Abstract
The objective of this prospective clinical study was to evaluate the accuracy of pulse oximetry and capnography in healthy and compromised horses during general anesthesia with spontaneous and controlled ventilation. Horses anesthetized in a dorsal recumbency position for arthroscopy (n = 20) or colic surgery (n = 16) were instrumented with an earlobe probe from the pulse oximeter positioned on the tip of the tongue and a sample line inserted at the Y-piece for capnography. The horses were allowed to breathe spontaneously (SV) for the first 20 min after induction, and thereafter ventilation was controlled (IPPV). Arterial blood, for blood gas analysis, was drawn 20 min after induction and 20 min after IPPV was started. Relationships between oxygen saturation as determined by pulse oximetry (SpO2), arterial oxygen saturation (SaO2), arterial carbon dioxide partial pressure (PaCO2), and end tidal carbon dioxide (P(et)CO2), several physiological variables, and the accuracy of pulse oximetry and capnography, were evaluated by Bland–Altman or regression analysis. In the present study, both SpO2 and P(et)CO2 provided a relatively poor indication of SaO2 and PaCO2, respectively, in both healthy and compromised horses, especially during SV. A difference in heart rate obtained by pulse oximetry, ECG, or palpation is significantly correlated with any pulse oximeter inaccuracy. If blood gas analysis is not available, ventilation to P(et)CO2 of 35 to 45 mmHg should maintain the PaCO2 within a normal range. However, especially in compromised horses, it should never substitute blood gas analysis.
Introduction
General anesthesia in the horse is often accompanied by respiratory depression with a variable degree of hypoxemia, hypercapnia, and respiratory acidosis (1,2,3,4). Decreased chemoreceptor sensitivity to increased arterial carbon dioxide partial pressure (PaCO2), and positional factors lead to alveolar hypoventilation (3). Furthermore, ventilation — perfusion mismatching, especially in dorsal recumbency, impairs gas exchange in the lungs, contributing to the hypoxemia and hypercapnia (5,6). Abdominal tympany and endotoxemia, frequently associated with equine colic, also impair ventilatory function resulting in hypoxemia (3,7).
The adequacy of pulmonary gas exchange is best assessed by measuring arterial oxygen partial pressure (PaO2 ) and PaCO2 with blood gas analysis (3). There are several reports on the clinical use of pulse oximetry (8,9,10,11,12,13) and capnography (4,14,15,16) in equine anesthesia. Most of these studies suggest a strong correlation between arterial blood gases using a regression analysis (4,9,10,12,13,15). However, this type of analysis often overstates the actual degree of accuracy that can be expected from individual measurements (17). Because many equine practices do not have access to blood gas analysis, reliable noninvasive monitoring of oxygenation and ventilation with pulse oximetry and capnography should provide useful information.
The purpose of this study was to evaluate the accuracy of pulse oximetry and capnography both in healthy and compromised horses during general anesthesia with spontaneous and controlled respiration, using the recommended system of analysis for 2 methods of measurement of a common variable in terms of bias and precision (17,18).
Materials and methods
Animals
This study involved 36 horses presented to the Ontario Veterinary College for arthroscopic procedures or for colic surgery between August 1996 and November 1996. Based on physical examination and pre-operative blood work the animals were assigned an American Society of Anesthesiologists (ASA) rating 1 to 5. Group 1 (arthroscopy, n = 20) consisted of 13 thoroughbreds, 4 standardbreds, 2 warmbloods, and 1 quarterhorse for arthroscopy, ranging in weight from 380 to 634 kg (mean 480 kg) and age from 1 to 14 y (mean 3.7 y). Ten horses were rated ASA 1 and the other 10 ASA 2. Group 2 (colic surgery, n = 16) consisted of 7 thoroughbreds, 4 quarterhorses, 2 standardbreds, 2 warmbloods, and 1 trotter for colic surgery, ranging in weight from 399 to 654 kg (mean 500 kg) and age from 2 to 24 y (mean 7.2 y). Four horses were rated ASA 3 and 12 horses ASA 4. Horses were premedicated with intravenous xylazine (0.3 mg/kg, Rompun; Bayer Inc. Agriculture Division Animal Health, Toronto, Ontario) or romifidine (0.03 mg/kg, Sedivet; Boehringer Ingelheim, Burlington, Ontario), sometimes in combination with butorphanol (0.02 