Abstract
The matrix metalloproteinases (MMPs) are a family of extracellular matrix-cleaving enzymes involved in ovarian remodeling. In many non-tropical species, including Siberian hamsters, ovarian remodeling is necessary for the functional changes associated with seasonal reproduction. We evaluated MMPs and their endogenous inhibitors (TIMPs), during photoperiod-induced ovarian recrudescence in Siberian hamsters. Hamsters were transferred from long-day (LD;16:8) to short-day (SD;8:16) photoperiods for 14wks, and then returned to LD for 0,1,2,4, or 8wks for collection of ovaries and plasma. Post-transfer (PT) LD exposure increased body and ovarian mass. Numbers of corpora lutea and antral, but not preantral follicles increased in PT groups. Plasma estradiol concentrations were lower in PT wks0−4, and returned to LD levels at PTwk8. No change was observed in relative MMP/TIMP mRNA levels at PTwk0 (SDwk14) as compared to LD. Photostimulation increased MMP-2 mRNA at PTwk8 as compared to PT wks 0−1. MMP-14 mRNA expression peaked at PTwks1−2 as compared to LD levels, while MMP-13 expression was low during this time. TIMP-1 mRNA peaked at PT wk8 as compared to PTwks0−4. No changes were noted in MMP-9 and TIMP-2 mRNA expression. In general, MMP/TIMP protein immunodetection followed the same patterns with most staining occurring in granulosa cells of follicles and corpora lutea. Our data suggest that mRNA and protein for several members of the MMP/TIMP families are expressed in Siberian hamster ovaries during recrudescence. Because of the variation observed in expression patterns, MMPs and TIMPs may be differentially involved with photo-stimulated return to ovarian function.
Keywords: Ovary, MMP, Seasonal Reproduction, Photoperiod, Hamster, Gonad Recrudescence
Introduction
Ovarian activity does not remain constant throughout the year in most non-tropical species, where reproductive function is limited seasonally to maximize breeding success. For photoperiodic Siberian hamsters (Phodopus sungorus), this entails full gonadal function during the summer, or long day (LD) months, and ovarian atrophy or regression during the winter, or short day (SD) months (Goldman et al., 2000).
In the laboratory, exposure to 12 weeks of short (<12.5 hrs of light per day) photoperiod induces ovarian atrophy, or regression, resulting in reduced or absent ovulation and decreased plasma estradiol concentrations (Moffatt-Blue et al., 2006; Schlatt et al., 1993). In contrast, maintenance of LD breeding rodents in chronic (18−30 weeks) SD, or transferring animals from 12−14 weeks of SD to stimulatory LD conditions (>12.5 hrs of light per day) induces gonadal recrudescence (Schlatt et al., 1993; Ross et al., 2005). At the level of the gonad, recrudescence is prompted by increases in serum FSH levels as the hypothalamic-pituitary axis restores function (Schlatt et al., 1993; Muteka et al., 2006; Schlatt et al., 1995). In male rodents, such as Djungarian hamsters (Phodopus sungorus campbelli), white-footed mice (Peromyscus leucopus), and Tete veld rats (Aethomys ineptus), recrudescence triggers the return of spermatogenesis and testicular steroidogenesis concomitant with increases in seminiferous tubule size and testis mass, and decreases in apoptotic cell death (Schlatt et al., 1995; Young, Zirkin, and Nelson, 2001; Muteka et al., 2006). While testicular restoration during recrudescence has been well examined (reviewed in Young and Nelson, 2001), few studies have explored the basis for the photoperiod-mediated return of ovarian function.
Successful ovarian function requires substantial cyclical tissue remodeling, specifically during follicular development, ovulation, and during the formation and regression of the corpus luteum. A family of extra-cellular matrix (ECM)-cleaving enzymes known as the matrix metalloproteinases (MMPs) is implicated in mediating aspects of ovarian tissue remodeling as a result of their differential and species-specific expression during the ovarian cycle (reviewed in Curry and Osteen, 2001; Smith et al., 1999).
MMP expression and activity in ovarian tissue occurs in multiple species and studies have investigated ovarian expression of many of the characterized MMP family members. Secretion of MMP-9 and TIMPs (Tissue Inhibitors of Matrix Metalloproteinases) -1 and -2 has been correlated to overall follicular health during in vitro bovine follicle development (McCaffery et al., 2000), and both MMP-2 and MMP-9 are distinctly active in growing follicles in rodents and goats (Curry et al., 2001; Jo et al., 2004; García et al., 1997). MMP-19 and TIMP-1 are induced by hCG administration in mice and humans (Hägglund et al., 1999; Lind et al., 2006) and MMPs-1, 2, 7, 9 and TIMP-1, -2 are upregulated by hCG administration in the periovulatory ovary of rhesus monkeys (Chaffin and Stouffer, 1999). MMP-13 (collagenase-3) is critical in the ovary because of its involvement in both peri-ovulatory (GnRH injection and natural cycle) follicles and in the corpus luteum (Bakke, et al., 2004), and the membrane bound collagenase MMP-14 increases in both peri-ovulatory and luteal tissue with the gonadotropin surge (Bakke et al., 2002). Indeed, an increase in collagenase activity prior to ovulation is reported in most mammals (reviewed in Goldman and Shalev, 2004). In mice and rhesus monkeys, MMPs are up-regulated during the transformation of follicular granulosa cells into functional luteal tissue, and again during luteal regression (Young and Stouffer, 2003; Liu et al., 1999). In addition to physical remodeling, the cleavage action of ovarian MMPs can release growth factors in vitro encouraging follicular development, and in turn, ovulation (Levi et al., 1996; Fowlkes et al., 1994; Gearing et al., 1995; Logan and Hill, 1992; Massova et al., 1998; Stouffer et al., 2007).
During the process of seasonal ovarian recrudescence, extensive remodeling of the ECM and release of necessary growth factors must occur for proper return of function. Given that ovarian recrudescence involves the return of organ function, MMPs are likely to play a key role in restoring ovarian performance. Because ovarian regression in Siberian hamsters is characterized by a reduction in follicle numbers, a decline in gonadal mass, and an absence of corpora lutea (Moffatt-Blue et al., 2006), recrudescence of non-functional ovarian tissue would likely show reciprocal changes during the return of ovarian function. We hypothesized that ovarian recrudescence would occur rapidly with LD stimulation of reproductively regressed females, and that return of function would be characterized by an increased number of tertiary follicles and corpora lutea as well as a subsequent increase in serum estradiol. Finally, we hypothesized that mRNA and protein expression of MMPs and their tissue inhibitors, TIMPs, would be altered during recrudescence of the ovary to attain full gonadal function by eight weeks following transfer to stimulatory LD photoperiod. As a first step to address this last hypothesis, we examined two gelatinases (MMPs-2 and -9) two collagenases (MMP-13 and membrane bound MMP-14), and two endogenous inhibitors of MMPs (TIMPs-1 and -2).
Methods
Animals
Adult Siberian Hamsters (Phodopus sungorus) were purchased from the colony of Dr. Katherine Wynne-Edwards, Queens University (Kingston, Ontario, Canada) and were housed at 20 ± 2 °C in individual polypropylene cages equipped with bedding. Access to food (a mixture of Lab Rodent Diet 5001 and Mazuri Hamster & Gerbil Diet both from Purina, Brentwood, MO) and tap water was provided ad libitum. All procedures were performed at California State University, Long Beach and complied with the CSULB and National Research Council guidelines for use of laboratory animals. Animals (n=70 females and 8 males) were acclimated for a two-week duration in long day (LD) conditions (16h light: 8h dark). Fifty-two female hamsters were then randomly assigned to a short day room (8L:16D), while the remainder (n=18) stayed in LD conditions as controls. In order to ensure complete reproductive regression with SD exposure, animals remained housed in these photoperiods for 14 weeks (Moffatt-Blue et al., 2006; Ross et al., 2005). Short day females were randomly assigned to one of five groups: post-transfer (PT) week 0, 1, 2, 4, or 8 (n=10−11 per group). At 14 weeks of SD, tissue was collected from the PT week 0 group and all other animals were transferred into LD photoperiod for the remainder of the study. LD female control hamsters were randomly assigned into 3 groups and tissue was collected at the same time as PT week 0, 2, and 8 as a baseline comparison (n=6 per group). Four male hamsters were placed among the female hamsters in each room to promote ovarian cyclicity, and estrous cycles were synchronized by placing soiled male bedding in to the female cages four days prior to tissue collection (Dodge et al., 2002), as we have done before (Moffatt-Blue et al., 2006). Body mass was measured, and the stage of the estrous cycle was determined by vaginal cytology on collection dates. A cotton swab dampened with saline solution was inserted into the vagina and vaginal cells were smeared over a microscope slide for cytology (Moffatt-Blue et al., 2006). For standardization purposes, only animals in Diestrus II were euthanized via cervical dislocation following a cocktail of ketamine (20mg/kg) and xylazine (200mg/kg) for tissue collection (Moffatt-Blue et al., 2006). Animals that were not in Diestrus II were replaced in their cage and reswabbed the following day(s) until in the correct stage. No animal was swabbed more than three consecutive days, with the majority of the animals euthanized the first day of the collection week. Following paired ovarian mass assessment, one ovary from each hamster was fixed in 10% neutral buffered formalin for 7 days; the contralateral organs were individually flash frozen for mRNA extraction. Blood samples were collected from the retro-orbital sinus, and plasma was stored at −80°C until radioimmunoassay to determine estradiol concentrations was completed.
