Abstract
The initial step of retinal ganglion cell (RGC) axon pathfinding involves directed growth of RGC axons toward the center of the retina, the optic disc, a process termed “intraretinal guidance”. Due to the accessibility of the system, and with various embryological, molecular, and genetic approaches, significant progress has been made in recent years toward understanding the mechanisms involved in the precise guidance of the RGC axons. As axons are extending from RGCs located throughout the retina, a multitude of factors expressed along with the differentiation wave are important for the guidance of the RGC axons. To ensure that the RGC axons are oriented correctly, restricted to the optic fiber layer (OFL) of the retina, and exit the eye properly, different sets of positive and negative factors cooperate in the process. Fasciculation mediated by a number of cell adhesion molecules (CAMs) and modulation of axonal response to guidance factors provide additional mechanisms to ensure proper guidance of the RGC axons. The intraretinal axon guidance thus serves as an excellent model system for studying how different signals are regulated, modulated and integrated for guiding a large number of axons in three-dimensional space.
Keywords: retinal ganglion cell axon, intraretinal guidance, guidance factors, optic disc, model system, fasciculation, review
1. Introduction
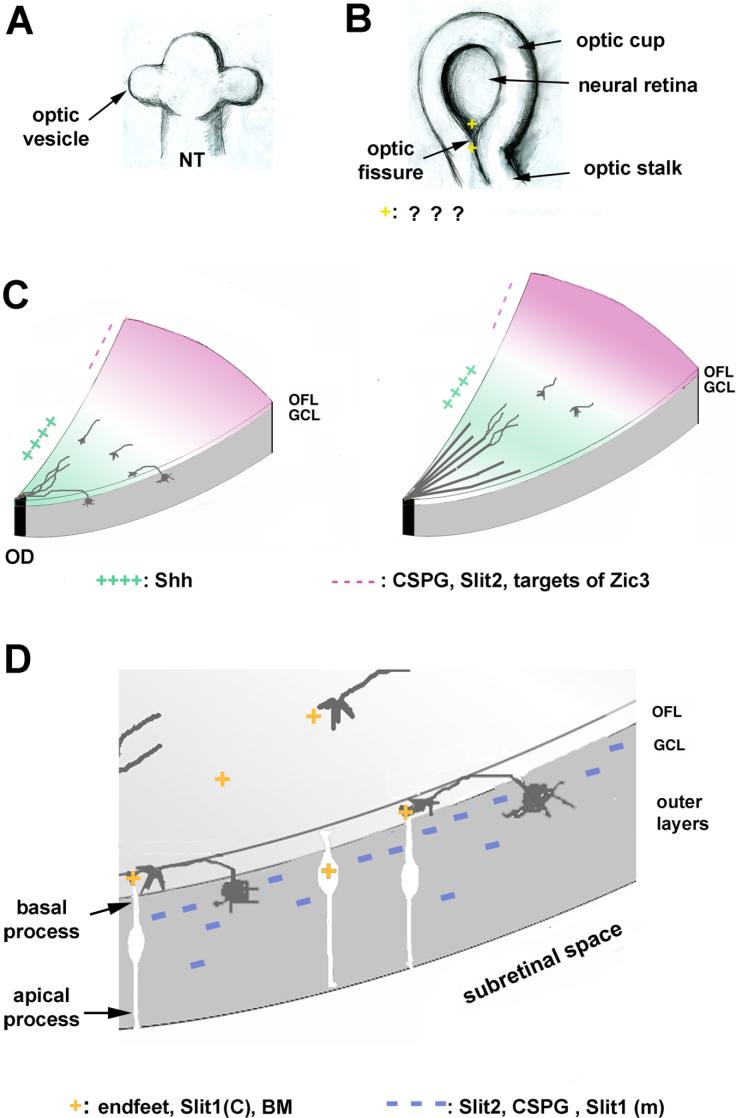
Proper wiring of the nervous system is of fundamental importance for the function of the nervous systems, and remains to be one of the fascinating and challenging areas in biology [101]. Due to its relative simplicity and accessibility, the vertebrate neural retina has been a favorite model system for both neurobiologists and developmental biologists. During embryonic development, bilateral evagination of the neural tube gives rise to the optic vesicle (Fig. 1A). Subsequent invagination of the optic vesicle results in the formation of a double-layered cup and the inner layer of the optic cup becomes the prospective neural retina (Fig. 1B). The process of invagination also forms a groove at the ventral side of the retina connecting with the optic stalk, called the “optic fissure”. Later, when the retinal tissues at both sides of the optic fissure expand and fuse, the first retinal ganglion cell (RGC) axons exit the eye through the optic fissure. In addition to the RGC axons, mesenchymal cells also migrate through the optic fissure into the eye cup to form the retinal artery. Surrounding the emerging blood vessels and the exiting axons, the “optic disc” is a circular structure that forms at the center of retina.
Figure 1.
Schematic diagrams of intraretinal axon pathfinding. Proper guidance of RGC axons inside the retina requires cooperation of a multitude of factors. A. Optic vesicles are derived from the evagination of the neural tube. NT: neural tube. B. Invagination of optic vesicles results in formation of the optic cup and also a ventral groove. The ventral groove is called the “optic fissure” that serves as the passage way for the exiting of the first axons and inward migration of the mesodermal cells for formation of retinal artery. The optic fissure fuses and the inner layer of the optic cup becomes the neural retina. Signals emitting from the optic fissure/disc may be required for guiding the early RGC axons to the optic fissure/disc. C. At later stages, when RGC axons are projected at a substantial distance away from the optic fissure/disc, a combination of positive guidance factors (shown in green) and negative factors (shown in pink) ensure the correct orientation of the RGC axons. Once the axons are oriented correctly, they can grow along with the other axons of the RGC cells at more central positions. OD: optic disc. D. Mechanisms are also present to restrict the RGC axonal growth within the OFL layer of the retina. The undifferentiated neuroepithelial cells are shown as white cells with bipolar processes, whereas the RGCs are shown as grey cells with their soma in the GCL. Glial/neuroepithelial endfeet and basal lamina (BM) provide positive/permissive substrates whereas Slit2 provides inhibitory signals to the RGC axons. Slit1 is a positive (or permissive) factor in chick (c), not in mouse (m).
Within the neural retina, multipotent progenitor cells differentiate into six major types of neurons and one type of glial cell [26,61,113]. The retinal ganglion cells are the first cell type to differentiate, and the differentiated ganglion cell soma reside in the ganglion cell layer (GCL) at the vitreal side of the retina. However, later in development, the GCL is not exclusively occupied by the ganglion cell soma. Approximately 50% of the cells in the GCL are displaced amacrine cells that synapse with the ganglion cells [23,52]. Shortly after the ganglion cells differentiate, the axons extend toward the vitreal surface and there, they turn and extend to form the optic fiber layer (OFL), along with the neuroepithelial endfoot processes (Fig. 1D) [37,46,84]. The optic fiber layer is thus very close to the vitreal surface, just below the inner limiting membrane, a thin basal lamina.
The retinal ganglion cells are the sole output neuron in the retina, relaying visual information by sending long axons to project to the brain. The long journey of the RGC axon pathfinding can be divided into the following segments. First, the axons from the RGCs distributed throughout the cup-shaped retina have to project toward the optic fissure/disc where they exit the eye. This precisely regulated process is called “intraretinal axon targeting” [30]. Next, after exiting the eye, the RGC axons arrive at the optic chiasm, where they interact with the scaffold of local cells in the midline and with axons from the contralateral eye. In mouse and human, a subset of the axons project to the contralateral side of the brain whereas the rest stay at the same side as the retina. In chick and fish, all RGC axons appear to cross to the contralateral side [75,83]. Finally, the axons reach the part of the brain, the optic tectum in non-mammalian vertebrates or the superior colliculus (SC) in mammals. Particular challenges that the RGC axons face are to establish specific connections in the tectum (or SC) to form a “topographic map”, so that the neighboring axons in the retina project onto the adjacent sites in the tectum (or SC). Remarkable progress has been made in recent years in understanding the molecular mechanisms involved in RGC axon pathfinding at the optic chiasm [63,64] and the establishment of the retinotectal map [29,58,66].
In this review article, I will focus on the first segment of the RGC axon navigation, the intraretinal axon guidance. There are approximately 50,000 RGCs in mouse, 1.5 million in human and 2.4 million in chick. The axons from the RGCs differentiated at different times and distributed throughout the retinal cup have to be guided to the optic disc. Intraretinal axon targeting thus serves as an excellent model system for studying how axons receive and integrate guidance information to navigate in three-dimensional space. This system can also be used for regeneration study in the hope of curing nerve damages occurred in injury or neurodegenerative diseases. A large number of molecules have been characterized to affect RGC axon growth in culture. However, the role of these molecules in intraretinal axon guidance requires additional consideration of proper spatiotemporal expression patterns and in vivo gain-of-function and loss-of-function experiments. I will thus place special emphasis on in vivo studies in the context of intraretinal axon pathfinding. Due to the limit of space, it is not possible to include all the relevant literature, and the readers are referred to several excellent reviews for detailed discussion of some of the earlier work [78,97,102].
2. Polarization of retinal ganglion cells and restricted growth of RGC axons in the OFL layer
2.1. Establishment of polarization of the RGCs
The ganglion cells are the first cells to differentiate inside the retina, which initiates shortly after the fusion of the optic fissure. Proliferating neuroepithelial cells inside the retina have both the apical (ventricular) and basal (vitreal) processes that span the full thickness of the retina (Fig. 1D). Starting at the early stage of differentiation, RGCs become polarized with axons oriented toward the basal (vitreal) side. The differentiating RGCs in the chick retina were observed to have a bipolar appearance by Ramón y Cajal, with a retracting apical process and an axon extending toward the basal side [86]. In mouse, it was suggested that RGC axons are derived from the basal process of the neuroepithelial precursor cells [44]. Basal emergence of RGC axons was also observed in Xenopus [46]. One of the important asymmetries of the retinal neuroepithelium is the presence of basal lamina at the vitreal side. Composed of laminin, nidogen, collagen IV, agrin, heparan sulfate proteoglycan (HSPG), condroitin sulfate proteoglycan (CSPG) and other unidentified components [97], the basal lamina has been shown to be an excellent supportive substratum for RGC axon growth [38,39].