mg/kg, Torbugesic; Ayerst, Montreal, Quebec) or diazepam (0.02 mg/kg, Diazepam; Sabex Inc., Boucherville, Quebec). The premedication administered depended upon the drugs used previously for analgesia. Anesthesia was induced with guaifenesin (100 mg/kg; B.I. Chemicals Inc., Petersburg, Virginia, USA) 10% intravenously until ataxia occurred, followed by an intravenous bolus of ketamine (2 mg/kg, Ketalean; Bimeda-MTC Animal Health Inc., Cambridge, Ontario). After endotracheal intubation, the horses were connected to a large animal breathing circuit (Narkovet-E-Large Animal Control Center; North American Draeger, Telford, Pennsylvania, USA) with an out-of-circuit halothane or isoflurane vaporizer (Vapor 19.1; North American Draeger) using 100% oxygen (O2) as the carrier gas. All horses were placed in dorsal recumbency and balanced fluids (Lactated Ringer's Injection USP; Baxter Corporation, Toronto, Ontario) administered by IV at a rate of at least 10 mL/kg/h. Electrodes for electrocardiogram monitoring were connected using a base-apex lead configuration and a catheter was placed in the facial or transverse facial artery to measure the arterial blood pressure and collect arterial blood samples. The blood pressure transducer was calibrated prior to measurement. The earlobe probe from the pulse oximeter (POET 1100; Criticare Systems, Waukesha, Wisconsin, USA) was positioned on the tip of the horse's tongue and provided continuous readings. The pulse oximeter probe was repositioned occasionally to alleviate tissue compression. The sample line for sidestream capnography (POET 1100; Criticare Systems) was screwed onto a receptor in the center on the Y-piece, and gases were sampled continuously at a flow rate of 150 mL/min. Using a standard gas (Anesthesia Calibration Gas; Scott Medical Products, Plumsteadville, Pennsylvania, USA), the capnograph was calibrated prior to each anesthetic event.
The horses were allowed to breathe the inhalational anesthetic in oxygen spontaneously (SV) for the first 20 min after induction, at which time ventilation was controlled (IPPV). The ventilator (Narkovet-E-Large Animal Control Center, North American Draeger) was initially set at a respiratory rate and tidal volume based on body weight (12 to 15 mL/kg ), excursion of the thorax, and clinical experience. The minute volume was varied by adjusting respiratory rate, tidal volume, or inspiratory pressure as indicated by blood gas analysis. Arterial blood pressure was maintained within acceptable limits (mean arterial blood pressure over 70 mmHg) by means of dobutamine infusion administration.
Arterial blood samples were anaerobically withdrawn in heparinized syringes (Aspirator A.B.G; Marquest Medical Products Inc., Englewood, Colarado, USA) and analyzed immediately for blood gases (ABL 500; Radiometer, Copenhagen, Denmark) 20 min after induction (SV) and 20 min after IPPV was started. The blood gas analyzer was corrected for the horses' rectal temperature. Arterial oxygen saturation (SaO2) was calculated from the measured blood gas values using a dissociation curve described for equine hemoglobin (19).
Simultaneous to the withdrawal of the arterial blood sample, the pulse oximeters oxygen saturation (SpO2) and heart rate (HRc) readings, and the endtidal CO2 (P(et)CO2) and inspiratory oxygen fraction (FiO2) readings from the capnograph, were recorded. Heart rate taken by the ECG (HRe) and mean arterial blood pressure (MAP) were also recorded. Daily barometric pressure (PB) was recorded and used in the calculation of PAO2 using the formula alveolar partial pressure of oxygen (PAO2) = [(PB − P H2O ) FiO2] − [PaCO2/0.8] (3).
The arterio-alveolar CO2-difference (P(a-A) CO2) and the alveolo-arterial O2-difference (P(A-a) O2) were calculated as previously described (3).
Statistical analysis
The SpO2 readings were compared with SaO2 values, and P(et)CO2 readings were compared with PaCO2 values by a method where differences between the indirect measurements and the arterial blood gas values were plotted against the arterial blood gas values (17,18). The mean, standard deviation (s), and limits of agreement were calculated for the differences. The limits of agreement were defined as the mean difference ± 2 s, which represented 95% of the differences. To study the influence of MAP, weight, age, P(A-a)O2, and the difference of the heart rate between pulse oximetry and ECG on the accuracy of the pulse oximeter and the capnograph, Pearson's correlation was performed.
