Abstract
Cytotoxic T lymphocyte-associated antigen 4 (CTLA-4) functions as a negative regulator of endogenous and vaccine-induced antitumor immunity. The administration of fully human anti-CTLA-4 blocking monoclonal antibodies to advanced-cancer patients increases immune-mediated tumor destruction in some subjects. Nonetheless, patients that respond also frequently manifest serious inflammatory pathologies, raising the possibility that the therapeutic and toxic effects of CTLA-4 blockade might be linked. Here we show that periodic infusions of anti-CTLA-4 antibodies after vaccination with irradiated, autologous tumor cells engineered to secrete GM-CSF (GVAX) generate clinically meaningful antitumor immunity without grade 3 or 4 toxicity in a majority of metastatic melanoma patients. The application of this sequential immunotherapy to advanced ovarian carcinoma patients also revealed that tumor destruction and severe inflammatory pathology could be dissociated, although further refinements are required to increase clinical responses and to minimize toxicity in this population. The extent of therapy-induced tumor necrosis was linearly related to the natural logarithm of the ratio of intratumoral CD8+ effector T cells to FoxP3+ regulatory T cells (Tregs) in posttreatment biopsies. Together, these findings help clarify the immunologic and clinical effects of CTLA-4 antibody blockade in previously vaccinated patients and raise the possibility that selective targeting of antitumor Tregs may constitute a complementary strategy for combination therapy.
Keywords: CTLA-4, regulatory T cells (Tregs), vaccine, GM-CSF
Tumors frequently provoke innate and adaptive immune recognition, but the development of clinically evident disease denotes a failure of host defense (1). The elucidation of specific mechanisms that contribute to tumor escape provides a strong foundation for crafting therapeutic strategies to improve host immunity. One major defect involves inefficient antigen presentation by tumor-infiltrating dendritic cells and macrophages (2). Soluble and membrane-bound factors produced in the tumor microenvironment impede the activation of these professional antigen-presenting cells, skewing host reactions toward immunosuppressive circuits and dysfunctional effector T cells. Diverse vaccination strategies, however, may enhance the acquisition and processing of tumor antigens by dendritic cells and thereby engender protective immunity (3). Among these approaches, vaccination with irradiated, autologous tumor cells engineered to secrete GM-CSF (GVAX) stimulates a coordinated cellular and humoral response that effectuates substantial tumor necrosis in many patients with solid or hematologic malignancies (4).
Whereas immunization augments the magnitude and breadth of host reactions, most vaccinated subjects eventually succumb to progressive disease, implying the existence of additional immune defects. In this context, a key role has emerged for cytotoxic T lymphocyte-associated antigen 4 (CTLA-4) as a negative regulator of antitumor responses (5). Upon engagement by B7–1 or B7–2, CTLA-4 expressed on the surface of activated T lymphocytes signals for cell-cycle arrest and attenuation of effector function (6–8). Therefore, the transient blockade of CTLA-4 with antibodies potentiates immune-mediated tumor destruction, particularly in combination with cancer vaccines (9–11). The therapeutic efficacy of CTLA-4 blockade in murine models involves enhanced antitumor cytotoxicity, which reflects an increased proportion of CD8+ T effector cells to FoxP3+ Tregs within intratumoral infiltrates (12). Although CTLA-4 may contribute to Treg-induced immune suppression in some settings (13, 14), the infusion of anti-CTLA-4 antibodies triggers an overall expansion of Treg numbers in tumor-bearing hosts, especially in regional lymph nodes (12). Thus, a complex dynamic between effector and regulatory pathways underlies the antitumor effects of CTLA-4 blockade.
The formulation of fully human anti-CTLA-4 blocking monoclonal antibodies suitable for clinical administration has provided an opportunity to investigate the activities of CTLA-4 in advanced-cancer patients (5). Several early-stage trials demonstrated that anti-CTLA-4 antibodies alone or in combination with melanosomal antigen-derived peptide vaccines or chemotherapy can accomplish durable tumor regressions in a minority of metastatic melanoma patients (15–19). However, responding subjects often develop significant toxicities, with gastrointestinal inflammation, manifested as diarrhea, abdominal pain, and rarely perforation, the most common serious adverse event. Endoscopic biopsies have documented mucosal disruption effectuated by mixed lymphocyte, granulocyte, and macrophage infiltrates (20), features consistent with a critical role for CTLA-4 in maintaining intestinal homeostasis in murine models (21). Symptomatic immune-mediated hypophysitis, hepatitis, glomerulonephritis, pneumonitis, and dermatitis have also been reported at lower frequencies.
The concordance of tumor regressions and serious inflammatory pathologies with CTLA-4 blockade has raised the possibility that the therapeutic and toxic effects of the antibody might be linked. Indeed, some current strategies for identifying the optimal dose and schedule of antibody treatment are based on the idea that tumor destruction might require a major breach of tolerance to normal self-antigens (22, 23). In contrast to this notion, though, the presence of a significant endogenous or vaccine-evoked host response might allow less intense levels of CTLA-4 blockade to accomplish selective amplification of tumor-specific immunity. Consistent with this idea, we previously demonstrated that a single infusion of anti-CTLA-4 antibody (3 mg/kg of MDX-010, Ipilumimab) triggered striking tumor necrosis without severe toxicity in five of five metastatic melanoma or ovarian carcinoma patients who were previously immunized with GVAX (24). To explore further whether tumor immunity and serious inflammatory toxicity might be uncoupled with CTLA-4 antibody blockade, we evaluated this sequential immunotherapy in a larger patient cohort.