Basal lamina is also crucial for polarization of the RGCs. Treatment of rat retinas with chondroitinase which cleaves the glycosaminoglycan chains from CSPGs, resulted in ectopic differentiation of the ganglion cells at the ventricular side of the retina, with disorganized axon projection [8]. Intriguingly, the polarity of RGC axonogenesis can also be completely reversed by exposing the ventricular surface of the organ-cultured rat retina to free chondroitin sulfate [10]. In contrast to the control retinas, all of the retinal ganglion cell bodies and axons were aberrantly localized to a new layer containing exogenous condroitin sulfate matrix at the ventricular margin, in spite of the apparently normal presence of basal lamina at the vitreal side. The mislocalized axons were not properly oriented toward the optic disc, but were morphologically indistinguishable from those that form normally during development. In chick retina, disruption of basal lamina by injection of collagenase led to retraction of the process (endfeet) of the neuroepithelial cells from the vitreal surface and caused disorganization of the ganglion cell layer [35]. In addition, RGC axons were no longer confined to the optic fiber layer and many appeared to orient toward the ventricular surface of the retina.
Recently, RGC differentiation and axonogenesis has been directly observed in zebrafish embryo, by using four-dimensional microscopy [119]. Shortly after the final cell division, the cell body of the RGC precursor cell is observed to move toward the basal side along the basal process, and the apical process begins to retract. After the onset of apical retraction but before its completion, the neurite emerges from the basal side and differentiates into an axon with the formation of a growth cone. While almost all axons formed at later stages arise from the basal processes of the precursor cells, half of the axons formed earlier (before 40 hpf) appeared to arise without visible basal processes. Furthermore, it was reported that components of the apical complex, such as Par-3, do not localize to the emerging axon, nor does the centrosome predict the site of axon emergence [119], differing from proposed models based on in vitro cell cultures. Morpholino knock-down of one of the slit family genes, slit1b, delayed retraction of the apical process and the migration of the nucleus to the basal side of the retina, but did not block extension of axons at the basal side. In the Slit1b knock-down samples, the apical processes were later retracted and a normal ganglion cell layer was formed. By mosaic analysis using zebrafish mutants, it was concluded that exposure to basal lamina is a critical cell extrinsic factor for RGC polarization [119].
2.2. Confined growth of the RGC axons in the optic fiber layer
After arriving at the optic fiber layer (OFL) at the vitreal side, RGC axons continue to grow toward the optic fissure/disc within the OFL (Fig. 1C, D). What is the mechanism for restricting the growth of the RGC axons in the OFL but not the outer layers of the retina? By electron micrographs, the RGC axons were observed to grow along the basally oriented processes (endfeet) of the neuroepithelial, or later, the müller glial cells [37,46,84]. By culturing retinal explants onto cryosections of the retina [96], the restrictive growth of the RGC axons in the OFL was shown not due to physical hindrance, or through axon fasciculation. Rather, the inner limiting membrane containing glial endfeet appears to be an excellent substratum, and the layers outside of the RGC and OFL appear inhibitory to RGC axon outgrowth [96]. In addition, the embryonic lens appears to produce strong repulsive activity to the RGC axons [76], possibly inhibiting the RGC axons from targeting toward the lens.
Two recent studies have shed some light on the molecular mechanisms of this process. In chick, Slit1 is expressed in a subset of cells in the ganglion cell layer distinct from the homeobox gene Irx4-expressing cells in the same layer [54]. Overexpression of Irx4 decreased, whereas expression of a dominant negative Irx4 construct increased, Slit1 expression [54], suggesting that Irx4 is an important regulator of slit1 expression. Slit proteins are a family of evolutionarily conserved proteins playing many important roles in embryonic development including axon guidance, cell migration and organogenesis [12,43,111]. By in ovo electroporation, the RGC axons were observed to project preferentially over the cells expressing Slit1, suggesting that Slit1 is a positive (or permissive) factor to the RGC axons (Fig. 1D) [54]. Decreasing the level of Slit1 resulted in abnormal RGC axon trajectory, excessive fasciculation, and a loss of the “honeycomb” projection pattern. Because the RGC axons tend to grow over the Slit1-expressing cells, it was postulated that the Slit1-expressing cells provide a shield from inhibitory activities emanating from the outer layer of the retina, a “stepping stone phenomenon” (Fig. 1D). As the Slit1 expression was only known at the messenger RNA level, not at the protein level, it is unclear whether Slit1 contributes to the growth promoting activity of the neuroepithelial/glial endfeet. Of note, despite the loss of “honeycomb” trajectory, the RGC axons still project toward the optic disc, suggesting that Slit1 is not a main guidance factor directing the RGC axons toward the optic disc. It is currently unclear as to why Slit1 appears to act positively to the RGC axons inside the retina, differing from its negative effect by in vitro assays [82,103]. It is notable that Slit2 has also been shown to stimulate axon elongation and branching [107]. A large number of factors, including the presence of ECM molecules, receptor combinations, and levels of second messengers in the cytosol of the neurons such as cAMP, cGMP, and Ca2+, have been shown to modulate the effects of the guidance molecules, converting between repulsion and attraction [42,47,48,73,95].
In another study, retinal ganglion axon pathfinding was examined in the Slit1 and Slit2-deficient mouse lines. Because the mouse lines were generated by replacing the second leucine rich repeat region with a cassette containing a stop codon and IRES-tauGFP [82], the expression of Slit1 and Slit2 genes was shown in the RGCs by using tau-GFP in the Slit1+/− and Slit2+/− embryos [103]. In wild-type mouse retinas, the RGC axons are restricted to the OFL at the inner surface of the retina [103]. In mice lacking slit1 alone, RGC axons remained restricted to the OFL. However, the number of misrouted RGC axon bundles in the outer retina was increased to 5 in the slit2-deficient mice, and to over 30 in slit1/2 double mutants [103]. The misrouted axons originated more in the ventral than dorsal retina. Thus, Slits appear to play an important role in restricting the RGC axons from projecting into the outer layers of the retina (Fig. 1D). Remarkably, despite being displaced from the OFL, the mislocalized axons were still directed toward the optic disc through the outer retina, suggesting that other cues are guiding the RGC axons toward the optic disc.
In summary, a number of factors are involved in keeping the growth of RGC axons within the OFL. Potential redundancy may explain why not all the RGC axons project to the outer layer of the retina when one factor is removed. As morpholino knock-down of slit1b caused a delay in retraction of the apical processes of the retinal ganglion cells in zebrafish, it remains to be shown whether the mislocalization of axons in the Slit-deficient mice was resulted from defects in initial polarization or subsequent straying from the OFL. The roles of the slit proteins also appear somewhat different among species. While slit1 is a positive factor in chick, its role in mouse intraretinal axon guidance is rather limited. As slit1 appears to be particularly involved in the “honeycomb” growth pattern of axons in chick, a transitory wavy pattern also reported in rat embryos [11,92] (see section 3.1), it is tempting to speculate that the “honeycomb” growth patterns may be especially important for retinas undergoing rapid growth during axonogenesis. The chick retina doubles its size, from ∼3 mm at E6 to ∼6 mm radius at E8. Wavy axons may accommodate better to the rapid growth rate than the straight ones. In addition, the transitional “honeycomb” pattern may help distribute the RGC axons more evenly in the OFL. It is also notable that the expression patterns of the slit genes are variable in different species. In mouse, slit1 and slit2 are shown to be expressed by the RGCs [28,74,88,115]. In chick, slit1 appears to be expressed by the displaced amacrine cells in RGC layer [54], whereas slit2 is expressed in a subset of cells in the inner nuclear layer (Jin and Bao, unpulished result). In zebrafish, slit1b is reported to be expressed in inner nuclear layer while slit1a is expressed at a very low level in the ganglion cell layer [49]. As these genes are highly similar in sequence, different expression patterns may render different roles to the proteins, like other reinventions in the evolution of the visual system.
3. Guidance of the RGC axons to the optic fissure/disc
3.1. Centripetal projection of the RGC axons
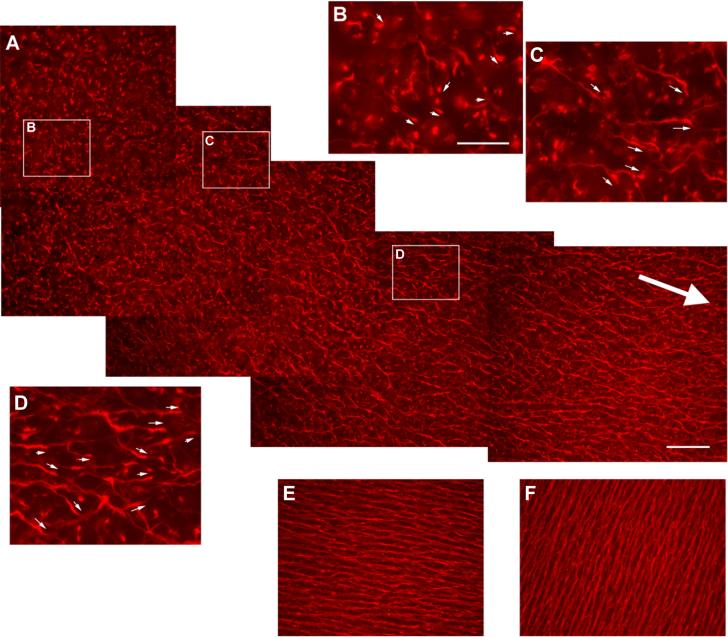
The differentiation of RGCs proceeds in a wave-like fashion, initiating at the vicinity of the optic fissure, followed by cells at more peripheral positions. The first RGC axons are thus in close proximity to the optic fissure, in contrast to the later differentiated RGC axons which are farther away and have to travel a greater distance (Fig. 1B). The large size of the chick retina allows examination of RGC axons on flat-mounted retinas at early stages (Fig. 2). A range of axonal growth patterns can be observed across the radius of the retina from E6 to E9, with immature patterns at the retinal periphery, a mature bundled pattern at the retinal center and a “honeycomb-like” appearance in between (Fig. 2). The newly emerged axons at the retinal periphery are short and have a degree of randomness in orientation (Fig. 2B). However, the axons quickly become oriented toward the direction of the optic disc, while remaining unfasciculated (Fig. 2C), similar as previously described in rat retina [11]. At more central positions, the axons appear to grow along briefly in groups of 2 or 3 axons and then pull away, resulting in wavy patterns resembling “honeycomb” (Fig. 2D). More mature axons close to the optic disc have already been growing for a few days (Fig. 2E). These axons start to form bundles and are also relatively straight like stretched “honeycombs”. Thus, except for the earliest outgrowth, the newly extended axons are oriented toward the optic fissure/disc, as reported previously [11,31,46], even though they have not yet fasciculated with the axons at the more central positions.
