Abstract
Whole-cell patch-clamp recordings of iontophoretically induced N-methyl-D-aspartate (NMDA) receptor-mediated currents (INMDA) in CA1 pyramidal cells in hippocampal slices from 1- to 40-day-old rats were used to characterize developmental changes in the Mg2+ sensitivity of NMDA receptors.
The dose-response relations for extracellular Mg2+ blockade of INMDA indicated a high affinity binding of Mg2+ to NMDA receptors at membrane potentials more negative than −60 mV, independent of postnatal age.
Depolarizing the cells unblocked NMDA receptors by decreasing their affinity for Mg2+. The efficacy of depolarization in unblocking NMDA receptors markedly increased after postnatal day 4 (P4), endowing the receptors with a greater voltage dependence.
The NR2B subunit-specific NMDA antagonist ifenprodil reduced INMDA in pyramidal cells of all ages. The sensitivity of INMDA to ifenprodil was greatest during the first postnatal week and decreased thereafter, indicating an enhanced contribution of NR2B subunit-containing NMDA receptors to INMDA in the first week after birth.
In the first postnatal week, the ifenprodil-insensitive INMDA component had a lower voltage dependence than the total INMDA. In older pyramidal cells, the voltage dependence of the ifenprodil-insensitive component and the total INMDA were similar.
In another set of CA1 pyramidal cells, single-cell reverse transcription and polymerase chain reaction (RT-PCR) were used to characterize concomitant developmental changes in NMDA subunit mRNA expression. The mRNA for the NR2D subunit was detected during the first postnatal week in 50% of the cells and disappeared thereafter. The proportion of cells expressing the NR2A and NR2B subunits remained relatively constant throughout the first five postnatal weeks.
We conclude that NMDA receptors in hippocampal CA1 pyramidal cells are effectively blocked by Mg2+ at all ages. After 4 days they become much less sensitive to Mg2+ at depolarized membrane potentials. This postnatal switch in voltage control of Mg2+ binding to NMDA receptors may be due to the downregulation of NR2D subunit expression in developing CA1 pyramidal cells.
The N-methyl-D-aspartate (NMDA) subtype of glutamate receptors plays a major role in central synaptic transmission (e.g. Armstrong James et al. 1993) and in various forms of neuronal plasticity (Collingridge & Singer, 1990; Constantine Paton et al. 1990; Bliss & Collingridge, 1993). The NMDA receptor is unique among the glutamate receptors in its voltage-dependent blockade by Mg2+ (Mayer et al. 1984; Nowak et al. 1984). It has been suggested that the sensitivity of NMDA receptors to blockade by Mg2+ (and hence to membrane potential) is absent or low at birth and increases thereafter (Bowe & Nadler, 1990; Kleckner & Dingledine, 1991). The resultant enhanced activity of NMDA receptors presumably underlies the increased susceptibility to NMDA receptor-mediated long-term potentiation during the first postnatal weeks (Harris & Teyler, 1984). However, in more recent studies, developmental changes in the Mg2+ sensitivity of NMDA receptors were not observed (Strecker et al. 1994; Khazipov et al. 1995; Kirson & Yaari, 1996).
Native NMDA receptors are believed to be multimeric proteins (for review see Mori & Mishina, 1995). Six subunits of the NMDA receptor have been cloned, namely NR1, NR2A-D and NR3A in the rat (Moriyoshi et al. 1991; Das et al. 1998) and the homologous ζ1, ε1–4 and χ1 in the mouse (Kutsuwada et al. 1992; Ciabarra et al. 1995). It has been shown that coexpression in host cells of the NR1/ζ1 subunit with any member of the NR2/ε family will form functional NMDA receptors (Mori & Mishina, 1995). The exact role of the NR3A/χ1 subunit has not yet been defined. However, the four NR1/ζ1-NR2/ε heterodimeric receptors differ in several functional properties, such as Mg2+ sensitivity, offset kinetics of glutamate-induced currents and affinity for different antagonists and modulators (Monyer et al. 1992, 1994; Williams, 1993; Williams et al. 1993). With respect to Mg2+ sensitivity, it was found that recombinant heterodimeric NMDA receptors containing the NR2A or NR2B subunits are more strongly blocked by Mg2+ than those containing the NR2C or NR2D subunits (Monyer et al. 1994; Kuner & Schoepfer, 1996). Given that the expression profile of the different NMDA receptor subunits throughout the brain changes with postnatal development (Mori & Mishina, 1995), it is possible that developmental changes in the Mg2+ sensitivity of NMDA receptors, if they exist, are a consequence of altered NMDA receptor subunit gene expression. Indeed, the developmental shortening of NMDA receptor-mediated excitatory postsynaptic currents has been shown to correlate with the postnatal increase in the fraction of NR2A subunit-containing receptors in both hippocampus (Kirson & Yaari, 1996) and neocortex (Flint et al. 1997).
In this study, we characterized quantitatively the postnatal development of NMDA receptor Mg2+ sensitivity. We describe an early postnatal increase in the voltage dependence of Mg2+ binding to NMDA receptors. This switch is paralleled by a decrease in the level of NR2D subunit mRNA expression. Therefore, the expression of NR2D may provide a molecular basis for the low voltage dependence of NMDA receptors early in postnatal development.
METHODS
Slice preparation
Experiments were performed on thin hippocampal slices obtained from 1- to 40-day-old Wistar rats. Methods for preparation of thin slices were similar to those described previously (Edwards et al. 1989; Kirson & Yaari, 1996). Briefly, rats were anaesthetized with ether and decapitated with a guillotine, according to guidelines of The Hebrew University Animal Care and Use Committee. The brain was removed and immediately immersed in ice-cold oxygenated (95% O2-5% CO2) dissection saline solution. The caudal two-thirds of one hemisphere (containing one hippocampus) were glued to the stage of a vibratome (Campden Instruments, UK). Transverse slices, 250 μm thick, were cut from the region of the hemisphere containing the anterior hippocampus. The hippocampal portion was dissected out of each slice and transferred to an incubation chamber containing the oxygenated incubation saline solution at 34°C. After an incubation period of 1 h, slices were transferred, one at a time, to a recording chamber, where they were continuously perfused (2·5 ml min−1) with oxygenated experimental saline solution at room temperature (21–24°C).