Results
Metastatic Melanoma.
In our initial studies of sequential immunotherapy for stage IV metastatic melanoma patients, the interval from completion of GVAX to anti-CTLA-4 antibody administration was several years (24). To examine the interactions of these approaches in more detail, we designed a new clinical protocol in which antibody infusion was moved temporally closer to vaccination. The 11 additional stage IV patients treated in this study received CTLA-4 blockade from 1 to 4 months after GVAX [supporting information (SI) Table 1]. Ipilumimab was infused at a fixed dose of 3 mg/kg, with subsequent treatments permitted at 2- to 3-month intervals as clinically indicated. Within this schema, the number of antibody infusions received by individual patients to date ranged from 1 to 16.
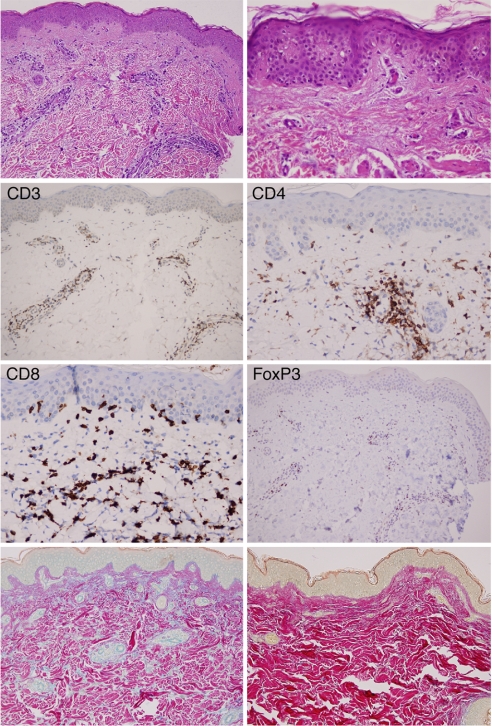
In contrast to other more intensive antibody doses and schedules (15–19), no grade 3 or 4 serious toxicities were observed in this melanoma cohort. Nonetheless, all subjects manifested mild inflammatory pathologies associated with low-level constitutional symptoms. The most common toxicity (10 of 11 subjects) was a grade I–II, reticular, and erythematous rash on the trunk and/or extremities that arose between 3 days and 3 weeks after antibody administration and then gradually resolved without specific intervention. Biopsies of involved skin revealed low-grade interface dermatitis, minor to moderate mononuclear infiltrates surrounding the superficial dermal vasculature, and increased mucin deposition (as revealed with colloidal iron staining) in the papillary and reticular dermis (Fig. 1). The interface dermatitis consisted almost exclusively of CD8+ T cells, few of which demonstrated significant epidermotropism. The perivascular infiltrate was composed of a mixture of CD4+ and CD8+ T cells. CD4+FoxP3+ Tregs were also abundantly represented in the superficial perivascular dermal infiltrates, where they might function to ameliorate the severity of the reaction. Collectively, these pathologic features resemble those observed in mild cutaneous forms of systemic lupus erythematosis. In accordance with this notion, two patients displayed transient, small increases in antinuclear antibody titers, but no evidence of systemic involvement including glomerulonephritis, serositis, cerebritis, or arthritis was noted.
Fig. 1.
CTLA-4 antibody blockade triggers a partial breach of tolerance to normal skin. (Top Left) Perivascular infiltrates (H&E). (Magnification: ×20.) (Top Right) Interface dermatitis (H&E). (Magnification: ×40.) (Middle) CD3 and FoxP3+ (×20), and CD4 and CD8 (×40). (Bottom Left) Colloidal iron stain reveals mucin accumulation in the dermis. (Magnification: ×20.) (Bottom Right) Colloidal iron stain, normal skin. (Magnification: ×20.)