Figure 2.
The trajectory of newly extended RGC axons at the peripheral chick retina. Retinas from E7 chick embryos were flat-mounted and the RGC axons were immunostained by an antibody specific for neurofilament, 270.7. A. Assembled photos taken at the dorsal peripheral retina are shown. The large arrow indicates the direction to the optic disc. Enlarged images are also shown for selected areas (B, C, D). B. In the most peripheral areas, the newly emerged axons exhibit a certain degree of randomness in orientation (arrows indicate the directions of axon projection). C. At slightly more central positions, the axons are a little longer, still unfasciculated, but with a clear orientation toward the optic disc (arrows). D. Further toward the retinal center, the RGC axons are now well oriented toward the optic disc (arrows), and grow along together briefly then apart, giving rise to the “honeycomb” appearance. E. At approximately 1/3 of the radius distance to the optic disc, the RGC axons appear straighter, like stretched “honeycomb”. F. In close proximity to the optic disc, axons are more heavily fasciculated and project straight toward the optic disc. Scale bar in A (same scale in E and F): 50 μm; scale bar in B (same for C and D): 20 μm.
3.2. The role of the optic fissure/disc in intraretinal axon guidance
So what are the signals that guide the RGC axons to the optic fissure, and later, the optic disc? As early as 1892, Ramón y Cajal proposed that long-range chemoattraction from the optic disc may play a central role in this process [85]. The experiments were later performed in Xenopus, by rotating the eye 180° before the RGC axons have extended. Axons appeared to be targeted to the ectopic optic fissure through abnormal routes and were able to exit from the site, suggesting a central role of the optic fissure in the guidance and the exit of the RGC axons [32]. As the earliest RGC axons project to the optic fissure at a close range, the optic fissure is a likely source for providing guidance factors for these early RGC axons (Fig. 1B). However, definitive evidence is still lacking to implicate a particular factor emanating from the optic fissure in guiding the RGC axons.
One of the challenges in identifying the guidance factors emitting from the optic fissure is to distinguish their effects on RGC axon guidance from those in formation or patterning of the optic disc. Recently, a member of the transforming growth factor beta (TGFβ) superfamily of secreted proteins, bone morphogenic protein 7 (BMP7), has been shown to be required for early development of the optic fissure [69]. Expression of BMP7 is dynamic in the developing eye, including specific segments of optic vesicles and optic cup, in the optic stalk, prospective retinal pigment epithelium and surrounding extraocular mesenchyme [25,69]. BMP7-null embryos exhibit microphthalmia with various degrees of penetrance of the anophthalmic phenotype, as a result of defective lens induction during early eye development [24,62,109]. Initiation of optic disc development has been shown defective in microphthalmic Bmp7-deficient embryos characterized by diminished expression of various markers of the optic disc. Failure in optic disc formation in microphthalmic Bmp7 null embryos was shown to be associated with optic nerve aplasia, intraretinal misrouting of RGC axons and absence of the hyaloid artery. The RGC axons projected aberrantly at the vitreal suface or in the subretinal space and failed to leave the eye [69]. The defect in intraretinal axon guidance was suggested to be a result of the loss of the optic disc and the guidance factors derived from it, rather than deficiency of BMP7 as a guidance molecule. Addition of BMP7 protein to retinal explant culture did not alter neurite outgrowth or directionality of RGC axon growth [69]. In the future, it will be interesting to identify the signals in the optic disc that guide the RGC axons and are responsible for the intraretinal axon targeting defects observed in the BMP7-null mice.
Detailed analysis is thus necessary to determine the primary cause for the RGC axon pathfinding phenotypes in animals with genetic mutations or gene perturbations. In zebrafish, the genes encoding stromal cell-derived factor (SDF-1), sdf-1a and sdf-1b, are expressed in the proximal optic stalk adjacent to the retina, whereas the gene encoding the SDF-1 receptor cxcr4b is expressed by the retinal ganglion cells during the period of axonogenesis [59]. Injection of antisense morpholino oligos targeted at the SDF1 and CXCR4 proteins resulted in misrouting of RGC axons inside the retina. Supporting the results of knock-down experiments, the RGC axons in the cxcr4b mutant embryos (odyt26035) also appeared misguided and formed abnormal tracts inside the retina [59]. It awaits further study to determine if the primary defect in the mutant is due to RGC axon misguidance, not a failure of axon exit. RGC axons that failed to exit at the optic disc can overshoot into the opposite side of the retina in abnormal tracts, as demonstrated in a number of studies. In mammals, the role of SDF-1/CXCR4 signaling in intraretinal axon pathfinding is less clear. SDF-1 has been shown to cause repulsive turning of the growth cones of rat cerebellar neurons and is required for normal axon pathfinding within the spinal cord by modulation of the effectiveness of multiple axon repellents [16,112]. However, SDF-1 does not have apparent attractive or repulsive effect on RGC or dorsal root ganglion (DRG) axons by itself in vitro [16]. SDF-1 promotes the survival of the cultured embryonic RGCs, and mouse embryos lacking the CXCR4 receptor have a reduced number of RGCs [15].
3.3. Factors expressed inside the retina along with the RGC differentiation wave in guiding the RGC axons
It has been established that the growth of RGC axons is not random, but mostly oriented toward the optic fissure/disc [11,30,46]. Because the RGCs are distributed throughout the surface of the retina, most of the RGCs are localized at quite a distance away from the optic disc when the axons are first elongating. This is particularly evident in animals with large eye sizes, such as chick and large mammals. For chick, when the RGCs at E8 or E9 peripheral retina start to extend their axons, the axons are 4−7 mm away from the optic disc. It is difficult thus to envision that a secreted molecule(s) from the optic disc can exert an effect at such a vast distance. A limited role of the optic disc in guiding the later RGC axons has been suggested by intraretinal grafting experiments in chick [33], and optic disc removal and mechanical deflection of RGC axons in rat [11]. In chick, removal or grafting of an optic disc to an ectopic site in a host retina had no overall effect on the navigation of the RGC axons. However, the host axons that encountered the grafted optic disc exited the optic fiber layer to the ventricular surface [33]. Similarly in rat, removal of the optic disc did not affect peripheral RGC axons, and axons mechanically deflected grew toward the retinal periphery appearing no different in growth cone morphology or growth velocities compared with the axons growing in centrally oriented direction, until reaching a peripheral zone with high levels of chondroitin sulfate proteoglycans [11]. In addition, RGC axons that have failed to exit at the optic disc in some mouse mutants or animals injected with blocking antibodies have been shown to grow in the direction toward the retinal periphery for quite a distance [1,22]. These results suggest that the local environment rather than long range cues from the optic disc plays a central role in later, more peripheral RGC axon pathfinding inside the retina.
One possible scenario is that later differentiated RGCs do not need specific guidance factors, but grow along with the more centrally-positioned RGC axons. However, shortly after the initial outgrowth, the chick RGC axons quickly become oriented toward the optic disc, similarly as observed in Xenopus and rat [11,46], without apparent fasciculation with other axons (Fig. 2). Subsequently, the axons go through a period of “honeycomb” pattern of growth, in which they grow along briefly and then apart, but maintaining the centrally oriented direction (Fig. 2D). Therefore, even though that fasciculation contributes to overall guidance of RGC axons inside the retina (see Section 3), other factors are required for orienting the newly extended axons. Because the RGC differentiation wave spreads from the optic disc toward the retinal periphery, the localization of the candidate guidance factors likely moves with the differentiation wave to ensure that they are in close range to the newly emerged RGC axons.
An earlier study showed that the expression pattern of chondroitin sulfate proteoglycans (CSPGs) moves with the differentiation wave. Expressed in a graded pattern high in the periphery and low in the retinal center in rat, the expression domain of CSPGs recedes over the course of retinal differentiation [8]. CSPGs are generally inhibitory and form barriers to the growth of axons including the RGC axons [14,41,93,94]. Enzymatic treatment with chondroitin sulfate ABC lyase resulted in ectopic and premature differentiation of the ganglion cells and abnormal orientation of RGC axons (section 2.1). Thus, the graded expression of CSPGs is important for controlling differentiation and polarization of ganglion cells and proper orientation of axon projection. The role of CSPGs in intraretinal axon pathfinding in chick, however, is less clear. In culture, CSPGs can potently inhibit chick RGC axon growth [41,94]. However, the expression pattern of CSPGs in chick retina does not show a perfect inverse correlation with the RGC differentiation wave [65]; the expression is peripheral to the differentiating RGCs in the ventral retina, but not in the dorsal retina [94].
To search for molecules that are involved in guiding the RGC axons inside the retina, a small-scale screen was carried out aimed at genes with expression patterns that move with the differentiation wave inside the retina. By using available cDNAs or cDNAs cloned by RT-PCRs, the expression of genes including those in the families of ephrins, semaphorins, wnts, hedgehog, and various transcription factors were examined by in situ hybridization [2,53,57,117] (and data not shown). Two genes were found to have graded expression patterns along the radial axis, Zic3 and Sonic Hedgehog (Shh). Zic3 encodes a zinc-finger transcription factor, and is expressed in a graded pattern high in the periphery and low in the center of the retina at the stage of active axon growth [117]. The graded expression of Zic3 recedes towards the periphery with the wave front of ganglion cell differentiation [117] (Fig. 1C). Misexpression of Zic3 by using retrovirus in chick retina resulted in abnormalities in RGC axon projection. Some RGC axons appeared to be blocked from projecting toward the optic disc while some others misrouted through the outer layers of the retina to the subretinal space in between the neural retina and retinal pigmented epithelium. However, no gross change in lamination or differentiation of retinal cell types was observed. By using stripe assay, overexpression of Zic3 was shown to induce the expression of an unknown inhibitory factor to the RGC axons [117]. Periphery-high and center-low distribution of Zic3 is thus important for inhibiting the RGC axons from projecting toward the periphery by regulating the expression of a negative factor.