Solutions
The dissection saline solution consisted of (mM): NaCl, 125; KCl, 2·5; NaHCO3, 26·7; Hepes, 13; NaH2PO4, 1·25; glucose, 6·3; CaCl2, 0·5; MgSO4, 4 (pH 7·3). The incubation saline solution was identical except for NaHCO3, which was 22·5 mM (to maintain pH 7·3 at 34°C). The standard experimental saline solution contained (mM): NaCl, 125; KCl, 2·5; NaHCO3, 26·7; CaCl2, 2·5; Hepes, 13; glucose, 12·5 (pH 7·3; osmolarity 300 mosmol l−1). MgCl2 was added at the indicated concentrations. The minimal concentration of MgCl2 tested was 30 μM in order to ensure that micromolar contamination of the solution by Mg2+ from the slice, estimated to be ∼2 μM (Perouansky & Yaari, 1993) would not influence the results. All saline solutions also contained 6-cyano-7-nitro-quinoxaline-2,3-dione (CNQX; 5 μM) to block non-NMDA receptors, tetrodotoxin (TTX; 0·4 μM) to block Na+ currents, CdCl2 (100 μM) to block Ca2+ currents, bicuculline methiodide (10 μM) to block GABA-mediated Cl− currents and glycine (5 μM) to saturate the glycine binding sites in NMDA receptors. The intracellular (pipette) solution consisted of (mM): CsF, 110; tetraethylamonium (TEA), 20; 1,2-bis(2-aminophenoxy)ethane-N,N,N′,N′-tetraacetic acid (BAPTA), 10; Hepes, 10; MgCl2, 2; CaCl2, 1 (pH 7·2; osmolarity ∼280 mosmol l−1). All drugs were purchased from Sigma Chemical Co. (Israel) with the exception of CNQX, which was from Tocris Neuramin (UK).
Whole-cell recordings
Cells in the CA1 field were visualized at ×640 magnification with Nomarski optics using a Zeiss Axioskop microscope. Pyramidal cells were identified by their position in the pyramidal layer, the pyramidal shape of their somata and their prominent apical dendrite. In a few slices, the apical dendritic arbors of the cells were severed using a sharp micropipette. The micropipette was dragged across the slice just apical to the pyramidal cell layer, and the slices were allowed to recuperate for at least an hour.
Recording pipettes were pulled from borosillicate glass on a vertical puller (List-medical, Germany) and coated with Sylgard resin (Dow Corning Chemical Co., USA). Pipette resistances were 1–5 MΩ when filled with intracellular solution. After establishing whole-cell recording configuration, series resistance was compensated for by setting the series resistance compensation control of the amplifier (Axopatch 200A) to 70–90%. Experiments in which the series resistance exceeded 20 MΩ were discarded.
Iontophoresis
A two-barrel electrode was used for NMDA iontophoresis. One barrel (∼100 MΩ) contained 100 mM NMDA and 50 mM NaCl; the second barrel (∼20 MΩ) contained 150 mM NaCl. The tip of the electrode was placed near to the soma of the patched cell. Iontophoretic ejection currents of ∼150 nA were applied for 60 s. A retaining current of up to 10 nA was used to avoid leak of NMDA.
Analysis and experimental protocol
Currents recorded were filtered on-line at 1 kHz, digitized at a sampling rate of 3 kHz and analysed off-line using a Pentium personal computer and software from Axon Instruments (USA). Analysis was performed on averages from two or three consecutive responses to voltage ramps.
The relation between NMDA-induced current (INMDA) and membrane voltage (INMDA-V relation) was obtained for each cell at four different Mg2+ concentrations using the following protocol (Fig. 1). The patched cell was held at an inter-episode membrane potential of −60 mV. The membrane potential was stepped to +30 mV for at least 1 s to allow for maximal inactivation of voltage-gated currents. Subsequently, the holding potential (Vh) was shifted to −100 mV at 0·1 mV ms−1 and then returned to −60 mV. The protocol was first executed in control saline solution, and then repeated after reaching a steady-state response to NMDA iontophoresis (Fig. 1A). The currents recorded in control condition were subtracted from those recorded during NMDA application. The subtracted currents for each Mg2+ concentration were normalized to the maximal outward current at a membrane potential of +30 mV. Finally, the +30 to −100 mV ramp portion of the resulting currents was plotted versus membrane potential (Fig. 1A).
Figure 1. Experimental protocol and analysis.

A, whole-cell patch-clamp recordings from an exemplary P21 pyramidal cell. The cell was held at +30 mV and stepped to −100 mV with a slow 1 s ramp. This protocol was repeated before and during a steady-state iontophoretic NMDA current. The Mg2+ concentration in the saline solution was 0·03, 0·1, 0·3 or 1 mM Mg2+. B, the control current trace was subtracted from the traces recorded in the presence of NMDA. The resultant INMDA values were then normalized to the peak outward current at +30 mV and plotted versus membrane potential. C, the normalized traces from B were sampled at 30 discrete voltage values. These values were normalized to a linear regression of the area of maximal NMDA conductance (dashed line in B), representing the predicted current values in Mg2+-free saline solution (Imax). At each membrane potential the normalized values (Inorm) were plotted versus the Mg2+ concentration in which they were recorded, and fitted with a simple dose-response curve (see Methods). Exemplary dose-response curves at −64 and −41 mV (▾ and •, respectively) are shown in C. The Kd value at each potential is plotted as a dotted line. D, the Kd values obtained above were plotted versus the corresponding membrane potential. The dotted lines represent the exemplary voltages from C. The Kd-V relation was fitted with both first (between −80 and −30 mV) and second order (over the entire voltage range) linear regressions (continuous lines; see Methods).
The INMDA-V relations obtained in each cell were reduced to thirty membrane potential samples each. The INMDA at each potential was then normalized to the predicted maximal current in the presence of 0 mM Mg2+ (Imax). The Imax values were obtained by fitting a linear regression line to the area of maximal NMDA conductance in the measured INMDA-V relations (∼-10 to +30 mV) and extrapolating it to the sampled membrane potentials (Fig. 1A). For each of these potentials, the relation between normalized INMDA (Inorm) and extracellular Mg2+ concentration ([Mg2+]o) was fitted using a simple dose-response equation (Fig. 1A):
where Inorm=INMDA/Imax, [Mg2+]o is the extracellular Mg2+ concentration and Kd is the [Mg2+]o of half-maximal inhibition. We used a Hill coefficient of unity, in concordance with a previous study of Mg2+ binding to different recombinant NMDA receptors (Kuner & Schoepfer, 1996), in order to fit Inorm to the four different [Mg2+]o (0·03, 0·1, 0·3 and 1 mM). The fit was constrained to Inorm= 1 (no Mg2+ block) at 0 mM [Mg2+]o. The Kd values thus obtained were plotted against membrane potential on a semi-logarithmic scale for each cell.
These Kd-V relations deviated slightly from linearity, as previously described for NMDA receptors expressed in Xenopus oocytes injected with native hippocampal mRNA (Kleckner & Dingledine, 1991) or with recombinant NMDA receptor subunit cRNA (Kuner & Schoepfer, 1996). They were fitted empirically with a second order linear regression (Fig. 1A):
where Vh is the membrane holding potential and a, b and c are constants.
In order to asses the relative position of the Mg2+ binding site within the transmembrane electric field, the voltage dependence of Mg2+ block of NMDA receptors was quantified by fitting the Woodhull equation (Woodhull, 1973) to the Kd-V relations:
where δ is the fraction of the electric field experienced by a Mg2+ ion, z is valency, V is membrane potential, Kd(0mV) is the affinity of the NMDA receptor for Mg2+ at 0 mV and R, T and F have their usual meanings. Because the Woodhull equation assumes linearity, we fitted only the −80 to −30 mV portion of our data where a good approximation of linearity could be assumed and the strength of fit of the Inorm-[Mg2+]o relations was best (r2 > 0·95; see also Kuner & Schoepfer, 1996).
