Abstract
The essential role of multispecific immune responses for the control of hepatitis B virus (HBV) infection implies the need of multimodal therapeutic strategies for chronic HBV infection, including antiviral chemotherapy and immunomodulation. This hypothesis was tested in the woodchuck model by a combination of lamivudine pretreatment and subsequent immunizations of woodchucks chronically infected with woodchuck hepatitis virus. The immunizations were performed with DNA vaccines or antigen-antibody immune complexes (IC)/DNA vaccines. Immunizations with IC/DNA vaccines led to an anti-woodchuck hepatitis virus surface antibody response and significant reductions of viral load and antigenemia, suggesting that such a strategy may be effective against chronic HBV infection.
Approximately 400 million people worldwide are chronically infected with hepatitis B virus (HBV). Therapy with interferon or nucleoside/tide analogs is not satisfactory due to the low responder rate and resistance development (16, 18, 20, 33). To date, immunotherapy against chronic hepatitis B has not yet achieved satisfactory results (7, 14, 17, 31, 32, 41, 44). Given the crucial role of cytotoxic T lymphocytes in the control of HBV infections, new therapeutic vaccines with the ability to stimulate vigorous, broad HBV-specific cytotoxic T lymphocyte responses are needed (3, 11, 26, 38, 39).
Several studies of therapeutic vaccinations have been carried out in the woodchuck model (reviewed in references 24, 25, 34, and 35) and demonstrated collectively that B- or T-cell responses to viral antigens could be induced in chronic woodchuck hepatitis virus (WHV) carriers (12, 13, 23, 30). Yet, none of these studies have demonstrated the capability of vaccines to suppress viral replication. Here, we carried out a proof-of-principle experiment using DNA vaccines alone and DNA vaccines combined with immune complexes (IC) in the woodchuck model. IC of HBsAg and anti-HBs have been tested in patients and in transgenic mice (42, 43, 45, 46). IC are more efficiently taken up by antigen-presenting cells than free antigens, leading to an improved presentation to T cells. DNA vaccines are potent inducers of T-cell responses. They could stimulate HBV-specific immune responses in humans and prevent hepadnaviral infections in the animal model (21, 22, 37). In addition, woodchucks were pretreated with lamivudine, a potent antiviral drug against HBV with the ability to enhance T-cell responses in chronically HBV-infected patients (1, 2).
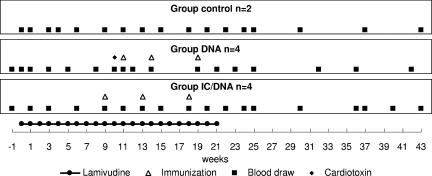
A total of 10 chronically WHV-infected woodchucks (from Northeastern Wildlife, Ithaca, NY) were first treated with 15 mg of lamivudine daily and randomly divided into three groups: the control group (n = 2), a group vaccinated with WHV surface antigen (WHsAg)-IC and DNA (n = 4), and a group vaccinated with DNA (n = 4). The immunization schedule is presented in Fig. 1.
FIG. 1.
Schema of treatment and immunizations. Ten woodchucks with chronic WHV infection were divided into three groups. For 21 weeks, 15 mg of lamivudine was given orally to all woodchucks. The first group (n = 2) served as the control and did not receive any vaccinations. The second group (n = 4) was immunized with the combination of three plasmids expressing WHsAg, WHcAg, and woodchuck gamma interferon. The third group (n = 4) received three immunizations with WHsAg-IC and plasmid pWHsIm.
The DNA vaccines consisted of an equimolar mixture of three plasmids, pWHsIm, pWHcIm, and pWIFN, expressing WHsAg, WHV core antigen (WHcAg), and woodchuck gamma interferon, respectively, as described previously (21, 37). To produce IC for woodchucks, antibodies to WHs (anti-WHs) and WHsAg were titrated by the checkerboard method to determine the stoichiometry of the antigen and antibodies. The appropriate concentration of WHsAg was chosen and incubated with antibodies at 37°C for 30 min and then incubated overnight at 4°C, resulting in a preparation with a final concentration of 80 μg WHsAg/ml in complex with anti-WHs. The vaccine consisted of 20 μg WHsAg-IC and 250 μg plasmid pWHsIm DNA per 0.5-ml dose. The DNA vaccines and IC-DNA vaccines were administered by intramuscular injections (21, 22).