Another gene, Sonic hedgehog (Shh), was found to be expressed in a complenentary pattern to that of the Zic3, high in the center and low in the periphery of the retina during the period of active axonogenesis. The expression domain of Shh also moves as a broad band from the center of the retina towards the periphery [57] (Fig. 1C). At E9, the expression of Shh was diminished in the central region of the retina and was detected only at the retinal periphery. The Shh expression appeared at a relatively central position to the differentiation wave front of the RGC cells, consistent with the reports that Shh is expressed in the nascent RGCs in fish, chick and mouse [72,108,118]. Shh encodes a secreted glycoprotein, which has been known to play many important roles in embryonic development, including tissue patterning, cell differentiation and organogenesis [45,51,67]. In contrast to the indirect role of Zic3 in regulation of RGC axon projection, Shh likely acts on the growth cones of RGC axons directly. It acts as a chemoattractant that collaborates with netrin-1 in midline axon guidance [17], and acts as a negative factor to direct RGC axonal growth at the diencephalic ventral midline [104]. In both cases, the effect of Shh on axon pathfinding has been shown not through re-patterning of the target tissues or gross alteration of neural differentiation, but through a direct effect of Shh on the growth cones of the targeted neurons.
When a range of concentrations of Shh protein were tested on chick RGC axons, Shh was shown to promote RGC axon growth at lower concentrations (0.3−0.6 μg/ml), and inhibit RGC axon growth at higher concentrations (2.5−3.0 μg/ml) [57]. Although it is difficult to determine the local Shh protein concentration that the RGC axons encounter inside the retina, the in situ hybridization signal detected with the Shh RNA probe was at least a few fold lower inside the retina than that in the floor plate. The positive effect of Shh on RGC axon growth at lower concentrations could be detected within 15−30 minutes after protein addition, suggesting that it is likely mediated by a transcription-independent pathway. In ovo overexpression of Shh protein caused excessive turning of the RGC axons and completely abolished their oriented growth, without apparently affecting the overall number of RGC axons [57]. In some areas, axons crossed over abnormally resembling ectopic “optic discs”, but did not appear to stray out of the layer. Similar phenotypes were observed by injecting an inhibitor of Shh signaling, cyclopamine, right before the RGC axon projection initiated, but not two days prior. Only small areas of RGC axons were affected in the cyclopamine-injected retinas, due to the fact that it was difficult to inject at later stages and cyclopamine leaked out of the retina shortly after injection. Thus, in chick, the effect of Shh on intraretinal axon guidance is likely mediated through its local action to the RGC axons, not through the effect of re-patterning.
The Shh-null mouse embryos had gross early patterning defects including cyclopia [18], making it difficult to analyze the later RGC axonal phenotypes. Conditional ablation of Shh in retinal precursor cells prior to RGC differentiation was achieved by using the Cre recombinase under the control of the Thy-1.2 promoter [20]. RGC axons of the ThyCreShhn/c mouse embyos were mis-targeted to sub-retinal spaces in some regions of the retina and at the optic disc. In some severe cases, misguidance of RGC axon resulted in appearance of multiple optic discs inside the retina [20]. Because the optic disc astrocyte precursor cells are absent in the conditional Shh mutant mice, it remains unclear whether the misguidance phenotype is due to defective development of the optic disc or loss of the local guidance by Shh, or both.
The guidance factors for directing RGC axon projection may even be derived from the lens, as the lens is juxtaposed to the peripheral retina during development. A recent analysis of the Slit1- and Slit2- deficient mice indicate that Slit 2 is also required for controlling the initial outgrowth of the peripheral dorsal RGC axons [103]. In the Slit2- or Slit1/2-deficient mice, the outgrowth of the RGC axons originating in most of the peripheral dorsal retina had an abnormal trajectory, which appeared to be corrected over the course of development. Because the phenotype was the same in the Slit2 and Slit1/2-deficient mice, this effect was attributed to Slit2 alone. By in vitro co-culture experiments, the lens from the wild type mice potently inhibited the outgrowth of the RGC axons. The lens from the Slit1/2-deficient mice, however, had reduced inhibition to the RGC axon growth [103], suggesting that the Slit2 secreted from the lens may be involved in inhibition of the dorsal RGC axons from projecting toward the periphery.
4. The role of axon fasciculation in intraretinal pathfinding
Fasciculation is an important mechanism for axons to project to their targets by forming bundles with pioneer axons [105]. Once the initial outgrowth of RGC axons is oriented correctly, it is possible to fasciculate with the axons from the RGCs at more central locations. Indeed, the RGC axons are fasciculated within the OFL inside the retina. However, in rat and chick embryos, the RGC axons form bundles only at a relatively mature stage close to the optic disc, not at initial outgrowth or the intermediate “honeycomb-like” stage (fig. 2) [11]. Due to the difficulty in preparing flat-mounted early retinal samples in species with small eye sizes, it remains unclear whether similar transition of RGC axon fasciculation patterns exists in other species.
Fasciculation is mediated by adhesive interactions among axons growing on less than optimal substrata. Therefore, perturbation of fasciculation can be achieved by manipulations affecting adhesion among axons and/or the repulsiveness of the substrate. A variety of molecules have been implicated in modulation of fasciculation of the RGC axons, including classic cell and substrate adhesion molecules (CAMs and SAMs, respectively) [105,106]. Because many of the CAMs are expressed along the axons and disruption of their function causes defects in axonal fasciculation [13,55], these molecules are discussed in this section. However, some of the CAMs have additional roles such as modulating the response of the RGC axons to other guidance factors [98]. Many CAMs are members of immunoglobulin super family which is comprised of a diverse group of molecules that contain immunoglobulin (Ig) domains [19]. The IgCAMs can be categorized into subfamilies based on the number of immunoglobulin domains and the number of fibronectin type III-like repeats. The prototype of the IgCAMs, NCAM, is composed of five immunoglobulin domains followed by two fibronectin type III repeats. In rat, treatment of anti-NCAM Fab fragments caused subsets of growth cones to speed up initially, but then stall and eventually halt [9]. However, the direction of the growth cones and the intraretinal projection pattern were not affected by treatment of the blocking antibody [9,36].
L1 is a representative member of the IgCAM subfamily which contains six Ig domains and three to five fibronectin type III repeats [19]. Other members of this subfamily include Nr-CAM, Ng-CAM, neurofascin, and neuroglian. In addition to homophilic interaction, L1 also binds heterophilically to other proteins such as DM1/GRASP, axonin1/TAG-1, contactin/F3/F11 [55], and integrins [68,114]. In rat, treatment of anti-L1 Fab fragments severely disrupted radial orientation of the RGC axon and the rate of outgrowth [9]. The axons were initially stalled, then changed orientation, and then elongated with a faster rate than that of the control axons. In goldfish, injection of blocking antibodies against the L1 family of cell adhesion molecule, E587, disrupted RGC axon fascicles, but not the oriented growth toward the optic disc [5]. Another molecule in the subfamily, NrCAM, has been shown to be a key component in RGC axon pathfinding in chick [116]. NrCAM combined with laminin provides a more supportive substratum for the growth of the RGC axons than laminin alone. Inhibition of NrCAM function by using function-blocking antibodies changed the dynamics of the RGC axon growth, increasing the length of pausing and frequency of retraction. Inhibition of NrCAM also resulted in defective RGC axon exit at the optic disc that the axons overshot into the opposite side of the retina [116].
DM-GRASP/SC1/BEN is an integral membrane protein that belongs to another subfamily of IgCAMs containing 5 immunoglobulin domains. It mediates homophilic interaction as well as heterophilic interactions with NgCAM, CD6 and high density lipoprotein [21,27,99]. Injection of blocking antibodies to the fish homologue of DM-GRASP/SC1/BEN, neurolin, disturbed axon fasciculation as well as caused aberrant pathways of RGC axons in the dorsal retina [5,79]. In the eyes of growing fish injected repeatedly with neurolin Fab fragments, small groups of axons in the dorsal retina dissociated from their fascicle, and turned 180° or in full circles close to the optic disc. These phenotypes suggest that the function of neurolin may be required for the growth of axons near the optic disc. In chick, DM-GRASP enhances RGC axon extension on laminin substrate [1]. Inhibition of DM-GRASP resulted in overshooting of axons into the opposite side of the retina, indicating a failure of RGC axon in exiting the optic disc [1]. RGC axon extension and navigation from the peripheral retina towards the optic fissure, however, was reported not to be affected. Mice lacking the DM-GRASP/BEN gene, also exhibited defects in fasciculation of retinal ganglion cell and motor axons [110]. The RGC axons in BEN−/− mice formed relatively loose and broader bundles compared to the control animals. Despite abnormal fasciculation, the RGC axons projected and exited the eye normally, with only occasional misdirection.
In mouse, the fasciculation patterns are similar for the RGC axon bundles in all four quadrants of the retina. In chicken, however, the RGC axons form thicker bundles in the ventral than in the dorsal retinas [50,70]. This asymmetry in fasciculation pattern was shown to be pre-patterned by a homeodomain transcription factor cVax/mVax2. cVax expression is restricted to the ventral eye cup and has been shown to be important for dorsal/ventral patterning in the retina [89]. Inactivation of mVax2 in mice resulted in an incomplete closure of the optic fissure and abnormal retinocollicular projections and projections at the chiasm [3,71]. Ectopic expression of cVax in chick caused dorsal RGC axons to switch to a ventral retinal fasciculation pattern characterized by thicker fascicles [70]. Because the Eph receptors EphB2 and EphB3 are known downstream targets of cVax, the authors tested the contribution of these molecules to RGC axon fasciculation by ectopically expressing ephrin-B2 [70]. Overepxression of ephrin-B2 resulted in disruption of uniform thickness of RGC axon fascicles suggesting that the ephrin/Eph molecules may be involved in the differential fasciculation patterns of the RGC axons.
