conjugate vaccine (PCV7) into the U.S. vaccination schedule in 2000. A decrease in invasive pneumococcal disease was found in children with herd immunity conferred to the adult population (11). Of concern however is the emergence of nonvaccine serotypes (NVS) which then may become increasingly drug resistant. This is especially evident with an increase in serotype 19A, which is not included in PCV7, is often antibiotic resistant, and is known to cause invasive disease (5, 11, 14). Serotype 19A was the most-common serotype included in our strain collection. The mef(A) genotype traditionally associated with lower level macrolide resistance is the most prevalent macrolide resistance mechanism in the United States, and the erm(B) genotype conferring higher level macrolide resistance is the most-common mechanism worldwide (6). However, there has been an increase in erm(B) plus mef(A) expression, especially in serogroup 19, which can also exhibit multidrug resistance (5, 6).
The increase in drug-resistant NVS is likely multifactorial, including antibiotic and immunologic selective pressure which may involve clonal expansion into niches previously occupied by vaccine serotypes, introduction of novel clones into the population, NVS acquisition of de novo resistance, and/or capsular switching (7, 14). Over time, there could be a development of prevaccine era drug resistance but with new clinically relevant serotypes. Therefore, new antimicrobial agent options will still be important clinically.
Our study has shown that AR-709 was highly active against this collection of invasive MDR pneumococcal strains, irrespective of the pattern of drug resistance. Potent activity of AR-709 was observed despite the fact that >85% of strains were resistant to SXT. Both AR-709 and trimethoprim (TMP) inhibit dihydrofolate reductase (DHFR). However, earlier studies showed that AR-709 exhibited inhibition of DHFR that was 50-fold and 500-fold better than that with TMP when tested against TMP-susceptible and TMP-resistant pneumococcal DHFRs, respectively (13). In addition, Ki measurements showed AR-709 exhibited 5- to 10-fold and 60- to 600-fold better inhibition of susceptible and resistant pneumococcal DHFRs, respectively (13). Thus, AR-709 is a promising new agent for potential therapy of MDR S. pneumoniae strains and further studies are warranted.
Acknowledgments
This study was supported in part by a grant from Arpida AG, Reinach, Switzerland.
We thank M. Leticia McElmeel and Letitia C. Fulcher for excellent technical assistance, CDC's Streptococcal Laboratory for serotyping data, and the ABCs principal investigators and sites for supplying the pneumococcal isolates.
The opinions or assertions contained herein are the private views of the authors and are not to be construed as official or reflecting the views of the Department of Defense, the Department of Health and Human Services, or the U.S. government.
Footnotes
Published ahead of print on 7 January 2008.
REFERENCES
- 1.Centers for Disease Control and Prevention. 2006. Active Bacterial Core surveillance report, Emerging Infections Program Network, Streptococcus pneumoniae, 2005. http://www.cdc.gov/ncidod/dbmd/abcs/survreports/spneu05.pdf.
- 2.Clinical and Laboratory Standards Institute. 2006. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically. Approved standard, M7-A7. Clinical and Laboratory Standards Institute, Wayne, PA.
- 3.Clinical and Laboratory Standards Institute. 2007. Performance standards for antimicrobial susceptibility testing; seventeenth informational supplement. CLSI document M100-S17. Clinical and Laboratory Standards Institute, Wayne, PA.
- 4.Doern, G. V., S. S. Richter, A. Miller, N. Miller, C. Rice, K. Heilmann, and S. Beekman. 2005. Antimicrobial resistance among Streptococcus pneumoniae in the United States: have we begun to turn the corner on resistance to certain antimicrobial classes? Clin. Infect. Dis. 41:139-148. [DOI] [PubMed] [Google Scholar]
- 5.Farrell, D. J., K. P. Klugman, and M. Pichichero. 2007. Increased antimicrobial resistance among nonvaccine serotypes of Streptococcus pneumoniae in the pediatric population after the introduction of 7-valent pneumococcal vaccine in the United States. Pediatr. Infect. Dis. J. 26:123-128. [DOI] [PubMed] [Google Scholar]
- 6.Felmingham, D., R. Canton, and S. G. Jenkins. 2007. Regional trends in β-lactam, macrolide, fluoroquinolone and telithromycin resistance among Streptococcus pneumoniae isolates 2001-2004. J. Infect. 55:111-118. [DOI] [PubMed] [Google Scholar]
- 7.Hanage, W. P., S. S. Huang, M. Lipsitch, C. J. Bishop, D. Godoy, S. I. Pelton, R. Goldstein, H. Huot, and J. A. Finkelstein. 2007. Diversity and antibiotic resistance among nonvaccine serotypes of Streptococcus pneumoniae carriage isolates in the post-heptavalent conjugate vaccine era. J. Infect. Dis. 195:347-352. [DOI] [PubMed] [Google Scholar]
- 8.Jacobs, M. R., C. Good, A. Windau, and S. Bajaksouzian. 2006. AR-709, a novel diaminopyrimidine compound: activity against Streptococcus pneumoniae, abstr. F1-1956. Abstr. 46th Intersci. Conf. Antimicrob. Agents Chemother.