Follicle Counts
Formalin fixed tissues were processed through a series of PBS washes, dehydrated through a graded series of ethanol, and embedded in paraffin wax. Six-mm thick ovarian sections were collected serially every 60 mm and mounted onto Superfrost-plus microscope slides (Fisher Scientific, Pittsburgh, PA). Tissues were stained with hematoxylin and eosin in order to differentiate ovarian structures. To avoid double counting preantral follicles, only those containing oocytes were counted. The follicles counted included preantral follicles, early antral follicles, corpora lutea, atretic follicles, advanced atretic follicles, and terminal atretic follicles (Moffatt-Blue et al., 2006). Because terminal atretic follicles have been reported in few species other than Siberian hamsters, we sought to investigate if disappearance of these unique structures correlated with MMP activity. The average number of ovarian structures was determined from six sections per ovary.
Estradiol Radioimmunoassay
Collected plasma was separated by centrifugation (5000 rpm for 5 min) and stored at −80°C until the estradiol radioimmunoassay using Ultra-Sensitive Estradiol RIA125I double antibody kits (Diagnostic Systems Laboratories, Inc., Webster, TX). Samples were assayed in duplicate and their radioactivity was measured using a Perkin-Elmer Cobra II gamma counter (Packard Instruments Co., Boston, MA). A standard curve was generated using a four-parameter logistic curve function (Sigma Plot software, SPSS Inc., Chicago, IL) and the final hormone concentrations were calculated using the Sigma Plot standard curve analysis function. Assay standards and controls were within the normal limits (Moffatt-Blue et al., 2006; Schmidt and Kelley, 2001), with a 5 pg/mL lower limit of detection and low (0.64−2.40%) cross-reactions to other steroids.
RT-PCR/ mRNA Analysis
Following tissue collection, total RNA was isolated from the frozen ovaries using the RNeasy Micro Kit (QIAGEN, Valencia, CA) according to QIAGEN standard protocol. cDNA was generated by performing reverse transcription on all samples containing sufficient RNA (see figure legends for n/group; ImProm Reverse Transcription System, Promega, Madison, WI). Hamster MMP primers were designed on Vector NTI software by evaluating the properties (e.g., GC content, annealing temperature, self-dimerization potential) of sections of conserved MMP sequences in the mouse and rat genomes, and by then empirically determining the optimum annealing temperature, cycle number, and primer concentration for each chosen pair. Semi-quantitative RT-PCR was completed using GoTaq Green Promega PCR reagents (Promega, Madison, WI). Reactions were conducted at 94°C for 30 sec, annealing at gene-specific optimal temperature (Table 1), and extension at 72°C for 1 min for a gene-specific number of cycles (Table 1).
Table 1.
MMP and TIMP Primers
|
Primer Sequence |
Tm |
Cycle Number |
bp |
|---|---|---|---|
| MMP2F: CACACCTGACCTGGACCCTGAAAC | 55°C | 28 cycles | 506bp |
|
MMP2R: TGCCCTGGAAGCGGAACG |
|
|
|
| MMP9F: GACGGGTATCCCTTCGACGG | 69°C | 35cycles | 383bp |
|
MMP9R: GTGGTGCAGGCRGAGTAGGA |
|
|
|
| MMP13F: GACTTCTACCCATTTGATGG | 56°C | 34 cycles | 362bp |
|
MMP13R: GTAATGGCATCAAGGGATARG |
|
|
|
| MMP14F: ATGGTTTACAAGTGACAGGCAAGG | 51°C | 33 cycles | 446bp |
|
MMP14R: GGGCTCRGCAGAATCAAA |
|
|
|
| TIMP1F: TCRASGCTGTGGGAAATG | 55°C | 34 cycles | 355bp |
|
TIMP1R: AGGCARGCAAAGTGAYSG |
|
|
|
| TIMP2F: AAAGCAGTGAGCGAGAAG | 52°C | 28 cycles | 363bp |
| TIMP2R: GGAGGAGATGTAGCAWGG |
For each female, 10μl of the PCR reaction was electrophoresed on a 2% agarose gel containing 1 μl of ethidium bromide to allow visualization. Gels were visualized using BioRad Gel Doc SR documentation system (BioRad, Hercules, CA), and global adjusted volume of bands analyzed using Quantity One software (The Discovery Series, BioRad). For each hamster, the global adjusted volume of each gene was normalized by division of the global adjusted volume for the loading standard ß-actin band to obtain the relative mRNA expression for each gene.
Immunohistochemistry
Sectioned ovary tissue (n=5 arbitrarily determined females per group) was deparaffinized in xylene, rehydrated through a graded series of ethanol, washed in phosphate buffer solution (PBS), and heated under pressure in a citra solution for 10 min to facilitate antigen retrieval. Sections were placed in a 3% hydrogen peroxide/methanol solution and blocked with normal goat or horse serum/Tween-20 (0.1%) for 40 min. Primary antibody (Chemicon, Temecula, CA) was applied to sections in an empirically determined dilution (1:200 for MMPs-2, -9, -13, and TIMP-1; 1:400 for MMP-14 and TIMP-1) and incubated at 4°C overnight. All primary antibodies recognized both pro and active forms of MMPs. Biotinylated goat anti-rabbit or anti-mouse immunoglobulin G (Vector Laboratories, Burlingame, CA) was then applied to sections according to company protocol, and incubated for 45min at room temperature, prior to incubation in an avidin-biotin peroxidase solution (Vectastain Elite ABC kit; Vector Laboratories). Vector NovaRed Substrate kit (Vector Laboratories) was used to visualize the antigen and sections were counterstained with hematoxylin. Sections processed in the absence of primary antibody were used as negative controls. Mouse ovary sections served as a positive control for each antibody used and showed staining patterns identical to the hamster tissue (data not shown). Characteristic staining intensity, extent of staining, and location of stain were noted for three sections per animal for each MMP/TIMP. A semiquantitative analysis of staining intensity and extent was conducted using three to six sections per animal. Sections were given a numerical value ranging from 0 to 3. A score of 0 indicated no staining; a score of 1 meant some faint staining was apparent in at least some of the cells, a score of 2 indicated medium-intense staining that extended throughout the section across multiple follicles, and a score of 3 specified intense staining throughout follicles and some stroma. Results for individual animals (n=5/group) were averaged per group and analyzed.
Statistical Analysis
All data were analyzed using one-way ANOVAs with the Prism 4 statistical software 240 package (GraphPad Software, Inc., San Diego, CA) and are represented by mean ± SEM. If results were significant (p<0.05) with a 95% confidence interval, a Neuman-Keuls post-hoc test was used to compare experimental groups. To reduce variance in the early antral, atretic, advanced atretic, and corpora lutea analyses, a square-root transform was used.
Results
No changes were observed in any parameters analyzed for LD hamsters. Because LD females serve as controls for our experiment, we pooled LD hamster data from week 0, 2, and 8 for all parameters analyzed (after Moffatt-Blue et al., 2006).