Statistics
Data are presented as means ±s.e.m. Significant differences between groups of samples were tested with either ANOVA with subsequent post hoc testing, Student's t test, or the Mann-Whitney U test. A significance level of α= 0·05 was used in all tests. Fitting procedures used the Marquardt-Levenberg algorithm to seek parameter values that minimized the sum of the squared differences between the observed and predicted values of the dependent variables.
Single-cell RT-PCR
In order to characterize the expression profile of NR2 subunit mRNA at different stages of development, pyramidal cell contents were harvested from a separate set of cells as described previously (Plant et al. 1997). Briefly, cells were harvested by applying steady negative pressure to the pipette in the whole-cell recording mode. The flow of the cytoplasm with or without the nucleus into the pipette was monitored visually. In some experiments a larger diameter pipette (2–3 μm) was pressed to the cell surface and the whole soma was aspired without previously obtaining a tight seal. In some of the latter experiments (n = 6), equivalent volumes of extracellular material were harvested from the same slices. No subunit expression was found in these samples, ruling out contamination of the pipette with mRNA from the debris of neighbouring cells. No differences were found between results obtained in the two different harvesting protocols. Under a dissecting microscope, the pipette tip was broken and the contents of the pipette expelled into a PCR tube by applying a positive pressure pulse (N2, 200–400 kPa). The tube contained 2·5 μl of a solution of hexamer random primers (final concentration 5 μM), the four deoxyribonucleotide triphosphates (final concentration 0·5 mM) and dithiothreitol (final concentration 10 mM), to which 20 units of ribonuclease inhibitor and 100 units of Moloney murine leukaemia virus reverse transcriptase (GIBCO-BRL, Eggenstein, Germany) were added (for details see Lambolez et al. 1992; Audinat et al. 1994). The ∼10 μl of reaction mix was incubated at 37°C for 1 h to synthesize single-stranded cDNA, then stored at −80°C until used for PCR amplification. Controls with water and with total RNA, prepared using a guanidium thiocyanate single-step method (Chomczynski & Sacchi, 1987), from the cerebellum of 15-day-old rats were performed in parallel.
The PCR amplification of NR2A-D was performed as described by Audinat et al. 1994. To test the same cell for NR2A-C and NR2D, the product of the RT reaction was divided into equal amounts and one half used for the PCR amplification of NR2A-C and the other for NR2D.
RESULTS
Developmental changes in voltage dependence of NMDA currents
To test whether the affinity of NMDA receptors for Mg2+ is developmentally regulated, we compared INMDA values in CA1 pyramidal cells from postnatal day 1 (P1) to postnatal day 40 (P40). Significant differences in INMDA-V relations were observed only in the first postnatal week. Therefore, the results from 37 neurones were grouped as follows: P1-P4, P5-P7, P8-P14 and P15-adult. Exemplary (upper panels) and averaged (lower panels) INMDA-V curves for these four age groups are shown in Fig. 2. It is evident from the INMDA-V curves in the youngest group (Fig. 2A), that NMDA receptors are already highly sensitive to extracellular Mg2+ at birth. Thus at membrane potentials negative to −25 mV, INMDA decreased as [Mg2+]o was elevated. However, the efficacy of depolarization in relieving the Mg2+ block, i.e. its voltage dependence, was much less apparent in P1-P4 than in the older age groups (Fig. 2A–D). Thus, in the neurone shown in Fig. 2Aa, depolarization from −60 to −30 mV in 1 mM Mg2+ caused only a small increase in INMDA, whereas a similar change in older neurones (Fig. 2Ba–Da) caused a greater than 5-fold increase in INMDA. The lesser voltage dependence of Mg2+ block of NMDA currents in the youngest neurones compared to those older than P4 was also apparent in the averaged INMDA-V relations (compare Ab to Bb-Db in Fig. 2). This consistent finding is further illustrated in Fig. 3 by superimposing the averaged INMDA-V relations in 0·1 mM (Fig. 3A) and 1 mM Mg2+ (Fig. 3A).
Figure 2. Postnatal changes in INMDA-V relations.

Exemplary (panel a) and averaged (means ±s.e.m.; panel b) INMDA-V relations. These relations were recorded from CA1 pyramidal cells from four different age groups: P1-4 (A; n = 5), P5-7 (B; n = 9), P8-14 (C; n = 8) and P15-adult (D; n = 15). In each neurone the INMDA-V relations were recorded in saline solution containing 0·03, 0·1, 0·3 and 1 mM Mg2+. Currents were normalized to the maximal outward current at +30 mV. Note that depolarizing the cell in the presence of > 0·1 mM Mg2+ caused almost no change in the Mg2+ block of INMDA in the youngest age group (A), whereas in the older age groups (B–D) depolarizing the cells from −60 to −20 mV caused a 3- to 5-fold increase in INMDA. The INMDA-V relations recorded from cells from the three older groups are almost identical.
Figure 3. Developmental profile of INMDA-V relations.

Average INMDA-V relations recorded from CA1 pyramidal cells in the presence of 0·1 mM (A) and 1 mM Mg2+ (B) from the four different age groups. Error bars are omitted for clarity. In the P1-P4 age group (•), depolarizing the cells from −90 to −20 mV caused almost no increase in the INMDA, indicating low voltage dependence of the Mg2+ block. In the P5-P7 groups (▾) an increase in the voltage dependence is seen, leading to a large increase in INMDA upon depolarization. Further changes in voltage dependence are not seen in the P8-P14 (▪) and the P15-adult groups (♦).
In order to quantify the developmental changes in the voltage dependence of NMDA receptors, we compared the Kd-V relations obtained for the four age groups studied (Fig. 4). At membrane potentials negative to −60 mV no significant differences in Kd of Mg2+ were found between the different age groups (P > 0·05, Mann-Whitney U test). In all cells tested, Kd was significantly dependent on membrane potential (P < 0·05, ANOVA). The Kd-V relations were nearly linear when plotted on a semi-logarithmic scale, though a better fit was achieved using a second order regression (Fig. 4; see Methods). The Kd-V relation of the first age group was significantly less voltage dependent than those of the older age groups (Fig. 4 and Table 1; P < 0·005, ANOVA). A slight decrease in voltage dependence of Mg2+ blockade was again seen after the second postnatal week (Fig. 4 and Table 1; P < 0·05, ANOVA).
Figure 4. Developmental profile of the voltage dependence of NMDA receptors.
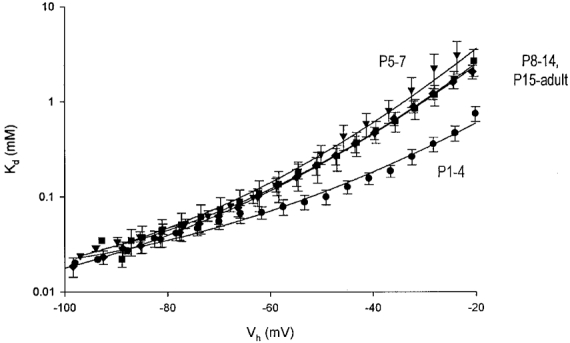
The average Kd-V relations calculated for all four age groups are shown superimposed on a semilogarithmic scale (means ±s.e.m.; P1-P4, •; P5-P7, ▾; P8-P14, ▪; P15-adult, ♦). The relations were fitted with a second order linear regression as described in Methods (continuous lines). A significant increase in Kd is seen after P4 (P < 0·05; ANOVA). The slope of the Kd-V relations increased after P4 and did not change significantly thereafter.