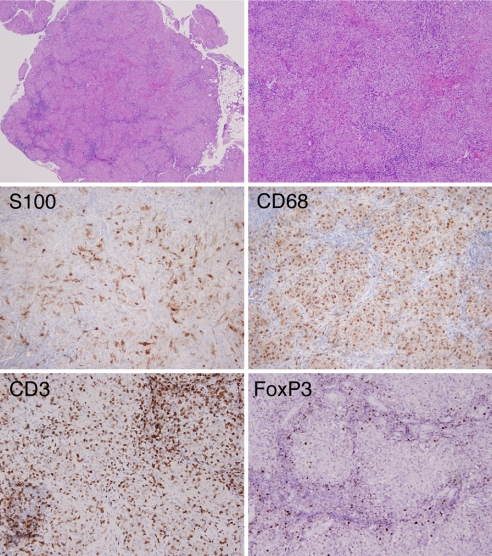
A key role for CTLA-4 in immune homeostasis was further revealed by other grade 1 or 2 inflammatory toxicities in individual patients. Bilateral, hilar lymphadenopathy without pulmonary symptoms was detected in one subject upon routine thoracic computerized tomography imaging. Biopsies of the enlarged nodes disclosed abundant epithelioid histiocytes that formed noncaseating granulomas, a pattern typical of sarcoidosis (Fig. 2). Although melanoma cells were not detected by immunohistochemistry for HMB-45 and MART-1 expression in these nodes (data not shown), abundant dendritic cells and macrophages were present, as revealed by antibody staining for S100 and CD68. Substantial numbers of CD3+ T cells and FoxP3+ Tregs were also evident, consistent with a recent report highlighting the involvement of these lymphocyte subsets in sarcoidosis (25). The patient continued to receive anti-CTLA-4 antibody treatments, and the lymphadenopathy waned without intervention. One subject experienced transient grade 2 diarrhea reflecting low-grade colitis, as documented by sigmoidoscopy, but this subsided in a few days without therapy. One patient showed mild, nonprogressive, focal hypopigmentation of the retinal pigment epithelium without alteration of visual acuity.
Fig. 2.
CTLA-4 antibody blockade precipitates a sarcoidosis-like pathology. (Top Left) Involved hilar lymph node (H&E). (Magnification: ×4.) (Top Right) Noncaseating granulomas (H&E). (Magnification: ×10.) Immunostains: S100, CD68, CD3, and FoxP3 (Magnification: ×20).
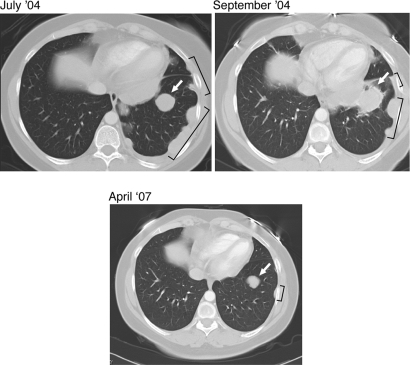
Notwithstanding the absence of clinical grade 3 or 4 inflammatory toxicities, 8 of the 11 subjects displayed meaningful antitumor effects, as measured by tumor regressions or prolonged stable disease, with associated improvements in disease-related morbidities (SI Table 1). By standard Response Evaluation Criteria in Solid Tumors (RECIST), three subjects demonstrated partial responses (Fig. 3). These regressions became apparent only after several months of therapy and were preceded by transient increases in tumor size early after antibody infusion. Regressions were observed in multiple visceral sites, including lung, pleura, intestine, and perirenal soft tissues, and these responses have proved durable (ongoing at 34, 24, and 21 months). By radiographic and clinical criteria, an additional five patients manifested stable disease for a minimum of 4 months and extending up to 25 months (the current length of follow-up). In light of the prior pace of disease progression and the significant tumor burden present in these patients upon initiation of anti-CTLA-4 antibody treatment (liver, lung, bone, visceral nodes, skin, and soft tissues), these antitumor effects are significant.
Fig. 3.
CTLA-4 antibody blockade evokes durable objective responses of metastatic melanoma with minimal toxicities. Serial thoracic CT scans document a transient increase in size of a pulmonary nodule (arrows) and pleural-based disease (brackets) followed by marked regression of the involved sites.
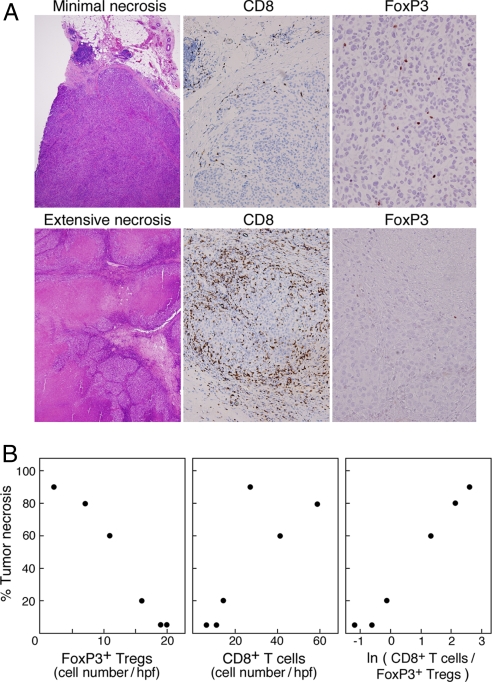
To examine the immune mechanisms underlying the antitumor activities of CTLA-4 blockade, we studied biopsies of metastatic lesions that were resected in six cases after Ipilumimab administration. These specimens revealed a spectrum of tumor necrosis ranging from minimal to extensive (Fig. 4A). All samples that exhibited significant necrosis contained a dense infiltration of CD8+ T cells, sometimes associated with acute inflammatory cells, particularly at the periphery of the lesion, where scattered viable melanoma cells were still evident. A vasculopathy composed of lymphocytes and neutrophils with rare fibrin thrombi was also found in areas of zonal necrosis. The degree of tumor cell destruction was inversely related to the number of intratumoral FoxP3+ Tregs (slope −5.27, P < 0.0001), but only weakly proportional to the intensity of intratumoral CD8+ T cells (slope 1.5, P = 0.054) (Fig. 4B). However, the overall extent of tumor necrosis was linearly related to the natural logarithm of the ratio of infiltrating CD8+ T cells to FoxP3+ cells (Necrosis = 26.25 + 24.6 * (ln(Ratio)), P < 0.0001, and R2 is 98.6%). These findings are in accordance with recent work demonstrating that combined GVAX and anti-CTLA-4 antibody treatment in the B16 murine melanoma model modulates the balance of antitumor effector T cells and Tregs (12). Our clinical results further raise the possibility that complementary strategies targeting intratumoral Tregs might increase the therapeutic efficacy of GVAX and CTLA-4 blockade in advanced melanoma patients.