Reproductive Organ and Body Mass
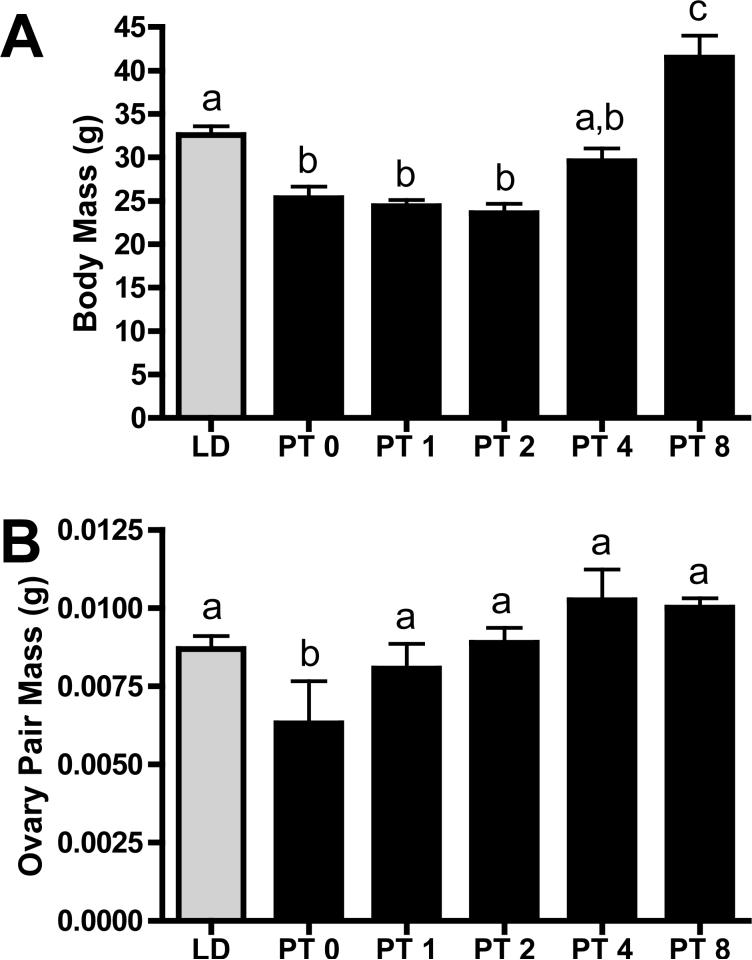
Exposure to 14 weeks of SD photoperiods reduced body mass, whereas transfer to LD photoperiod significantly increased the mass of these hamsters (p<0.05; Figure 1A). After remaining constant from PT week 0 to 2, body mass increased 1.7 fold from week 2 to week 8 (p<0.05; Figure 1A). Ovarian mass also decreased with exposure to SD (p< 0.05; Figure 1B). Transfer to stimulatory photoperiod rapidly increased ovarian mass 1.3 fold from PT weeks 0 to 1, and remained at LD levels thereafter (p<0.05; Figure 1B).
Figure 1. Body mass (A) and paired ovary mass (B).
Mean ± S.E.M. A) body mass (g) and B) ovarian mass (g) in Siberian hamsters exposed to long day (LD; n=18)), or post-transfer (PT) from short to long day photoperiod for 0 (n=10), 1 (n=10), 2 (n=10), 4 (n=11), and 8 (n=11) weeks. Groups with different letters are significantly different (P<0.05).
Follicular Analysis
Histological examination revealed high numbers of corpora lutea (CL) in LD controls, indicative of ovulation (Figure 2A). In addition, ovaries from all LD females contained preantral and antral follicles (Figure 2A). In contrast, CL and antral follicles were absent in PT week 0 (SD week 14), although these ovaries did contain preantral (mostly primary) follicles (Figure 2B). Ovaries from the PT week 0 hamsters contained high levels of the terminal atretic follicles that are characteristic of reproductive regression, and had fewer follicles undergoing atresia typical of the normal ovarian cycle. Follicle development was stimulated in PT weeks 1 and 2, and an abundance of antral and preantral follicles were noted; however, low numbers of CL were observed these groups (Figure 2C, D). At PT weeks 4 and 8, CL were notably more abundant, in addition to the pool of growing and atretic follicles (Figure 2E, F).
Figure 2. Representative cross sections of Siberian hamster ovaries stained with hematoxylin and eosin.
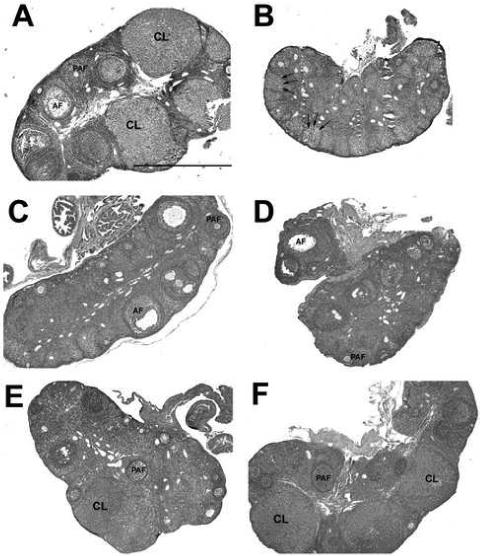
A) Long day (LD) control (n=18). B) Post-transfer (PT) week 0 (same as SD week 14). C) PT week 1. D) PT week 2. E) PT week 4. F) PT week 8. CL, corpus luteum; AF, antral follicles; PAF, preantral follicles; → indicate terminal atretic follicles. All photos taken at 4x magnification; scale bar pertains to all sections and represents 1.0mm. N per group the same as listed for Figure 1.
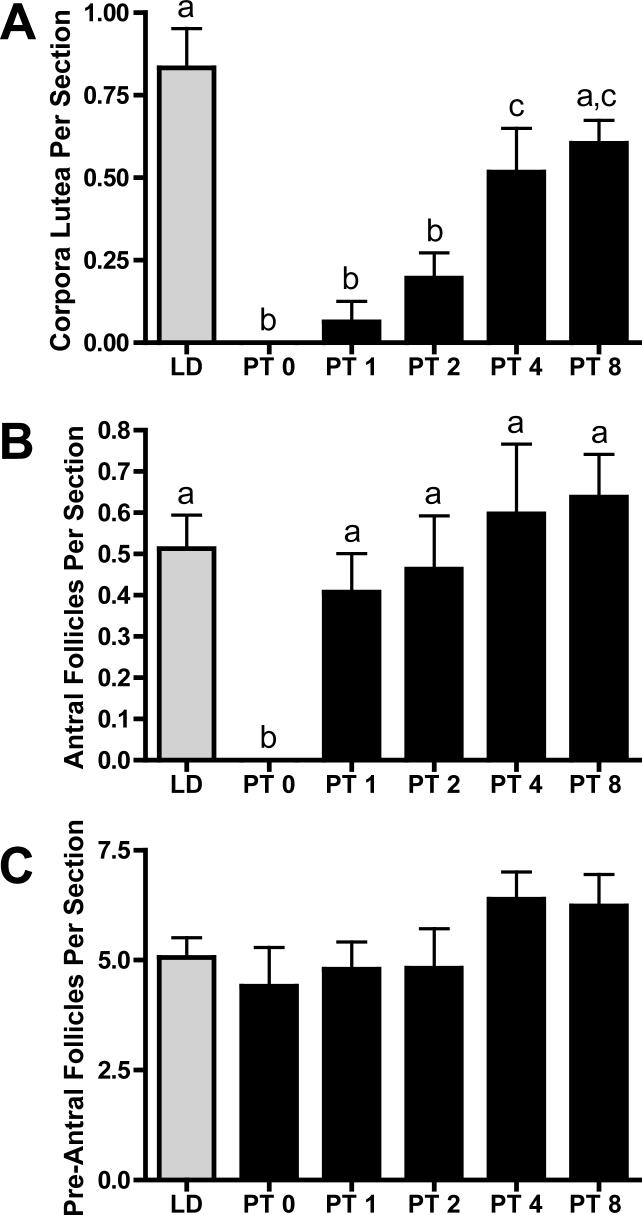
Follicle counts were quantified for the number of CL, antral, and preantral follicles. In addition, in order to potentially correlate MMP activity with atresia, the number of atretic follicles was counted and separated into standard atretic follicles, advanced atretic follicles, and the unique terminal atretic follicles that occur in hamsters with SD exposure (Moffatt-Blue et al., 2006). The number of CL present in the ovarian sections changed with exposure to photoperiod. LD females maintained ovaries with high numbers of CL, with nearly one unique CL per section quantified. However, exposure to SD for 14 weeks decreased the number of CL to zero in the PT week 0 group (p<0.05; Figure 3A). Following transfer to LD, the number of CL increased; there was a rapid 3-fold increase in the number of CL between PT weeks 1 and 2 (p<0.05; Figure 3A), leading to an overall 10-fold increase from PT week 0 to 8 (p<0.001; Figure 3A). By PT week 8, the number of CL had returned to levels not significantly different from LD values (p>0.05; Figure 3A).
Figure 3. Number of corpora lutea (A), antral follicles (B), and preantral follicles (C) per section.
Mean ± S.E.M. A) corpora lutea, B) antral follicles, and C) pre-antral follicles per ovarian section in Siberian hamsters exposed to long day (LD), or post-transfer (PT) from short to long day photoperiod for 0, 1, 2, 4, and 8 weeks. Groups with different letters are significantly different (P<0.05). N per group the same as listed for Figure 1.
