Abstract
Although numerous quantitative trait loci (QTL) influencing disease-related phenotypes have been detected through gene mapping and positional cloning, identification of the individual gene(s) and molecular pathways leading to those phenotypes is often elusive. One way to improve understanding of genetic architecture is to classify phenotypes in greater depth by including transcriptional and metabolic profiling. In the current study, we have generated and analyzed mRNA expression and metabolic profiles in liver samples obtained in an F2 intercross between the diabetes-resistant C57BL/6 leptinob/ob and the diabetes-susceptible BTBR leptinob/ob mouse strains. This cross, which segregates for genotype and physiological traits, was previously used to identify several diabetes-related QTL. Our current investigation includes microarray analysis of over 40,000 probe sets, plus quantitative mass spectrometry-based measurements of sixty-seven intermediary metabolites in three different classes (amino acids, organic acids, and acyl-carnitines). We show that liver metabolites map to distinct genetic regions, thereby indicating that tissue metabolites are heritable. We also demonstrate that genomic analysis can be integrated with liver mRNA expression and metabolite profiling data to construct causal networks for control of specific metabolic processes in liver. As a proof of principle of the practical significance of this integrative approach, we illustrate the construction of a specific causal network that links gene expression and metabolic changes in the context of glutamate metabolism, and demonstrate its validity by showing that genes in the network respond to changes in glutamine and glutamate availability. Thus, the methods described here have the potential to reveal regulatory networks that contribute to chronic, complex, and highly prevalent diseases and conditions such as obesity and diabetes.
Author Summary
Although numerous quantitative trait loci (QTL) influencing disease-related phenotypes have been detected through gene mapping and positional cloning, identifying individual genes and their potential roles in molecular pathways leading to disease remains a challenge. In this study, we include transcriptional and metabolic profiling in genomic analyses to address this limitation. We investigated an F2 intercross between the diabetes-resistant C57BL/6 leptinob/ob and the diabetes-susceptible BTBR leptinob/ob mouse strains that segregates for genotype and diabetes-related physiological traits; blood glucose, plasma insulin and body weight. Our study shows that liver metabolites (comprised of amino acids, organic acids, and acyl-carnitines) map to distinct genetic regions, thereby indicating that tissue metabolites are heritable. We also demonstrate that genomic analysis can be integrated with liver mRNA expression and metabolite profiling data to construct causal, testable networks for control of specific metabolic processes in liver. We apply an in vitro study to confirm the validity of this integrative method, and thus provide a novel approach to reveal regulatory networks that contribute to chronic, complex, and highly prevalent diseases and conditions such as obesity and diabetes.
Introduction
Genetic linkage and association studies have the power to establish a causal link between gene loci and physiological traits. These studies can make novel connections between biological processes that would not otherwise be predictable based on current knowledge. The pace of gene discovery has greatly accelerated in recent years, and numerous quantitative trait loci (QTL) influencing disease-related phenotypes have been identified through gene mapping and positional cloning. While it has become relatively straightforward to map a phenotype to a broad genomic region, identification of the individual gene(s) responsible for the phenotype remains difficult. Consequently, only a few percent of the many QTL that have been mapped have had their underlying gene(s) identified [1]–[7]. Another limitation of traditional QTL mapping is that it is based on association with a physiological phenotype, but often does not reveal the molecular pathways leading to that phenotype.
One way to uncover molecular mechanisms of disease states is to broadly expand the types of phenotypes analyzed in genetic screens. For example, with microarray technology, one can measure the abundance of virtually all mRNAs in a segregating sample. Importantly, mRNA abundance shows sufficient heritability in outbred populations and experimental crosses to allow mapping of gene loci that control gene expression, termed expression QTL (eQTL) [8],[9]. When eQTL co-localize with a physiological QTL, one can hypothesize a shared regulator and offer a potential pathway leading to the physiological trait [9],[10].
The pathway between a QTL and a physiological trait often involves changes in the steady-state levels of metabolic intermediates, in addition to changes in mRNA abundance. These metabolites can correlate with the genetic, transcriptional, translational, post-translational, and environmental influences on phenotype [7],[11]. Moreover, metabolites are intermediates in signaling pathways that can regulate gene expression. For example, fatty acids act as ligands for several of the PPAR nuclear hormone receptors, bile acids activate FXR in liver, and diacylglycerol regulates protein kinase C [12]–[14]. Metabolite abundance reflects a biological response to exogenous and endogenous inputs, and when investigating pathways from genotype to phenotype, metabolites can provide a powerful complement to gene expression data and give novel insights into disease pathogenesis mechanisms [7], [11], [15]–[25].
Our laboratories have begun to apply targeted metabolic profiling to study mechanisms underlying obesity-induced diabetes [15]–[20], but have not yet attempted to integrate these methods with genotyping and transcriptional profiling. This has included the application of gas chromatography/mass spectrometry (GC/MS) and tandem mass spectrometry (MS/MS) for measurements of acyl-carnitine, organic acid, amino acid, free fatty acid, and long and medium-chain acyl-CoA metabolites in tissue extracts and bodily fluids. Herein, we have applied these methods to measure various metabolites in liver samples from mouse strains that differ in susceptibility to obesity-induced diabetes.
C57BL/6 (B6) leptinob/ob mice are obese but essentially resistant to diabetes, whereas BTBR leptinob/ob mice are severely diabetic [22]. In an F2 cohort derived from these parental strains, we have shown that the range of blood glucose, insulin levels, and body weight exceeds that of either the C57BL/6 (B6) leptinob/ob or BTBR leptinob/ob parental strains. We went on to identify several diabetes-related QTL in this F2 sample [21],[22]. In the current study, we focused on a subset of 60 F2 mice that have previously been evaluated in detail with regard to liver gene expression profiles [24] to ask if the abundances of hepatic metabolic intermediates would show sufficient heritability to enable us to map metabolic QTL (mQTL). Because we previously performed mRNA expression profiling on liver samples from this F2 sample, we were also able to investigate the potential for integrative analysis of the expression profiling and metabolite data sets.
We show that liver metabolites do map to distinct genetic regions, thereby demonstrating that tissue metabolite profiles are heritable. In addition, we show that mQTL co-localize with eQTL, suggesting common genetic regulators. Finally, as a proof of principle of the practical significance of this multi-disciplinary approach, we illustrate the construction of a specific causal network that links gene expression and metabolic changes, and demonstrate its validity by targeted gene expression analysis.
Results
Metabolites of Similar Function Are Highly Correlated across the F2 Population
We determined the concentration of 67 liver metabolites, comprised of 15 amino acids and urea cycle intermediates, 45 acyl-carnitines, and 7 organic acids (TCA cycle intermediates and related metabolites) in the F2 sample. The specific analytes are summarized in Table S1.
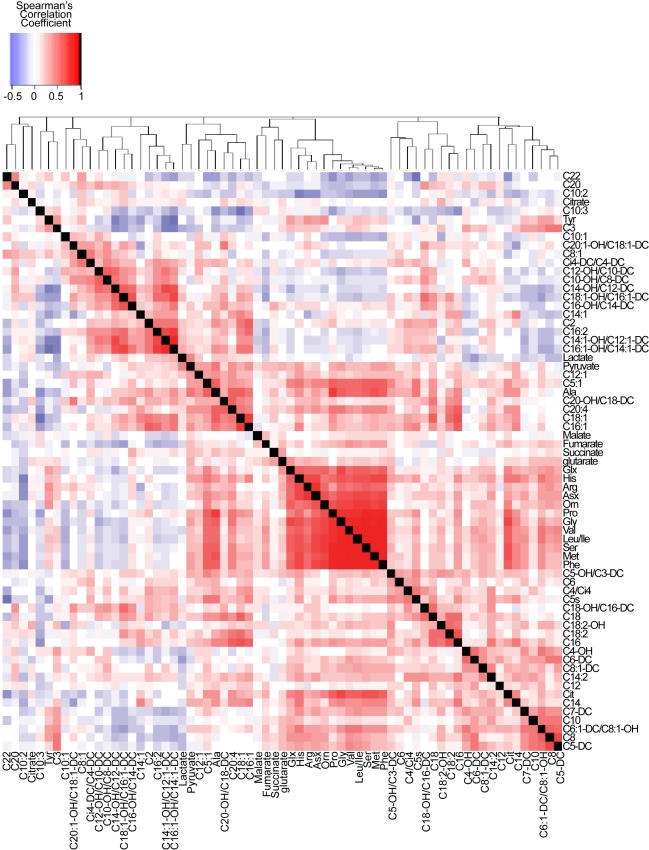
We created a correlation matrix of all pairwise comparisons among individual metabolites. Unsupervised hierarchical clustering revealed several “hot spots” of highly correlated metabolites (Figure 1). It is striking that several hot spots correspond to the biochemical pathway to which the metabolites belong. For example, 12 of the 15 amino acids cluster in this matrix. Moreover, when we consider pairwise correlations between all amino acids, 75% had absolute correlation coefficients greater than 0.5 (p<0.01) (Table S2). Permutation analysis of these pairwise correlations confirm that the 15 amino acids correlate as a functional group (p<0.001). Several specific acyl-carnitine derivatives are also clustered, such as hexadecadienoyl carnitine (C16∶2), 3-hydroxy-tetradecanoyl carnitine or dodecenedioyl carnitine (C14∶1-OH/C12∶1-DC), and 3-hydroxy-palmitoleoyl carnitine or cis-5-tetradecenedioyl carnitine (C16∶1-OH/C14∶1-DC). The fact that metabolites of a common functional group are highly correlated suggests that there are potential regulators of these biochemical pathways segregating in this F2 sample.