The following parameters were determined by the indicated methods: serum WHV DNA concentrations as genome equivalents (GE) by real-time PCR with the LightCycler DNA Master Sybr green kit (Roche) (22), WHsAg concentrations by the electroimmunodiffusion technique using a polyvalent anti-WHV antiserum (10, 40), anti-WHs concentrations by enzyme-linked immunosorbent assay (23), and lymphoproliferative responses to WHV proteins and peptides by immunofluorescence (29).
Lamivudine therapy alone.
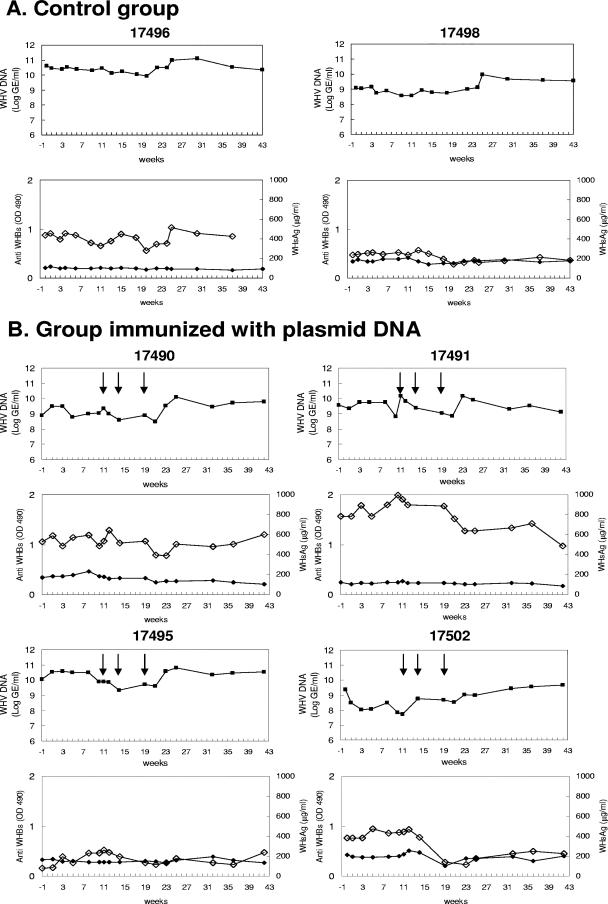
In both control animals, the viral loads decreased slightly during treatment and reached 8.1 × 109 GE/ml for woodchuck WH17496 and 5.6 × 108 GE/ml for woodchuck WH17498 at week 21, corresponding to reductions of 0.7 log and 0.32 log, respectively (Fig. 1; Table 1). Following the discontinuation of lamivudine treatment, the viral loads rebounded and increased to peak values of 9. 7 × 1010 GE/ml (WH17496) and 8.8 × 109 GE/ml (WH17498) at week 25 and then returned to the pretreatment levels for both animals. The serum WHsAg concentrations in WH17496 and WH17498 were 438 and 239 μg/ml, respectively, prior to the lamivudine treatment. The WHsAg concentrations in the control animals decreased to 281 μg/ml and 140 μg/ml at week 21 and rebounded to 516 μg/ml and 170 μg/ml, respectively, at week 25. These results are consistent with the published data (28). No anti-WHs antibodies were detected in either control woodchuck.
TABLE 1.
Viral loads and WHsAg concentrations in the treated woodchucks
| Group | Woodchuck | Baseline viral load (108 GE/ml) | Reduction of WHV DNA concna (log) | Baseline WHsAg concn (μg/ml) | Reduction of WHsAg concna (%) |
|---|---|---|---|---|---|
| Control | 17496 | 400 | 0.7 | 438 | 36 |
| 17498 | 12 | 0.32 | 239 | 25 | |
| DNA | 17490 | 7.87 | 0.42 | 528 | 25 |
| 17491 | 37.4 | 0.72 | 779 | 3 | |
| 17495 | 108 | 0.44 | 80 | +47.5b | |
| 17502 | 22.8 | 0.85 | 383 | 64 | |
| IC and DNA | 17492 | 70 | 1.3 | 158 | 92 |
| 17493 | 25 | 2.9 | 556 | 71 | |
| 17494 | 7.3 | 2.2 | 535 | 67 | |
| 17497 | 490 | 1.44 | 250 | 85 |
The reductions of the viral loads and WHsAg concentrations in the woodchucks were calculated using the baseline values measured before the treatment and the values measured from weeks 18 to 21 after the completion of three vaccinations with DNA or IC and DNA.