Photoperiod also induced changes in antral and atretic follicle counts. LD females maintained a high level of antral follicles, whereas 14 weeks of SD (PT week 0) resulted in ovaries with no antral follicles (p<0.05; Figure 3B). Interestingly, alterations in the number of antral follicles was rapid, increasing from zero at PT week 0 to 0.4 at PT week 1, and maintaining these levels typical of LD animals at PT weeks 2, 4, and 8 (p>0.05; Figure 3B). In contrast, there were no changes in preantral follicles across any group (p>0.05; Figure 3C). The number of atretic follicles, typical of the ovarian cycle was moderate in functional LD ovaries; however, exposure to SD for 14 weeks (PT week 0) decreased atretic follicle numbers. Atretic follicle counts did not recover to LD levels until PT week 2 and continued to increase (5-fold) at PT weeks 4 and 8 as compared to PT weeks 0 and 1 (p<0.05; Table 2). In contrast, there were no changes in the number of advanced atretic follicles; these follicles were common in all groups (p>0.05; Table 2). While LD ovaries did not contain terminal atretic follicles, females exposed to 14 weeks of SD (PT week 0) had significantly higher numbers of terminal atretic follicles, structures unique to regressed hamster ovaries (Moffatt-Blue et al., 2006; Schlatt et al., 1993), as compared to all other groups (p<0.05; Table 2). Transfer to LD decreased the number of terminal atretic follicles over 10-fold from PT week 0 to 2; however, these structures did not decline to LD levels until PT week 2 and were no longer apparent after PT week 4 (Table 2).
Table 2.
Atretic Follicle Counts in Recrudescing Hamster Ovaries
| Photoperiod group | Atretic | Advanced Atretic | Terminal Atretic |
|---|---|---|---|
| LD | 0.88±0.1a | 0.43±0.1 | 0.00±0.0a |
| PT 0 | 0.22±0.2b | 0.32±0.1 | 19.06±4.1b |
| PT 1 | 0.46±0.2b | 0.60±0.2 | 5.44±1.8c |
| PT 2 | 0.62±0.2a,b | 0.67±0.1 | 1.81±2.1a |
| PT 4 | 1.00±0.2a | 0.66±0.1 | 0.00±0.0a |
| PT 8 | 1.02±0.2a | 0.59±0.1 | 0.00±0.0a |
a-cValues with different superscript letters differ significantly (P<0.05).
Estradiol Radioimmunoassay
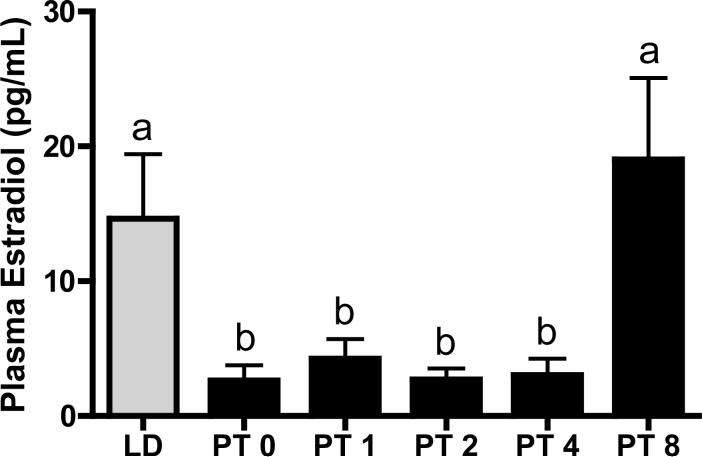
Plasma estradiol concentrations were measured by radioimmunoassay, and were at typical levels in the LD females (Figure 4). Exposure to 14 weeks of SD in the PT week 0 group induced a decrease in plasma estradiol concentrations that was not recovered until PT week 8. During early recrudescence (PT week 1 through 4), estradiol concentrations remained constant, then increased 6.2-fold from PT week 4 to 8, returning to long day control values (p<0.05; Figure 4).
Figure 4. Plasma estradiol concentrations (pg/mL) in recrudescing Siberian hamsters.
Mean ± S.E.M. plasma estradiol concentrations (pg/mL) in Siberian hamsters exposed to long day (LD), or post-transfer (PT) from short to long day photoperiod for 0, 1, 2, 4, and 8 weeks. Groups with different letters are significantly different (P<0.05). N per group the same as listed for Figure 1.
RT-PCR/ mRNA Analysis
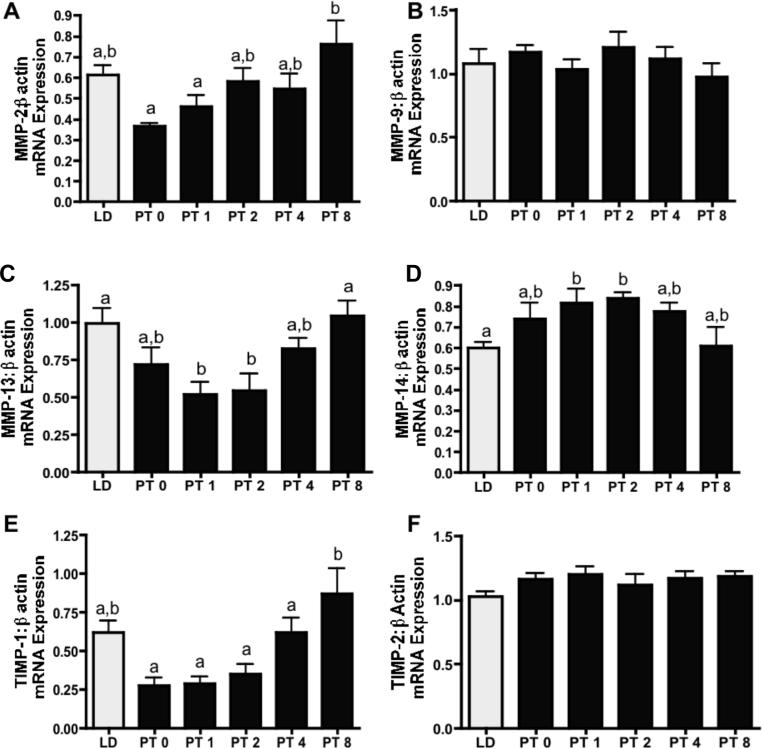
To determine the contribution of MMPs and TIMPs in photostimulated return to ovarian function, total ovarian mRNA was analyzed using MMP-2, -9, -13, and -14 and TIMP-1 and -2 specific primers (Figure 5). Interestingly, among all genes examined, exposure to 14 weeks of SD (PT week 0) had no effect on gene expression as compared to LD controls (p>0.05 in all cases; Figure 6). However, the subsequent photostimulation by transfer to LD did affect mRNA expression in a number of MMPs/TIMPs. Despite a declining trend, relative ovarian MMP-2 (gelatinase-A) levels did not change with transfer to SD; however, MMP-2 increased progressively from PT weeks 1 through 8 (p<0.05; Figure 6A). Relative levels of MMP-9 (gelatinase-B) remained constant throughout the study period (p>0.05; Figure 6B. Relative MMP-13 mRNA levels declined after PT week1 and PT week 2 as compared to LD and PT week 8 values (p<0.05; Figure 6C). Relative MMP-14 (mt-MMP-1) expression increased at PT week 1 and 2 as compared to LD (p<0.05), levels for this membrane type collagenase then returned to LD levels at PT weeks 4 and 8 (p>0.05; Figure 6D). TIMP-1 levels appeared to decline at PT week 0; however, this change was not significant (p>0.05; Figure 6E). Relative TIMP-1 mRNA expression levels were significantly increased 3.5-fold in PT week 8 as compared to PT week 0. There was no change in TIMP-2 mRNA expression among the PT or the LD control groups (p>0.05; Figure 6F).
Figure 5. mRNA expression for MMPs and TIMPs.
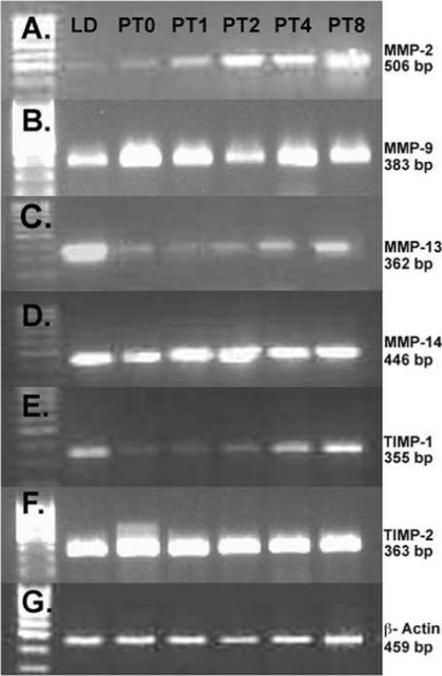
Semi-quantitative RT-PCR expression of MMP and TIMP mRNA in Siberian hamsters exposed to long day (LD), or post-transfer (PT) from short to long day photoperiod for 0, 1, 2, 4, and 8 weeks. A) MMP-2, B) MMP-9, C) MMP-13, D) MMP-14 (mt-MMP-1), E) TIMP-1, F) TIMP-2, and G) β-actin used as a control gene for all RT-PCR reactions.
Figure 6. Relative mRNA expression levels for MMPs and TIMPs.
