Whether you indulge in a few bars of chocolate or run a marathon, the pancreatic islets of Langerhans ensure that the swings in your blood sugar level are not large. Each islet consists of a central core of insulin-secreting β-cells, surrounded by an outer mantle of cells that secrete glucagon (α-cells), somatostatin (δ-cells) or pancreatic polypeptide (PP-cells). A rise in the plasma glucose level stimulates insulin and somatostatin secretion, but inhibits glucagon secretion; these hormonal changes serve to return the blood glucose to its resting level.
The β-cells compose more than 60 % of the islet volume, so it is perhaps not surprising that the mechanism by which glucose stimulates insulin secretion is now well established (Ashcroft & Rorsman, 1989). Insulin exocytosis is produced by elevation of intracellular calcium, which results from Ca2+ influx through voltage-gated (L-type) Ca2+ channels. In the resting β-cell, the membrane is kept at a hyperpolarized level through the activity of ATP-sensitive potassium (KATP) channels. Glucose uptake and metabolism lead to the closure of the KATP channels, triggering a membrane depolarization that activates the L-type Ca2+ channels and electrical activity. Metabolic regulation of KATP channel activity is thought to be mediated by changes in the intracellular concentrations of ATP and ADP, which inhibit and activate the channel, respectively.
Much less is known of how glucose stimulates somatostatin secretion and inhibits glucagon release. Two papers by Göpel and colleagues in this issue of The Journal of Physiology remedy this lacuna (Göpel et al. 2000a,b). In the first of these, they show that, as in β-cells, glucose-dependent somatostatin release from δ-cells is triggered by closure of KATP channels, membrane depolarization and initiation of electrical activity (Göpel et al. 2000a). Their studies involved the use of a novel method that enables patch-clamp measurements of membrane currents from cells within intact islets. This technique is, in itself, a valuable addition to our range of tools for studying islet cells because several studies have shown that chemically dispersed cells do not always exhibit the properties of cells within the intact islet.
Figure 1.
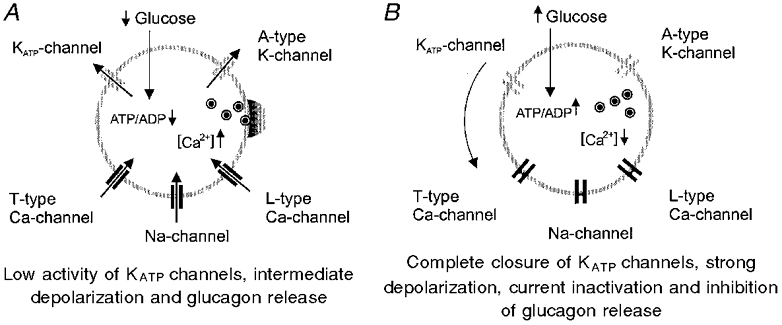
As in β- and δ-cells, a rise in intracellular calcium, brought about by Ca2+ influx through voltage-gated Ca2+ channels, also triggers glucagon secretion from α-cells. The mechanism by which glucose produces inhibition of α-cell electrical activity, [Ca2+]i influx and glucagon secretion has, however, remained obscure. One particularly puzzling finding is that α-cells also possess KATP channels (Bokvist et al. 1999). Yet if both α- and β-cells possess KATP channels with similar properties how can they respond to glucose in opposite ways? In a seminal paper in this issue, Göpel and colleagues propose a new model of glucagon release (Göpel et al. 2000b).
The model Göpel et al. propose thus suggests that the KATP channel in all three types of islet cells serves a similar function – the regulation of electrical activity in response to cell metabolism. It is the very different voltage-gated ion channels that the cells express that produces the opposite effects of glucose on electrical activity and secretion. The model also makes some interesting predictions. Thus, for example, in addition to stimulating insulin release, the sulphonylurea tolbutamide (which blocks KATP channels) should block glucagon release. Because glucagon secretion is often elevated in diabetes this might constitute part of the therapeutic effect of sulphonylureas. Second, low concentrations of the KATP channel opener diazoxide, which do not fully depolarize the α-cell, should actually stimulate glucagon secretion because they do not induce depolarization block of voltage-gated currents. Importantly, both of these predictions were confirmed experimentally.
Precisely how metabolism regulates KATP channels in α-cells remains unclear. Although the glucose uptake transporter differs in α-cells (GLUT1) and β-cells (GLUT2), glucokinase appears to serve as the rate-limiting step for glucose metabolism in both cell types (Heimberg et al. 1995). However, because changes in cytosolic ATP and ADP levels are not detected in response to glucose in α-cells, the mechanism of coupling metabolism to KATP channel activity in α-cells may differ from that of β-cells.
In conclusion, KATP channel closure produces activation or inhibition of electrical activity in α-cells, depending on the degree of channel inhibition and the resulting magnitude of the depolarization. Why does this not happen in β-cells? One reason may be that the L-type Ca2+ channel (unlike the Na+ channel) does not exhibit voltage-dependent inactivation, but is inactivated by Ca2+. Another is that the β-cell shows a bursting pattern of electrical activity over the physiological glucose range, so that the membrane is not permanently depolarized and recovery from inactivation can occur during the interburst hyperpolarizations.
References
- Ashcroft FM, Rorsman P. Progress in Biophysics and Molecular Biology. 1989;54:87–143. doi: 10.1016/0079-6107(89)90013-8. [DOI] [PubMed] [Google Scholar]
- Bokvist K, et al. Pflügers Archiv. 1999;438:428–436. doi: 10.1007/s004249900076. [DOI] [PubMed] [Google Scholar]
- Göpel SO, et al. Journal of Physiology. 2000a;528:497–507. doi: 10.1111/j.1469-7793.2000.00497.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Göpel SO, et al. Journal of Physiology. 2000b;528:509–520. doi: 10.1111/j.1469-7793.2000.00509.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heimberg H, et al. Journal of Biological Chemistry. 1995;270:8971–8975. doi: 10.1074/jbc.270.15.8971. [DOI] [PubMed] [Google Scholar]