Figure 1. Heat map of correlations between liver metabolites.
Each square represents the Spearman's correlation coefficient between the metabolite of the column with that of the row (|r|>0.254, p<0.05; |r|>0.330, p<0.01). Metabolite order is determined as in hierarchical clustering using the distance function 1-correlation. Self-self correlations are identified in black. Acyl-carnitines are annotated according to clinical acyl-carnitine profile shorthand and amino acids by three letter code; other metabolite abbreviations are found in Table S1. Individual correlation coefficients can be found in Table S2.
In another cluster, pyruvate correlates most highly with alanine (r = 0.53, p<0.01), and also with lactate and tiglyl carnitine (C5∶1) (p<0.01). Alanine and short-chain acyl-carnitines are products of peripheral protein and fatty acid catabolism, respectively, and are delivered to the liver. The liver uses alanine, along with pyruvate and lactate, as gluconeogenic substrates and rapidly interconverts these metabolites through transamination and oxidation/reduction. The clustering of these metabolites based on their relative concentration in F2 animals suggests that static metabolic profiling can be used as a marker for changes in flux through certain metabolic pathways. All metabolite-metabolite correlation coefficients are listed in Table S2.
It has been demonstrated that mRNA abundance, as determined with microarray technology, is sufficiently heritable to map QTL [7], [8], [10], [23]–[27]. Lan et. al. showed that using expression mapping, specifically in this F2 intercross, can uncover mechanisms that explain correlations between specific transcripts [8]. We therefore sought to determine if metabolite abundance, as measured in F2 liver samples by mass spectrometry, was similarly heritable. If so, resulting metabolic QTL (mQTL) could be integrated with expression QTL (eQTL) to form network models of gene expression that might ultimately help to explain diabetes susceptibility and resistance in the BTBR leptinob/ob and B6 leptinob/ob strains, respectively [28],[29].
We found that individual metabolites mapped to specific regions of the genome. By permutation analysis, 21% of the metabolites map significantly to genomic regions (LOD>5.0, p<0.05), indicating those genomic regions could potentially influence (either directly or indirectly) the abundance of these metabolites. We used LOD threshold of 3.0 to investigate both major and minor putative mQTL where groups of metabolites map. Figure 2 displays a heat map, with metabolites organized by hierarchical clustering as in Figure 1. The twelve amino acids that clustered based on correlation (citrulline, tyrosine, and alanine are the exceptions) map to common mQTL, e.g., an overlapping region of chromosome 9. Amino acids that act together in specific pathways show additional common mQTL. For example glx (glutamine+glutamate) and urea cycle intermediates arginine, asx (asparagine+aspartate), and ornithine, map to a common region of chromosome 7. The gluconeogenic substrates alanine and pyruvate have a mapping profile distinct from the majority of amino acids in that they lack the prominent mQTL on chromosome 9 (Figure 2). This unique alanine/pyruvate mQTL may explain why alanine clusters with pyruvate rather than the amino acids in the correlation matrix (Figure 1).
Figure 2. Linkage hot spots for metabolic quantitative trait loci (mQTL).
Each row represents a marker; each column represents a metabolite. Metabolites are ordered as in hierarchical clustering using the distance function 1-correlation (as in Figure 1). The LOD color scale is indicated, showing blue (red) when the B6 (BTBR) allele at that marker results in an elevated level of metabolite.
Expected and Novel Correlations between Transcripts and Metabolites
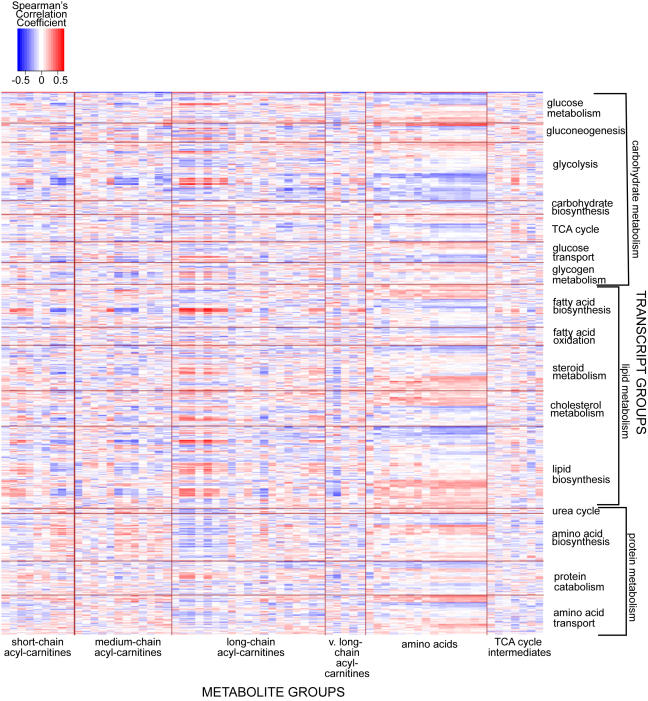
The foregoing results demonstrate that metabolites of a functional class often are correlated with one another and have common mQTL. To better understand how gene expression and metabolites are related, we adopted the approach used by Carrari [30] and created a correlation matrix between liver metabolites and selected liver transcripts of our 60 F2 mice. Three categories of transcripts were chosen, based on gene ontology terms relating to the biological process in which they play a role: 1) carbohydrate metabolism (glucose metabolism, gluconeogenesis, glycolysis, carbohydrate biosynthesis, TCA cycle, glucose transport, and glycogen metabolism); 2) lipid metabolism (fatty acid biosynthesis, fatty acid oxidation, steroid metabolism, cholesterol metabolism and biosynthesis, and lipid biosynthesis); and 3) protein metabolism (urea cycle, amino acid biosynthesis, protein catabolism, and amino acid transport). We organized the metabolites into functional classes to reveal whether biochemical groups of metabolites correlated in a specific pattern with transcripts of a particular pathway (Figure 3).
Figure 3. Heat map of correlations between liver metabolites and select liver transcripts.
Each square represents the Spearman's correlation coefficient between the metabolite of the column with the transcript of the row (|r|>0.254, p<0.05; |r|>0.330, p<0.01). Metabolites are organized into their biochemical class; transcripts are selected based on gene ontology terms relating to biological processes in which they play a role. Correlation coefficients between individual amino acids with select transcripts are found in Table S3.
We found evidence for correlations among functionally similar metabolites and transcripts when organized by biological process. For example, several long-chain acyl-carnitine species show a positive correlation with groups of transcripts involved in glycolysis, fatty acid biosynthesis, steroid metabolism, cholesterol metabolism, and lipid biosynthesis. In contrast, a subset of medium-chain acyl-carnitines and short chain acyl-carnitines exhibit a negative correlation to these same individual transcripts. These findings are consistent with recent studies from our laboratories showing that long-chain acyl-carnitines accumulate in muscle of animals with diet-induced obesity at the expense of short-chain acyl-carnitines, and that this abnormality is resolved when obese animals are exercised [17].
The 15 amino acids displayed a common correlation pattern with mRNA transcripts in pathways of protein metabolism, as well as glycolysis, the TCA cycle, and several lipid metabolism transcripts. These amino acids are very tightly correlated with one another, leading us to investigate the role played by individual transcripts in control of amino acid abundance. Our data show that two very highly correlated metabolites often correlate with the same set of individual transcripts. However, we also see that within this metabolite group, subsets of amino acids will have a unique transcript correlation pattern (Table S3, Table S4). For example, thirteen of fifteen amino acids correlate (r>0.35, p<0.01) with Slc38a3, a sodium-dependent transporter that mediates entry of a select group of amino acids across the plasma membrane. There are pathways by which the few known Slc38a3 amino acid substrates (alanine, asparagine, histidine, and glutamine) could serve as precursors for biosynthesis of non-substrate amino acids that also correlate with this transporter [31],[32]. In contrast, only valine and leucine+isoleucine correlate as highly (r>0.35, p<0.01) with Ppargc1a mRNA, and could represent a unique metabolic pathway involving the branched-chain amino acids.