Table 1.
Comparison of NMDA receptor sensitivity to Mg2+ during development in the presence and absence of ifenprodil
| Control | 10 μm ifenprodil | |||
|---|---|---|---|---|
| δ | Kd(0mV) (mm) | δ | Kd(0mV) (mm) | |
| P1–P4 | 0.65 ± 0.04(5) | 1.4 ± 0.2(5) | — | — |
| P5–P7 | 1.10 ± 0.04(9) | 21 ± 2.2(9) | 0.79 ± 0.02(5) | 3.6 ± 0.3(5) |
| P8–P14 | 0.95 ± 0.02(8) | 9.3 ± 0.6(8) | 0.88 ± 0.04(6) | 7.2 ± 0.8(6) |
| P15–adult | 0.99 ± 0.02(15) | 10.7 ± 0.5(15) | 1.1 ± 0.08(8) | 21 ± 4.5(8) |
* Data are presented as means ± s.e.m. The number of cells in each group is shown in parentheses.
To estimate the Kd values for Mg2+ block in the absence of an electric field (Kd(0mV)) and the fraction of the electric field felt by the blocking Mg2+ ions (δ), we fitted the linear (-80 to −30 mV) portion of the Kd-V relations with the Woodhull equation and extrapolated it to 0 mV (see Methods; Woodhull, 1973). In the youngest neurones (P1-P4), Kd(0mV) was 1·4 mM and δ was 0·65, whereas in neurones older than P4, Kd(0mV) was greater than 9 mM and δ was ∼1·0 (Fig. 4; Table 1).
Taken together, these data suggest that at birth Mg2+ blocks NMDA receptor channels with high affinity but the voltage dependence of this block is low. After P4, the affinity of Mg2+ for the NMDA receptor channels decreases somewhat, whereas its voltage dependence is markedly augmented.
Evaluation of space-clamp errors
During postnatal development the dendritic arbors of CA1 pyramidal cells increase in both size and complexity (Pokorny & Yamamoto, 1981; Kirson & Yaari, 1996). Even though we applied NMDA to the somata of the cells, age-dependent changes in the quality of space-clamp control could possibly confound our results. In order to evaluate this possibility, we compared data obtained from intact adult pyramidal cells to that obtained from pyramidal cells whose apical dendrites were severed about 50 μm from the soma (see Methods). No significant differences were found in the INMDA-V relations (Fig. 5A and B) and corresponding Kd-V relations (Fig. 5A) obtained from the two groups of pyramidal cells (P = 0·97, ANOVA).
Figure 5. Evaluation of space-clamp control.

INMDA-V relations were recorded from adult pyramidal cells at four Mg2+ concentrations (0·03, 0·1, 0·3, 1 mM). In some cells the apical dendrite was severed selectively. Exemplary INMDA-V relations are shown from a control cell (A) and from a cell with a severed apical dendrite (B). C, average Kd-V relations (means ±s.e.m.) from cells with severed apical dendrites (○; n = 4) and from control cells (•; n = 9). No differences in NMDA receptor voltage dependence were seen between the two groups (P = 0·97; ANOVA).
Effect of ifenprodil
The switch in voltage dependence of Mg2+ block of NMDA receptors may reflect a developmental change in the subunit composition of receptors. If that is the case, then the voltage dependence of the INMDA component that is sensitive to subunit-selective NMDA receptor antagonists may differ from the voltage dependence of the insensitive INMDA component. We tested this notion with ifenprodil, an NMDA receptor antagonist that preferentially blocks NR2B subunit-containing NMDA receptors (Williams, 1993, 1995; Williams et al. 1993; Hess et al. 1998). We reasoned that because the latter receptors are highly voltage dependent (Monyer et al. 1994; Kuner & Schoepfer, 1996), then blocking them selectively with ifenprodil may unmask an INMDA component which is less sensitive to voltage. This would be expected if NR2C or NR2D subunit-containing NMDA receptors contribute substantially to the total INMDA, given that depolarization is less effective in removing the Mg2+ block of these receptors (Kuner & Schoepfer, 1996).
Bath application of 10 μM ifenprodil (a concentration which entirely blocks recombinant NR2B subunit-containing NMDA receptors; Williams, 1993; Williams et al. 1993), significantly reduced INMDA values (P < 0·05, t test). This effect was maximal during the first postnatal week and decreased thereafter (Fig. 6). Thus, the magnitude of ifenprodil block of INMDA for the P1-P4, P5-P7, P8-P14 and P15-adult age groups was 85, 84, 53 and 28%, respectively. This age-dependent effect of ifenprodil is consistent with our previous report of a postnatal decrease in the ifenprodil sensitivity of synaptic NMDA receptors in CA1 pyramidal cells (Kirson & Yaari, 1996), presumably consequent to a reduction in hippocampal NR2B mRNA expression after birth (Monyer et al. 1994; Mori & Mishina, 1995; Zhong et al. 1995).
Figure 6. Developmental profile of NMDA receptor block by ifenprodil.

The INMDA values were recorded in the presence of nominally Mg2+-free saline solution. Ifenprodil (10 μM) blocked NMDA currents significantly (P < 0·05; Mann-Whitney U test) in all cells tested. The average ratio of INMDA in the presence of ifenprodil divided by control INMDA is plotted versus postnatal day. The extent of inhibition decreased dramatically after P9, reaching minimal values at about P15.
We then compared INMDA-V relations obtained in control saline solution and in the presence of 10 μM ifenprodil in the age groups P5-P7, P8-P14 and P15-adult (n = 19). This comparison could not be done for P1-P4, because INMDA values obtained at this age were very small (< 50 pA) and, as shown above (Fig. 6), were almost completely blocked by 10 μM ifenprodil. The sensitivity of NMDA receptors to extracellular Mg2+ persisted in the presence of 10 μM ifenprodil (Fig. 7). However, unlike the Kd-V relations in control saline solution (Fig. 4), the voltage dependence of the Mg2+ block was less apparent in the P5-P7 group than in the older age groups (Fig. 7A). Indeed, the Kd-V relations from P5-P7 cells in the presence of 10 μM ifenprodil were similar to those from control P1-P4 cells, and differed significantly (P < 0·05, ANOVA) from control P5-P7 cells (control P1-P4, δ= 0·65; ifenprodil P5-P7, δ= 0·79; control P5-P7, δ= 1·1; Table 1 and Fig. 7A). In P8-P14 cells and in P15-adult cells, the Kd-V relations of the ifenprodil-insensitive INMDA component were not significantly different from the control Kd-V relations (Table 1 and Fig. 7A).
Figure 7. Effect of ifenprodil on the voltage dependence of NMDA receptors.