The WHsAg concentration was increased in WH17495; therefore the change is indicated with +.
DNA vaccination.
Four woodchucks, WH17490, WH17491, WH17495, and WH17502, had initial WHV loads at 7.87 × 108, 3.74 × 109, 1.08 × 1010, and 2.28 × 109 GE/ml, respectively (Fig. 2B). Pretreatment with cardiotoxin was performed at week 10 and at the DNA immunizations at weeks 11, 14, and 19. The viral loads decreased slightly in all four woodchucks without relation to vaccinations and rebounded at week 21 with the end of lamivudine treatment (Fig. 2B). In woodchuck WH17491, the WHV DNA concentration was reduced from 1.5 × 1010 to 7.2 × 108 GE/ml from weeks 11 to 21. In woodchuck WH17502, the viral load declined to 5.4 × 107 GE/ml after the start of lamivudine treatment but showed no response to vaccinations. The serum WHsAg concentrations in WH17490, WH17491, and WH17502 showed decreases of 25%, 3%, and 64%, respectively, at week 21. For unknown reasons, WHsAg increased in WH17495 to 118 μg/ml, corresponding to 147.5% of the baseline. No anti-WHs antibodies were detected in these woodchucks.
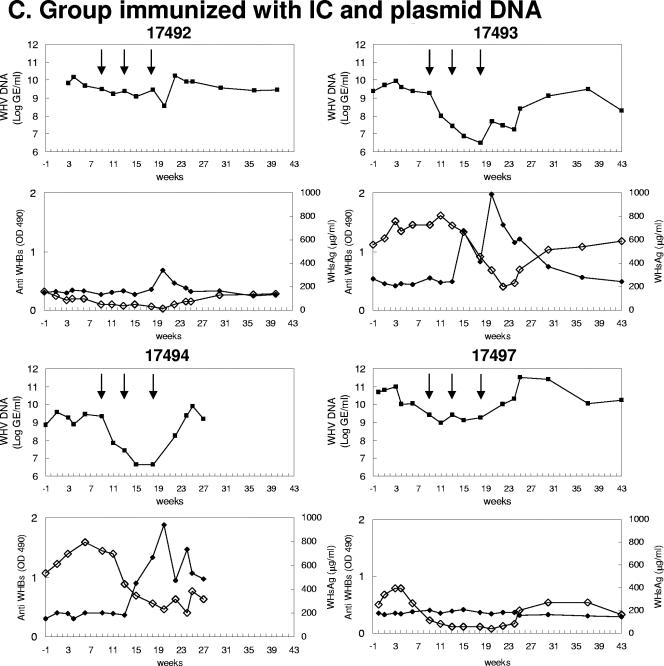
FIG. 2.
Therapeutic vaccinations of chronically WHV-infected woodchucks. The serum WHV DNA concentrations (▪), anti-WHs antibody response (Anti WHBs) (♦), and serum WHsAg concentrations (⋄) in the treated woodchucks were measured. The duration of lamivudine treatment is indicated with a line. (A) Control group; (B) group immunized with plasmid DNA; (C) group immunized with IC and plasmid DNA. The serum WHV DNA concentrations are presented as GE/ml in the log scale. The anti-WHs were measured by enzyme-linked immunosorbent assay and are presented as values of optical density at 490 nm. WHsAg is given as μg/ml. The animal WH17494 was in a bad condition due to the biopsies and therefore was not included in further monitoring after week 27.
Vaccination with WHs-IC and plasmid DNA.