- 9.Kaplan, S. L., and E. O. Mason. 1998. Management of infections due to antibiotic-resistant Streptococcus pneumoniae. Clin. Microbiol. Rev. 11:628-644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Klugman, K. P., and J. R. Lonks. 2005. Hidden epidemic of macrolide-resistant pneumococci. Emerg. Infect. Dis. 11:802-807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kyaw, M. H., R. Lynfield, W. Schaffner, A. S. Craig, J. Hadler, A. Reingold, A. R. Thomas, L. H. Harrison, N. M. Bennett, M. M. Farley, R. R. Facklam, J. H. Jorgensen, John Besser, E. R. Zell, A. Schuchat, and C. G. Whitney. 2006. Effect of introduction of the pneumococcal conjugate vaccine on drug-resistant Streptococcus pneumoniae. N. Engl. J. Med. 354:1455-1463. [DOI] [PubMed] [Google Scholar]
- 12.Lappin, G., S. Warrington, D. Honeybourne, D. Sanghera, S. Dowen, N. Lister, K. Islam, and S. Lociuro. 2007. Concentrations of AR-709 in plasma and key compartments of the lungs after microdosing, abstr. F1-938. Abstr. 47th Intersci. Conf. Antimicrob. Agents Chemother.
- 13.Mukhija, S., M. Bandera, S. Parisi, S. Rigo, S. Lieb, S. Lociuro, D. Gillessen, and K. Islam. 2006. AR-709—an investigational diaminopyrimidine: inhibition, binding and mode of action, abstr. F1-1955. Abstr. 46th Intersci. Conf. Antimicrob. Agents Chemother.
- 14.Pai, R., M. R. Moore, T. Pilishvili, R. E. Gertz, C. G. Whitney, B. Beall, and the Active Bacterial Core Surveillance Team. 2005. Postvaccine genetic structure of Streptococcus pneumoniae serotype 19A from children in the United States. J. Infect. Dis. 192:1988-1995. [DOI] [PubMed] [Google Scholar]
- 15.Pallares, R., F. Gudiol, J. Linares, J. Ariza, G. Rufi, L. Murgui, J. Dorca, and P. F. Viladrich. 1987. Risk factors and response to antibiotic therapy in adults with bacteremic pneumonia caused by penicillin-resistant pneumococci. N. Engl. J. Med. 317:18-22. [DOI] [PubMed] [Google Scholar]
- 16.Rzeszutek, M., A. Wierzbowski, D. J. Hoban, J. Conly, W. Bishai, and G. G. Zhanel. 2004. A review of clinical features associated with macrolide-resistant Streptococcus pneumoniae. Int. J. Antimicrob. Agents 24:95-104. [DOI] [PubMed] [Google Scholar]
- 17.Vanderkooi, O. G., D. E. Low, K. Green, J. E. Powis, and A. McGeer. 2005. Predicting antimicrobial resistance in invasive pneumococcal infections. Clin. Infect. Dis. 40:1288-1297. [DOI] [PubMed] [Google Scholar]
- 18.Yu, V. L., C. C. Chiou, C. Feldman, A. Ortqvist, J. Rello, A. J. Morris, L. M. Baddour, C. M. Luna, D. R. Snydman, M. Ip, W. Chien Ko, M. B. F. Chedid, A. Andremont, and K. P. Klugman. 2003. An international prospective study of pneumococcal bacteremia: correlation with in vitro resistance, antibiotics administered, and clinical outcome. Clin. Infect. Dis. 37:230-237. [DOI] [PubMed] [Google Scholar]