A, the average Kd-V relations obtained from neurones in the P5-P7 (•), P8-P14 (▾) and > P15 (▪) age groups are shown superimposed on a semilogarithmic scale (means ±s.e.m.). The relations were fitted with a second order linear regression as described in Methods (continuous lines). B, the P5-P7 average Kd-V relation obtained in the presence of ifenprodil (•) was similar to the P1-P4 average Kd-V relation obtained in control saline solution (lower continuous line; P = 0·99, ANOVA), but differed significantly from the P5-P7 average Kd-V relation obtained in control saline solution (upper continuous line; P < 0·05, ANOVA). C, the P8-P14 average Kd-V relations obtained in the presence of ifenprodil (▾) and in control saline solution were identical (continuous line; P = 0·99, ANOVA).
Single-cell RT-PCR of NMDA receptor subunits
The lesser voltage dependence of Mg2+ binding to NMDA receptors at P1-P4 compared to ≥P5 may be imparted by NMDA receptors comprising the NR2C or NR2D subunits, which are less sensitive to Mg2+ block than those containing the NR2A or NR2B subunits (Monyer et al. 1992, 1994; Kuner & Schoepfer, 1996). However, in situ hybridization for NMDA receptor subunits disclosed only NR2A and NR2B mRNA in postnatal CA1 pyramidal cells (Watanabe et al. 1992; Monyer et al. 1994). We examined this issue using single cell RT-PCR (see Methods). Separate groups of single CA1 pyramidal cells were harvested in the first (n = 8), third (n = 10) and fifth postnatal weeks (n = 11). After NR2A-C amplification, all cells showed a single band of 547 bp (Fig. 8A and B). The amplified product was cut with Bpm I, Bfa I and Sca I, restriction enzymes specific for the NR2A (226 and 321 bp fragments), NR2B (392 and 155 bp fragments) and NR2C (361 and 186 bp fragments) subunits. After NR2D amplification, less than half the cells from all ages showed a single band of 465 bp (Fig. 8A). In these cells, the amplification product was cut with Xho I, a restriction enzyme specific for the NR2D subunit (283 and 182 bp fragments). Almost all cells expressed the NR2A and NR2B subunits, whereas, less than half expressed the NR2D subunit. The NR2C subunit was found only in one cell.
Figure 8. Expression of NR2 subunits in neonate versus adult pyramidal cells.

Agarose gel electrophoresis of cDNA amplified from single neonate (A) and adult (B) pyramidal cells. Lane 1 (M) low molecular weight marker Hae III digest of ΦX174. Lane 2, single band 547 bp amplified product of the first PCR for NR2A-C. Lanes 3–5, results of digestion with Bpm I, Bfa I and Sca I, specific for NR2A, NR2B and NR2C, respectively, following purification of the product of the first PCR and a second PCR amplification with the same primers. Lane 6, single band 465 bp amplified product of the first PCR for NR2D. Lane 7 (in A), results of digestion with Xho I following purification of the product of the first PCR and a second PCR amplification with the same primer. Note that in the neonate, NR2B and NR2D were detected (A) whereas in the adult, NR2A and NR2B were detected (B). Numbers to the left of the gels are base pairs (bp).
The PCR and restriction analysis results are shown for exemplary first (Fig. 8A) and fifth (Fig. 8A) week pyramidal cells. These cells expressed the NR2B and NR2D subunits and the NR2A and NR2B subunits, respectively. In the first week, 75% of cells expressed NR2A, 100% NR2B and 50% NR2D. In the third week, 100% of cells expressed NR2A, 90% NR2B and 10% NR2D. In the fifth week, 100% of cells expressed NR2A and NR2B and no cells expressed NR2D. The decrease in expression of the NR2D subunit was statistically significant (P < 0·05; Kruskal-Wallis ANOVA). These results are presented graphically in Fig. 9.
Figure 9. Developmental changes in expression of NR2 subunits.

Single pyramidal cells were harvested for RT-PCR and the expression of each of the four NMDA NR2 subunit mRNA was determined in each cell. The fraction of cells expressing each of the NR2 subunits is plotted as a histogram. The cells were grouped by age into first week (n = 8), third week (n = 10) and fifth week (n = 11). The proportion of cells expressing NR2A mRNA (□, A) increases with age while NR2B mRNA expression ( , B) remains relatively constant. NR2D mRNA (
, B) remains relatively constant. NR2D mRNA ( , D) expression decreases from 50% in the youngest age group to nil in the oldest age group. NR2C mRNA (▪, C) was detected in one adult cell only.
, D) expression decreases from 50% in the youngest age group to nil in the oldest age group. NR2C mRNA (▪, C) was detected in one adult cell only.
DISCUSSION
The main finding in this report is that NMDA receptors in CA1 pyramidal cells are highly sensitive to Mg2+ block at birth and that the voltage dependence of this block is developmentally regulated. In the first postnatal days (P1-P4), the Mg2+ block is only slightly affected by depolarization, whereas after P5 it becomes more voltage dependent. This switch in NMDA receptor function may reflect a developmental change in NMDA receptor subunit composition, possibly downregulation of the NR2D subunit.
Developmental changes in Mg2+ block of NMDA receptors in CA1 pyramidal cells
At all ages examined, NMDA receptors in CA1 pyramidal cells were strongly blocked by Mg2+ at membrane potentials more negative than −60 mV. This blockade was relieved with depolarization in all cells tested, albeit in an age-dependent manner. Using the Woodhull model of voltage-dependent channel block (Woodhull, 1973), we calculated δ values ranging between 0·65 and 1·1. Previous studies in both cultured and dissociated brain neurones have shown native NMDA receptors to have similar voltage dependencies (δ= 1, Ascher & Nowak, 1988; δ= 0·8, Jahr & Stevens, 1990; δ= 0·73, Chen & Huang, 1992). These δ values imply that the Mg2+ binding site is situated near the cytoplasmic opening of the receptor channel. It should be noted, however, that δ values greater than one cannot be explained by the Woodhull model. Also, this model predicts a linear relationship between logKd for Mg2+ binding and membrane voltage, whereas our data show that this relationship deviates from linearity (see also Kleckner & Dingledine, 1991; Kuner & Schoepfer, 1996). These discrepancies may be due to displacement of Mg2+ from its binding site by Ca2+ ions (Ascher & Nowak, 1988) or to permeation of the channel by Mg2+ itself (Mayer & Westbrook, 1987).
We found that before P4 the NMDA receptor channel blockade by 1 mM Mg2+ was barely relieved by depolarization (δ= 0·65), whereas after P4 depolarization unblocked the channel more effectively (δ≈ 1). This early developmental change in voltage control of Mg2+ binding to NMDA receptors could be due to a shift of the Mg2+ binding site closer to the cytoplasmic opening of the channel. Such a shift may also account for the concurrent decrease in the affinity of NMDA receptors for Mg2+ in the absence of an electric field (from Kd(0mV) of 1·4 mM in P1-P4 receptors to Kd(0mV) of > 9 mM in receptors older than P5).