WH17492, WH17493, WH17494, and WH17497 had initial viral loads of 7.0 × 109, 2.5 × 109, 7.3 × 108, and 4.9 × 1010 GE/ml, respectively, and received the combined IC/DNA vaccine at weeks 9, 13, and 18. In woodchucks WH17492 and WH17497, the viral loads were reduced to 3.6 × 108 and 1.33 × 109 GE/ml, respectively, after the second boost (Fig. 2C). The viral loads in WH17493 and WH17494 dropped strongly to 3 × 106 and 4 × 106 GE/ml, respectively, at week 18. The maximal reduction levels of the viral loads in woodchucks after vaccinations with IC/DNA ranged between 1.3 and 2.9 log (Table 1). A rebound of the viral loads occurred after the discontinuation of lamivudine treatment at week 21.
Uniformly, the vaccinations with IC and DNA led to significant decreases of the serum WHsAg concentrations of 67% to 92% compared with the baseline in all four woodchucks (Fig. 2C; Table 1). After the start of lamivudine treatment, the serum WHsAg concentrations fell from 159 and 250 μg/ml in WH17492 and WH17497 to 12.5 and 37.7 μg/ml, respectively, after three vaccinations. WH17493 and WH17494 had serum WHsAg concentrations over 500 μg/ml at the beginning. The WHsAg concentrations reached about 200 μg/ml in these two animals after three vaccinations.
Strikingly, woodchucks WH17492, WH17493, and WH17494 developed a detectable anti-WHs antibody response after boosts, though the antibody response was not sustained (Fig. 2C). One woodchuck, WH17497, did not show any detectable anti-WHs antibody response despite the reduction of serum WHsAg concentrations. A reciprocal correlation between the amount of anti-WHs and WHV DNA or WHsAg was clearly observed in animals WH17493 and WH17494. With the rise of anti-WHs antibodies at week 13, the WHV DNA and the serum WHsAg concentrations were reduced to low levels in the woodchucks. Both markers for WHV replication increased again as the anti-WHs antibody titer decreased. Surprisingly, the anti-WHs antibodies were detected at higher levels in woodchucks WH17493 and WH17494, which had higher initial serum WHsAg concentrations. WH17497 had a relatively low serum WHsAg concentration at 250 μg/ml and the lowest level at 37.7 μg/ml. However, no anti-WHs antibodies were detected in this animal.
Lymphoproliferative responses in vaccinated woodchucks.
WHV-specific lymphoproliferative responses were not detectable in the majority of chronically WHV-infected woodchucks in this study, even in woodchucks that were immunized with IC/DNA vaccines and exhibited virological responses (data not shown). Significant lymphoproliferative responses to WHV antigens were measured only in woodchuck WH17502 after three vaccinations with associations of significant reductions of WHV markers (Table 1). These results were largely consistent with previous results (23). The time points of sampling are unlikely to be the reason for the negative results, since assays were performed every 2 weeks. These results may indicate the inability of vaccines to induce specific cellular responses. However, it is also possible that specific T cells were sequestered to the liver and therefore not detectable in peripheral blood.
Liver histology in immunized woodchucks.
The liver biopsy specimens taken after the completion of the treatment at week 25 revealed mild or moderate hepatitis in all animals, according to the Ishak score (15) (Table 2), similar to untreated carrier woodchucks (5, 6). The levels of severity of portal inflammation varied in different animals, from a minimal portal infiltrate (grade 0) to a mild periportal infiltrate to a maximal periportal infiltrate with mild piecemeal necrosis (grade 3). The lobular inflammation also varied in different animals from grade 1 to grade 2 (mild inflammation but no necrosis or with focal necrosis), resulting in a range of the total score from minimal 1 to maximal 5. The two control animals showed a score of 2 and 3. The scores were between 3 and 5 or 1 and 5 for animals vaccinated with DNA and IC/DNA, respectively. In summary, immunizations with either of the protocols did not result in severe liver disease in woodchucks.
TABLE 2.
Histological examination of liver tissues of treated woodchucks
| Group | Woodchuck | Scorea
|
||
|---|---|---|---|---|
| Portal inflammation | Lobular inflammation | Sum | ||
| Control | 17496 | 2 | 1 | 3 |
| 17498 | 0 | 2 | 2 | |
| DNA | 17490 | 2 | 1 | 3 |
| 17491 | 3 | 2 | 5 | |
| 17495 | 2 | 1 | 3 | |
| 17502 | 1 | 2 | 3 | |
| IC and DNA | 17492 | 3 | 2 | 5 |
| 17493 | 0 | 1 | 1 | |
| 17494 | 2 | 1 | 3 | |
| 17497 | 3 | 1 | 4 | |
The histological evaluation was done according to the Ishak scoring method (15). The grade 0 for portal inflammation indicates a minimal portal infiltrate.