Correlations and Co-Mapping of Transcripts and Metabolites Produce Causal Network Models
One hypothesis that follows from our results is that unique genetic regulators could affect the abundance of clusters of metabolites. Unlike mRNA transcripts, metabolites can be interconverted with other metabolites, generating a cluster to which the precursor metabolite will be highly correlated [33]. The downstream product metabolites will also be correlated with the regulatory transcript and co-map with the eQTL of the regulatory transcript [7],[34].
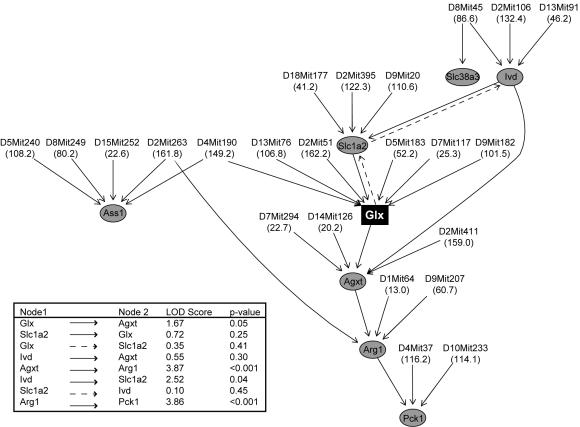
Glutamate is a substrate and product in amino acid catabolic and biosynthetic pathways. Glutamate can act either as an ammonium donor or acceptor in transamination reactions (via α-ketoglutarate) and the glutamate dehydrogenase reaction, and can also be rapidly synthesized from glutamine via glutaminase, thus providing precursor metabolites for the generation of other organic acids and amino acids. Glutamine can also act as a signaling molecule to alter expression of urea cycle and gluconeogenic enzymes [35]–[39]. Given that glutamine and glutamate (glx) can generate a network of related metabolites and can also change gene expression, we focused on glx as the start-point for building a proof-of-principle causal network from the F2 liver expression and metabolite profiling data sets. We generated a network featuring glx and a limited number of transcripts that passed multiple, stringent selection filters (see materials and methods). This provided a testable network that would enable us to gain insights into metabolite-transcript relationships.
Transcript nodes of the network are highly correlated to glx (p<0.05 by 10,000 permutations) as well as other amino acids (Table 1, Table S4). Table 1 depicts the overlap of the glx mQTL interval and the physical location of the transcripts or their eQTL encompassing a 1.5 LOD support interval around LOD peaks that are at least 3.0 [40],[41]. We note that glx is correlated with mRNA of two transporters: sodium-dependent amino acid transporter Slc38a3 and glutamate transporter Slc1a2, whose genes are located on chromosomes 9 (102.5 Mb) and 2 (107.5 Mb), respectively. Additionally, the glx mQTL on chromosome 9 spans a region containing Slc38a3 and the mQTL on chromosome 2 and 9 overlaps with the eQTL of Slc1a2 (Table 1). We hypothesize that both Slc1a2 and Slc38a3 could mediate the entry of glx into liver cells, but that Slc1a2 may also have expression regulated by glx abundance.
Table 1. Glx network correlations and mapping.
| Gene/Metabolite Symbol (Affy Primer Set)</emph> | GLX | Agxt 1418833_at | Arg11419549_at | Asl1448350_at | Ass11416239_at | Ivd1418238_at | Pck11423439_at | Pck11439617_s_at | Slc1a21451627_a_at | Slc38a31418706_atz | ||
| Spearman's Correlation to Glx | 1.00 | 0.55 | 0.35 | 0.23 | 0.40 | 0.33 | 0.36 | 0.39 | 0.49 | 0.42 | ||
| mQTL/eQTL (LOD>3±1.5 LOD) (Mb) | Chromosome | 1 | 95.0 | 13.0–171.5 | 13.0–156.6 | |||||||
| 2 | 114.0–178.5 | 68.2–178.5 | 145.3–171.9 | 31.3;149.7–178.4 | 118.6;68.9–151.9 | 172.8 | 172.8 | 102.6;105.2–141.8 | ||||
| 3 | ||||||||||||
| 4 | 3.5–150.1 | 22.6–150.1 | 40.8–121.5 | 3.6–144.6 | ||||||||
| 5 | 39.3–117.7 | 130.3 | 47.7–129.2 | |||||||||
| 6 | ||||||||||||
| 7 | 13.9–35.1 | 13.9–28.7 | ||||||||||
| 8 | 48.5–119.5 | 46.0–170.7 | 36.6–100.0 | |||||||||
| 9 | 85.1–112.0 | 54.2–74.5 | 90.2–117.6 | 107.5 | ||||||||
| 10 | 24.6 | 107.2–120.4 | ||||||||||
| 11 | ||||||||||||
| 12 | ||||||||||||
| 13 | 81.4–112.0 | 33.1–86.4 | ||||||||||
| 14 | 19.2–23.1 | 21.1–98.3 | ||||||||||
| 15 | 3.3–91.8 | 3.3–76.0 | ||||||||||
| 16 | ||||||||||||
| 17 | ||||||||||||
| 18 | 15.5–85.7 | 30.7–48.3 | ||||||||||
| 19 | ||||||||||||
The Spearman's correlation coefficient of glx with the transcripts of the network is recorded (|r|>0.254, p<0.05; |r|>0.330, p<0.01). The physical location (Mb) of each transcript is noted in bold. For chromosomes containing eQTL or mQTL LOD>3.0, the transcript eQTL and glx mQTL ±1.5 LOD interval are given in Mb. Spearman's correlation coefficients between network transcripts and all fifteen amino acids are found in Table S4.
Table 1 also shows that glx is significantly correlated to argininosuccinate synthetase 1 (Ass1), arginase 1 (Arg1), phosphoenolpyruvate carboxykinase 1 (Pck1), isovaleryl coenzyme A dehydrogenase (Ivd) and alanine∶glyoxylate aminotransferase (Agxt) mRNAs. The physical location and/or mapping location of these transcripts with respect to the glx mQTL indicates that the metabolite-transcript relationship may go beyond correlation. For example, on chromosome 2, we see that the glx mQTL co-maps with the eQTL for Agxt, Arg1, Ass1, and Ivd [41]. This is consistent with network models in which the QTL regulates glx, which then regulates gene expression or conversely, the QTL regulates mRNA abunance of the four transcripts, which then regulate glx [9].
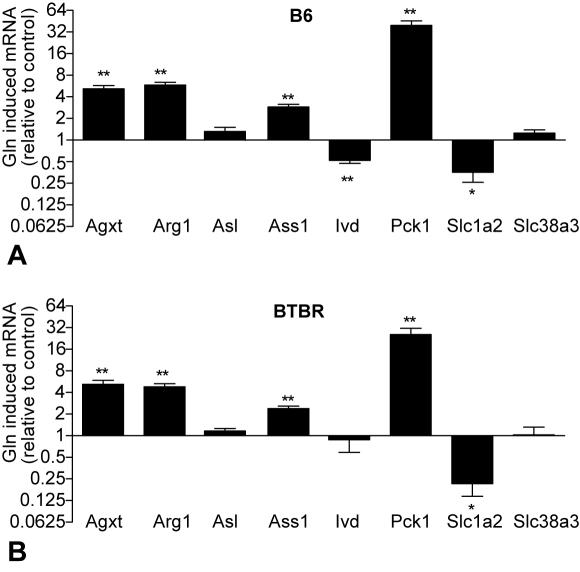
Using the method described by Chaibub et al. (in review), we generated a causal network consisting of glx and these highly correlated transcripts (FDR = 0.014), incorporating mQTL and eQTL to determine directionality between the nodes (Figure 4). This network model predicts that modulation of glutamine and/or glutamate levels should lead to a change in the expression of Agxt, Arg1, and Pck1. To test this prediction, we isolated hepatocytes from lean B6 and BTBR parental strains and measured changes in gene expression as a result of addition of 10 mM glutamine to the cultured cells. Glutamine exposure changed transcript abundance, and no transcript-specific strain differences in glutamine effect on gene expression were found (p = 0.53) (Figure 5). Glutamine significantly increased expression of Agxt, Arg1, Pck1, and Ass1 in both strains (p<0.05 for both strains); the increases in Pck1 and Ass1 confirm prior studies [36]–[39].
Figure 4. Glx network.