Mean ± S.E.M. relative levels mRNA expression in Siberian hamsters exposed to long day (LD), or post-transfer (PT) from short to long day photoperiod for 0, 1, 2, 4, and 8 weeks. A) MMP-2:β-actin mRNA expression. B) MMP-9:β-actin mRNA expression. C) MMP-13:β-actin mRNA expression. D) MMP-14:β-actin mRNA expression. E) TIMP-1:β-actin mRNA expression. F) TIMP-2:β-actin mRNA expression. Groups with different letters are significantly different (P<0.05). LD (n=14), PT 0 (n=5), PT 1 (n=9), PT 2 (n=7), PT 4 (n=11), PT 8 (n=9).
Immunohistochemistry
Primary anti-mouse antibodies for MMPs-2, 9, 13, and 14 and TIMPs-1, and -2 were used on paraffin embedded tissue sections. No staining was evident in control sections processed without primary antibodies (negative control insets, Figure 7). Staining patterns in mouse ovaries (positive controls, data not shown) matched what was observed in hamsters for all antibodies.
Figure 7. MMP and TIMP protein immunodetection during recrudescence in Siberian hamsters.
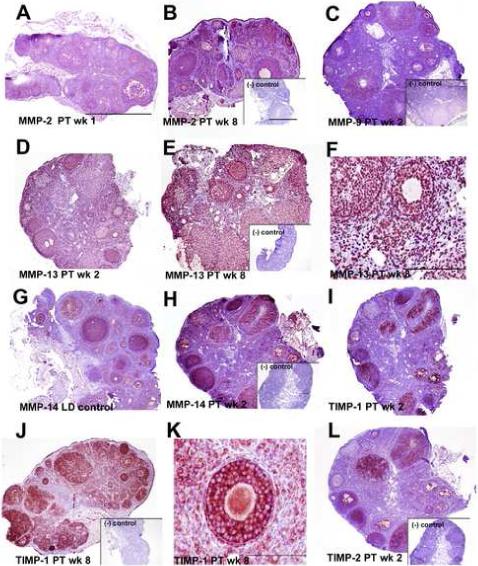
Immunohistochemical staining (red/pink stain on purple background) for MMPs and TIMPs. Scale bars (A, inset B) depict 1.0mm with photographs taken at 4× magnification unless stated otherwise (micrographs in F and K). A) Tissue section from post-transfer (PT) week 2 demonstrating diffuse, cytoplasmic immunodetection of MMP-2 typical of PT weeks 0, 1, and 2. B) MMP-2 immunolabeling was stronger during PT weeks 4 and 8, and strongly abundant in granulosa cells (PT week 8 shown). C) Section depicting MMP-9 protein levels during ovarian recrudescence; cytoplasmic MMP-9 staining was consistent among all tested groups (PT week 2 shown). D) Typical lower levels of punctate, peri-nuclear MMP-13 staining observed in PT week 2. E) Ovary section illustrating high levels of punctate, peri-nuclear MMP-13 staining observed in PT week 8. F) Detail of puntate, perinuclear staining observed with the MMP-13 antibody; photo taken at 20×, scale bar indicates 200μm. G) Cytoplasmic MMP-14 immunolabeling in the LD control group. H) MMP-14 staining in the recrudescing ovary; MMP-14 protein levels appeared elevated in post-transfer weeks 0, 1, 2, and 4 (PT week 2 shown). I) Tissue section showing cytoplasmic TIMP-1 immunolabeling typical of the recrudescing ovary during weeks 0, 1, 2, and 4, post-transfer (PT week 2 shown). J) TIMP-1 labeling was more extensive at PT week 8. K) Cytoplasmic diffuse staining typical of MMPs-2, -9, -14 and TIMPs-1 and -2, shown here in a 40× magnification photo of TIMP-1 at PT week 8. Scale bar indicates 100μm. L) Section illustrating cytoplasmic TIMP-2 immunolabeling in the ovary during recrudescence, TIMP-1 intensity did not appear to change in response to stimulatory photoperiod. Insets on B, C, E, H, J, and L depict negative control sections processed without primary antibody.
Gelatinases
MMP-2 and -9 immunostaining was observed in LD and PT weeks 0, 1, 2, 4, and 8; representative sections are shown (Figure 7 A-C). In LD, PT week 4, and PT week 8 ovaries (PT week 8 shown in Figure 7B), MMP-2 immunolabeling was abundant in granulosa cells in follicles and CL, and was present, although less plentiful, in theca cells. Less robust MMP-2 staining was also noted in stroma in LD and PT weeks 4 and 8, and oocytes also showed positive reactivity. In contrast, the extent and intensity of MMP-2 staining was reduced in PT weeks 0, 1, and 2, and diffuse cytoplasmic detection was more typical in these weeks (Figure 7A, PT week 2 ovaries shown). When quantified using an immunostaining index, extent and intensity of staining was significantly lower in PT week 1 as compared to PT weeks 4 and 8 (p<0.052; Figure 8A). A consistent pattern of diffuse cytoplasmic MMP-9 immunodetection was noted in granulosa cells throughout the preantral and antral follicles, as well as in the CL (Figure 7C, representative PT week 2 ovary shown). Only low levels or complete absence of MMP-9 staining were noted in theca cells and stroma, although oocytes showed some positive immunoreaction. Upon quantification, no differences were observed across any group for MMP-9 immunostaining (p=0.08; Figure 8B).
Figure 8. Extent and Intensity of Immunostaining for MMPs and TIMPs.
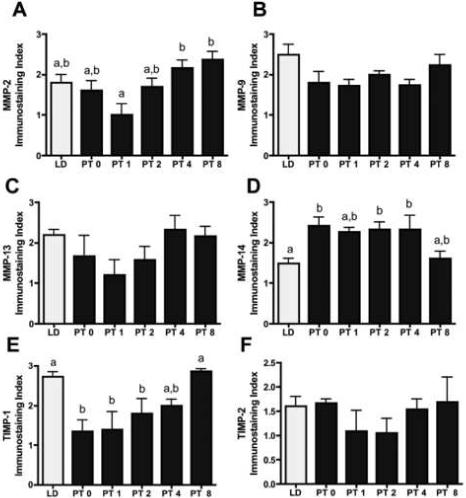
Mean ± S.E.M. immunostaining index levels (scores of 0−3) indicating extent and intensity of red/pink stained cells in Siberian hamsters exposed to long day (LD), or post-transfer (PT) from short to long day photoperiod for 0, 1, 2, 4, and 8 weeks. A) MMP-2. B) MMP-9. C) MMP-13. D) MMP-14. E) TIMP-1. F) TIMP-2. N=5/group. Groups with different letters are significantly different (P<0.05).
Collagenases
MMP-13 immunostaining was detected throughout all groups, although it appeared to increase in PT week 8 (Figures 7D-F). Strong, punctate, perinuclear expression was detected for MMP-13 throughout follicles, CL, and stroma, but not in oocytes (detail of perinuclear staining in Figure 7F). Qualitative analysis showed that staining intensity was slightly less intense in PT weeks 1, 2, and 4, however when quantified no significant differences were noted due to variability across sections (p=0.1; Figure 8C). MMP-14 (mt-MMP-1) immunostaining was cytoplasmic and present in granulosa cells in follicles (preantral and antral), oocytes, and CL in LD and PT weeks 0 to 8 (Figure 7G, H). Staining was confined to the cytoplasm and, while present to some extent in stroma tissue, was notably reduced in thecal cells. MMP-14 immunostaining appeared reduced in LD and PT week 8 as compared to PT weeks 0, 1, and 2 (Figure 7G, LD ovary shown, 7H, PT week 2 ovary shown). When quantified, MMP-14 immunostaining was reduced in LD animals as compared to those in PT weeks 0, 2, and 4 (p<0.05; Figure 8D).
TIMPs
TIMP-1 showed an intense cytoplasmic staining predominantly in granulosa cells, although oocytes and select cells in the stroma were also immunoreactive in all sections (Figure 7I-K with detail of typical cytoplasmic staining in 7K). Qualitatively, TIMP-1 staining was less extensive in PT weeks 0, 1, 2, and 4 (Figure 7I, PT week 2 ovary shown), but increased in PT week 8 where diffuse staining was also noted in thecal cells (Figure 7J). Following immunostaining quantification, TIMP-1 reactivity was reduced at PT weeks 0, 1 and 2 as compared to LD and PT week 8 individuals (p<0.05; Figure 8E). TIMP-2 staining was present selectively in granulosa cells in both follicles and CL; however, TIMP-2 staining was absent from the majority of stroma and theca cells (Figure 7L). No changes among groups were noted for extent and intensity of TIMP-2 immunoreactivty (p>0.05; Figure 8F).