Several experimental approaches have been used previously to study developmental changes in the voltage dependence of NMDA receptor blockade by Mg2+. In most studies, the affinity of Mg2+ for the NMDA receptor and its voltage dependence were deduced from INMDA-V curves obtained at only one or two [Mg2+]o, which may account for their contradictory results. Both the NMDA receptor affinity for Mg2+ and its voltage dependence were reported to increase in CA3 pyramidal cells after P10 (Ben-Ari et al. 1988) and in neocortical neurones after P5 (Burgard & Hablitz, 1994). An increase in the voltage dependence of Mg2+ block was seen also in nucleus tractus solitarii neurones after P4 (Nabekura et al. 1994). A delayed (after the second to fourth postnatal weeks) increase in the overall Mg2+ sensitivity of NMDA receptors was found in CA1 pyramidal cells (Bowe & Nadler, 1990; Morrisett et al. 1990) and in other neocortical neurones (Kato & Yoshimura, 1993), but the voltage dependence of this change was not studied. In contrast, in a study in which INMDA-V relations were examined in several different [Mg2+]o, NMDA receptors expressed in oocytes following hippocampal mRNA injection displayed a prominent decrease in their affinity for Mg2+ after P1-P2 without a change in their voltage dependence (Kleckner & Dingledine, 1991). Our present data are consistent with the early postnatal increase in voltage dependence of Mg2+ blockade of NMDA receptors described in some of these studies (Nabekura et al. 1994; Burgard & Hablitz, 1994).
Developmental profile of NMDA receptor subunit expression
Using single-cell RT-PCR, we have shown that NMDA receptor subunit mRNA expression in CA1 pyramidal cells changes with development. As described previously for both mRNA and protein levels (Watanabe et al. 1992; Monyer et al. 1994; Zhong et al. 1995; Portera Cailliau et al. 1996; Wenzel et al. 1997), we found that the fraction of cells expressing NR2A mRNA increased between the first and third postnatal weeks, while the fraction expressing NR2B mRNA remained relatively constant. The NR2C mRNA was not significantly expressed in all age groups. Though previous studies failed to find significant NR2D mRNA expression in CA1 pyramidal cells at any age (Watanabe et al. 1992; Monyer et al. 1994; Wenzel et al. 1996), we found that 50% of these neurones express this mRNA in the first postnatal week. However, this expression declined to nil in the subsequent 4 weeks.
Relationship between voltage dependence and subunit composition of NMDA receptors
The developmental switch in voltage dependence of NMDA receptor Mg2+ blockade may be imparted by alteration in the subunits that assemble to form a receptor. Previous studies have shown that the Mg2+ block of recombinant NMDA receptors containing the NR2D subunit is much less sensitive to voltage (δ= 0·75) than that of heterodimers containing either NR2A or NR2B subunits (δ= 1·05 and 1·07, respectively; Kuner & Schoepfer, 1996). Therefore, the low voltage dependence of P1-P4 NMDA receptors may be related to the transient postnatal expression of the NR2D subunit. Consistent with this hypothesis we found that the INMDA component remaining after the pharmacological block of NR2B subunit-containing NMDA receptors with ifenprodil in P5-P7 pyramidal cells, which still express the NR2D subunit, was less voltage-dependent (δ= 0·79) than the total INMDA (δ= 1·1). This effect was not seen after P7, presumably due to the downregulation of NR2D subunits and the concurrent increase in the fraction of NR2A subunit-containing NMDA receptors, which are much less sensitive to ifenprodil.
It should be noted, however, that the above hypothesis is incongruent with the postnatal decrease in the NMDA receptor affinity for Mg2+ at 0 mV (expressed above as an increase in Kd(0mV)), because all recombinant heterodimeric NMDA receptors were shown to have a similar Kd(0mV) (∼4 mM; Kuner & Schoepfer, 1996). Perhaps native NMDA receptors incorporate more than one type of NR2 subunit, yielding heterotrimeric assemblies with different ligand binding affinities (Wafford et al. 1993; Chazot et al. 1994; Chazot & Stephenson, 1997). Indeed, the formation of heterotrimeric receptors comprising the NR2D and NR2B subunits in the first postnatal days, may also explain why INMDA values in P1-P4 pyramidal cells are almost completely blocked by 10 μM ifenprodil. In this respect, it is interesting that NR2D subunits preferentially assemble with NR2B subunits to form heterotrimeric receptors when the NR1, NR2D and NR2B subunits are coexpressed in oocytes (Buller & Monaghan, 1997).
It has recently been suggested that the NR2B subunit is mainly a component of extrasynaptic NMDA receptors, expressed early in cultured hippocampal neurones, and that the formation of functional synapses coincides with the incorporation of an ifenprodil-insensitive subunit (i.e. NR2A) into the newly formed synaptic receptors (Tovar & Westbrook, 1999). Our data are not sufficient to distinguish between synaptic versus extrasynaptic changes in voltage dependence of NMDA receptors.
Functional implications
It is widely believed that the voltage dependence of Mg2+ block of NMDA receptors plays a critical role in activity-dependent modifications of excitatory synapses that are seen during long-term potentiation and synapse formation (Bliss & Collingridge, 1993; Durand et al. 1996). In the developing hippocampus, activation of synaptic NMDA receptors has been shown to convert ‘silent’ synapses into functional connections by inducing the expression of α-amino-3-hydroxy-5-methyl-4-isoxasoleproprionate (AMPA) receptors (Durand et al. 1996). The low voltage dependence of NMDA receptors in P1-P4 pyramidal cells described here would entail a low propensity for NMDA-dependent synaptic plasticity in the first postnatal days and may impede synapse formation. Thus, the developmental switch from low to high voltage dependence of NMDA receptors, which may be imparted by a decrease in NR2D expression, may be the basis for the accelerated formation and strengthening of functional excitatory synapses in the postnatal hippocampus.
Acknowledgments
This work was supported by the German Israeli Foundation for Scientific Research and Development (GIF), Israeli Ministry of Science and the Israel Science Foundation founded by the Israel Academy of Sciences and Humanities. E.D.K. was supported by fellowships from the Foulkes Foundation, Teva Pharmaceuticals Inc. and EMBO (short term fellowship).