This network consists of a select number of transcripts (grey circles) among the 250 mRNA that are most correlated to glx (black rectangle) (p<0.002). The microsatellite marker (Mb) for peak eQTL or mQTL altering levels of transcripts and metabolites, respectively, are given. For any two phenotypes connected by an edge, the direction LOD score and p-value are indicated (insert). The best solution was determined by an approximate Bayes factor (BF) and indicated in solid lines, the second best solution in dotted lines.
Figure 5. Glutamine changes hepatic gene expression.
Hepatocytes from 10-week old lean B6 (A) and BTBR (B) were treated overnight +/− 10 mM glutamine (n = 5 per strain). Transcripts were measured by RT-PCR and expression was normalized to Actb control. Significance calculated based on the difference of delta CT value of each transcript between the untreated and glutamine treated hepatocytes for each individual animal (*p<0.05, **p<0.005).
Given its role as a glutamate transporter, it is not surprising that Slc1a2 is upstream of glx in the best proposed causal network (BF = 163) (Figure 4, solid lines). However, glutamine exposure in vitro reduced Slc1a2 expression in isolated hepatocytes from either mouse strain, supporting the second-best causal network solution (Figure 4, dotted lines). Glutamine also reduced Ivd expression in the B6 strain but showed no effect in the BTBR strain, despite Ivd being upstream of glx in our best causal network. Our causal network predicts Slc38a3 should be unchanged by glutamine treatment. Our hepatocyte experiments confirm this prediction (Figure 5). Argininosuccinate lyase (Asl), which is neither correlated nor co-maps with glx, served as a negative control and indeed was not altered by glutamine treatment.
Discussion
Genomics, transcriptomics, proteomics, and metabolomics have delivered large arrays of data, allowing one to correlate physiological states with patterns of gene expression, protein levels, and metabolite abundance. A major challenge in the analysis and interpretation of this data is delivering models of causation from correlations [9],[42]. Mouse models of diabetes provide a unique method for exploring correlation structure since metabolic dysregulation creates a window for simultaneous application of multiple “omic” technologies.
We have previously shown that diabetes traits show strong heritability in an F2 intercross between the diabetes-resistant C57BL/6 leptinob/ob and the diabetes-susceptible BTBR leptinob/ob mouse strains. We assume that the disease phenotype is brought about by a complex pattern of gene expression changes in key tissues [21],[22]. However, we also recognize the complexity inherent in discriminating the gene expression changes that cause diabetes from those that occur as a consequence of the disease. For example, many genes are known to be responsive to elevated blood glucose levels [43]. Through correlation alone, it is difficult to distinguish these “reactive” genes from ones that are “causal” for the disease.
We have taken advantage of the high heritability of mRNA abundance phenotypes, and via microarray technology, have mapped gene loci controlling gene expression at the genome-wide level [8]. This establishes at least one node in a network simply because genetic variation leads to changes in gene expression and not vice versa. However, it does not establish whether the link between a locus and a phenotype is direct or via multiple steps and pathways [27],[44].
The purpose of the current study was to explore the possibility that the levels of metabolites in tissues are sufficiently heritable in an F2 intercross to provide significant linkage signals, leading to metabolic QTL. Given that many pathways converge upon common metabolites and that these pathways have multiple controllers, any one genetic locus may not alter metabolite levels significantly, and therefore may not be identified as a metabolite QTL. Nonetheless, in our F2 sample, we found significant linkage signals, including some that are quite strong (e.g. tyrosine: LOD>7, p<0.005; chromosome 2).
Our results reveal that metabolites can be mapped to distinct genetic regions, much like mRNA transcripts. Although QTL mapping in an F2 sample does not provide sufficient resolution to identify individual genes with high certainty, it can yield novel information about regulatory networks. Phenotypes mapping to the same locus can be hypothesized to be co-regulated by that locus. With our definition of “phenotype” now including transcripts, metabolites, and physiological traits, we can begin to devise relationships between these phenotypes and genetic regions.
This F2 study provides evidence of co-regulation of biologically related pathways. An example is the correlations we found between amino acids and short-chain acyl-carnitine derivatives. These findings are consistent with our understanding of metabolic physiology. In a catabolic, “glucose starved” state, muscle degrades proteins and delivers amino acids to the liver for glucose production. The liver transaminates amino acids to corresponding α-keto acid gluconeogenic substrates. Alpha-ketoglutarate is often the α-keto acid acceptor for these transaminase reactions, generating glutamate as a product. Glutamate, which can also be generated from glutamine in the glutaminase reaction, is then deaminated to produce ammonia by glutamate dehydrogenase, to be fixed through the urea cycle. Additionally, hepatic fatty acid oxidation and amino acid catabolism yield even and odd-numbered short-chain acyl CoAs, which can be used for fuel and for production of ketone bodies. These short-chain acyl-CoA species are readily converted to the cognate carnitine esters, which we have profiled by MS/MS in this study.
The amino acid metabolites provide the most striking evidence of functional clustering. We see in both the correlation matrix (Figure 1) and the genetic linkage data (Figure 2) that the majority of amino acids group together. However, a subset of the amino acids, asx, glx, arginine, and ornithine uniquely map to chromosome 7. Our data predict that these metabolites are driven by different genetic regulators, leading to a unique mapping signature, even within a group of highly correlated metabolites. The C/EBP transcription factors have been shown to alter expression of enzymes acting in the urea cycle and gluconeogenic pathway [45]–[51], and the C/EBPα isoform is encoded on chromosome 7. Although we cannot determine that metabolites are mapping to the same individual genes, we can identify genetic regions that coordinate groups of metabolites and transcripts and contain plausible candidate genes.
The relationship between mRNA transcripts and metabolites, however, can be bi-directional. Our network identifies a specific metabolite, glx that regulates gene expression. This is consistent with previous studies where glutamine alone increases hepatic expression of argininosuccinate synthetase and phosphoenolpyruvate carboxykinase, but when combined with other essential amino acids, alters additional transcripts of urea cycle and gluconeogenic pathways [36]–[38],[52]. Our work extends these prior observations by showing that glutamine also changes expression of Agxt, Arg1, Ivd, and Slc1a2, but does not alter Slc38a3, despite the positive correlation with this transcript. The combination of pathway construction based on transcriptional and metabolic profiling and direct model testing in living cells provides evidence for a new pathway by which glx can regulate a key gluconeogenic enzyme. Future studies will be needed to investigate if this pathway is perturbed in development of diabetes.
The glutamine induced reduction in Slc1a2 expression was unexpected given that this glutamate transporter is upstream of glx in the best-proposed causal network (Figure 4, solid lines). Slc1a2 mRNA abundance, however, maps in trans (to a locus distinct from the physical location of the gene) to chromosome 9, its eQTL overlapping with the glx mQTL. It is therefore possible that glutamine could regulate Slc1a2, as indicated by the second causal network (Figure 4, dotted lines). Several studies have shown that Slc1a2 expression in astrocytes is reduced by increased ammonia [45]–[47], [51], [53]–[55]. Despite the positive correlation between Slc1a2 and glx in vivo, the glutamine-treated hepatocytes produce ammonia via glutaminase, and could decrease expression of hepatic Slc1a2 in vitro. We also did not predict altered expression of Ivd, an enzyme of leucine oxidation. It is interesting to note that Ivd is a case where a gene maps both in cis (to the locus containing the Ivd gene) and in trans, here overlapping with the glx mQTL on chromosomes 2 and 13. Studies have shown that glutamine has an inverse relationship with leucine oxidation, and this could be mediated by glutamine-induced decreased Ivd expression [48],[50].
We show that the combined use of eQTL and mQTL, with correlations allows one to derive a network and establish data-driven hypotheses about metabolite and gene expression relationships. For example, glycine and serine are the two amino acids most highly correlated with glx, and the transcript most highly correlated with glx is Agxt (Table 1, Table S2). Indeed, in our experiments, Agxt was upregulated by glutamine. We hypothesize that the upregulation by glx of Agxt is one mechanism by which glx is correlated with glycine and serine since Agxt catalyzes the transamination of glyoxalate to form glycine, which can then be converted to serine. In further support of this hypothesis, in the F2 sample, serine and glycine correlate (r>0.5, p<0.01) to Agxt.
The concurrent use of transcriptomics and metabolomics is not limited to one biochemical pathway. For example, the correlation between amino acids and transcripts of carbohydrate and lipid metabolism might reflect a broader signaling function of amino acids beyond pathways of protein metabolism. Furthermore, this correlation, co-mapping, and causal network analysis can uncover roles for transcripts of unknown function. We note Riken clones and ESTs are among the transcripts highly correlated to individual metabolites (Table S3). By incorporating these transcripts of unknown function as nodes into causal networks, along with transcripts from known pathways, we may infer the functions of these previously unidentified mRNA species.