5. Exit of the RGC axons through the optic disc
RGC axons converge at the optic disc and make a sharp turn to exit the eye [31,34,91]. A number of mouse mutants have been characterized that affect the guidance of RGC axons at the optic disc. A gene encoding a secreted guidance factor, netrin-1, has been shown to be expressed specifically in the neuroepethelial cells at the optic disc surrounding the exiting RGC axons, while its receptor, DCC (deleted in colorectal cancer), is expressed on the RGC axons [22]. First identified by its activity in promotion of commissural axon projection and turning [56,90], netrin-1 also induced the outgrowth of RGC axons by interacting with DCC by in vitro culture assays [22]. Mouse mutants deficient of netrin-1 or DCC exhibit very similar phenotypes in eye development and RGC axon projection. In netrin-1-deficient or DCC-deficient mouse embryos, hypopigmented streaks were present in the retinal pigment epithelium, and the optic disc size was reduced, despite overall normal size and shape of the eye. The number of RGC axons was decreased in the mutant optic nerve at the early stage of optic nerve development, suggesting a primary defect in RGC axon exit from the retina [22]. By DiI labeling, the RGC axons appeared to project normally to the vicinity of the disc. However, many labeled axons were splayed out at the optic disc and failed to exit into the optic nerve. Axon bundles that failed to exit at the optic disc appeared to grow ectopically into other regions of the mutant eye, through the outer layers of the retina or the subretinal space [22]. These results indicate that netrin-1/DCC signaling is required at the disc for proper exit of RGC axons from the eye.
Semaphorin5A (Sema5A), a transmembrane protein of the semaphorin family. is also expressed in the developing optic disc neuroepithelial cells and along the developing optic nerve [77]. Semaphorins comprise one of the largest families of axon guidance cues and have broad effects in axon guidance, cell migration, and morphogenesis [81,87]. Sema5A was shown to inhibit the growth of RGC axons by itself and in combination with netrin, laminin, or L1 [77], suggesting that it likely acts as a negative factor to the RGC axons along the visual pathway. Perturbation of Sema5A function by administering a function-blocking antibody resulted in some axons that strayed away from the main retinal pathway. Some axons left the optic disc and projected erroneously inside for short distances within the retina, whereas some strayed from the main optic nerve bundle.
The Eph family proteins are a family of receptor tyrosine kinases that interact with membrane-anchored ligands, called ephrins (reviewed in [80]). Eph/ephrin signaling is involved in multiple axon pathfinding processes, particularly in the establishment of retinotectal mapping [29,66]. EphB2 and EphB3 are expressed uniformly along the dorsal-ventral axis early during development but EphB2 expression becomes high in ventral and low in dorsal retina at later stages [6]. In mutant mice lacking both the EphB2 and EphB3 proteins, RGC axons originating from the dorsal retina failed to exit at the optic disc and abnormally grew into the opposite half of the retina. The mutant EphB2 lacking the kinase domain rescued the pathfinding defect in the dorsal RGC axons suggesting that the role of EphB2 in intraretinal targeting is not dependent on the kinase domain [6]. The extracellular domains of the EphB receptors were subsequently shown to act directly as inhibitory factors to the RGC axons, distinct from their conventional role as receptors for the ephrin protein ligands [7]. Based on these observations, a model was proposed that high levels of EphB expression in the ventral retina ensure precise targeting of the RGC axons at the exit point by helping to maintain tight fasciculation of the axons originating from the dorsal retina and preventing the axons from growing into the ventral side [7]. A similar phenotype, but only in the axons arising from the ventral side of the retina, has been described in the mouse mutant deficient of BMP receptor 1b [60]. The RGC axons originating from the ventral retina made abrupt turns at the optic disc and failed to enter the optic disc. BmprIb is expressed exclusively in the ventral retina. However, the axonal phenotype of the BmprIb–deficient mouse mutants was not attributed to gross alterations in dorsal-ventral patterning. Because the expression of four genes known to be involved in axon guidance, ephrinA2, Brn 3b, netrin-1 and Zic2, was reduced in the BmprIb−/− mice, BmprIb appears to function in a specific pathway to regulate axon guidance molecules for ventral RGC axon targeting.
The response of the RGC axons to netrin-1 and EphB molecules can also be modulated by laminin and L1. Laminin is localized in the OFL and optic disc region [48] and L1 is present on RGC axons [4,40]. In Xenopus, laminin-1 or a soluble peptide fragment of laminin-1, YIGSR, converts netrin-mediated attraction into repulsion [48]. Application of YIGSR to the developing retina resulted in failure of RGC axons to converge into a single fascicle at the optic nerve head. Axon trajectories appeared disorganized near the optic disc and some axons failed to exit the eye [48]. In another study, the response of RGC axons to the extracellular domains of EphB was shown to be modulated by laminin and L1. Extracellular domains of EphB induce a typical growth cone collapse to the RGC axons on laminin alone. In the presence of both laminin and L1, however, EphB triggers a unique inhibitory pause, with normal growth cone morphology and filopodial activity [98]. This growth cone pause is reminiscent of the in vivo observation that retinal growth cones pause but maintain prominent filopodia and a complex morphology when approaching the optic disc [11,46]. The optic disc is a key juncture where axons have to pause and make a sharp turn in order to exit the eye. These studies reveal a molecular paradigm involving multiple factors and complex regulatory relationships to ensure the precise exit of the RGC axons.
6. Summary and perspectives
In this review, we summarized some recent progress toward the understanding of the mechanisms underlying the RGC axon pathfinding inside the retina. Due to easy accessibility of the visual system, the RGC axon has long been a great culture model for studying axon growth and guidance. With the recent development in genetic techniques in mouse and zebrafish, combined with local perturbation techniques by microinjection, a molecular paradigm starts to emerge that ensures precise guidance of the RGC axons inside the three-dimensional space and proper exit from the eye. Molecules that have been found to play important roles in the spinal axon pathfinding turn out to be the key players in the intraretinal guidance as well, including members from the families of netrin, hedgehog, and Slits. Additionally, multiple members of the IgCAMs, semaphorins, ephrin/Eph, and proteoglycans are also involved. In the future, it will be interesting to study the cellular and signaling mechanisms by which the axons can sense and integrate multiple signals that are present in the environment. Elucidation of molecular mechanisms of RGC axon guidance will lead to a better understanding of the causes of congenital anomalies of the visual system such as optic nerve aplasia and optic nerve hypoplasia [78,100]. Increasingly, the RGC axons have been used as a model system for studying the mechanisms involved in axon degeneration and regeneration. A comparison of expression of guidance cues and receptors of the adult retina with that of the embryonic visual system may reveal the molecular basis for axonal plasticity. With the accessibility and powerful experimental approaches available to the visual system, a future of vision restoration to correct congenital defects and degenerative diseases may be in view.
Acknowledgement
We thank Dr. Virginia Lee for generously providing anti-neurofilament antibody, 270.7, Jun Jiang for technical assistance, and Adrianne Kolpak for critical reading of the manuscript. The work in Z. Z. Bao's laboratory is supported by National Institutes of Health (National Eye Institute), American Heart Association, Worcester Foundation for Biomedical Sciences and Hood Foundation.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Avci HX, Zelina P, Thelen K, Pollerberg GE. Role of cell adhesion molecule DM-GRASP in growth and orientation of retinal ganglion cell axons. Dev Biol. 2004;271:291–305. doi: 10.1016/j.ydbio.2004.03.035. [DOI] [PubMed] [Google Scholar]
- 2.Bao ZZ, Jin Z. Sema3D and Sema7A have distinct expression patterns in chick embryonic development. Dev Dyn. 2006;235:2282–2289. doi: 10.1002/dvdy.20882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Barbieri AM, Broccoli V, Bovolenta P, Alfano G, Marchitiello A, Mocchetti C, Crippa L, Bulfone A, Marigo V, Ballabio A, Banfi S. Vax2 inactivation in mouse determines alteration of the eye dorsal-ventral axis, misrouting of the optic fibres and eye coloboma. Development. 2002;129:805–813. doi: 10.1242/dev.129.3.805. [DOI] [PubMed] [Google Scholar]