Results
Mean arterial blood pressure, arterial blood gas values, FiO2 values, SaO2 values, P(A-a)O2 values, and heart rates are presented in Table I. The curves of the capnograph (capnogram) appeared normal (flat) in all cases, supporting our assumption is that P(et)CO2 reflects PACO2.
Table I.
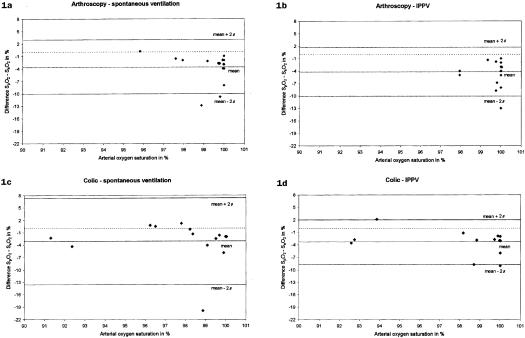
During SV, the modified Bland–Altmann analysis showed that the pulse oximeter underestimated SaO2 by a mean differences of 3.6% (arthroscopy group; Figure 1a) and 3.1% (colic group; Figure 1c). The pulse oximeter produced limits of agreement of −10% and +2.96% (arthroscopy group), and −13.6% and +7.3% (colic group). During IPPV the pulse oximeter underestimated SaO2 by a mean differences of 4.2% (arthroscopy group; Figure 1b) and 3.3% (colic group; Figure 1d). The limits of agreement were −10.1% and +1.6% (arthroscopy group) and −8.71% and +2.03% (colic group).
Figure 1. Difference in pulse oximeter oxygen saturation and arterial oxygen saturation readings compared to arterial blood gas values in healthy (arthroscopy group) and compromised (colic group) horses during spontaneous and controlled ventilation. The mean difference (bias) and limits of agreement (± 2 s) are shown.
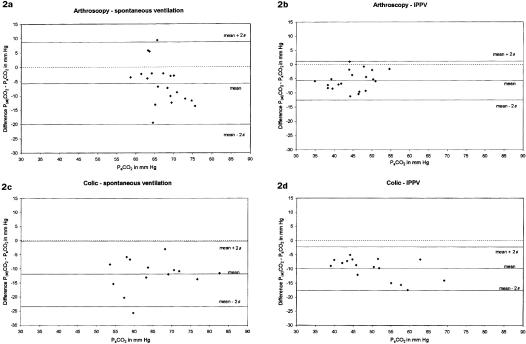
During SV the Bland–Altmann analysis showed that P(et)CO2 underestimated PaCO2 by a mean difference of 5.7 mmHg in the arthroscopy group (Figure 2a) and 11.9 mmHg in the colic group (Figure 2c). The limits of agreement were −20.14 mmHg and +8.72 mmHg (arthroscopy group), and −23.54 mmHg and +0.35 mmHg (colic group). During IPPV, P(et)CO2 underestimated PaCO2 by a mean difference of 5.7 mmHg (arthroscopy group; Figure 2b) and 9.9 mmHg (colic group; Figure 2d). The limits of agreement were −12.53 mmHg and +1.1 mmHg in the arthroscopy group, and −17.4 mmHg and −2.34 mmHg in the colic group.
Figure 2. Differences in endtidal CO2 and arterial CO2 readings compared to arterial blood gas values in healthy (arthroscopy group) and compromised (colic group) horses during spontaneous and controlled ventilation. The mean difference (bias) and limits of agreement (± 2 s) are shown.
No significant effects were found for MAP, age, weight, and P(A-a)O2 on the accuracy of pulse oximetry and capnography. Further, no significant correlation between P(A-a)O2, MAP, and age on P(a-A)CO2 was found. Correlation was significant (P < 0.0001) between differences in HRc and HRe (formula: HRc − HRe), and the inaccuracy of pulse oximetry (SpO2−SaO2) during SV, but not during IPPV.