Fig. 4.
The ratio of tumor-infiltrating CD8+ T cells to FoxP3+ Tregs after Ipilumimab infusion is tightly correlated with the extent of tumor necrosis. (A Upper) Minimal necrosis of melanoma metastasis. (Lower) Extensive necrosis of melanoma metastasis. (Magnification: H&E, ×4; CD8, ×20; FoxP3, ×40.) (B) Numbers of intratumoral FoxP3+ Tregs and CD8+ T cells versus tumor necrosis.
Metastatic Ovarian Carcinoma.
We previously reported that a single infusion of Ipilumimab (3 mg/kg) after GVAX in two stage IV ovarian carcinoma patients was well tolerated and triggered a decrease or stabilization of CA-125 levels of several month's duration (24). To define the toxicity and antitumor effects of this sequential immunotherapy in more detail, we treated an additional nine stage IV ovarian carcinoma subjects by using the same antibody dose and schedule that was explored in the metastatic melanoma cohort (with the exception of one patient, who was treated at 3- to 6-month intervals as described below). The time from completion of GVAX to Ipilumimab for the ovarian group varied from 1 month to 3 years, and the number of antibody infusions administered to individual patients to date ranged from 1 to 11 (SI Table 2).
In contrast to the melanoma patients, two cases of grade 3 inflammatory toxicities that involved the gastrointestinal tract were observed in the ovarian population. In both instances, significant diarrhea followed a single infusion of antibody, and endoscopic biopsies revealed mucosal damage associated with abundant granulocytes, macrophages, CD4+ and CD8+ T cells, and FoxP3+ Tregs. A brief course of oral corticosteroids accomplished rapid amelioration of symptoms in one subject, whereas the other case gradually resolved over several months. This latter subject also manifested Sweet's syndrome, with a marked peripheral neutrophilia and a characteristic rash. Biopsy of the involved skin revealed an intense dermal infiltrate of neutrophils, which over time evolved to palisading granuloma annulare-like features in the deep dermis with marked edema of the papillary dermis (SI Fig. 6).
The remaining seven patients showed only minor inflammatory toxicities. Three subjects developed rashes consisting of papular, erythematous patches, which upon biopsy displayed elements similar to the melanoma cohort (data not shown), whereas four patients generated urticarial-like reactions restricted to sites of prior vaccination. One subject mounted a transient increase in antinuclear antibody titers.
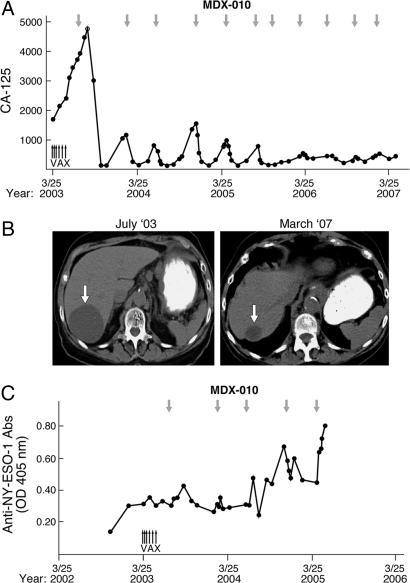
Although less common than the melanoma group, significant antitumor effects were observed in a minority of the ovarian carcinoma patients. Among these, the most striking was patient OV5, who achieved a dramatic fall in CA-125 levels several months after an initial dose of Ipilumimab (Fig. 5A). Although the response was not maintained, a second infusion resulted in a more rapid decline in CA-125 values. Interestingly, this acceleration of the antitumor reaction was mirrored by the kinetics of the Ipilumimab-triggered papular rash, which arose over several weeks with the first antibody treatment, but after only a few days with the second. Nine additional infusions of the anti-CTLA-4 antibody spaced at 3- to 5-month intervals over nearly 4 years have maintained profound disease control, with grade 1 rash as the only adverse event. Moreover, this patient's course was initially characterized by a remarkable periodicity to the rise and fall of CA-125 levels, but this gradually dampened as a function of time and/or prolonged therapy. Consistent with the reduction in the circulating tumor marker, radiographic studies documented substantial regression of a large, cystic hepatic metastasis and the complete resolution of mesenteric lymphadenopathy and thickening of the gastrocolic ligament (Fig. 5B and data not shown). Although the antitumor response did not involve the generation of anti-CA-125 antibodies (data not shown), increased humoral reactions to NY-ESO-1, a cancer-testis antigen ectopically expressed in many ovarian carcinomas (26), were associated with therapeutic activity (Fig. 5C).