References
- Armstrong James M, Welker E, Callahan CA. The contribution of NMDA and non-NMDA receptors to fast and slow transmission of sensory information in the rat SI barrel cortex. Journal of Neuroscience. 1993;13:2149–2160. doi: 10.1523/JNEUROSCI.13-05-02149.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ascher P, Nowak L. The role of divalent cations in the N-methyl-D-aspartate responses of mouse central neurones in culture. The Journal of Physiology. 1988;399:247–266. doi: 10.1113/jphysiol.1988.sp017078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Audinat E, Lambolez B, Rossier J, Crepel F. Activity-dependent regulation of N-methyl-D-aspartate receptor subunit expression in rat cerebellar granule cells. European Journal of Neuroscience. 1994;6:1792–1800. doi: 10.1111/j.1460-9568.1994.tb00572.x. [DOI] [PubMed] [Google Scholar]
- Ben-Ari Y, Cherubini E, Krnjevic K. Changes in voltage dependence of NMDA currents during development. Neuroscience Letters. 1988;94:88–92. doi: 10.1016/0304-3940(88)90275-3. [DOI] [PubMed] [Google Scholar]
- Bliss TV, Collingridge GL. A synaptic model of memory: long-term potentiation in the hippocampus. Nature. 1993;361:31–39. doi: 10.1038/361031a0. [DOI] [PubMed] [Google Scholar]
- Bowe MA, Nadler JV. Developmental increase in the sensitivity to magnesium of NMDA receptors on CA1 hippocampal pyramidal cells. Developmental Brain Research. 1990;56:55–61. doi: 10.1016/0165-3806(90)90164-t. [DOI] [PubMed] [Google Scholar]
- Buller AL, Monaghan DT. Pharmacological heterogeneity of NMDA receptors: characterization of NR1a/NR2D heteromers expressed in Xenopus oocytes. European Journal of Pharmacology. 1997;320:87–94. doi: 10.1016/s0014-2999(96)00880-1. [DOI] [PubMed] [Google Scholar]
- Burgard EC, Hablitz JJ. Developmental changes in the voltage-dependence of neocortical NMDA responses. Developmental Brain Research. 1994;80:275–278. doi: 10.1016/0165-3806(94)90113-9. [DOI] [PubMed] [Google Scholar]
- Chazot PL, Coleman SK, Cik M, Stephenson FA. Molecular characterization of N-methyl-D-aspartate receptors expressed in mammalian cells yields evidence for the coexistence of three subunit types within a discrete receptor molecule. Journal of Biological Chemistry. 1994;269:24403–24409. [PubMed] [Google Scholar]
- Chazot PL, Stephenson FA. Molecular dissection of native mammalian forebrain NMDA receptors containing the NR1 C2 exon: direct demonstration of NMDA receptors comprising NR1, NR2A, and NR2B subunits within the same complex. Journal of Neurochemistry. 1997;69:2138–2144. doi: 10.1046/j.1471-4159.1997.69052138.x. [DOI] [PubMed] [Google Scholar]
- Chen L, Huang LY. Protein kinase C reduces Mg2+ block of NMDA-receptor channels as a mechanism of modulation. Nature. 1992;356:521–523. doi: 10.1038/356521a0. [DOI] [PubMed] [Google Scholar]
- Chomczynski P, Sacchi N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Annals of Biochemistry. 1987;162:156–159. doi: 10.1006/abio.1987.9999. [DOI] [PubMed] [Google Scholar]
- Ciabarra AM, Sullivan JM, Gahn LG, Pecht G, Heinemann S, Sevarino KA. Cloning and characterization of chi-1: a developmentally regulated member of a novel class of the ionotropic glutamate receptor family. Journal of Neuroscience. 1995;15:6498–6508. doi: 10.1523/JNEUROSCI.15-10-06498.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collingridge GL, Singer W. Excitatory amino acid receptors and synaptic plasticity. Trends in Pharmacological Sciences. 1990;11:290–296. doi: 10.1016/0165-6147(90)90011-v. [DOI] [PubMed] [Google Scholar]
- Constantine Paton M, Cline HT, Debski E. Patterned activity, synaptic convergence, and the NMDA receptor in developing visual pathways. Annual Review of Neuroscience. 1990;13:129–154. doi: 10.1146/annurev.ne.13.030190.001021. [DOI] [PubMed] [Google Scholar]
- Das S, Sasaki YF, Rothe T, Premkumar LS, Takasu M, Crandall JE, Dikkes P, Conner DA, Rayudu PV, Cheung W, Chen HS, Lipton SA, Nakanishi N. Increased NMDA current and spine density in mice lacking the NMDA receptor subunit NR3A. Nature. 1998;393:377–381. doi: 10.1038/30748. [DOI] [PubMed] [Google Scholar]
- Durand GM, Kovalchuk Y, Konnerth A. Long-term potentiation and functional synapse induction in developing hippocampus. Nature. 1996;381:71–75. doi: 10.1038/381071a0. [DOI] [PubMed] [Google Scholar]
- Edwards FA, Konnerth A, Sakmann B, Takahashi T. A thin slice preparation for patch clamp recordings from neurones of the mammalian central nervous system. Pflügers Archiv. 1989;414:600–612. doi: 10.1007/BF00580998. [DOI] [PubMed] [Google Scholar]
- Flint AC, Maisch US, Weishaupt JH, Kriegstein AR, Monyer H. NR2A subunit expression shortens NMDA receptor synaptic currents in developing neocortex. Journal of Neuroscience. 1997;17:2469–2476. doi: 10.1523/JNEUROSCI.17-07-02469.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harris KM, Teyler TJ. Developmental onset of long-term potentiation in area CA1 of the rat hippocampus. The Journal of Physiology. 1984;346:27–48. doi: 10.1113/jphysiol.1984.sp015005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hess SD, Daggett LP, Deal C, Lu CC, Johnson EC, Velicelebi G. Functional characterization of human N-methyl-D-aspartate subtype 1A/2D receptors. Journal of Neurochemistry. 1998;70:1269–1279. doi: 10.1046/j.1471-4159.1998.70031269.x. [DOI] [PubMed] [Google Scholar]
- Jahr CE, Stevens CF. A quantitative description of NMDA receptor-channel kinetic behavior. Journal of Neuroscience. 1990;10:1830–1837. doi: 10.1523/JNEUROSCI.10-06-01830.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kato N, Yoshimura H. Reduced Mg2+ block of N-methyl-D-aspartate receptor-mediated synaptic potentials in developing visual cortex. Proceedings of the National Academy of Sciences of the USA. 1993;90:7114–7118. doi: 10.1073/pnas.90.15.7114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khazipov R, Ragozzino D, Bregestovski P. Kinetics and Mg2+ block of N-methyl-D-aspartate receptor channels during postnatal development of hippocampal CA3 pyramidal neurons. Neuroscience. 1995;69:1057–1065. doi: 10.1016/0306-4522(95)00337-i. [DOI] [PubMed] [Google Scholar]
- Kirson ED, Yaari Y. Synaptic NMDA receptors in developing mouse hippocampal neurones: functional properties and sensitivity to ifenprodil. The Journal of Physiology. 1996;497:437–455. doi: 10.1113/jphysiol.1996.sp021779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kleckner NW, Dingledine R. Regulation of hippocampal NMDA receptors by magnesium and glycine during development. Molecular Brain Research. 1991;11:151–159. [PubMed] [Google Scholar]