Taken together, the findings demonstrated that a combination of lamivudine treatment and immunizations with a vaccine containing IC and plasmid DNA was able to reduce the viral load up to 2.9 log and the serum WHsAg load up to 92% and induced specific anti-WHs antibodies in chronic carrier woodchucks. The lamivudine contributed to the suppression of viral loads despite its low effectiveness in woodchucks (28), as a viral rebound uniformly occurred immediately after the discontinuation of lamivudine treatment.
DNA immunization is considered to be a powerful method of inducing cellular immune responses to pathogens. However, the DNA vaccinations in our study did not clearly show any additional therapeutic effect compared to the lamivudine treatment alone. Similarly, DNA vaccination failed to induce viral clearance in chronically infected ducks (8, 19, 36). A DNA vaccine expressing HBsAg has been tested in patients for immunotherapy and appeared to enhance T-cell responses (27). Further studies on DNA vaccines are needed to improve their effectiveness for immunotherapies.
The present protocol with the lamivudine treatment and IC/DNA immunizations led to only a transient response in woodchucks. More potent antivirals, like entecavir (4, 9), and multiple vaccinations of more than three injections could further enhance specific immune responses and yield better results.
Acknowledgments
We are grateful for the editorial assistance of Delia Cosgrove.
This work was partly supported by the Bai-Yu-Lan program of the Shanghai Municipal Government (Y.W. and M.L). D.G. and W.G. were supported by DFG grant SFB535 A2. M.R., M.L., and Y.X. are supported by DFG grant GRK1045/1.
Footnotes
Published ahead of print on 26 December 2007.
REFERENCES
- 1.Boni, C., A. Bertoletti, A. Penna, A. Cavalli, M. Pilli, S. Urbani, and P. Scognamiglio. 1998. Lamivudine treatment can restore T cell hyporesponsiveness in chronic hepatitis B: new perspectives for immune therapy. J. Clin. Investig. 102968-975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Boni, C., A. Penna, G. S. Ogg, A. Bertoletti, M. Pilli, C. Cavallo, and A. Cavalli. 2001. Lamivudine treatment can overcome cytotoxic T-cell hyporesponsiveness in chronic hepatitis B: new perspectives for immune therapy. Hepatology 33963-971. [DOI] [PubMed] [Google Scholar]
- 3.Chisari, F. V., and C. Ferrari. 1995. Hepatitis B virus immunopathogenesis. Annu. Rev. Immunol. 1329-60. [DOI] [PubMed] [Google Scholar]
- 4.Colonno, R. J., E. V. Genovesi, I. Medina, L. Lamb, S. K. Durham, M. L. Huang, and L. Corey. 2001. Long-term entecavir treatment results in sustained antiviral efficacy and prolonged life span in the woodchuck model of chronic hepatitis infection. J. Infect. Dis. 1841236-1245. [DOI] [PubMed] [Google Scholar]
- 5.Dahmen, U., O. Dirsch, J. Li, M. Fiedler, M. Lu, K. Rispeter, M. Picucci, C. E. Broelsch, and M. Roggendorf. 2004. Adoptive transfer of immunity: a new strategy to interfere with severe hepatitis virus reinfection after woodchuck liver transplantation. Transplantation 77965-972. [DOI] [PubMed] [Google Scholar]
- 6.Dahmen, U., J. Li, O. Dirsch, M. Fiedler, M. Lu, M. Roggendorf, and C. E. Broelsch. 2002. Liver transplantation in woodchuck: a newly established animal model of hepatitis B reinfection. Transplantation 74374-380. [Google Scholar]
- 7.Dikici, B., A. G. Kalayci, F. Ozgenc, M. Bosnak, M. Davutoglu, A. Ece, and T. Ozkan. 2003. Therapeutic vaccination in the immunotolerant phase of children with chronic hepatitis B infection. Pediatr. Infect. Dis. J. 22345-349. [DOI] [PubMed] [Google Scholar]