Fig. 5.
CTLA-4 antibody blockade accomplishes the durable regression of advanced ovarian carcinoma in the absence of significant toxicity. (A) CA-125 levels as a function of therapy. Small upward arrows indicate GVAX; downward arrows denote Ipilumimab infusions. (B) Arrow indicates a large hepatic metastasis that underwent marked regression. (C) MDX-010 augmented IgG antibodies to NY-ESO-1.
Three additional subjects achieved stable disease of 6+ (ongoing), 4, and 2 months' duration, as measured by CA-125 levels and radiographic criteria, in the absence of serious toxicities. The patient that developed Sweet's syndrome and inflammatory colitis underwent a needle biopsy of pelvic disease, and this revealed extensive hemorrhagic tumor necrosis.
Discussion
Substantial evidence delineates CTLA-4 as a critical negative regulator of antitumor immunity and a compelling target for cancer immunotherapy (5). However, CTLA-4 also is an essential mediator of immune homeostasis, as illuminated by the fatal lymphoproliferative disorder that arises in young CTLA-4-deficient mice (27, 28). In light of these complex activities, a major challenge for the clinical development of anti-CTLA-4-blocking monoclonal antibodies is to define a favorable therapeutic index that strikes an optimum balance between tumor immunity and inflammatory disease. The concordance of tumor regressions and serious toxicities in several previous trials of CTLA-4 blockade has raised the spectre that these two treatment effects might be linked (15–19). Indeed, because some molecularly defined cancer antigens are nonmutated differentiation proteins expressed in normal tissues (29), tumor immunity and autoreactivity may be tightly associated in particular contexts.
Notwithstanding this close relationship, cancer genome sequencing experiments have uncovered a multiplicity of mutated gene products that derive from defects in tumor cell DNA repair mechanisms (30, 31). These alterations constitute neo-epitopes that may be presented in the context of MHC molecules as unique antigens for T cell recognition. If the host harbored sufficient reactivity to these mutated proteins, or other gene products aberrantly expressed in tumor cells, such as cancer-testes antigens (29), then less-intensive dosing and/or schedules of CTLA-4 blockade might be able to expand tumor-specific T cells preferentially, without compromising major homeostatic circuits that maintain tolerance to normal tissues.
To explore this idea, we administered 3 mg/kg of Ipilumimab at 2- to 3-month intervals to patients who previously received GVAX. This autologous tumor-cell-based vaccination strategy consistently enhances specific and long-lasting immunity, which likely encompasses at least some tumor-restricted epitopes (4). Intriguingly, none of the 11 stage IV metastatic melanoma patients treated with this regimen manifested grade 3 or 4 inflammatory toxicities, as compared with 15%–25% of melanoma subjects that received higher levels of CTLA-4 blockade on other clinical trials (15–19). Nonetheless, larger numbers of melanoma patients will need to be studied to define more fully the incidence of severe toxicities with this combination regimen. Mild pathologies indicative of a partial breach of tolerance were evident, however, and these included a self-limited rash in most subjects and individual cases of asymptomatic bilateral hilar lymphadenopathy, focal retinal pigment epithelium hypopigmentation, and low-grade colitis. In contrast to the melanoma cohort, though, a single dose of 3 mg/kg Ipilumimab triggered two cases of grade 3 gastrointestinal inflammation in a group of nine ovarian carcinoma patients previously immunized with GVAX. Although the number of subjects is somewhat limited, these findings raise the possibility that there may be disease-related factors that influence susceptibility to the toxicities of CTLA-4 blockade. Future investigations of genetic polymorphisms and the complex patterns of alternative splicing in the CTLA-4 locus might help identify high-risk patients (19, 32), for whom even lower doses of antibody or alternative therapies could be contemplated.
The antitumor responses accomplished with CTLA-4 blockade display a relatively slow tempo of onset and may even be preceded by a transient period of increased lesion size. Pathologic examination of metastases resected after Ipilumimab infusion indicate that this unusual course may reflect pronounced inflammation and edema induced by tumor-infiltrating lymphocytes. This profile of therapeutic activity is not adequately captured by the current RECIST criteria for monitoring tumor response, which primarily are based on target lesion volume (33). Thus, although overall survival remains the most important parameter of clinical efficacy, the adoption of new indexes that incorporate recent insights into the concept of immune equilibrium should be strongly considered (34).