Discussion
The objective of this study was to determine the accuracy of pulse oximetry and capnography in clinically healthy (arthroscopy) and clinically compromised (colic surgery) horses. There are definite limits to the accuracy of pulse oximetry to predict SaO2 as tested in the present study. In this study, there was a general tendency for the pulse oximeter to underestimate SaO2 values that are over 90%, only 2 measurements overestimated SaO2 values. Because clinical patients were studied, deliberate hypoxemia was not induced. As a result the spread of SaO2 values was small with a range of 95% to 100% in healthy horses, and 91% to 100% in compromised horses. A PaO2 less than 60 mmHg is considered as hypoxemia, because rapid desaturation of hemoglobin occurs below this level. This is equivalent to an equine SaO2 of approximately 93% (19) and treatment is recommended. Since most SaO2 measurements are obtained from blood gas analyzers using a human oxygen-hemoglobin-dissociation curve, aggressive treatment should be initiated at SaO2 below 90%. Six SpO2 readings in healthy horses and 3 SpO2 readings in compromised horses would have caused us to initiate treatment unnecessarily, because SpO2 readings were lower then the actual SaO2, suggesting hypoxemia. Other equine studies of pulse oximetry have also found SpO2 to underestimate SaO2 (10,13). It has been reported that below 90% SaO2, the pulse oximeter can either overestimate (9,11) or underestimate SaO2 (13) in horses. Interestingly, in dogs, sheep, and pigs, pulse oximeters tend to overestimate SaO2 (20,21).
The magnitude of mean differences between SpO2 and SaO2 obtained in this study are similar to findings from previous studies (9,13), but some report a smaller mean difference (10,12). One reason for the discrepancies among studies may be that different pulse oximeters were used. Manufacturers commonly specify that pulse oximeters have a standard deviation, of 1.5 to 2.5% at saturations in the range 90 to 100%, when compared to a “gold standard” determined by co-oximetry. Thus, 95% of readings should be within 1.96 times the standard deviation or 2.9 to 4.9%. Clayton et al (22) compared the performance of 20 pulse oximeters under conditions of poor perfusion in humans and only 2 (Datex Satlite and Criticare CSI 503) out of the 20 were within the manufacturers' range. Maier and Wintzer (9) used the same pulse oximeter used in this study (POET, Criticare) on the horses tongue and found a similar mean difference. However, the regression analysis, used in their study makes comparison to our data difficult.
The present study has also demonstrated that the inaccuracy of the pulse oximeter in obtaining the exact heart rate correlates significantly (P < 0.0001) with the inaccuracy of SpO2 readings. Failure to detect arterial blood pulsation occurs when the components of the transmitted red and infrared light beams are small, compared to the static components and noise. This may be the result of vasoconstriction, vasodilation, venous congestion, or other factors (23). It seems that poor signal quality is the major reason for inaccuracy of pulse oximeters. Clayton et al (22) showed that 10 out of 20 pulse oximeters failed to give readings at least 10% of the time. The choice of the probe site may influence the accuracy of pulse oximetry. A human earlobe probe was used on the equine tongue in this study. When pulse oximeters (Biox 3700; Lifestat 1600; Medtronic, Redmond, Washington, USA) with human earlobe probes were applied to either the equine tongue or ear, the oximeter results tended to underestimate the SaO2 with mean differences of −3.7% on the tongue and −6.0% on the ear (13). Moens et al (11) also found that positioning of a modified pulse oximeter sensor on the rostral nasal septum resulted in underestimation of SaO2 with a mean difference of −3.9% in the saturation range 90 to 100%. In humans, ear, nose, and forehead probes respond more rapidly to rapid desaturation than finger probes, but are generally less accurate and less precise (23).
In the present study, P(et)CO2 provided a relatively poor indication of PaCO2 in both healthy and compromised horses, especially during SV. In the anesthetized horse wide P(a-A)CO2 gradients have commonly been reported (2,4,14,15). Increase in P(a-A) CO2 gradients are due to ventilation-perfusion mismatch (V/Q mismatch), caused by disease or anesthesia and recumbency (3). In this study, after 20 min of spontaneous ventilation the PaCO2 of healthy horses was between 58.6 and 75.6 mmHg (mean = 67.11 mmHg), and the mean difference between PaCO2 and P(et)CO2 was −5.7 mmHg. In compromised horses the PaCO2 was between 53.5 and 82.6 mmHg (mean = 64.79 mmHg) and with a mean difference between PaCO2 and P(et)CO2 of −11.9 mmHg. In the healthy, standing horse, P(a-A)CO2 is small and, thus, similar to other species (24). The P(a-A)CO2 was remarkably greater in compromised horses compared to healthy horses. Abdominal distension and poor perfusion, both likely to be present in horses with colic, may increase V/Q mismatch and physiological dead space (25,26). Meyer et al (2) described a highly significant difference between PaCO2 and P(et)CO2 after 90 min of halothane anesthesia in spontaneously breathing ponies, which was not present with isoflurane. Of 16 colic cases in our study, 6 were maintained with isoflurane, as was 1 of the 20 arthroscopy cases. No difference in the P(a-A)CO2 gradient between isoflurane or halothane was noted. Furthermore, there was no apparent increase present when PaCO2 was over 60 to 70 mmHg, as reported by Meyer et al (2), who used an experimental study design and obtained PaCO2 values as high as 109 ± 35 mmHg, which is unacceptable for clinical cases.