- 4.Bartsch U, Kirchhoff F, Schachner M. Immunohistological localization of the adhesion molecules L1, N-CAM, and MAG in the developing and adult optic nerve of mice. J Comp Neurol. 1989;284:451–462. doi: 10.1002/cne.902840310. [DOI] [PubMed] [Google Scholar]
- 5.Bastmeyer M, Ott H, Leppert CA, Stuermer CA. Fish E587 glycoprotein, a member of the L1 family of cell adhesion molecules, participates in axonal fasciculation and the age-related order of ganglion cell axons in the goldfish retina. J Cell Biol. 1995;130:969–976. doi: 10.1083/jcb.130.4.969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Birgbauer E, Cowan CA, Sretavan DW, Henkemeyer M. Kinase independent function of EphB receptors in retinal axon pathfinding to the optic disc from dorsal but not ventral retina. Development - Supplement. 2000;127:1231–1241. doi: 10.1242/dev.127.6.1231. [DOI] [PubMed] [Google Scholar]
- 7.Birgbauer E, Oster SF, Severin CG, Sretavan DW. Retinal axon growth cones respond to EphB extracellular domains as inhibitory axon guidance cues. Development. 2001;128:3041–3048. doi: 10.1242/dev.128.15.3041. [DOI] [PubMed] [Google Scholar]
- 8.Brittis PA, Canning DR, Silver J. Chondroitin sulfate as a regulator of neuronal patterning in the retina. Science. 1992;255:733–736. doi: 10.1126/science.1738848. [DOI] [PubMed] [Google Scholar]
- 9.Brittis PA, Lemmon V, Rutishauser U, Silver J. Unique changes of ganglion cell growth cone behavior following cell adhesion molecule perturbations: a time-lapse study of the living retina. Molecular & Cellular Neurosciences. 1995;6:433–449. doi: 10.1006/mcne.1995.1032. [DOI] [PubMed] [Google Scholar]
- 10.Brittis PA, Silver J. Exogenous glycosaminoglycans induce complete inversion of retinal ganglion cell bodies and their axons within the retinal neuroepithelium. Proc Natl Acad Sci U S A. 1994;91:7539–7542. doi: 10.1073/pnas.91.16.7539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Brittis PA, Silver J. Multiple factors govern intraretinal axon guidance: a time-lapse study. Mol Cell Neurosci. 1995;6:413–432. doi: 10.1006/mcne.1995.1031. [DOI] [PubMed] [Google Scholar]
- 12.Brose K, Tessier-Lavigne M. Slit proteins: key regulators of axon guidance, axonal branching, and cell migration. Current Opinion in Neurobiology. 2000;10:95–102. doi: 10.1016/s0959-4388(99)00066-5. [DOI] [PubMed] [Google Scholar]
- 13.Brummendorf T, Rathjen FG. Cell adhesion molecules 1: immunoglobulin superfamily. Protein Profile. 1995;2:963–1108. [PubMed] [Google Scholar]
- 14.Carulli D, Laabs T, Geller HM, Fawcett JW. Chondroitin sulfate proteoglycans in neural development and regeneration. Curr Opin Neurobiol. 2005;15:116–120. doi: 10.1016/j.conb.2005.01.014. [DOI] [PubMed] [Google Scholar]
- 15.Chalasani SH, Baribaud F, Coughlan CM, Sunshine MJ, Lee VM, Doms RW, Littman DR, Raper JA. The chemokine stromal cell-derived factor-1 promotes the survival of embryonic retinal ganglion cells. J Neurosci. 2003;23:4601–4612. doi: 10.1523/JNEUROSCI.23-11-04601.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chalasani SH, Sabelko KA, Sunshine MJ, Littman DR, Raper JA. A chemokine, SDF-1, reduces the effectiveness of multiple axonal repellents and is required for normal axon pathfinding. J Neurosci. 2003;23:1360–1371. doi: 10.1523/JNEUROSCI.23-04-01360.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Charron F, Stein E, Jeong J, McMahon AP, Tessier-Lavigne M. The morphogen Sonic Hedgehog is an axonal chemoattractant that collaborates with Netrin-1 in midline axon guidance. Cell. 2003;113:11–23. doi: 10.1016/s0092-8674(03)00199-5. [DOI] [PubMed] [Google Scholar]
- 18.Chiang C, Litingtung Y, Lee E, Young KE, Corden JL, Westphal H, Beachy PA. Cyclopia and defective axial patterning in mice lacking Sonic hedgehog gene function. Nature. 1996;383:407–413. doi: 10.1038/383407a0. [DOI] [PubMed] [Google Scholar]
- 19.Crossin KL, Krushel LA. Cellular signaling by neural cell adhesion molecules of the immunoglobulin superfamily. Dev Dyn. 2000;218:260–279. doi: 10.1002/(SICI)1097-0177(200006)218:2<260::AID-DVDY3>3.0.CO;2-9. [DOI] [PubMed] [Google Scholar]
- 20.Dakubo GD, Wang YP, Mazerolle C, Campsall K, McMahon AP, Wallace VA. Retinal ganglion cell-derived sonic hedgehog signaling is required for optic disc and stalk neuroepithelial cell development. Development. 2003;130:2967–2980. doi: 10.1242/dev.00515. [DOI] [PubMed] [Google Scholar]
- 21.DeBernardo AP, Chang S. Heterophilic interactions of DM-GRASP: GRASP-NgCAM interactions involved in neurite extension. J Cell Biol. 1996;133:657–666. doi: 10.1083/jcb.133.3.657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Deiner MS, Kennedy TE, Fazeli A, Serafini T, Tessier-Lavigne M, Sretavan DW. Netrin-1 and DCC mediate axon guidance locally at the optic disc: loss of function leads to optic nerve hypoplasia. Neuron. 1997;19:575–589. doi: 10.1016/s0896-6273(00)80373-6. [DOI] [PubMed] [Google Scholar]
- 23.Drager UC, Olsen JF. Ganglion cell distribution in the retina of the mouse. Investigative Ophthalmology & Visual Science. 1981;20:285–293. [PubMed] [Google Scholar]
- 24.Dudley AT, Lyons KM, Robertson EJ. A requirement for bone morphogenetic protein-7 during development of the mammalian kidney and eye. Genes Dev. 1995;9:2795–2807. doi: 10.1101/gad.9.22.2795. [DOI] [PubMed] [Google Scholar]
- 25.Dudley AT, Robertson EJ. Overlapping expression domains of bone morphogenetic protein family members potentially account for limited tissue defects in BMP7 deficient embryos. Dev Dyn. 1997;208:349–362. doi: 10.1002/(SICI)1097-0177(199703)208:3<349::AID-AJA6>3.0.CO;2-I. [DOI] [PubMed] [Google Scholar]
- 26.Dyer MA, Cepko CL. Regulating proliferation during retinal development. Nat Rev Neurosci. 2001;2:333–342. doi: 10.1038/35072555. [DOI] [PubMed] [Google Scholar]
- 27.el-Deeb S, Thompson SC, Covault J. Characterization of a cell surface adhesion molecule expressed by a subset of developing chick neurons. Dev Biol. 1992;149:213–227. doi: 10.1016/0012-1606(92)90278-o. [DOI] [PubMed] [Google Scholar]
- 28.Erskine L, Williams SE, Brose K, Kidd T, Rachel RA, Goodman CS, Tessier-Lavigne M, Mason CA. Retinal ganglion cell axon guidance in the mouse optic chiasm: expression and function of robos and slits. Journal of Neuroscience. 2000;20:4975–4982. doi: 10.1523/JNEUROSCI.20-13-04975.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Flanagan JG. Neural map specification by gradients. Curr Opin Neurobiol. 2006;16:59–66. doi: 10.1016/j.conb.2006.01.010. [DOI] [PubMed] [Google Scholar]
- 30.Goldberg S, Coulombre AJ. Topographical development of the ganglion cell fiber layer in the chick retina. A whole mount study. Journal of Comparative Neurology. 1972;146:507–518. doi: 10.1002/cne.901460406. [DOI] [PubMed] [Google Scholar]
- 31.Goldberg S, Coulombre AJ. Topographical development of the ganglion cell fiber layer in the chick retina. A whole mount study. J Comp Neurol. 1972;146:507–518. doi: 10.1002/cne.901460406. [DOI] [PubMed] [Google Scholar]
- 32.Grant P, Rubin E. Disruption of optic fibre growth following eye rotation in Xenopus laevis embryos. Nature. 1980;287:845–848. doi: 10.1038/287845a0. [DOI] [PubMed] [Google Scholar]
- 33.Halfter W. Intraretinal grafting reveals growth requirements and guidance cues for optic axons in the developing avian retina. Developmental Biology. 1996;177:160–177. doi: 10.1006/dbio.1996.0153. [DOI] [PubMed] [Google Scholar]
- 34.Halfter W, Deiss S. Axon growth in embryonic chick and quail retinal whole mounts in vitro. Developmental Biology. 1984;102:344–355. doi: 10.1016/0012-1606(84)90199-4. [DOI] [PubMed] [Google Scholar]
- 35.Halfter W, Dong S, Balasubramani M, Bier ME. Temporary disruption of the retinal basal lamina and its effect on retinal histogenesis. Dev Biol. 2001;238:79–96. doi: 10.1006/dbio.2001.0396. [DOI] [PubMed] [Google Scholar]
- 36.Halfter W, Fua CS. Immunohistochemical localization of laminin, neural cell adhesion molecule, collagen type IV and T-61 antigen in the embryonic retina of the Japanese quail by in vivo injection of antibodies. Cell Tissue Res. 1987;249:487–496. doi: 10.1007/BF00217320. [DOI] [PubMed] [Google Scholar]
- 37.Halfter W, Newgreen DF, Sauter J, Schwarz U. Oriented axon outgrowth from avian embryonic retinae in culture. Dev Biol. 1983;95:56–64. doi: 10.1016/0012-1606(83)90006-4. [DOI] [PubMed] [Google Scholar]
- 38.Halfter W, Reckhaus W, Kroger S. Nondirected axonal growth on basal lamina from avian embryonic neural retina. J Neurosci. 1987;7:3712–3722. doi: 10.1523/JNEUROSCI.07-11-03712.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Halfter W, Schurer B. Disruption of the pial basal lamina during early avian embryonic development inhibits histogenesis and axonal pathfinding in the optic tectum. Journal of Comparative Neurology. 1998;397:105–117. doi: 10.1002/(sici)1096-9861(19980720)397:1<105::aid-cne8>3.0.co;2-4. [DOI] [PubMed] [Google Scholar]
- 40.Hankin MH, Lagenaur CF. Cell adhesion molecules in the early developing mouse retina: retinal neurons show preferential outgrowth in vitro on L1 but not N-CAM. J Neurobiol. 1994;25:472–487. doi: 10.1002/neu.480250503. [DOI] [PubMed] [Google Scholar]