- 8.Foster, W. K., D. S. Miller, P. L. Marion, R. J. Colonno, I. Kotlarski, and A. R. Jilbert. 2003. Entecavir therapy combined with DNA vaccination for persistent duck hepatitis B virus infection. Antimicrob. Agents Chemother. 472624-2635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Genovesi, E. V., L. Lamb, I. Medina, D. Taylor, M. Seifer, S. Innaimo, and R. J. Colonno. 1998. Efficacy of the carbocyclic 2′-deoxyguanosine nucleoside BMS-200475 in the woodchuck model of hepatitis B virus infection. Antimicrob. Agents Chemother. 423209-3217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gerlich, W. H., U. Wend, and D. Glebe. 2004. Quantitative assay of hepatitis B surface antigen in serum or plasma using Laurell electrophoresis, p. 57-63. In J. Lau and R. Hamatake (ed.), Hepatitis B virus protocols. Humana Press, Totowa, NJ. [DOI] [PubMed]
- 11.Guidotti, L. G., R. Rochford, J. Chung, M. Shapiro, R. Purcell, and F. V. Chisari. 1999. Viral clearance without destruction of infected cells during acute HBV infection. Science 284825-829. [DOI] [PubMed] [Google Scholar]
- 12.Hervas-Stubbs, S., J. J. Lasarte, P. Sarobe, J. Prieto, J. M. Cullen, M. Roggendorf, and F. Borras-Cuesta. 2001. T-helper cell response to woodchuck hepatitis virus antigens after therapeutic vaccination of chronically-infected animals treated with lamivudine. J. Hepatol. 35105-111. [DOI] [PubMed] [Google Scholar]
- 13.Hervas-Stubbs, S., J. J. Lasarte, P. Sarobe, I. Vivas, L. Condreay, J. M. Cullen, and J. Prieto. 1997. Therapeutic vaccination of woodchucks against chronic woodchuck hepatitis virus infection. J. Hepatol. 27726-737. [DOI] [PubMed] [Google Scholar]
- 14.Horiike, N., S. M. Fazle Akbar, K. Michitaka, K. Joukou, K. Yamamoto, N. Kojima, and Y. Hiasa. 2003. In vivo immunization by vaccine therapy following virus suppression by lamivudine: a novel approach for treating patients with chronic hepatitis B. J. Clin. Virol. 32156-161. [DOI] [PubMed] [Google Scholar]
- 15.Ishak, K., A. Baptista, L. Bianchi, F. Callea, J. De Groote, F. Gudat, and H. Denk. 1995. Histological grading and staging of chronic hepatitis. J. Hepatol. 22696-699. [DOI] [PubMed] [Google Scholar]
- 16.Janssen, H. L., M. van Zonneveld, H. Senturk, S. Zeuzem, U. S. Akarca, Y. Cakaloglu, C. Simon, et al. 2005. Pegylated interferon alfa-2b alone or in combination with lamivudine for HBeAg-positive chronic hepatitis B: a randomised trial. Lancet 365123-129. [DOI] [PubMed] [Google Scholar]
- 17.Jung, M. C., N. Gruner, R. Zachoval, W. Schraut, T. Gerlach, H. Diepolder, and C. A. Schirren. 2002. Immunological monitoring during therapeutic vaccination as a prerequisite for the design of new effective therapies: induction of a vaccine-specific CD4+ T-cell proliferation response in chronic hepatitis B carriers. Vaccine 203598-3612. [DOI] [PubMed] [Google Scholar]
- 18.Lau, G. K., T. Piratvisuth, K. X. Luo, P. Marcellin, S. Thongsawat, G. Cooksley, E. Gane, et al. 2005. Peginterferon alfa-2a, lamivudine, and the combination for HBeAg-positive chronic hepatitis B. N. Engl. J. Med. 3522682-2695. [DOI] [PubMed] [Google Scholar]
- 19.Le Guerhier, F., A. Thermet, S. Guerret, M. Chevallier, C. Jamard, C. S. Gibbs, and C. Trepo. 2003. Antiviral effect of adefovir in combination with a DNA vaccine in the duck hepatitis B virus infection model. J. Hepatol. 3328-334. [DOI] [PubMed] [Google Scholar]
- 20.Locarnini, S., and W. S. Mason. 2006. Cellular and virological mechanisms of HBV drug resistance. J. Hepatol. 44422-431. [DOI] [PubMed] [Google Scholar]