Our pathologic analysis uncovered a striking linear relationship between the extent of tumor necrosis in posttreatment biopsies and the natural logarithm of the ratio of intratumoral CD8+ T cells and FoxP3+ Tregs. The balance of these T cell subsets was previously highlighted in mechanistic studies of combined GVAX/CTLA-4 blockade in the B16 murine melanoma model and as a major prognostic factor in advanced ovarian carcinoma patients (12, 26). Together, these findings suggest that complementary strategies that target intratumoral Tregs might extend the clinical benefits of GVAX and CTLA-4 blockade (35). In this context, we recently showed that GM-CSF promotes the development of FoxP3+ Tregs during tumor vaccination through a pathway involving the milk fat globule-EGF protein 8 (MFG-E8)-mediated uptake of apoptotic cells by antigen-presenting cells (36). Inhibition of MFG-E8 function in conjunction with GM-CSF delivery intensified tumor destruction by suppressing antitumor Tregs and amplifying cytotoxic effectors. Studies examining the interplay of this scheme with the administration of anti-CTLA-4 antibodies are underway.
The design of our clinical trial does not permit a definitive determination of the individual contributions of GVAX and CTLA-4 blockade to the biologic activity of the sequential immunotherapy. However, we previously reported that some patients responding to GVAX alone or GVAX followed by Ipilumimab infusion generated high titer antibodies against MHC class I-related chain A (MICA) (37). These humoral reactions antagonized the immune suppression evoked by soluble MICA and augmented innate and adaptive antitumor cytotoxicity. The correlation uncovered here between a heightened antibody response to NY-ESO-1 and the regression of advanced ovarian carcinoma further supports the idea that CTLA-4 blockade might unleash preexisiting or vaccine-induced antitumor effector and/or memory reactions. Whether concurrent administration of GVAX and CTLA-4 blockade might also enhance immune priming and/or alter toxicity profiles are important issues for future studies.
Materials and Methods
Clinical Protocols.
The Ipilumimab protocol received approval from the National Cancer Institute's Cancer Therapy Evaluation Program and the Dana–Farber/Harvard Cancer Center Institutional Review Board, and all patients provided informed consent for enrollment. Subjects were required to have received GVAX, be at least 4 weeks from prior therapy, have measurable disease, a life expectancy of at least 12 weeks, adequate end organ function, and an Eastern Cooperative Oncology Group (ECOG) performance status of 0–2. Melanoma patients were all stage IV, whereas ovarian patients harbored persistent or recurrent disease after primary surgery and chemotherapy. Exclusion criteria included autoimmune disease requiring immunosuppressive treatment, active infection, CNS metastases unless previously treated and stable for at least 3 months, prior Ipilumimab therapy, and use of systemic corticosteroids.
Ipilumimab was drawn through a 0.22 μM filter, diluted in normal saline to a concentration of 2.5 mg/ml, and administered intravenously by using a volumetric pump at a dose of 3.0 mg/kg over 90 min. Diagnostic imaging was performed at bimonthly intervals, and blood was collected regularly for laboratory analysis. Patients with a clinical response or up to a 40% increase in the sum of the longest diameters of target lesions and no more than two new target lesions were eligible for additional infusions at 2-month intervals.
Pathologic Analysis.
Biopsy tissue was fixed in 10% buffered formalin (Fisher Scientific) and processed for permanent paraffin embedding on a Leica ASP 300 tissue processor (Leica Microsystems); 5-μm paraffin sections were stained with hematoxylin and eosin by using a Leica Autostainer XL. Immunohistochemistry was performed on 5-μm paraffin sections as per standard technique on a Ventana Benchmark XT autostainer (Ventana Medical Systems) by using antibodies against CD3, CD4, CD8, S100, CD68 (Ventana Medical Systems), MART-1 (Signet), HMB-45 (Dako), and FoxP3 (Abcam). Colloidal iron staining was performed on a NexEs special stainer (Ventana Medical Systems). Histological samples were analyzed by using an Olympus BX41 microscope, and microscopic images were taken with a SPOT digital camera and SPOT software (Diagnostic Instruments).
CD8+ and FoxP3+ tumor-infiltrating lymphocytes were counted by using an Olympus BX41 microscope and a 2-key Laboratory Counter. Tissue sections for this analysis were cut from a single representative block for each patient, and all viable tumor tissue on the slide was counted (at 40× objective, 10 to 334 fields per slide). Mean values were calculated from all scored fields, and least squares regression analysis was used to model the effect of CD8+ T cells and Tregs on the degree of tumor necrosis.
Serology.
ELISAs were performed as previously described in ref. 38 by using purified recombinant His-tagged NY-ESO-1 protein (kindly provided by Lloyd Old, Ludwig Institute for Cancer, New York) in PBS. Plates were blocked with 5% nonfat dry milk, and serial aliquots of OV5 sera diluted 1:50,000 were tested.
Supplementary Material
ACKNOWLEDGMENTS.
We thank Lloyd Old (Ludwig Institute for Cancer, New York) for kindly providing recombinant NY-ESO-1 protein. This work was supported by the Cancer Therapy Evaluation Program, National Institutes of Health Grants CA105776 and CA111506, and the Thalma Cohen Gruszka Fund.
Footnotes
Conflict of interest statement: I.L. and A. Korman are employees of Medarex, Inc. J.P.A. is a consultant to Medarex, Inc. F.S.H. has served as a nonpaid consultant for Medarex, Inc., and Bristol–Myers Squibb. F.S.H. receives support for company-sponsored trials from Medarex, Inc., and Bristol–Myers Squibb.