In conclusion, this study shows that metabolites, in addition to transcripts and physiological traits, can be mapped to genetic regions, providing a powerful tool to establish connections between genetic loci and physiological traits. The groups of metabolites and transcripts that are correlated or co-map to physiological traits in our F2 sample may offer insight into metabolic pathways that are causal or reactive to diabetes pathology.
Materials and Methods
Animals
BTBR, B6, and B6-ob/+ mice were purchased from The Jackson Laboratory (Bar Harbor, ME) and bred at the University of Wisconsin. The lineage and characteristics of the BTBR strain have been reviewed by Ranheim et al. Mice were housed in an environmentally controlled facility (12-hour light and dark cycles) and were weaned at 3 weeks of age onto a 6% fat diet (Purina; #5008). Mice had ad libitum access to food and water, except for 4 hour fasting periods before blood draws and killing (by CO2 asphyxiation). Plasma glucose levels were measured using a commercially available kit (994-90902; Wako Chemicals). Plasma insulin levels were measured by radioimmunoassay (RI-13K; Linco Research).
The facilities and research protocols were approved by the University of Wisconsin Institutional Animal Care and Use Committee.
Genotyping
Sixty F2 leptinob/ob mice ranging in age from 13 to 26 weeks were genotyped as previously described [22]. Mapmaker/EXP was used to compile genotype data into framework map.
RNA Collection and Microarray
Liver RNA was arrayed as described in Lan et. al [8]. Ten to 12 week old male and female F2 leptinob/ob mice were killed by CO2 asphyxiation after a 4-h fast. Total RNA from sixty F2 mice using RNAzol reagent (Tel-Test) and was further purified using an RNeasy kit (Qiagen). The sample labeling, microarray hybridization, washing, and scanning were performed according to the manufacturer's protocols (Affymetrix). Labeled cRNA was prepared and hybridization assay procedures including preparation of solutions were carried out as described in the Affymetrix GeneChip Expression Analysis Technical Manual. A total of 60 MOE430A and MOE430B arrays were used to monitor the expression levels of approximately 45,000 genes or ESTs. The distribution of fluorescent material on the array was obtained using G2500A GeneArray Scanner (Affymetrix). Microarray Suite (MAS) version 5.0 and GeneChip Operating Software (GCOS) supplied by Affymetrix was used to perform gene expression analysis. Expression levels of all the transcripts were estimated using the RMA algorithm [49].
Liver Metabolite Quantification
Amino acids, acyl-carnitines and organic acids were measured using stable isotope dilution techniques [15],[18],[56]. Amino acids and acyl-carnitine species were measured using flow injection tandem mass spectrometry and sample preparation methods described previously [15],[56]. Briefly, samples were equilibrated with a cocktail of internal standards, de-proteinated by precipitation with methanol, aliquoted supernatants were dried, and then esterified with hot, acidic methanol (acyl-carnitines) or n-butanol (amino acids). The data were acquired using a Micromass Quattro micro TM system equipped with a model 2777 autosampler, a model 1525 µ HPLC solvent delivery system and a data system controlled by MassLynx 4.0 operating system (Waters, Milford, MA) [15],[56]. Organic acids were quantified using a previously described method that utilizes Trace GC Ultra coupled to a Trace DSQ MS operating under Excalibur 1.4 (Thermo Fisher Scientific, Austin, TX) [18].
Sixty-seven liver metabolites were measured, comprised of 15 amino acids and urea cycle intermediates, 45 acyl-carnitine derivatives, and 7 organic acids (TCA cycle intermediates and related analytes). The specific metabolites are listed in Table S1. All MS analyses employed stable-isotope-dilution. The standards serve both to help identify each of the analyte peaks and provide the reference for quantifying their levels. Quantification was facilitated by addition of mixtures of known quantities of stable-isotope internal standards from Isotec (St. Louis, MO), Cambridge Isotope Laboratories (Andover, MA), and CDN Isotopes (Pointe-Claire, Quebec, CN) to samples, as follows: Acyl-carnitine assays–D3-acetyl, D3-propionyl, D3-butyryl, D9-isovaleryl, D3-octanoyl, and D3-palmitoyl carnitines; Amino acid assays–15N1,13C1-glycine, D4-alanine, D8-valine, D7-proline, D3-serine, D3-leucine, D3-methionine, D5-phenylalanine, D4-tyrosine, D3-aspartate, D3-glutamate, D2-ornithine, D2-citrulline, and D5-arginine; Organic acid assays–D3-lactate, D3-pyruvate, 13C4-succinate, D2-fumarate, D4-glutarate, 13C1-malate, D6-alpha-ketoglutarate, and D3-citrate. In addition to mass, analytes are identified on the basis of the particular MS/MS transitions that we monitor for each class of metabolites. For example, all acyl-carnitine methyl esters produce a fragment m/z 99. We make the assumption that all even mass precursors ions of m/z 99 are acyl-carnitines to which we assign plausible molecular structures. We differentiate isobaric structures e.g., dicarboxylic and hydroxylated acyl-carnitines, by comparing of MS/MS spectra for precursors of m/z 85 butylated acyl-carnitine species. We can infer whether the original compound had one or two carboxyl groups on the basis of the mass change from methyl to butyl esters.
Given our sample size, we initially analyzed metabolite abundance by hierarchical clustering using the distance function 1-correlation [40], [57]–[60]. Pairwise Spearman correlation coefficients of r>0.254 and r>0.330 reflected p-values p<0.05 and p<0.01, respectively. To test whether the 15 amino acids are significantly correlated as a group, groups of 15 metabolites were permuted 1,000 times and the percentage of pairwise correlations exceeding 0.5 was recorded for each group. The fifteen amino acids cluster significantly as a group based on 1,000 permutations (p<0.001).
QTL Analysis
Detection and mapping of QTL was performed as previously described [8],[22]. Briefly, genotypes of 512 F2 mice at 293 markers were assembled using MAPMAKER/EXP [61]. A previously established subset of 60 mice with transcript data was used for expression QTL analysis [24]. Interval mapping methods adjusted for sex as implemented in R/qtl [62] were used to compute linkage to the traits of interest and to investigate mode of inheritance. The traits included the 45,265 probe sets surveyed by microarray analysis, and the 67 liver metabolites assayed by MS methods.
We used standard interval mapping implemented in R/qtl to map each of the transcripts and liver metabolites at 1-cM resolution with age as additive covariates and sex as both additive and interactive covariates [62]. A LOD threshold of 5.0 is required to reach a level of p<0.05 in this data set with sample size 60 based on 10,000 permutations. We used threshold of 3.0 in order to highlight genetic regions to which groups of metabolites map. To visualize regions of mQTL co-localization in highly correlated metabolites (Figure 2), we constructed heat maps where metabolites are ordered as in hierarchical clustering using 1-correlation, as in Figure 1. When mice with the B6 allele at a marker have greater levels of metabolites on average than mice with the BTBR allele at that marker, the LOD score at that marker is multiplied by −1. This adjustment allows us to visualize whether the B6 or BTBR allele results in elevated metabolite abundance.
In Vitro Hepatocyte Experiments
Hepatocytes from 10-week lean male and female BTBR and B6 parental strain mice (n = 5 for each genotype) were isolated by liver perfusion [63]. Hepatocytes were seeded at subconfluency (3.5 × 106 cells/6 well plate) in low glucose DMEM (GIBCO) supplemented with FBS (10% vol/vol; GIBCO), pen/strep antibiotic (1%, GIBCO), glutamine (2 mM; GIBCO), and pyruvate (1 mM; GIBCO). Cells were left to attach for 3 hours in an incubator at 37°C, 5% CO2. After a wash with PBS, the cells were treated with unsupplemented DMEM (Sigma) with 1 g/L glucose, pen/strep (1%), and +/− 10 mM glutamine. Cells were treated for 24 hours.
RNA was extracted from hepatocytes using RNeasy kits (Qiagen) after treatment described above. Hepatocytes in 6-well plates were homogenized in 0.35 ml of RLT buffer and stored at −80 C. RNA was purified using RNeasy-mini columns (Qiagen) according to the manufacturer's directions. The ratio of the optical densities from RNA samples measured at 260 and 280 nm was used to evaluate nucleic acid purity and total RNA concentrations were determined by the absorbance at 260 nm. The quality of total RNA was estimated based on the integrity of 28S and 18S rRNA separated using 1% agarose gel electrophoresis.
Gene expression was measured using a 7500 fast real-time PCR system (Applied Biosystems). cDNA was synthesized from 1 ug of total RNA using the SuperScriptIII first-strand cDNA synthesis kit (Invitrogen) primed with a mixture of oligo-dT and random hexamers. Primers were obtained from Integrated DNA Technologies and MWG Biotechnology. The SYBR Green PCR core reagent kit (Applied Biosystems) was used to determine relative expression. The housekeeping gene Actb was used as a normalization control. Primer sequences and gene accession codes for transcripts of the glx network are provided in Table S5.