- 21.Lu, M., G. Hilken, J. Kruppenbacher, T. Kemper, R. Schirmbeck, J. Reimann, and M. Roggendorf. 1999. Immunization of woodchucks with plasmids expressing woodchuck hepatitis virus (WHV) core antigen and surface antigen suppresses WHV infection. J. Virol. 73281-289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lu, M., M. Isogawa, Y. Xu, and G. Hilken. 2005. Immunization with the gene expressing woodchuck hepatitis virus nucleocapsid protein fused to cytotoxic-T-lymphocyte-associated antigen 4 leads to enhanced specific immune responses in mice and woodchucks. J. Virol. 796368-6376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lu, M., R. Klaes, S. Menne, W. Gerlich, B. Stahl, H. P. Dienes, U. Drebber, and M. Roggendorf. 2003. Induction of antibodies to the PreS region of surface antigens of woodchuck hepatitis virus (WHV) in chronic carrier woodchucks by immunizations with WHV surface antigens. J. Hepatol. 39405-413. [DOI] [PubMed] [Google Scholar]
- 24.Lu, M., S. Menne, D. Yang, Y. Xu, and M. Roggendorf. 2007. Immunomodulation as an option for the treatment of chronic hepatitis B virus infection: preclinical studies in the woodchuck model. Expert Opin. Investig. Drugs 16787-801. [DOI] [PubMed] [Google Scholar]
- 25.Lu, M., and M. Roggendorf. 2001. Evaluation of new approaches to prophylactic and therapeutic vaccinations against hepatitis B viruses in the woodchuck model. Intervirology 44124-131. [DOI] [PubMed] [Google Scholar]
- 26.Maini, M. K., C. Boni, C. K. Lee, J. R. Larrubia, S. Reignat, G. S. Ogg, A. S. King, and J. Herberg. 2000. The role of virus-specific CD8(+) cells in liver damage and viral control during persistent hepatitis B virus infection. J. Exp. Med. 1911269-1280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Mancini-Bourgine, M., H. Fontaine, D. Scott-Algara, S. Pol, C. Brechot, and M. L. Michel. 2004. Induction or expansion of T-cell responses by a hepatitis B DNA vaccine administered to chronic HBV carriers. Hepatology 40874-882. [DOI] [PubMed] [Google Scholar]
- 28.Mason, W. S., J. Cullen, G. Moraleda, J. Saputelli, C. E. Aldrich, D. S. Miller, and B. Tennant. 1998. Lamivudine therapy of WHV-infected woodchucks. Virology 24518-32. [DOI] [PubMed] [Google Scholar]
- 29.Menne, S., J. Maschke, T. K. Tolle, M. Lu, and M. Roggendorf. 1997. Characterization of T-cell response to woodchuck hepatitis virus core protein and protection of woodchucks from infection by immunization with peptides containing a T-cell epitope. J. Virol. 7165-74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Menne, S., C. A. Roneker, B. E. Korba, J. L. Gerin, B. C. Tennant, and P. J. Cote. 2002. Immunization with surface antigen vaccine alone and after treatment with 1-(2-fluoro-5-methyl-β-l-arabinofuranosyl)-uracil (l-FMAU) breaks humoral and cell-mediated immune tolerance in chronic woodchuck hepatitis virus infection. J. Virol. 765305-5314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Pol, S., F. Driss, M. L. Michel, B. Nalpas, P. Berthelot, and C. Brechot. 1994. Specific vaccine therapy in chronic hepatitis B infection. Lancet 344342. [DOI] [PubMed] [Google Scholar]
- 32.Pol, S., B. Nalpas, F. Driss, M. L. Michel, P. Tiollais, J. Denis, and C. Brechot. 2001. Efficacy and limitations of a specific immunotherapy in chronic hepatitis B. J. Hepatol. 34917-921. [DOI] [PubMed] [Google Scholar]
- 33.Raney, A. K., R. K. Hamatake, and Z. Hong. 2003. Agents in clinical development for the treatment of chronic hepatitis B. Expert. Opin. Investig. Drugs 121281-1295. [DOI] [PubMed] [Google Scholar]
- 34.Roggendorf, M., and M. Lu. 2005. Woodchuck hepatitis virus, p. 210-224. In T. H. Thomas, A. Zuckermann, and S. Lemon (ed.), Viral hepatitis, 3rd ed. Blackwell Publishing Ltd. Oxford, United Kingdom.