This article contains supporting information online at www.pnas.org/cgi/content/full/0712237105/DC1.
References
- 1.Dranoff G. Cytokines in cancer pathogenesis and cancer therapy. Nat Rev Cancer. 2004;4:11–22. doi: 10.1038/nrc1252. [DOI] [PubMed] [Google Scholar]
- 2.Rabinovich GA, Gabrilovich D, Sotomayor EM. Immunosuppressive strategies that are mediated by tumor cells. Annu Rev Immunol. 2007;25:267–296. doi: 10.1146/annurev.immunol.25.022106.141609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gilboa E. DC-based cancer vaccines. J Clin Invest. 2007;117:1195–1203. doi: 10.1172/JCI31205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hodi FS, Dranoff G. Combinatorial cancer immunotherapy. Adv Immunol. 2006;90:337–360. doi: 10.1016/S0065-2776(06)90009-1. [DOI] [PubMed] [Google Scholar]
- 5.Korman A, Peggs K, Allison JP. Checkpoint blockade in cancer immunotherapy. Adv Immunol. 2006;90:293–335. doi: 10.1016/S0065-2776(06)90008-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Thompson CB, Allison JP. The emerging role of CTLA-4 as an immune attenuator. Immunity. 1997;7:445–450. doi: 10.1016/s1074-7613(00)80366-0. [DOI] [PubMed] [Google Scholar]
- 7.Doyle AM, et al. Induction of cytotoxic T lymphocyte antigen 4 (CTLA-4) restricts clonal expansion of helper T cells. J Exp Med. 2001;194:893–902. doi: 10.1084/jem.194.7.893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Salomon B, Bluestone JA. Complexities of CD28/B7: CTLA-4 costimulatory pathways in autoimmunity and transplantation. Annu Rev Immunol. 2001;19:225–252. doi: 10.1146/annurev.immunol.19.1.225. [DOI] [PubMed] [Google Scholar]
- 9.van Elsas A, Hurwitz A, Allison J. Combination immunotherapy of B16 melanoma using anti-cytotoxic T lymphocyte-associated antigen 4 (CTLA-4) and granulocyte/macrophage colony-stimulating factor (GM-CSF)-producing vaccines induces rejection of subcutaneous and metastatic tumors accompanied by autoimmune depigmentation. J Exp Med. 1999;190:355–366. doi: 10.1084/jem.190.3.355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hurwitz A, Yu T, Leach D, Allison J. CTLA-4 blockade synergizes with tumor-derived granulocyte-macrophage colony-stimulating factor for treatment of an experimental mammary carcinoma. Proc Natl Acad Sci USA. 1998;95:10067–10071. doi: 10.1073/pnas.95.17.10067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hurwitz AA, et al. Combination immunotherapy of primary prostate cancer in a transgenic mouse model using CTLA-4 blockade. Cancer Res. 2000;60:2444–2448. [PubMed] [Google Scholar]
- 12.Quezada SA, Peggs KS, Curran MA, Allison JP. CTLA4 blockade and GM-CSF combination immunotherapy alters the intratumor balance of effector and regulatory T cells. J Clin Invest. 2006;116:1935–1945. doi: 10.1172/JCI27745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Read S, et al. Blockade of CTLA-4 on CD4+CD25+ regulatory T cells abrogates their function in vivo. J Immunol. 2006;177:4376–4383. doi: 10.4049/jimmunol.177.7.4376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Reuben JM, et al. Biologic and immunomodulatory events after CTLA-4 blockade with ticilimumab in patients with advanced malignant melanoma. Cancer. 2006;106:2437–2444. doi: 10.1002/cncr.21854. [DOI] [PubMed] [Google Scholar]
- 15.Phan GQ, et al. Cancer regression and autoimmunity induced by cytotoxic T lymphocyte-associated antigen 4 blockade in patients with metastatic melanoma. Proc Natl Acad Sci USA. 2003;100:8372–8377. doi: 10.1073/pnas.1533209100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Attia P, et al. Autoimmunity correlates with tumor regression in patients with metastatic melanoma treated with anti-cytotoxic T-lymphocyte antigen-4. J Clin Oncol. 2005;23:6043–6053. doi: 10.1200/JCO.2005.06.205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Maker AV, et al. Intrapatient dose escalation of anti-CTLA-4 antibody in patients with metastatic melanoma. J Immunother (1997) 2006;29:455–463. doi: 10.1097/01.cji.0000208259.73167.58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ribas A, et al. Antitumor activity in melanoma and anti-self responses in a phase I trial with the anti-cytotoxic T lymphocyte-associated antigen 4 monoclonal antibody CP-675,206. J Clin Oncol. 2005;23:8968–8977. doi: 10.1200/JCO.2005.01.109. [DOI] [PubMed] [Google Scholar]
- 19.Sanderson K, et al. Autoimmunity in a phase I trial of a fully human anti-cytotoxic T-lymphocyte antigen-4 monoclonal antibody with multiple melanoma peptides and Montanide ISA 51 for patients with resected stages III, IV melanoma. J Clin Oncol. 2005;23:741–750. doi: 10.1200/JCO.2005.01.128. [DOI] [PubMed] [Google Scholar]