Networks
Causal networks were constructed using the methods of Chaibub, et al. (in review). Although the network has the ability to accommodate 100 or more transcripts, we chose a limited number of transcripts passing several selection filters. The transcripts for the glx network were derived from the top 250 most correlated transcripts (p<0.002) according to the WebQTL software (www.genenetwork.org). A hypergeometric test was performed and identified the GO term category “metabolism” as one of the two processes significantly enriched by these correlates (p<0.004). Transcripts were chosen from this category, with an additional requirement being that they have at least one eQTL overlapping with the glx mQTL (Table 1). QTL in the genetic region encompassing a 1.5 LOD support interval around LOD peaks that are at least 3.0 are also included [41],[42]. Based on 10,000 permutations for each of the transcripts, the LOD threshold is significantly higher to reach significance (LOD>5.0 is required for p<0.05), but the 3.0 threshold was used include major and minor putative QTL [8],[24].
If more than one probe set was used to identify a transcript of interest, only probe sets with a grade A annotation on Affymetrix were considered. For these probe sets, only those with all eleven oligonucleotides aligning (via BLAST) to their appropriate target sequence provided by the National Center for Biotechnology Information (www.ncbi.nlm.nih.gov) were considered acceptable. If more than one primer set still identified the transcript, an average of the probe sets in the network.
We built an undirected dependency graph (UDG) of order 6 with glx and these transcripts as nodes with a two-tailed significance level of 0.05 [64]. We remove edges that are based on spurious or partial correlations, and then orient causal edges between all pairs of connected phenotypes using associated multiple QTLs to break likelihood equivalence. Quantitative trait loci for glx and the selected transcripts were identified with R/qtl [62] using a 3.0 LOD cutoff; the marker closest to each peak provided key information for inferring causal direction. We oriented phenotype edges using our QTL-directed dependency graph (QDG) approach. For any two phenotypes connected by an edge, the direction LOD score was computed by regressing these phenotypes on each other and on their respective multiple QTLs, adjusting for age and for QTL-sex interactions, and by other phenotypes that might be directly connected to either phenotype by an UDG edge. For each edge, we evaluate a LOD score comparing the two possible orientations and we orient the edge in favor of the direction with the higher likelihood in the ratio. P-values for the direction of the edges were computed using 10,000 permutations. Our QDG algorithm used random starts to converge to possible solutions. The best solution was determined by an approximate Bayes factor (BF) [65],[66]. A detailed materials and methods section describing the construction of causal networks is provided in Supporting Protocols (Protocol S1).
We estimated network parameters from the true data and simulated synthetic data according to the causal network in Figure 4. We simulated 1,000 realizations from the causal network and for each edge, we recorded the percentage of undirected edges recovered by the UDG algorithm and the percentage correctly inferred direction by the orientation steps of the QDG algorithm. Overall, the average percentage of true recovered edges was 75% and the average percentage of correctly inferred direction was 83%. False edges were detected at a rate below 2%. To calculate the false discovery rate for the network, we simulated 1,000 data sets from the true network. For each data set, the UDG algorithm was used to infer the network topology, and computed the fraction of false edges (those detected that do not exist in the true network) relative to the total number edges detected by the UDG algorithm. The FDR for the network topology, computed as the average fraction for these 1,000 simulations, is 0.014.
Gene Expression Analysis
The fold changes relative to the untreated hepatocytes for each animal were calculated. An overall ANOVA analysis was performed with gene transcripts nested within subject; interest focused on gene transcript effects and possible gene transcript differences between strains. This analysis showed that glutamine-induced expression change differed by gene (p<0.0001). Significant overall gene transcript effects allows separate transcript-specific paired t-tests between the difference in delta CT values of untreated and glutamine induced gene expression (relative to Actb) in each strain separately. Statistics on these data were analyzed with Prism software version 4.02 (Graph Pad Software) and the aov command in R (www.r-project.org).
Supporting Information
Causal network reconstruction.
(0.05 MB DOC)
Metabolite codes. Acyl-carnitines are annotated according to clinical acyl-carnitine profile shorthand; conventional amino acids are annotated by their three-letter code. In the cases where two acyl-carnitine derivatives were unable to be distinguished by MS analysis alone, both are reported, indicating that either analyte could be the predominant species.
(0.02 MB XLS)
Metabolite correlation matrix. All pairwise correlations between the 67 metabolites were calculated and recorded as the Spearman's correlation coefficient (|r|>0.254, p<0.05; |r|>0.330, p<0.01). Metabolites are ordered according to the strength of their correlation as in Figure 1.
(0.10 MB XLS)
Correlation of amino acids and individual transcripts metabolic pathways. Transcripts that had Spearman's correlation r>0.4 to any individual amino acid are included (|r|>0.254, p<0.05; |r|>0.330, p<0.01). The physical location of the gene is provided (Mb). Amino acids are ordered as in Figure 3.
(0.81 MB XLS)
Glx network correlations. The Spearman's correlation coefficient of the 15 individual amino acids with the transcripts of the glx network is recorded (|r|>0.254, p<0.05; |r|>0.330, p<0.01). Amino acids are ordered as in Figure 3.
(0.02 MB XLS)
Primer sequences. Integrated DNA Technologies (Agxt, Pck1, Slc38a3) and MWG Biotechnology (Actb, Arg1, Asl, Ass1, Ivd, Slc1a2).
(0.02 MB XLS)
Acknowledgments
We would like to acknowledge Angie Tebon Oler for her very generous technical help with hepatocyte experiments. We would also like to thank Katherine Scheuler, Donald Stapleton, and the animal care facility at the University of Wisconsin for their services.
Footnotes
The authors have declared that no competing interests exist.
Funding was supported by grants DK58037 and DK06639 (ADA), and the NIDDK grant PO1 DK58398 (CBN). Funding was also provided from the National Institute of General Medical Sciences through the Duke Medical Scientist Training Program grant 2T32GM007171 and CNPq, Brazil.
References
- 1.DiPetrillo K, Wang X, Stylianou IM, Paigen B. Bioinformatics toolbox for narrowing rodent quantitative trait loci. Trends Genet. 2005;21:683–692. doi: 10.1016/j.tig.2005.09.008. [DOI] [PubMed] [Google Scholar]
- 2.Glazier AM, Nadeau JH, Aitman TJ. Finding genes that underlie complex traits. Science. 2002;298:2345–2349. doi: 10.1126/science.1076641. [DOI] [PubMed] [Google Scholar]
- 3.Guo SW, Lange K. Genetic mapping of complex traits: promises, problems, and prospects. Theor Popul Biol. 2000;57:1–11. doi: 10.1006/tpbi.2000.1449. [DOI] [PubMed] [Google Scholar]
- 4.Hirschhorn JN, Daly MJ. Genome-wide association studies for common diseases and complex traits. Nat Rev Genet. 2005;6:95–108. doi: 10.1038/nrg1521. [DOI] [PubMed] [Google Scholar]
- 5.Nadeau JH, Frankel WN. The roads from phenotypic variation to gene discovery: mutagenesis versus QTLs. Nat Genet. 2000;25:381–384. doi: 10.1038/78051. [DOI] [PubMed] [Google Scholar]
- 6.Petretto E, Liu ET, Aitman TJ. A gene harvest revealing the archeology and complexity of human disease. Nat Genet. 2007;39:1299–1301. doi: 10.1038/ng1107-1299. [DOI] [PubMed] [Google Scholar]