- 35.Roggendorf, M., I. Schulte, Y. Xu, and M. Lu. 2007. Therapeutic vaccination in chronic hepatitis B: pre-clinical studies in the woodchuck model. J. Viral Hepat. 14(Suppl. 1)51-57. [DOI] [PubMed] [Google Scholar]
- 36.Rollier, C., C. Sunyach, L. Barraud, N. Madani, C. Jamard, C. Trepo, and L. Cova. 1999. Protective and therapeutic effect of DNA-based immunization against hepadnavirus large envelope protein. Gastroenterology 116658-665. [DOI] [PubMed] [Google Scholar]
- 37.Siegel, F., M. Lu, and M. Roggendorf. 2001. Coadministration of gamma interferon with DNA vaccine expressing woodchuck hepatitis virus (WHV) core antigen enhances the specific immune response and protects against WHV infection. J. Virol. 755036-5042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Tang, T. J., J. Kwekkeboom, S. Mancham, R. S. Binda, R. A. de Man, S. W. Schalm, and J. G. Kusters. 2005. Intrahepatic CD8+ T-lymphocyte response is important for therapy-induced viral clearance in chronic hepatitis B infection. J. Hepatol. 4345-52. [DOI] [PubMed] [Google Scholar]
- 39.Thimme, R., S. Wieland, C. Steiger, J. Ghrayeb, K. A. Reimann, R. H. Purcell, and F. V. Chisari. 2003. CD8+ T cells mediate viral clearance and disease pathogenesis during acute hepatitis B virus infection. J. Virol. 7768-76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Tolle, T. K., D. Glebe, M. Linder, D. Linder, S. Schmitt, R. Geyer, and W. H. Gerlich. 1998. Structure and glycosylation patterns of surface proteins from woodchuck hepatitis virus. J. Virol. 729978-9985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Vandepapelière, P., G. K. Lau, G. Leroux-Roels, Y. Horsmans, E. Gane, T. Tawandee, M. I. Merican, K. M. Win, C. Trepo, G. Cooksley, M. Wettendorff, C. Ferrari, and The Therapeutic HBV Vaccine Group of Investigators. 2007. Therapeutic vaccination of chronic hepatitis B patients with virus suppression by antiviral therapy: a randomized, controlled study of co-administration of HBsAg/AS02 candidate vaccine and lamivudine. Vaccine [Epub ahead of print.] doi: 10.1016/j.vaccine.2007.09.072. [DOI] [PubMed]
- 42.Wen, Y. M., X. H. Wu, D. C. Hu, Q. P. Zhang, and S. Q. Guo. 1995. Hepatitis B vaccine and anti-HBs complex as approach for vaccine therapy. Lancet 3451575-1576. [DOI] [PubMed] [Google Scholar]
- 43.Xu, D. Z., K. L. Huang, K. Zhao, L. F. Xu, N. Shi, Z. H. Yuan, and Y. M. Wen. 2005. Vaccination with recombinant HBsAg-HBIG complex in healthy adults. Vaccine 232658-2664. [DOI] [PubMed] [Google Scholar]
- 44.Yalcin, K., M. Acar, and H. Degertekin. 2003. Specific hepatitis B vaccine therapy in inactive HBsAg carriers: a randomized controlled trial. Infection 31221-225. [DOI] [PubMed] [Google Scholar]
- 45.Yao, X., B. Zheng, J. Zhou, D. Z. Xu, K. Zhao, S. H. Sun, Z. H. Yuan, and Y. M. Wen. 2007. Therapeutic effect of hepatitis B surface antigen-antibody complex is associated with cytolytic and non-cytolytic immune responses in hepatitis B patients. Vaccine 251771-1779. [DOI] [PubMed] [Google Scholar]
- 46.Zheng, B. J., M. H. Ng, L. F. He, X. Yao, K. W. Chan, K. Y. Yuen, and Y. M. Wen. 2001. Therapeutic efficacy of hepatitis B surface antigen-antibodies-recombinant DNA composite in HBsAg transgenic mice. Vaccine 194219-4225. [DOI] [PubMed] [Google Scholar]