- 20.Beck KE, et al. Enterocolitis in patients with cancer after antibody blockade of cytotoxic T-lymphocyte-associated antigen 4. J Clin Oncol. 2006;24:2283–2289. doi: 10.1200/JCO.2005.04.5716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Read S, Malmstrom V, Powrie F. Cytotoxic T lymphocyte-associated antigen 4 plays an essential role in the function of CD25+CD4+ regulatory cells that control intestinal inflammation. J Exp Med. 2000;192:295–302. doi: 10.1084/jem.192.2.295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kapadia D, Fong L. CTLA-4 blockade: Autoimmunity as treatment. J Clin Oncol. 2005;23:8926–8928. doi: 10.1200/JCO.2005.07.012. [DOI] [PubMed] [Google Scholar]
- 23.Ernstoff MS. Self-recognition and tumor response to immunotherapy. J Clin Oncol. 2005;23:5875–5877. doi: 10.1200/JCO.2005.95.029. [DOI] [PubMed] [Google Scholar]
- 24.Hodi FS, et al. Biologic activity of cytotoxic T lymphocyte-associated antigen 4 antibody blockade in previously vaccinated metastatic melanoma and ovarian carcinoma patients. Proc Natl Acad Sci USA. 2003;100:4712–4717. doi: 10.1073/pnas.0830997100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Miyara M, et al. The immune paradox of sarcoidosis and regulatory T cells. J Exp Med. 2006;203:359–370. doi: 10.1084/jem.20050648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sato E, et al. Intraepithelial CD8+ tumor-infiltrating lymphocytes and a high CD8+/regulatory T cell ratio are associated with favorable prognosis in ovarian cancer. Proc Natl Acad Sci USA. 2005;102:18538–18543. doi: 10.1073/pnas.0509182102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Waterhouse P, et al. Lymphoproliferative disorders with early lethality in mice deficient in Ctla-4. Science. 1995;270:985–988. doi: 10.1126/science.270.5238.985. [DOI] [PubMed] [Google Scholar]
- 28.Tivol EA, et al. Loss of CTLA-4 leads to massive lymphoproliferation and fatal multiorgan tissue destruction, revealing a critical negative regulatory role of CTLA-4. Immunity. 1995;3:541–547. doi: 10.1016/1074-7613(95)90125-6. [DOI] [PubMed] [Google Scholar]
- 29.Boon T, Coulie PG, Van den Eynde BJ, van der Bruggen P. Human T cell responses against melanoma. Annu Rev Immunol. 2006;24:175–208. doi: 10.1146/annurev.immunol.24.021605.090733. [DOI] [PubMed] [Google Scholar]
- 30.Sjoblom T, et al. The consensus coding sequences of human breast and colorectal cancers. Science. 2006;314:268–274. doi: 10.1126/science.1133427. [DOI] [PubMed] [Google Scholar]
- 31.Thomas RK, et al. High-throughput oncogene mutation profiling in human cancer. Nat Genet. 2007;39:347–351. doi: 10.1038/ng1975. [DOI] [PubMed] [Google Scholar]
- 32.Ueda H, et al. Association of the T-cell regulatory gene CTLA4 with susceptibility to autoimmune disease. Nature. 2003;423:506–511. doi: 10.1038/nature01621. [DOI] [PubMed] [Google Scholar]
- 33.Michaelis LC, Ratain MJ. Measuring response in a post-RECIST world: From black and white to shades of grey. Nat Rev Cancer. 2006;6:409–414. doi: 10.1038/nrc1883. [DOI] [PubMed] [Google Scholar]
- 34.Smyth MJ, Dunn GP, Schreiber RD. Cancer immunosurveillance and immunoediting: the roles of immunity in suppressing tumor development and shaping tumor immunogenicity. Adv Immunol. 2006;90:1–50. doi: 10.1016/S0065-2776(06)90001-7. [DOI] [PubMed] [Google Scholar]
- 35.Sutmuller RP, et al. Synergism of cytotoxic T lymphocyte-associated antigen 4 blockade and depletion of CD25+ regulatory T cells in antitumor therapy reveals alternative pathways for suppression of autoreactive cytotoxic T lymphocyte responses. J Exp Med. 2001;194:823–832. doi: 10.1084/jem.194.6.823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Jinushi M, et al. MFG-E8 mediated uptake of apoptotic cells by APCs links the pro- and anti-inflammatory activities of GM-CSF. J Clin Invest. 2007;117:1902–1913. doi: 10.1172/JCI30966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Jinushi M, Hodi FS, Dranoff G. Therapy-induced antibodies to MHC class I chain-related protein A antagonize immune suppression and stimulate antitumor cytotoxicity. Proc Natl Acad Sci USA. 2006;103:9190–9195. doi: 10.1073/pnas.0603503103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hirano N, et al. Autoantibodies frequently detected in patients with aplastic anemia. Blood. 2003;102:4567–4575. doi: 10.1182/blood-2002-11-3409. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.