- 7.Wentzell AM, Rowe HC, Hansen BG, Ticconi C, Halkier BA, et al. Linking metabolic QTLs with network and cis-eQTLs controlling biosynthetic pathways. PLoS Genet. 2007;3:1687–1701. doi: 10.1371/journal.pgen.0030162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lan H, Chen M, Flowers JB, Yandell BS, Stapleton DS, et al. Combined expression trait correlations and expression quantitative trait locus mapping. PLoS Genet. 2006;2:e6. doi: 10.1371/journal.pgen.0020006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Schadt EE, Lamb J, Yang X, Zhu J, Edwards S, et al. An integrative genomics approach to infer causal associations between gene expression and disease. Nat Genet. 2005;37:710–717. doi: 10.1038/ng1589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kliebenstein DJ, West MA, van Leeuwen H, Loudet O, Doerge RW, et al. Identification of QTLs controlling gene expression networks defined a priori. BMC Bioinformatics. 2006;7:308. doi: 10.1186/1471-2105-7-308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fiehn O. Metabolomics–the link between genotypes and phenotypes. Plant Mol Biol. 2002;48:155–171. [PubMed] [Google Scholar]
- 12.Graber R, Sumida C, Nunez EA. Fatty acids and cell signal transduction. J Lipid Mediat Cell Signal. 1994;9:91–116. [PubMed] [Google Scholar]
- 13.Quest AF, Ghosh S, Xie WQ, Bell RM. DAG second messengers: molecular switches and growth control. Adv Exp Med Biol. 1997;400A:297–303. doi: 10.1007/978-1-4615-5325-0_42. [DOI] [PubMed] [Google Scholar]
- 14.Wang H, Chen J, Hollister K, Sowers LC, Forman BM. Endogenous bile acids are ligands for the nuclear receptor FXR/BAR. Mol Cell. 1999;3:543–553. doi: 10.1016/s1097-2765(00)80348-2. [DOI] [PubMed] [Google Scholar]
- 15.An J, Muoio DM, Shiota M, Fujimoto Y, Cline GW, et al. Hepatic expression of malonyl-CoA decarboxylase reverses muscle, liver and whole-animal insulin resistance. Nat Med. 2004;10:268–274. doi: 10.1038/nm995. [DOI] [PubMed] [Google Scholar]
- 16.Haqq AM, Lien LF, Boan J, Arlotto M, Slentz CA, et al. The Study of the Effects of Diet on Metabolism and Nutrition (STEDMAN) weight loss project: Rationale and design. Contemp Clin Trials. 2005;26:616–625. doi: 10.1016/j.cct.2005.09.003. [DOI] [PubMed] [Google Scholar]
- 17.Koves TR, Li P, An J, Akimoto T, Slentz D, et al. Peroxisome proliferator-activated receptor-gamma co-activator 1alpha-mediated metabolic remodeling of skeletal myocytes mimics exercise training and reverses lipid-induced mitochondrial inefficiency. J Biol Chem. 2005;280:33588–33598. doi: 10.1074/jbc.M507621200. [DOI] [PubMed] [Google Scholar]
- 18.Jensen MV, Joseph JW, Ilkayeva O, Burgess S, Lu D, et al. Compensatory responses to pyruvate carboxylase suppression in islet beta-cells. Preservation of glucose-stimulated insulin secretion. J Biol Chem. 2006;281:22342–22351. doi: 10.1074/jbc.M604350200. [DOI] [PubMed] [Google Scholar]
- 19.Ronnebaum SM, Ilkayeva O, Burgess SC, Joseph JW, Lu D, et al. A pyruvate cycling pathway involving cytosolic NADP-dependent isocitrate dehydrogenase regulates glucose-stimulated insulin secretion. J Biol Chem. 2006;281:30593–30602. doi: 10.1074/jbc.M511908200. [DOI] [PubMed] [Google Scholar]
- 20.Monetti M, Levin MC, Watt MJ, Sajan MP, Marmor S, et al. Dissociation of hepatic steatosis and insulin resistance in mice overexpressing DGAT in the liver. Cell Metab. 2007;6:69–78. doi: 10.1016/j.cmet.2007.05.005. [DOI] [PubMed] [Google Scholar]
- 21.Stoehr JP, Byers JE, Clee SM, Lan H, Boronenkov IV, et al. Identification of major quantitative trait loci controlling body weight variation in ob/ob mice. Diabetes. 2004;53:245–249. doi: 10.2337/diabetes.53.1.245. [DOI] [PubMed] [Google Scholar]
- 22.Stoehr JP, Nadler ST, Schueler KL, Rabaglia ME, Yandell BS, et al. Genetic obesity unmasks nonlinear interactions between murine type 2 diabetes susceptibility loci. Diabetes. 2000;49:1946–1954. doi: 10.2337/diabetes.49.11.1946. [DOI] [PubMed] [Google Scholar]
- 23.Deutsch S, Lyle R, Dermitzakis ET, Attar H, Subrahmanyan L, et al. Gene expression variation and expression quantitative trait mapping of human chromosome 21 genes. Hum Mol Genet. 2005;14:3741–3749. doi: 10.1093/hmg/ddi404. [DOI] [PubMed] [Google Scholar]
- 24.Kendziorski CM, Chen M, Yuan M, Lan H, Attie AD. Statistical methods for expression quantitative trait loci (eQTL) mapping. Biometrics. 2006;62:19–27. doi: 10.1111/j.1541-0420.2005.00437.x. [DOI] [PubMed] [Google Scholar]
- 25.Lum PY, Chen Y, Zhu J, Lamb J, Melmed S, et al. Elucidating the murine brain transcriptional network in a segregating mouse population to identify core functional modules for obesity and diabetes. J Neurochem. 2006 doi: 10.1111/j.1471-4159.2006.03661.x. [DOI] [PubMed] [Google Scholar]
- 26.Bao L, Wei L, Peirce JL, Homayouni R, Li H, et al. Combining gene expression QTL mapping and phenotypic spectrum analysis to uncover gene regulatory relationships. Mamm Genome. 2006;17:575–583. doi: 10.1007/s00335-005-0172-2. [DOI] [PubMed] [Google Scholar]
- 27.Li H, Chen H, Bao L, Manly KF, Chesler EJ, et al. Integrative genetic analysis of transcription modules: towards filling the gap between genetic loci and inherited traits. Hum Mol Genet. 2006;15:481–492. doi: 10.1093/hmg/ddi462. [DOI] [PubMed] [Google Scholar]
- 28.Chesler EJ, Lu L, Shou S, Qu Y, Gu J, et al. Complex trait analysis of gene expression uncovers polygenic and pleiotropic networks that modulate nervous system function. Nat Genet. 2005;37:233–242. doi: 10.1038/ng1518. [DOI] [PubMed] [Google Scholar]
- 29.Schauer N, Semel Y, Roessner U, Gur A, Balbo I, et al. Comprehensive metabolic profiling and phenotyping of interspecific introgression lines for tomato improvement. Nat Biotechnol. 2006;24:447–454. doi: 10.1038/nbt1192. [DOI] [PubMed] [Google Scholar]
- 30.Carrari F, Baxter C, Usadel B, Urbanczyk-Wochniak E, Zanor MI, et al. Integrated analysis of metabolite and transcript levels reveals the metabolic shifts that underlie tomato fruit development and highlight regulatory aspects of metabolic network behavior. Plant Physiol. 2006;142:1380–1396. doi: 10.1104/pp.106.088534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gu S, Langlais P, Liu F, Jiang JX. Mouse system-N amino acid transporter, mNAT3, expressed in hepatocytes and regulated by insulin-activated and phosphoinositide 3-kinase-dependent signalling. Biochem J. 2003;371:721–731. doi: 10.1042/BJ20030049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Gu S, Villegas CJ, Jiang JX. Differential regulation of amino acid transporter SNAT3 by insulin in hepatocytes. J Biol Chem. 2005;280:26055–26062. doi: 10.1074/jbc.M504401200. [DOI] [PubMed] [Google Scholar]
- 33.Steuer R. Review: on the analysis and interpretation of correlations in metabolomic data. Brief Bioinform. 2006;7:151–158. doi: 10.1093/bib/bbl009. [DOI] [PubMed] [Google Scholar]
- 34.Jansen RC, Nap JP. Genetical genomics: the added value from segregation. Trends Genet. 2001;17:388–391. doi: 10.1016/s0168-9525(01)02310-1. [DOI] [PubMed] [Google Scholar]
- 35.Brosnan JT. Glutamate, at the interface between amino acid and carbohydrate metabolism. J Nutr. 2000;130:988S–990S. doi: 10.1093/jn/130.4.988S. [DOI] [PubMed] [Google Scholar]
- 36.Curi R, Lagranha CJ, Doi SQ, Sellitti DF, Procopio J, et al. Glutamine-dependent changes in gene expression and protein activity. Cell Biochem Funct. 2005;23:77–84. doi: 10.1002/cbf.1165. [DOI] [PubMed] [Google Scholar]
- 37.Curi R, Lagranha CJ, Doi SQ, Sellitti DF, Procopio J, et al. Molecular mechanisms of glutamine action. J Cell Physiol. 2005;204:392–401. doi: 10.1002/jcp.20339. [DOI] [PubMed] [Google Scholar]
- 38.Curi R, Newsholme P, Procopio J, Lagranha C, Gorjao R, et al. Glutamine, gene expression, and cell function. Front Biosci. 2007;12:344–357. doi: 10.2741/2068. [DOI] [PubMed] [Google Scholar]