- Kuner T, Schoepfer R. Multiple structural elements determine subunit specificity of Mg2+ block in NMDA receptor channels. Journal of Neuroscience. 1996;16:3549–3558. doi: 10.1523/JNEUROSCI.16-11-03549.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kutsuwada T, Kashiwabuchi N, Mori H, Sakimura K, Kushiya E, Araki K, Meguro H, Masaki H, Kumanishi T, Arakawa M, Mishina M. Molecular diversity of the NMDA receptor channel. Nature. 1992;358:36–41. doi: 10.1038/358036a0. [DOI] [PubMed] [Google Scholar]
- Lambolez B, Audinat E, Bochet P, Crepel F, Rossier J. AMPA receptor subunits expressed by single Purkinje cells. Neuron. 1992;9:247–258. doi: 10.1016/0896-6273(92)90164-9. [DOI] [PubMed] [Google Scholar]
- Mayer ML, Westbrook GL. Permeation and block of N-methyl-D-aspartic acid receptor channels by divalent cations in mouse cultured central neurones. The Journal of Physiology. 1987;394:501–527. doi: 10.1113/jphysiol.1987.sp016883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayer ML, Westbrook GL, Guthrie PB. Voltage-dependent block by Mg2+ of NMDA responses in spinal cord neurones. Nature. 1984;309:261–263. doi: 10.1038/309261a0. [DOI] [PubMed] [Google Scholar]
- Monyer H, Burnashev N, Laurie DJ, Sakmann B, Seeburg PH. Developmental and regional expression in the rat brain and functional properties of four NMDA receptors. Neuron. 1994;12:529–540. doi: 10.1016/0896-6273(94)90210-0. [DOI] [PubMed] [Google Scholar]
- Monyer H, Sprengel R, Schoepfer R, Herb A, Higuchi M, Lomeli H, Burnashev N, Sakmann B, Seeburg PH. Heteromeric NMDA receptors: molecular and functional distinction of subtypes. Science. 1992;256:1217–1221. doi: 10.1126/science.256.5060.1217. [DOI] [PubMed] [Google Scholar]
- Mori H, Mishina M. Structure and function of the NMDA receptor channel. Neuropharmacology. 1995;34:1219–1237. doi: 10.1016/0028-3908(95)00109-j. [DOI] [PubMed] [Google Scholar]
- Moriyoshi K, Masu M, Ishii T, Shigemoto R, Mizuno N, Nakanishi S. Molecular cloning and characterization of the rat NMDA receptor. Nature. 1991;354:31–37. doi: 10.1038/354031a0. [DOI] [PubMed] [Google Scholar]
- Morrisett RA, Mott DD, Lewis DV, Wilson WA, Swartzwelder HS. Reduced sensitivity of the N-methyl-D-aspartate component of synaptic transmission to magnesium in hippocampal slices from immature rats. Developmental Brain Research. 1990;56:257–262. doi: 10.1016/0165-3806(90)90090-l. [DOI] [PubMed] [Google Scholar]
- Nabekura J, Kawamoto I, Akaike N. Developmental change in voltage dependency of NMDA receptor-mediated response in nucleus tractus solitarii neurons. Brain Research. 1994;648:152–156. doi: 10.1016/0006-8993(94)91915-1. [DOI] [PubMed] [Google Scholar]
- Nowak L, Bregestovski P, Ascher P, Herbet A, Prochiantz A. Magnesium gates glutamate-activated channels in mouse central neurones. Nature. 1984;307:462–465. doi: 10.1038/307462a0. [DOI] [PubMed] [Google Scholar]
- Perouansky M, Yaari Y. Kinetic properties of NMDA receptor-mediated synaptic currents in rat hippocampal pyramidal cells versus interneurones. The Journal of Physiology. 1993;465:223–244. doi: 10.1113/jphysiol.1993.sp019674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plant T, Schirra C, Garaschuk O, Rossier J, Konnerth A. Molecular determinants of NMDA receptor function in GABAergic neurones of rat forebrain. The Journal of Physiology. 1997;499:47–63. doi: 10.1113/jphysiol.1997.sp021910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pokorny J, Yamamoto T. Postnatal ontogenesis of hippocampal CA1 area in rats. II. Development of ultrastructure in stratum lacunosum and moleculare. Brain Research Bulletin. 1981;7:121–130. doi: 10.1016/0361-9230(81)90076-9. [DOI] [PubMed] [Google Scholar]
- Portera Cailliau C, Price DL, Martin LJ. N-methyl-D-aspartate receptor proteins NR2A and NR2B are differentially distributed in the developing rat central nervous system as revealed by subunit-specific antibodies. Journal of Neurochemistry. 1996;66:692–700. doi: 10.1046/j.1471-4159.1996.66020692.x. [DOI] [PubMed] [Google Scholar]
- Strecker GJ, Jackson MB, Dudek FE. Blockade of NMDA-activated channels by magnesium in the immature rat hippocampus. Journal of Neurophysiology. 1994;72:1538–1548. doi: 10.1152/jn.1994.72.4.1538. [DOI] [PubMed] [Google Scholar]
- Tovar KR, Westbrook GL. The incorporation of NMDA receptors with a distinct subunit composition at nascent hippocampal synapses in vitro. Journal of Neuroscience. 1999;19:4180–4188. doi: 10.1523/JNEUROSCI.19-10-04180.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wafford KA, Bain CJ, Le Bourdelles B, Whiting PJ, Kemp JA. Preferential co-assembly of recombinant NMDA receptors composed of three different subunits. NeuroReport. 1993;4:1347–1349. doi: 10.1097/00001756-199309150-00015. [DOI] [PubMed] [Google Scholar]
- Watanabe M, Inoue Y, Sakimura K, Mishina M. Developmental changes in distribution of NMDA receptor channel subunit mRNAs. NeuroReport. 1992;3:1138–1140. doi: 10.1097/00001756-199212000-00027. [DOI] [PubMed] [Google Scholar]
- Wenzel A, Fritschy JM, Mohler H, Benke D. NMDA receptor heterogeneity during postnatal development of the rat brain: differential expression of the NR2A, NR2B, and NR2C subunit proteins. Journal of Neurochemistry. 1997;68:469–478. doi: 10.1046/j.1471-4159.1997.68020469.x. [DOI] [PubMed] [Google Scholar]
- Wenzel A, Villa M, Mohler H, Benke D. Developmental and regional expression of NMDA receptor subtypes containing the NR2D subunit in rat brain. Journal of Neurochemistry. 1996;66:1240–1248. doi: 10.1046/j.1471-4159.1996.66031240.x. [DOI] [PubMed] [Google Scholar]
- Williams K. Ifenprodil discriminates subtypes of the N-methyl-D-aspartate receptor: selectivity and mechanisms at recombinant heteromeric receptors. Molecular Pharmacology. 1993;44:851–859. [PubMed] [Google Scholar]
- Williams K. Pharmacological properties of recombinant N-methyl-D-aspartate (NMDA) receptors containing the epsilon 4 (NR2D) subunit. Neuroscience Letters. 1995;184:181–184. doi: 10.1016/0304-3940(94)11201-s. [DOI] [PubMed] [Google Scholar]
- Williams K, Russell SL, Shen YM, Molinoff PB. Developmental switch in the expression of NMDA receptors occurs in vivo and in vitro. Neuron. 1993;10:267–278. doi: 10.1016/0896-6273(93)90317-k. [DOI] [PubMed] [Google Scholar]
- Woodhull AM. Ionic blockage of sodium channels in nerve. Journal of General Physiology. 1973;61:687–708. doi: 10.1085/jgp.61.6.687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhong J, Carrozza DP, Williams K, Pritchett DB, Molinoff PB. Expression of mRNAs encoding subunits of the NMDA receptor in developing rat brain. Journal of Neurochemistry. 1995;64:531–539. doi: 10.1046/j.1471-4159.1995.64020531.x. [DOI] [PubMed] [Google Scholar]