Discussion
The current study shows for the first time that photostimulation following inhibitory photoperiod exposure can alter matrix metalloproteinase expression in the ovary. MMPs and their tissue inhibitors, TIMPs, are expressed in the ovaries of Siberian hamsters throughout both LD and SD photoperiods. Although 14 weeks of SD photo-inhibition did not significantly alter MMP expression, MMPs and TIMPs showed differential changes during recrudescence following transfer from SD to LD photoperiods. In addition, this is the first study to systematically characterize ovarian activity through a photostimulated recrudescence paradigm.
Exposure to SD photoperiods reduced reproductive function after 14 weeks. This is congruent with similar studies examining photo-inhibited loss of ovarian function, where SD exposure decreased CL numbers, estradiol concentrations, ovarian cycling, fertility, and mating behavior (Moffatt-Blue et al., 2006; Beery et al., 2007). Interestingly, subsequent transfer to LD induced a rapid return of ovarian function; ovarian mass and antral follicle numbers returned to LD levels after one-week post LD transfer in the present study. This rapid (∼ 7 days) response to stimulating photoperiod suggests that mechanisms are in place to immediately exploit conditions conducive to breeding. Increases in antral follicles were followed by significantly increased numbers of CL noted at PT week 4 as compared to SD females and PT weeks 1 and 2 (Figure 2). At PT week 2 the number of atretic follicles (common in cycling ovaries) returns to levels no different than LD, again suggesting a restoration of normal cyclic function. By PT week 4, the number of advanced atretic follicles, structures unique to the regressed ovary in Siberian hamsters, return to the LD value of none present (Table 2). These prompt responses may be attributed to the constant supply of preantral (predominantly primary) follicles maintained in SD ovaries observed in this study. It may be that regressed females are ready to respond rapidly to photoperiod stimulation due to increased sensitivity of the hypothalamic pituitary gonadal (HPG) axis, and an upregulation of ovarian gonadotropin receptors is likely. Indeed, SD exposure increases testicular sensitivity to FSH stimulation in male Djungarian hamsters (Chandrashekar et al., 1994), and serum but not pituitary levels of FSH change in female Djungarian hamsters exposed to SD, although increases in both pituitary and serum FSH occur with spontaneous recrudescence (Schlatt et al., 1993). Alternatively, the rapid response may be related to housing conditions. The hamsters in the present study were housed in SD conditions with secondary cues (food availability, temperature, presence of males in the room) considered non-inhibitory to reproductive function. As a result of the laboratory conditions, females in the current study are likely to respond far more rapidly to increased day lengths as compared to the gradual gonadal recrudescence observed in a more natural environment, or during spontaneous recrudescence (e.g., Young, Zirkin, and Nelson, 2001).
Despite this rapid initial response, full ovarian function indicated by estradiol concentrations and ovulation events (number of CL present in the ovary), does not return to LD levels until PT week 8. The ovaries are ovulating as early as PT week 1, but at a reduced level as compared to LD and PT week 8, and complete restoration of ovarian cyclic activity appears to require several weeks of transformation. Indeed, the number of CL noted per ovary during spontaneous recrudescence (30 weeks in SD) in Djungarian hamsters never recovered to LD values (Schlatt et al., 1993). Our data suggest that this transition is not mediated by estradiol; however, because the estradiol concentrations increase with increased numbers of developing follicles, the spike in estradiol at PT week 8 corresponds with the timeframe of full return of ovulatory function (Figures 2 - 4).
Our results show that gelatinases (MMP-2 and 9), collagenases (MMP-13 and 14) and TIMP-1 and 2 mRNA and protein are expressed in the hamster ovaries. However, no clear pattern of their expression emerged in spite of a progressive increase in MMP-2, MMP-13 and TIMP-1 mRNA during recrudescence (Figures 6 - 7). This inconsistent expression suggests that these genes may have multiple roles in the ovary during normal LD estrous cycling, maintenance of regressed ovarian tissue, and photostimulated re-growth. Interestingly, no apparent differences were observed in MMP-9 and TIMP-2 mRNA or protein expression between PT wks 0 to 8 or compared to LD controls (Figure 6 - 7). These results may be partially explained at the protein level by the use of primary antibodies in this study that react with both the pro and active forms of these proteins. MMPs are synthesized as proenzymes and are converted to active forms as needed. Therefore, it is difficult to fully describe changes in their expression or their relative balance between the pro and active forms in the current data. The relative mRNA expression for both MMP-9 and TIMP-2 was actually high as compared to other genes in this study, which may suggest a continual or constitutive role in hamster ovaries, such as is observed in TIMP-2 expression in periovulatory mouse ovaries (Hägglund et al., 1999).
Other MMPs had divergent expression patterns, with PT week 8 often significantly different from time points earlier in the recrudescing timeline. MMP-2 and TIMP-1 mRNA expression and characteristic protein immunodetection both increased after 8 weeks in post-transfer photostimulation as compared to earlier post-transfer weeks. MMP-13 mRNA expression also showed an increase at PT week 8 as compared to PT weeks 1 and 2, although the typical immunostaining showed only a slight increase at PT week 8 as compared to earlier weeks. In these three genes, the change occurs only after long-term (8 weeks) exposure to LD. This change at PT week 8 may be similar to the hyper-responsiveness noted in other recrudescing females. Syrian hamsters undergoing spontaneous recrudescence produce larger litters than age-matched females chronically-housed in long days (Beery et al., 2007) and Siberian hamsters raised in SD and transferred to LD maintain higher levels of fertility than hamsters raised in LD (Place et al., 2004). The increase at PT week 8 extended also to body mass, and may be attributable, in part, to ovarian steroids, as levels of estradiol remain repressed in this experiment until PT week 8.
While our data suggest a role for some MMPs in the tissue remodeling of photo-stimulated recrudescence, the very role for MMPs in cyclic ovarian function is debated. Although inhibition of ovarian MMPs can reduce or inhibit ovulation, this appears to be dependent on species and methodology. Synthetic inhibitors of collagenase significantly reduced ovulation in PMSG or eCG-primed perfused rat ovaries (Brännström et al., 1988; Butler et al., 1991), and ovulation was blocked with administration of an anti-MMP-2 antibody in PGF (2alpha) and GnRH primed sheep (Gottsch et al., 2002). Despite these correlations, other studies do not clearly identify a role for MMPs during ovulation or corpus luteum function. Use of a broad-spectrum inhibitor of MMPs, GM6001, reduced, but did not prevent ovulation, and did not alter corpus luteum development in wild type and plasminogen activator deficient mice (Liu et al., 2006; Wahlberg et al., 2007). Similarly, no significant changes in MMP-2 and MMP-9 were noted in human or equine ovaries prior to ovulation (Lind et al., 2006; Riley et al., 2004), and the absence of MMP-9 and MMP-13 in fertile mice suggests that these MMPs are not essential for ovulation (Hägglund et al., 1999; Johnson et al., 1999). Continued functional examination of MMPs and other enzymes that regulate tissue remodeling will elucidate which genes/gene families are critical and necessary for changes occurring: 1) in the normal estrous cycle and 2) during ovarian regression and recrudescence.
MMPs are regulated at a number of levels, and transcriptional/translational control, endogenous inhibitors, and hormones can mediate their activity. At the protein level, the endogenous TIMPs can closely regulate the activity of MMPs, and while the balance between protease and inhibitor can influence (cellular) growth or death within the ovary, high levels of TIMP expression do not always preclude high levels of MMPs (e.g., Curry et al., 2001; Li et al., 2002). In the present study, MMP mRNA and protein detection did not appear to be limited by the relatively high amounts of TIMP-2 inhibitor. TIMP-1 mRNA was at its lowest levels at PT week 0 and immediately following LD transfer, and typical TIMP-1 protein immunostaining appeared to be consistent with this pattern. However, this MMP inhibitor was detected in all groups to some extent. Because both TIMP-1 and -2 were expressed at some level in all groups, and because the antibodies used in the present study detected both pro and active forms of MMPs, the extent of regulation of MMPs by TIMPs in the recrudescing ovary cannot be fully determined. In addition, there are likely to be other regulators of ovarian MMPs during recrudescence such as cytokines, interleukins and other growth factors, and the extracellular matrix metalloproteinase inducer (EMMPRIN). Examining the roles of these potential regulators on MMP activity during recrudescence, in addition to assessing other members of the MMP and TIMP families, are logical next steps for continued elucidation of the role of MMP action during the photoperiod-stimulated return of ovarian function.