- 39.Curthoys NP, Watford M. Regulation of glutaminase activity and glutamine metabolism. Annu Rev Nutr. 1995;15:133–159. doi: 10.1146/annurev.nu.15.070195.001025. [DOI] [PubMed] [Google Scholar]
- 40.Eisen MB, Spellman PT, Brown PO, Botstein D. Cluster analysis and display of genome-wide expression patterns. Proc Natl Acad Sci U S A. 1998;95:14863–14868. doi: 10.1073/pnas.95.25.14863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Manichaikul A, Dupuis J, Sen S, Broman KW. Poor performance of bootstrap confidence intervals for the location of a quantitative trait locus. Genetics. 2006;174:481–489. doi: 10.1534/genetics.106.061549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Drake TA, Schadt EE, Lusis AJ. Integrating genetic and gene expression data: application to cardiovascular and metabolic traits in mice. Mamm Genome. 2006;17:466–479. doi: 10.1007/s00335-005-0175-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lan MS, Wang HW, Chong J, Breslin MB. Coupling of glucose response element from L-type pyruvate kinase and G6Pase promoter enhances glucose responsive activity in hepatoma cells. Mol Cell Biochem. 2007;300:191–196. doi: 10.1007/s11010-006-9383-4. [DOI] [PubMed] [Google Scholar]
- 44.Hall R, Beale M, Fiehn O, Hardy N, Sumner L, et al. Plant metabolomics: the missing link in functional genomics strategies. Plant Cell. 2002;14:1437–1440. doi: 10.1105/tpc.140720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Desjardins P, Belanger M, Butterworth RF. Alterations in expression of genes coding for key astrocytic proteins in acute liver failure. J Neurosci Res. 2001;66:967–971. doi: 10.1002/jnr.10045. [DOI] [PubMed] [Google Scholar]
- 46.Hazell AS, Butterworth RF. Hepatic encephalopathy: An update of pathophysiologic mechanisms. Proc Soc Exp Biol Med. 1999;222:99–112. doi: 10.1046/j.1525-1373.1999.d01-120.x. [DOI] [PubMed] [Google Scholar]
- 47.Ye ZC, Sontheimer H. Modulation of glial glutamate transport through cell interactions with the extracellular matrix. Int J Dev Neurosci. 2002;20:209–217. doi: 10.1016/s0736-5748(02)00048-5. [DOI] [PubMed] [Google Scholar]
- 48.Hankard RG, Haymond MW, Darmaun D. Effect of glutamine on leucine metabolism in humans. Am J Physiol. 1996;271:E748–754. doi: 10.1152/ajpendo.1996.271.4.E748. [DOI] [PubMed] [Google Scholar]
- 49.Irizarry RA, Hobbs B, Collin F, Beazer-Barclay YD, Antonellis KJ, et al. Exploration, normalization, and summaries of high density oligonucleotide array probe level data. Biostatistics. 2003;4:249–264. doi: 10.1093/biostatistics/4.2.249. [DOI] [PubMed] [Google Scholar]
- 50.Le Bacquer O, Mauras N, Welch S, Haymond M, Darmaun D. Acute depletion of plasma glutamine increases leucine oxidation in prednisone-treated humans. Clin Nutr. 2007;26:231–238. doi: 10.1016/j.clnu.2006.09.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Zhou BG, Norenberg MD. Ammonia downregulates GLAST mRNA glutamate transporter in rat astrocyte cultures. Neurosci Lett. 1999;276:145–148. doi: 10.1016/s0304-3940(99)00816-2. [DOI] [PubMed] [Google Scholar]
- 52.Li Z, Yarmush ML, Chan C. Insulin concentration during preconditioning mediates the regulation of urea synthesis during exposure to amino acid-supplemented plasma. Tissue Eng. 2004;10:1737–1746. doi: 10.1089/ten.2004.10.1737. [DOI] [PubMed] [Google Scholar]
- 53.Albrecht J, Jones EA. Hepatic encephalopathy: molecular mechanisms underlying the clinical syndrome. J Neurol Sci. 1999;170:138–146. doi: 10.1016/s0022-510x(99)00169-0. [DOI] [PubMed] [Google Scholar]
- 54.Butterworth RF. Neurotransmitter dysfunction in hepatic encephalopathy: new approaches and new findings. Metab Brain Dis. 2001;16:55–65. doi: 10.1023/a:1011614528751. [DOI] [PubMed] [Google Scholar]
- 55.Butterworth RF. Glutamate transporters in hyperammonemia. Neurochem Int. 2002;41:81–85. doi: 10.1016/s0197-0186(02)00027-x. [DOI] [PubMed] [Google Scholar]
- 56.Wu JY, Kao HJ, Li SC, Stevens R, Hillman S, et al. ENU mutagenesis identifies mice with mitochondrial branched-chain aminotransferase deficiency resembling human maple syrup urine disease. J Clin Invest. 2004;113:434–440. doi: 10.1172/JCI19574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Davidov E, Clish CB, Oresic M, Meys M, Stochaj W, et al. Methods for the differential integrative omic analysis of plasma from a transgenic disease animal model. Omics. 2004;8:267–288. doi: 10.1089/omi.2004.8.267. [DOI] [PubMed] [Google Scholar]
- 58.Liu X, Gershenfeld HK. An exploratory factor analysis of the Tail Suspension Test in 12 inbred strains of mice and an F2 intercross. Brain Res Bull. 2003;60:223–231. doi: 10.1016/s0361-9230(03)00033-9. [DOI] [PubMed] [Google Scholar]
- 59.Musani SK, Zhang HG, Hsu HC, Yi N, Gorman BS, et al. Principal component analysis of quantitative trait loci for immune response to adenovirus in mice. Hereditas. 2006;143:189–197. doi: 10.1111/j.2006.0018-0661.01925.x. [DOI] [PubMed] [Google Scholar]
- 60.Nishihara E, Tsaih SW, Tsukahara C, Langley S, Sheehan S, et al. Quantitative trait loci associated with blood pressure of metabolic syndrome in the progeny of NZO/HILtJxC3H/HeJ intercrosses. Mamm Genome. 2007;18:573–583. doi: 10.1007/s00335-007-9033-5. [DOI] [PubMed] [Google Scholar]
- 61.Lander ES, Green P, Abrahamson J, Barlow A, Daly MJ, et al. MAPMAKER: an interactive computer package for constructing primary genetic linkage maps of experimental and natural populations. Genomics. 1987;1:174–181. doi: 10.1016/0888-7543(87)90010-3. [DOI] [PubMed] [Google Scholar]
- 62.Broman KW, Wu H, Sen S, Churchill GA. R/qtl: QTL mapping in experimental crosses. Bioinformatics. 2003;19:889–890. doi: 10.1093/bioinformatics/btg112. [DOI] [PubMed] [Google Scholar]
- 63.Princen HM, Huijsmans CM, Kuipers F, Vonk RJ, Kempen HJ. Ketoconazole blocks bile acid synthesis in hepatocyte monolayer cultures and in vivo in rat by inhibiting cholesterol 7 alpha-hydroxylase. J Clin Invest. 1986;78:1064–1071. doi: 10.1172/JCI112662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Shipley B. Cause and Correlation in Biology. New York: Cambridge University Press; 2000. [Google Scholar]
- 65.Krass RE, Raftery AE. Bayes factors. University of Washington; 1995. pp. 773–795. [Google Scholar]
- 66.Sartor MA, Tomlinson CR, Wesselkamper SC, Sivaganesan S, Leikauf GD, et al. Intensity-based hierarchical Bayes method improves testing for differentially expressed genes in microarray experiments. BMC Bioinformatics. 2006;7:538. doi: 10.1186/1471-2105-7-538. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Causal network reconstruction.
(0.05 MB DOC)
Metabolite codes. Acyl-carnitines are annotated according to clinical acyl-carnitine profile shorthand; conventional amino acids are annotated by their three-letter code. In the cases where two acyl-carnitine derivatives were unable to be distinguished by MS analysis alone, both are reported, indicating that either analyte could be the predominant species.
(0.02 MB XLS)
Metabolite correlation matrix. All pairwise correlations between the 67 metabolites were calculated and recorded as the Spearman's correlation coefficient (|r|>0.254, p<0.05; |r|>0.330, p<0.01). Metabolites are ordered according to the strength of their correlation as in Figure 1.
(0.10 MB XLS)
Correlation of amino acids and individual transcripts metabolic pathways. Transcripts that had Spearman's correlation r>0.4 to any individual amino acid are included (|r|>0.254, p<0.05; |r|>0.330, p<0.01). The physical location of the gene is provided (Mb). Amino acids are ordered as in Figure 3.
(0.81 MB XLS)
Glx network correlations. The Spearman's correlation coefficient of the 15 individual amino acids with the transcripts of the glx network is recorded (|r|>0.254, p<0.05; |r|>0.330, p<0.01). Amino acids are ordered as in Figure 3.
(0.02 MB XLS)
Primer sequences. Integrated DNA Technologies (Agxt, Pck1, Slc38a3) and MWG Biotechnology (Actb, Arg1, Asl, Ass1, Ivd, Slc1a2).
(0.02 MB XLS)