- 41.Henke-Fahle S, Wild K, Sierra A, Monnier PP. Characterization of a new brain-derived proteoglycan inhibiting retinal ganglion cell axon outgrowth. Mol Cell Neurosci. 2001;18:541–556. doi: 10.1006/mcne.2001.1034. [DOI] [PubMed] [Google Scholar]
- 42.Henley J, Poo MM. Guiding neuronal growth cones using Ca2+ signals. Trends Cell Biol. 2004;14:320–330. doi: 10.1016/j.tcb.2004.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hinck L. The versatile roles of “axon guidance” cues in tissue morphogenesis. Dev Cell. 2004;7:783–793. doi: 10.1016/j.devcel.2004.11.002. [DOI] [PubMed] [Google Scholar]
- 44.Hinds JW, Hinds PL. Early ganglion cell differentiation in the mouse retina: an electron microscopic analysis utilizing serial sections. Dev Biol. 1974;37:381–416. doi: 10.1016/0012-1606(74)90156-0. [DOI] [PubMed] [Google Scholar]
- 45.Ho KS, Scott MP. Sonic hedgehog in te nervous system: functions, modifications and mechanisms. Curr. Opin. Neurobiol. 2002;12:57–63. doi: 10.1016/s0959-4388(02)00290-8. [DOI] [PubMed] [Google Scholar]
- 46.Holt CE. A single-cell analysis of early retinal ganglion cell differentiation in Xenopus: from soma to axon tip. J Neurosci. 1989;9:3123–3145. doi: 10.1523/JNEUROSCI.09-09-03123.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hong K, Hinck L, Nishiyama M, Poo M, Tessier-Lavigne M, Stein E. A ligand-gated association between cytoplasmic domains of UNC5 and DCC family receptors converts netrin-induced growth cone attraction to repulsion. [see comments.] Cell. 1999;97:927–941. doi: 10.1016/s0092-8674(00)80804-1. [DOI] [PubMed] [Google Scholar]
- 48.Hopker VH, Shewan D, Tessier-Lavigne M, Poo M, Holt C. Growth-cone attraction to netrin-1 is converted to repulsion by laminin-1. Nature. 1999;401:69–73. doi: 10.1038/43441. [DOI] [PubMed] [Google Scholar]
- 49.Hutson LD, Jurynec MJ, Yeo SY, Okamoto H, Chien CB. Two divergent slit1 genes in zebrafish. Dev Dyn. 2003;228:358–369. doi: 10.1002/dvdy.10386. [DOI] [PubMed] [Google Scholar]
- 50.Imagawa T, Fujita Y, Kitagawa H, Uehara M. Quantitative studies of the optic nerve fiber layer in the chicken retina. J Vet Med Sci. 1999;61:883–889. doi: 10.1292/jvms.61.883. [DOI] [PubMed] [Google Scholar]
- 51.Ingham PW, Placzek M. Orchestrating ontogenesis: variations on a theme by sonic hedgehog. Nat Rev Genet. 2006;7:841–850. doi: 10.1038/nrg1969. [DOI] [PubMed] [Google Scholar]
- 52.Jeon CJ, Strettoi E, Masland RH. The major cell populations of the mouse retina. J Neurosci. 1998;18:8936–8946. doi: 10.1523/JNEUROSCI.18-21-08936.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Jin Z, Chau MD, Bao ZZ. Sema3D, Sema3F, and Sema5A are expressed in overlapping and distinct patterns in chick embryonic heart. Dev Dyn. 2006;235:163–169. doi: 10.1002/dvdy.20614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Jin Z, Zhang J, Klar A, Chedotal A, Rao Y, Cepko CL, Bao ZZ. Irx4 regulation of Slit1 expression contributes to the definition of early axonal paths inside the retina. Development. 2003;130:1037–1048. doi: 10.1242/dev.00326. [DOI] [PubMed] [Google Scholar]
- 55.Kamiguchi H, Lemmon V. Neural cell adhesion molecule L1: signaling pathways and growth cone motility. J Neurosci Res. 1997;49:1–8. doi: 10.1002/(sici)1097-4547(19970701)49:1<1::aid-jnr1>3.0.co;2-h. [DOI] [PubMed] [Google Scholar]
- 56.Kennedy TE, Serafini T, de la Torre JR, Tessier-Lavigne M. Netrins are diffusible chemotropic factors for commissural axons in the embryonic spinal cord. Cell. 1994;78:425–435. doi: 10.1016/0092-8674(94)90421-9. [DOI] [PubMed] [Google Scholar]
- 57.Kolpak A, Zhang J, Bao ZZ. Sonic hedgehog has a dual effect on the growth of retinal ganglion axons depending on its concentration. J Neurosci. 2005;25:3432–3441. doi: 10.1523/JNEUROSCI.4938-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Lemke G, Reber M. Retinotectal mapping: new insights from molecular genetics. Annu Rev Cell Dev Biol. 2005;21:551–580. doi: 10.1146/annurev.cellbio.20.022403.093702. [DOI] [PubMed] [Google Scholar]
- 59.Li Q, Shirabe K, Thisse C, Thisse B, Okamoto H, Masai I, Kuwada JY. Chemokine signaling guides axons within the retina in zebrafish. J Neurosci. 2005;25:1711–1717. doi: 10.1523/JNEUROSCI.4393-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Liu J, Wilson S, Reh T. BMP receptor 1b is required for axon guidance and cell survival in the developing retina. Dev Biol. 2003;256:34–48. doi: 10.1016/s0012-1606(02)00115-x. [DOI] [PubMed] [Google Scholar]
- 61.Livesey FJ, Cepko CL. Vertebrate neural cell-fate determination: lessons from the retina. Nature Reviews Neuroscience. 2001;2:109–118. doi: 10.1038/35053522. [DOI] [PubMed] [Google Scholar]
- 62.Luo G, Hofmann C, Bronckers AL, Sohocki M, Bradley A, Karsenty G. BMP-7 is an inducer of nephrogenesis, and is also required for eye development and skeletal patterning. Genes Dev. 1995;9:2808–2820. doi: 10.1101/gad.9.22.2808. [DOI] [PubMed] [Google Scholar]
- 63.Mann F, Holt CE. Control of retinal growth and axon divergence at the chiasm: lessons from Xenopus. Bioessays. 2001;23:319–326. doi: 10.1002/bies.1046. [DOI] [PubMed] [Google Scholar]
- 64.Mason C, Erskine L. Growth cone form, behavior, and interactions in vivo: retinal axon pathfinding as a model. J Neurobiol. 2000;44:260–270. doi: 10.1002/1097-4695(200008)44:2<260::aid-neu14>3.0.co;2-h. [DOI] [PubMed] [Google Scholar]
- 65.McAdams BD, McLoon SC. Expression of chondroitin sulfate and keratan sulfate proteoglycans in the path of growing retinal axons in the developing chick. Journal of Comparative Neurology. 1995;352:594–606. doi: 10.1002/cne.903520408. [DOI] [PubMed] [Google Scholar]
- 66.McLaughlin T, O'Leary DD. Molecular gradients and development of retinotopic maps. Annu Rev Neurosci. 2005;28:327–355. doi: 10.1146/annurev.neuro.28.061604.135714. [DOI] [PubMed] [Google Scholar]
- 67.McLoon SC, Barnes RB. Early differentiation of retinal ganglion cells: an axonal protein expressed by premigratory and migrating retinal ganglion cells. Journal of Neuroscience. 1989;9:1424–1432. doi: 10.1523/JNEUROSCI.09-04-01424.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Montgomery AM, Becker JC, Siu CH, Lemmon VP, Cheresh DA, Pancook JD, Zhao X, Reisfeld RA. Human neural cell adhesion molecule L1 and rat homologue NILE are ligands for integrin alpha v beta 3. J Cell Biol. 1996;132:475–485. doi: 10.1083/jcb.132.3.475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Morcillo J, Martinez-Morales JR, Trousse F, Fermin Y, Sowden JC, Bovolenta P. Proper patterning of the optic fissure requires the sequential activity of BMP7 and SHH. Development. 2006;133:3179–3190. doi: 10.1242/dev.02493. [DOI] [PubMed] [Google Scholar]
- 70.Muhleisen TW, Agoston Z, Schulte D. Retroviral misexpression of cVax disturbs retinal ganglion cell axon fasciculation and intraretinal pathfinding in vivo and guidance of nasal ganglion cell axons in vivo. Dev Biol. 2006;297:59–73. doi: 10.1016/j.ydbio.2006.04.466. [DOI] [PubMed] [Google Scholar]
- 71.Mui SH, Hindges R, O'Leary DD, Lemke G, Bertuzzi S. The homeodomain protein Vax2 patterns the dorsoventral and nasotemporal axes of the eye. Development. 2002;129:797–804. doi: 10.1242/dev.129.3.797. [DOI] [PubMed] [Google Scholar]
- 72.Neumann CJ, Nuesslein-Volhard C. Patterning of the zebrafish retina by a wave of sonic hedgehog activity. Science. 2000;289:2137–2139. doi: 10.1126/science.289.5487.2137. [DOI] [PubMed] [Google Scholar]
- 73.Nguyen-Ba-Charvet KT, Brose K, Marillat v., Sotelo C, Tessier-Lavigne M, Chedotal A. Sensory axon response to substrate-bound Slit2 is modulated by lamiin and cyclic GMP. Mol. Cell. Neurosci. 2001;17:1048–1058. doi: 10.1006/mcne.2001.0994. [DOI] [PubMed] [Google Scholar]
- 74.Niclou SP, Jia L, Raper JA. Slit2 is a repellent for retinal ganglion cell axons. Journal of Neuroscience. 2000;20:4962–4974. doi: 10.1523/JNEUROSCI.20-13-04962.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.O'Leary DM, Gerfen CR, Cowan WM. The development and restriction of the ipsilateral retinofugal projection in the chick. Brain Research. 1983;312:93–109. doi: 10.1016/0165-3806(83)90124-4. [DOI] [PubMed] [Google Scholar]
- 76.Ohta K, Tannahill D, Yoshida K, Johnson AR, Cook GM, Keynes RJ. Embryonic lens repels retinal ganglion cell axons. Dev Biol. 1999;211:124–132. doi: 10.1006/dbio.1999.9312. [DOI] [PubMed] [Google Scholar]
- 77.Oster SF, Bodeker MO, He F, Sretavan DW. Invariant Sema5A inhibition serves an ensheathing function during optic nerve development. Development. 2003;130:775–784. doi: 10.1242/dev.00299. [DOI] [PubMed] [Google Scholar]
- 78.Oster SF, Deiner M, Birgbauer E, Sretavan DW. Ganglion cell axon pathfinding in the retina and optic nerve. Semin Cell Dev Biol. 2004;15:125–136. doi: 10.1016/j.semcdb.2003.09.006. [DOI] [PubMed] [Google Scholar]
- 79.Ott H, Bastmeyer M, Stuermer CA. Neurolin, the goldfish homolog of DM-GRASP, is involved in retinal axon pathfinding to the optic disk. Journal of Neuroscience. 1998;18:3363–3372. doi: 10.1523/JNEUROSCI.18-09-03363.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Pasquale EB. Eph receptor signalling casts a wide net on cell behaviour. Nat Rev Mol Cell Biol. 2005;6:462–475. doi: 10.1038/nrm1662. [DOI] [PubMed] [Google Scholar]