In the current study, high levels of MMP mRNA and protein detection were noted at PT week 8, the only time point in the study where plasma estradiol concentrations matched LD levels. Estradiol concentrations may be correlated with mRNA expression of MMP-2, MMP-13, and in particular TIMP-1, as these genes had increased expression at the PT week 8 time point. While high levels of estradiol may influence expression of these genes, it has been shown that TIMP-1 stimulates ovarian steroid production in vitro (Nothnick et al., 1997), and it may be that the increased concentrations of TIMP-1 observed only at LD and PT week 8 have a facilitative effect on estradiol synthesis or secretion. Although we lacked sufficient plasma to assay progesterone concentrations in the current study, corpus luteum function may not have been fully active until PT week 8 as mice lacking TIMP-1 have low progesterone levels during the estrous cycle (Nothnick, 2000), as well as reduced luteal development (Nothnick, 2003). Hormones of the HPG axis may interact with MMP-2 expression as well; LH stimulation promotes MMP-2 production and activity in cultured bovine thecal cells (Smith et al., 2005), and abundant proMMP-2 correlates positively with estradiol and negatively with progesterone concentrations in bovine follicular fluid (Imai et al., 2003).
In summary, our data suggest that several members of the MMP (-2, -9, -13, and -14) family are expressed at both the mRNA and protein levels during the major tissue remodeling that occurs in the recrudescing ovary of Siberian hamsters. Inhibitory SD exposure induced reproductive regression, including loss of antral follicle development, ovulation, and estradiol production. Because an apparent pool of preantral (predominantly primary) follicles remained constant through 14 weeks of SD exposure, ovaries of Siberian hamsters were able to respond rapidly to LD transfer. Females showed signs of ovulation after one-week post LD transfer, and full ovarian recovery took place over an 8-week post-transfer period. While neither MMPs or TIMP-1 or -2 appear to be affected by chronic transfer to SD, it may be that these proteases are affected only at earlier time points during SD-induced regression. Instead, our findings show that MMPs and TIMPs may be involved with tissue transformation that occurs as a result of reproductively stimulating SD to LD transfer. Future studies examining MMPs during regression, the estrous cycle, and perhaps earlier points following reproductive recrudescence should be conducted to further understand the role that these proteases play during restoration of the photo-inhibited ovary.
Acknowledgements
We thank the CSULB Reproductive Biology Laboratory, particularly Melissa Bagnell, Jen Buehler, Hsin-Jan Peng, and Chantelle Moffatt-Blue for aid in tissue collection and data processing. This project was supported by NIH SCORE grant no. 2506GM06119-05 (KAY), and the Research Initiative For Scientific Enhancement Fellows (RISE) program grant no. GM071638 (JJS).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Bakke LJ, Dow MP, Cassar CA, Peters MW, Pursley JR, Smith GW. Effect of the preovulatory gonadotropin surge on matrix metalloproteinase (MMP)-14, MMP-2, and tissue inhibitor of metalloproteinases-2 expression within bovine periovulatory follicular and luteal tissue. Biol Reprod. 2002;66:1627–34. doi: 10.1095/biolreprod66.6.1627. [DOI] [PubMed] [Google Scholar]
- Bakke LJ, Li Q, Cassar CA, Dow MP, Pursley JR, Smith GW. Gonadotropin surge-induced differential upregulation of collagenase-1 (MMP-1) and collagenase-3 (MMP-13) mRNA and protein in bovine preovulatory follicles. Biol Reprod. 2004;71:605–12. doi: 10.1095/biolreprod.104.027185. [DOI] [PubMed] [Google Scholar]
- Beery AK, Trumbull JJ, Tsao JM, Costantini RM, Zucker I. Sex differences in the onset of seasonal reproductive quiescence in hamsters. Proc. Roy. Soc. 2007;274:281–286. doi: 10.1098/rspb.2006.3726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brännström M, Woessner JF, Koos RD, Sear CH, LeMaire WJ. Inhibitors of mammalian tissue collagenase and metalloproteinases suppress ovulation in the perfused rat ovary. Endocrinology. 1988;122:1715–1721. doi: 10.1210/endo-122-5-1715. [DOI] [PubMed] [Google Scholar]
- Butler TA, Zhu C, Mueller RA, Fuller GC, Lemaire WJ, Woessner JF. Inhibition of ovulation in the perfused rat ovary by the synthetic collagenase inhibitor SC 44463. Biol. Reprod. 1991;44:1183–1188. doi: 10.1095/biolreprod44.6.1183. [DOI] [PubMed] [Google Scholar]
- Chaffin CL, Stouffer RL. Expression of matrix metalloproteinases and their tissue inhibitor messenger ribonucleic acids in macaque periovulatory granulosa cells: time course and steroid regulation. Biol. Reprod. 1999;61:14–21. doi: 10.1095/biolreprod61.1.14. [DOI] [PubMed] [Google Scholar]
- Chandrashekar V, Majumdar SS, Bartke A. Assessment of the role of follicle-stimulating hormone and prolactin in the control of testicular endocrine function in adult Djungarian hamsters (Phodopus sungorus) exposed to either short or long photoperiod. Biol. Reprod. 1994;50:82–87. doi: 10.1095/biolreprod50.1.82. [DOI] [PubMed] [Google Scholar]
- Curry TE, Jr, Osteen KG. Cyclic changes in the matrix metalloproteinase system in the ovary and uterus. Biol. Reprod. 2001;64:1285–96. doi: 10.1095/biolreprod64.5.1285. [DOI] [PubMed] [Google Scholar]
- Curry TE, Song L, Wheeler SE. Cellular localization of gelatinases and tissue inhibitors of metalloproteinases during follicular growth, ovulation, and early luteal formation in the rat. Biol. Reprod. 2001;65:855–865. doi: 10.1095/biolreprod65.3.855. [DOI] [PubMed] [Google Scholar]
- Dodge JC, Kristal MB, Badura LL. Male-induced estrus synchronization in the female Siberian hamster (Phodopus sungorus sungorus). Physiological Behavior. 2002;7:227–231. doi: 10.1016/s0031-9384(02)00851-x. [DOI] [PubMed] [Google Scholar]
- Fowlkes JL, Enghild JJ, Suzuki K, Nagase H. Matrix metalloproteinases degrade insulin-like growth factor-binding protein 3 in dermal fibroblast cultures. J. Biol. Chem. 1994;269:25742–25746. [PubMed] [Google Scholar]
- García R, Ballesteros LM, Hernández-Pérez O, Rosales AM, Espinosa R, Soto H, Díaz de León L, Rosado A. Metalloproteinase activity during growth, maturation and atresia in the ovarian follicles of the goat. Anim. Reprod. Sci. 1997;47:211–228. doi: 10.1016/s0378-4320(96)01637-5. [DOI] [PubMed] [Google Scholar]
- Gearing AJH, Beckett P, Christodoulow M. Processing of tumor necrosis factor-alpha precursor by metalloproteinases. Nature. 1995;370:555–557. doi: 10.1038/370555a0. [DOI] [PubMed] [Google Scholar]
- Goldman SL, Dhandapani K, Goldman BD. Genetic and environmental influences on short-day responsiveness in Siberian hamsters (Phodopus sungorus). J. Biol. Rhythms. 2000;15:417–428. doi: 10.1177/074873000129001503. [DOI] [PubMed] [Google Scholar]
- Goldman S, Shalev E. MMPS and TIMPS in ovarian physiology and pathophysiology. Front. Biosci. 2004;9:2474–2483. doi: 10.2741/1409. [DOI] [PubMed] [Google Scholar]
- Gottsch ML, Van Kirk EA, Murdoch WJ. Role of matrix metalloproteinase 2 in the ovulatory folliculo-luteal transition of ewes. Reproduction. 2002;124:347–352. doi: 10.1530/rep.0.1240347. [DOI] [PubMed] [Google Scholar]
- Hägglund AC, Ny A, Leonardsson G, Ny T. Regulation and localization of matrix metalloproteinases and tissue inhibitors of metalloproteinases in the mouse ovary during gonadotropin-induced ovulation. Endocrinology. 1999;140:4351–4358. doi: 10.1210/endo.140.9.7002. [DOI] [PubMed] [Google Scholar]
- Imai K, Khandoker MA, Yonai M, Takahashi T, Sato T, Ito A, Hasegawa Y, Hashizume K. Matrix metalloproteinases-2 and -9 activities in bovine follicular fluid of different-sized follicles: relationship to intra-follicular inhibin and steroid concentrations. Domest. Anim. Endocrinol. 2003;24:171–183. doi: 10.1016/s0739-7240(02)00235-7. [DOI] [PubMed] [Google Scholar]