- 81.Pasterkamp RJ, Kolodkin AL. Semaphorin junction: making tracks toward neural connectivity. Curr Opin Neurobiol. 2003;13:79–89. doi: 10.1016/s0959-4388(03)00003-5. [DOI] [PubMed] [Google Scholar]
- 82.Plump AS, Erskine L, Sabatier C, Brose K, Epstein CJ, Goodman CS, Mason CA, Tessier-Lavigne M. Slit1 and Slit2 cooperate to prevent premature midline crossing of retinal axons in the mouse visual system. Neuron. 2002;33:219–232. doi: 10.1016/s0896-6273(01)00586-4. [DOI] [PubMed] [Google Scholar]
- 83.Polyak S. The Vertebrate Visual System. University of Chicago Press; Chicago: 1957. [Google Scholar]
- 84.Prada C, Puelles L, Genis-Galvez JM. A golgi study on the early sequence of differentiation of ganglion cells in the chick embryo retina. Anat Embryol (Berl) 1981;161:305–317. doi: 10.1007/BF00301828. [DOI] [PubMed] [Google Scholar]
- 85.Ramon y Cajal S. la retine des vertebres. La Cellule. 1892;9:121–255. [Google Scholar]
- 86.Ramon y Cajal S, Glickstein M, Thorpe SA. The stucture of the retina. C. C. Thomas; Springfield, Ill.: 1972. [Google Scholar]
- 87.Raper JA. Semaphorins and their receptors in vertebrates and invertebrates. Current Opinion in Neurobiology. 2000;10:88–94. doi: 10.1016/s0959-4388(99)00057-4. [DOI] [PubMed] [Google Scholar]
- 88.Ringstedt T, Braisted JE, Brose K, Kidd T, Goodman C, Tessier-Lavigne M, O'Leary DD. Slit inhibition of retinal axon growth and its role in retinal axon pathfinding and innervation patterns in the diencephalon. Journal of Neuroscience. 2000;20:4983–4991. doi: 10.1523/JNEUROSCI.20-13-04983.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Schulte D, Furukawa T, Peters MA, Kozak CA, Cepko CL. Misexpression of the Emx-related homeobox genes cVax and mVax2 ventralizes the retina and perturbs the retinotectal map. Neuron. 1999;24:541–553. doi: 10.1016/s0896-6273(00)81111-3. [DOI] [PubMed] [Google Scholar]
- 90.Serafini T, Kennedy TE, Galko MJ, Mirzayan C, Jessell TM, Tessier-Lavigne M. The netrins define a family of axon outgrowth-promoting proteins homologous to C. elegans UNC-6. Cell. 1994;78:409–424. doi: 10.1016/0092-8674(94)90420-0. [DOI] [PubMed] [Google Scholar]
- 91.Silver J, Robb RM. Studies on the development of the eye cup and optic nerve in normal mice and in mutants with congenital optic nerve aplasia. Dev Biol. 1979;68:175–190. doi: 10.1016/0012-1606(79)90252-5. [DOI] [PubMed] [Google Scholar]
- 92.Silver J, Rutishauser U. Guidance of optic axons in vivo by a preformed adhesive pathway on neuroepithelial endfeet. Dev Biol. 1984;106:485–499. doi: 10.1016/0012-1606(84)90248-3. [DOI] [PubMed] [Google Scholar]
- 93.Snow DM, Lemmon V, Carrino DA, Caplan AI, Silver J. Sulfated proteoglycans in astroglial barriers inhibit neurite outgrowth in vitro. Exp Neurol. 1990;109:111–130. doi: 10.1016/s0014-4886(05)80013-5. [DOI] [PubMed] [Google Scholar]
- 94.Snow DM, Watanabe M, Letourneau PC, Silver J. A chondroitin sulfate proteoglycan may influence the direction of retinal ganglion cell outgrowth. Development - Supplement. 1991;113:1473–1485. doi: 10.1242/dev.113.4.1473. [DOI] [PubMed] [Google Scholar]
- 95.Song H, Poo MM. The cell biology of neuronal navigation. Nature Cell Biology. 2001;3:E81–E88. doi: 10.1038/35060164. [DOI] [PubMed] [Google Scholar]
- 96.Stier H, Schlosshauer B. Axonal guidance in the chicken retina. Development. 1995;121:1443–1454. doi: 10.1242/dev.121.5.1443. [DOI] [PubMed] [Google Scholar]
- 97.Stuermer CA, Bastmeyer M. The retinal axon's pathfinding to the optic disk. Prog Neurobiol. 2000;62:197–214. doi: 10.1016/s0301-0082(00)00012-5. [DOI] [PubMed] [Google Scholar]
- 98.Suh LH, Oster SF, Soehrman SS, Grenningloh G, Sretavan DW. L1/Laminin modulation of growth cone response to EphB triggers growth pauses and regulates the microtubule destabilizing protein SCG10. J Neurosci. 2004;24:1976–1986. doi: 10.1523/JNEUROSCI.1670-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Tanaka H, Matsui T, Agata A, Tomura M, Kubota I, McFarland KC, Kohr B, Lee A, Phillips HS, Shelton DL. Molecular cloning and expression of a novel adhesion molecule, SC1. Neuron. 1991;7:535–545. doi: 10.1016/0896-6273(91)90366-8. [DOI] [PubMed] [Google Scholar]
- 100.Taylor D. Optic nerve axons: life and death before birth. Eye. 2005;19:499–527. doi: 10.1038/sj.eye.6701857. [DOI] [PubMed] [Google Scholar]
- 101.Tessier-Lavigne M, Goodman CS. The molecular biology of axon guidance. Science. 1996;274:1123–1133. doi: 10.1126/science.274.5290.1123. [DOI] [PubMed] [Google Scholar]
- 102.Thanos S, Mey J. Development of the visual system of the chick. II. Mechanisms of axonal guidance. Brain Research - Brain Research Reviews. 2001;35:205–245. doi: 10.1016/s0165-0173(01)00049-2. [DOI] [PubMed] [Google Scholar]
- 103.Thompson H, Camand O, Barker D, Erskine L. Slit proteins regulate distinct aspects of retinal ganglion cell axon guidance within dorsal and ventral retina. J Neurosci. 2006;26:8082–8091. doi: 10.1523/JNEUROSCI.1342-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Trousse F, Marti E, Gruss P, Torres M, Bovolenta P. Control of retinal ganglion cell axon growth: a new role for Sonic hedgehog. Development - Supplement. 2001;128:3927–3936. doi: 10.1242/dev.128.20.3927. [DOI] [PubMed] [Google Scholar]
- 105.Van Vactor D. Adhesion and signaling in axonal fasciculation. Curr Opin Neurobiol. 1998;8:80–86. doi: 10.1016/s0959-4388(98)80011-1. [DOI] [PubMed] [Google Scholar]
- 106.Walsh FS, Doherty P. Neural cell adhesion molecules of the immunoglobulin superfamily: role in axon growth and guidance. Annu Rev Cell Dev Biol. 1997;13:425–456. doi: 10.1146/annurev.cellbio.13.1.425. [DOI] [PubMed] [Google Scholar]
- 107.Wang KH, Brose K, Arnott D, Kidd T, Goodman CS, Henzel W, Tessier-Lavigne M. Biochemical purification of a mammalian slit protein as a positive regulator of sensory axon elongation and branching. Cell. 1999;96:771–784. doi: 10.1016/s0092-8674(00)80588-7. [DOI] [PubMed] [Google Scholar]
- 108.Wang Y, Dakubo GD, Thurig S, Mazerolle CJ, Wallace VA. Retinal ganglion cell-derived sonic hedgehog locally controls proliferation and the timing of RGC development in the embryonic mouse retina. Development. 2005;132:5103–5113. doi: 10.1242/dev.02096. [DOI] [PubMed] [Google Scholar]
- 109.Wawersik S, Purcell P, Rauchman M, Dudley AT, Robertson EJ, Maas R. BMP7 acts in murine lens placode development. Dev Biol. 1999;207:176–188. doi: 10.1006/dbio.1998.9153. [DOI] [PubMed] [Google Scholar]
- 110.Weiner JA, Koo SJ, Nicolas S, Fraboulet S, Pfaff SL, Pourquie O, Sanes JR. Axon fasciculation defects and retinal dysplasias in mice lacking the immunoglobulin superfamily adhesion molecule BEN/ALCAM/SC1. Mol Cell Neurosci. 2004;27:59–69. doi: 10.1016/j.mcn.2004.06.005. [DOI] [PubMed] [Google Scholar]
- 111.Wong K, Park HT, Wu JY, Rao Y. Slit proteins: molecular guidance cues for cells ranging from neurons to leukocytes. Curr Opin Genet Dev. 2002;12:583–591. doi: 10.1016/s0959-437x(02)00343-x. [DOI] [PubMed] [Google Scholar]
- 112.Xiang Y, Li Y, Zhang Z, Cui K, Wang S, Yuan XB, Wu CP, Poo MM, Duan S. Nerve growth cone guidance mediated by G protein-coupled receptors. Nat Neurosci. 2002;5:843–848. doi: 10.1038/nn899. [DOI] [PubMed] [Google Scholar]
- 113.Yang XJ. Roles of cell-extrinsic growth factors in vertebrate eye pattern formation and retinogenesis. Semin Cell Dev Biol. 2004;15:91–103. doi: 10.1016/j.semcdb.2003.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Yip PM, Zhao X, Montgomery AM, Siu CH. The Arg-Gly-Asp motif in the cell adhesion molecule L1 promotes neurite outgrowth via interaction with the alphavbeta3 integrin. Mol Biol Cell. 1998;9:277–290. doi: 10.1091/mbc.9.2.277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Yuan W, Zhou L, Chen JH, Wu JY, Rao Y, Ornitz DM. The mouse SLIT family: secreted ligands for ROBO expressed in patterns that suggest a role in morphogenesis and axon guidance. Developmental Biology. 1999;212:290–306. doi: 10.1006/dbio.1999.9371. [DOI] [PubMed] [Google Scholar]
- 116.Zelina P, Avci HX, Thelen K, Pollerberg GE. The cell adhesion molecule NrCAM is crucial for growth cone behaviour and pathfinding of retinal ganglion cell axons. Development. 2005;132:3609–3618. doi: 10.1242/dev.01934. [DOI] [PubMed] [Google Scholar]
- 117.Zhang J, Jin Z, Bao ZZ. Disruption of gradient expression of Zic3 resulted in aberrant projection of the retinal ganglion axons to the optic disc. Development. 2004;131:1553–1562. doi: 10.1242/dev.01041. [DOI] [PubMed] [Google Scholar]
- 118.Zhang XM, Yang XJ. Regulation of retinal ganglion cell production by Sonic hedgehog. Development - Supplement. 2001;128:943–957. doi: 10.1242/dev.128.6.943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Zolessi FR, Poggi L, Wilkinson CJ, Chien CB, Harris WA. Polarization and orientation of retinal ganglion cells in vivo. Neural Develop. 2006;1:2–22. doi: 10.1186/1749-8104-1-2. [DOI] [PMC free article] [PubMed] [Google Scholar]