- Jo M, Thomas LE, Wheeler SE, Curry TE. Membrane type 1-matrix metalloproteinase (MMP)-associated MMP-2 activation increases in the rat ovary in response to an ovulatory dose of human chorionic gonadotropin. Biol. Reprod. 2004;70:1024–1032. doi: 10.1095/biolreprod.103.023499. [DOI] [PubMed] [Google Scholar]
- Johnson ML, Murdoch J, Van Kirk EA, Kaltenbach JE, Murdoch WJ. Tumor necrosis factor alpha regulates collagenolytic activity in preovulatory ovine follicles: relationship to cytokine secretion by the oocyte-cumulus cell complex. Biol. Reprod. 1999;61:1581–1585. doi: 10.1095/biolreprod61.6.1581. [DOI] [PubMed] [Google Scholar]
- Levi E, Fridman R, Miao HQ, Ma YS, Yayon A, Vlodavsky I. Matrix metalloproteinase 2 releases active soluble ectodomain of fibroblast growth factor receptor 1. Proc. Natl. Acad. Sci. U S A. 1996;93:7069–7074. doi: 10.1073/pnas.93.14.7069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li QL, Wang HM, Lin HY, Liu DL, Zhang X, Liu GY, Qian D, Zhu C. Expression of gelatinases and their tissue inhibitors in rat corpus luteum during pregnancy and postpartum. Mol. Reprod. Dev. 2002;63:273–281. doi: 10.1002/mrd.10155. [DOI] [PubMed] [Google Scholar]
- Lind AK, Dahm-Kähler P, Weijdegård B, Sundfeldt K, Brännström M. Gelatinases and their tissue inhibitors during human ovulation: increased expression of tissue inhibitor of matrix metalloproteinase-1. Mol. Hum. Reprod. 2006;12:725–736. doi: 10.1093/molehr/gal086. [DOI] [PubMed] [Google Scholar]
- Liu K, Olofsson JI, Wahlberh P, Ny T. Distinct expression of gelatinase A [matrix metalloproteinase (MMP)-2], collagenase-3 (MMP-13), membrane type MMP 1 (MMP-14), and tissue inhibitor of MMPs type 1 mediated by physiological signals during formation and regression of the rat corpus luteum. Endocrinology. 1999;140:5330–5338. doi: 10.1210/endo.140.11.7096. [DOI] [PubMed] [Google Scholar]
- Liu K, Rajareddy S, Liu L, Jagarlamudi K, Boman K, Selstam G, Reddy P. Control of mammalian oocyte growth and early follicular development by the oocyte PI3 kinase pathway: New roles for an old timer. Dev. Biol. 2006;299:1–11. doi: 10.1016/j.ydbio.2006.07.038. [DOI] [PubMed] [Google Scholar]
- Logan A, Hill DJ. Bioavailability: is this a key event in regulating the actions of peptide growth factors? J. Endocrinol. 1992;134:157–161. doi: 10.1677/joe.0.1340157. [DOI] [PubMed] [Google Scholar]
- Massova I, Kotra LP, Fridman R, Mobashery S. Matrix metalloproteinases: structures, evolution, and diversification. The FASEB Journal. 1998;12:1075–1095. [PubMed] [Google Scholar]
- McCaffery FH, Leask R, Riley SC, Telfer EE. Culture of bovine preantral follicles in a serum-free system: markers for assessment of growth and development. Biol. Reprod. 2000;63:267–273. doi: 10.1095/biolreprod63.1.267. [DOI] [PubMed] [Google Scholar]
- Moffatt-Blue CS, Sury JJ, Young KA. Short photoperiod-induced ovarian regression is mediated by apoptosis in Siberian hamsters (Phodopus sungorus). Reproduction. 2006;131:771–782. doi: 10.1530/rep.1.00870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muteka SP, Chimimba CT, Bennett NC. Reproductive photoresponsiveness in Aethomys ineptus and A. Namaquensis (Rodentia: Muridae) from southern Africa. J. Zool. 2006;268:225–231. [Google Scholar]
- Muteka SP, Chimimba CT, Bennett NC. Reproductive Seasonality in Aethomys namaquensis (Rodentia: Muridae) from southern Africa. J. Mammalogy. 2006;87:67–74. [Google Scholar]
- Nothnick WB. Disruption of the Tissue Inhibitor of Metalloproteinase-1 Gene Results in Altered Reproductive Cyclicity and Uterine Morphology in Reproductive-Age Female Mice. Biol. Reprod. 2000;63:905–912. doi: 10.1095/biolreprod63.3.905. [DOI] [PubMed] [Google Scholar]
- Nothnick WB. Tissue Inhibitor of Metalloproteinase-1 (TIMP-1) Deficient Mice Display reduced Serum Progesterone Levels During Corpus Luteum Development. Endocrinology. 2003;1:5–8. doi: 10.1210/en.2002-220963. [DOI] [PubMed] [Google Scholar]
- Nothnick WB, Soloway P, Curry TE., Jr Assessment of the role of tissue inhibitor of metalloproteinase-1 (TIMP-1) during the periovulatory period in female mice lacking a functional TIMP-1 gene. Biol. Reprod. 1997;56:1181–1188. doi: 10.1095/biolreprod56.5.1181. [DOI] [PubMed] [Google Scholar]
- Place NJ, Tuthill CR, Schoomer EE, Tramontin AD, Zucker I. Short Day Lengths Delay Reproductive Aging. Biol. Reprod. 2004;71:987–992. doi: 10.1095/biolreprod.104.029900. [DOI] [PubMed] [Google Scholar]
- Riley SC, Thomassen R, Bae SE, Leask R, Pedersen HG, Watson ED. Matrix metalloproteinase-2 and -9 secretion by the equine ovary during follicular growth and prior to ovulation. Anim. Reprod. Sci. 2004;81:329–339. doi: 10.1016/j.anireprosci.2003.10.011. [DOI] [PubMed] [Google Scholar]
- Ross AW, Bell LM, Littlewood PA, Mercer JG, Barrett P, Morgan PJ. Temporal changes in gene expression in the arcuate nucleus precede seasonal responses in adiposity and reproduction. Endocrinology. 2005;146:1940–1947. doi: 10.1210/en.2004-1538. [DOI] [PubMed] [Google Scholar]
- Schlatt S, De Geyter M, Kliesch S, Nieschlag E, Bergmann M. Spontaneous recrudescence of spermatogenesis in the photoinhibited male Djungarian hamster, Phodopus sungorus. Biol. Reprod. 1995;53:1169–1177. doi: 10.1095/biolreprod53.5.1169. [DOI] [PubMed] [Google Scholar]
- Schlatt S, Niklowitz P, Hoffmann K, Nieschlag E. Influence of short photoperiods on reproductive organs and estrous cycles of normal and pinealectomized female Djungarian hamster, Phodopus sungorus. Biol. Reprod. 1993;49:243–250. doi: 10.1095/biolreprod49.2.243. [DOI] [PubMed] [Google Scholar]
- Schmidt KE, Kelley KM. Down-regulation in the insulin-like growth factor (IGF) axis during hibernation in the golden-mantled ground squirrel, Spermophilus lateralis: IGF-I and the IGF-binding proteins (IGFBPs). J. Exp. Zool. 2001;289:66–73. doi: 10.1002/1097-010x(20010101/31)289:1<66::aid-jez7>3.0.co;2-q. [DOI] [PubMed] [Google Scholar]
- Smith MF, Gutierrez CG, Ricke WA, Armstrong DG, Webb R. Production of matrix metalloproteinases by cultured bovine theca and granulosa cells. Reproduction. 2005;129:75–87. doi: 10.1530/rep.1.00381. [DOI] [PubMed] [Google Scholar]
- Smith MF, McIntush EW, Ricke WA, Kojima FN, Smith GW. Regulation of ovarian extracellular matrix remodeling by metalloproteinases and their tissue inhibitors: effects on follicular development, ovulation and luteal function. J. Reprod. Fertil. Supp. 1999;54:367–381. [PubMed] [Google Scholar]
- Stouffer RL, Xu F, Duffy DM. Molecular control of ovulation and luteinization in the primate follicle. Front. Biosci. 2007;12:297–307. doi: 10.2741/2065. [DOI] [PubMed] [Google Scholar]
- Wahlberg P, Bodén I, Paulsson J, Lund LR, Liu K, Ny T. Functional Corpora Lutea Are Formed in Matrix Metalloproteinase Inhibitor-Treated Plasminogen-Deficient Mice. Endocrinology. 2007;148:1226–1234. doi: 10.1210/en.2006-0669. [DOI] [PubMed] [Google Scholar]
- Young KA, Nelson RJ. Mediation of seasonal testicular regression by apoptosis. Reproduction. 2001;122:677–685. doi: 10.1530/rep.0.1220677. [DOI] [PubMed] [Google Scholar]
- Young KA, Stouffer RL. Gonadotropin and Steroid Regulation of Matrix Metalloproteinases and Their Endogenous Tissue Inhibitors in the Developed Corpus Luteum of the Rhesus Monkey During the Menstrual Cycle. Biol. Reprod. 2003;70:244–252. doi: 10.1095/biolreprod.103.022053. [DOI] [PubMed] [Google Scholar]
- Young KA, Zirkin BR, Nelson RJ. Testicular apoptosis is down-regulated during spontaneous recrudescence in white-footed mice (Peromyscus leucopus). J. Biol. Rhythms. 2001;16:479–88. doi: 10.1177/074873001129002150. [DOI] [PubMed] [Google Scholar]