Interestingly, in the present study, 3 readings during spontaneous ventilation in healthy horses had P(et)CO2 higher than PaCO2. While a positive P(et)CO2 to PaCO2 gradient is unusual, others have reported this observation in horses and humans (27,28). This phenomenon seems to be associated with large tidal volumes, comparatively low functional residual capacity, and alveoli with low V/Q ratios (28).
Moens (4) described a significant influence of BW (P < 0.0001) on P(a-A) CO2 and alveolar dead space; however, no correlation between BW and the P(a-A) CO2 was demonstrated in the present study.
In conclusion, capnography seems to be useful as a trend indicator for assessment of the ventilation status for horses maintained with IPPV, but not during SV. This is in agreement with Neto et al (29), who reported that P(et)CO2 is not a predictable method for estimating PaCO2 during SV in healthy horses. We agree with Cribb (15) that if blood gas analysis is not available, ventilation to P(et)CO2 of 35 to 45 mmHg should maintain the PaCO2 within a normal range. However, especially in compromised horses, it should never substitute blood gas analysis. When arterial blood gas analysis is not available, pulse oximetry may be used to monitor oxygen saturation; however, when the heart rate, measured by either ECG or palpation, does not correlate with the heart rate obtained by the pulse oximeter, the SpO2 value is most likely not reliable.
Footnotes
Dr. Valverde's current address is the Department of Large Animal Clinical Sciences, College of Veterinary Medicine, University of Florida, Gainesville, Florida, USA.
Address all correspondence and reprint requests to Dr. Judith Koenig; telephone: (519) 823-8840; fax: (519) 763-9544; e-mail: jkoenig@ovc.uoguelph.ca
Received May 3, 2002. Accepted September 24, 2002.
References
- 1.Gillespie JR, Tyler WS, Hall LW. Cardiopulmonary dysfunction in anaesthetised laterally recumbent horses. Am J Vet Res 1969;30:61–72. [PubMed]
- 2.Meyer RE, Short CE. Arterial to end-tidal CO2 tension and alveolar dead space in halothane — or isoflurane — anesthetized ponies. Am J Vet Res 1985;46:597–599. [PubMed]
- 3.McDonell W. Respiratory System. In: Thurmon JC, Tranquili WJ, Benson JG, eds. Lumb and Jones' Veterinary Anesthesia. 3rd ed., Wiliams & Wilkins, 1996;115–147.
- 4.Moens Y. Arterio-alveolar carbon-dioxide tension difference and alveolar dead space in halothane anaesthetized horses. Equine Vet J 1989;6:282–284. [DOI] [PubMed]
- 5.Nyman G, Hedenstierna G. Ventilation-perfusion relationships in the anaesthetized horse. Equine Vet J 1989;21:274–281. [DOI] [PubMed]
- 6.Schatzmann U. Pulmonary perfusion and ventilation: a mismatch? Equine Vet J 1995;27:80–81. [DOI] [PubMed]
- 7.Moore JN, Garner HE, Shapland JE, Hatfield DG. Prevention of endotoxin – induced arterial hypoxemia and lactic acidosis with flunixin meglumine in the conscious pony. Equine Vet J 1981;13:95–98. [DOI] [PubMed]
- 8.Alef M, Oechtering G. Nichtinvasive Patientenüberwachung in der Tiermedizin: Pulsoxymetrie und Kapnographie. Teil I: Pulsoxymetrie. Tierärztl Prax 1994;22:596–606. [PubMed]
- 9.Maier FP, Wintzer HJ. Möglichkeiten und Grenzen der Pulsoximetrie bei der Narkoseüberwachung des Pferdes. Berl MünchTierärtzl Wschr 1994;107:7–11. [PubMed]
- 10.Matthews NS, Hartsfield SM, Sanders EA, Light GS, Slater MS. Evaluation of pulse oximetry in horses surgically treated for colic. Equine Vet J 1994;26:114–116. [DOI] [PubMed]
- 11.Moens Y, Gootjes P, Lagerweij E, Van Dijk. Überwachung der Sauerstoffsättigung von Pferden in Halothannarkose durch Pulsoximetrie am Septum Nasale. Berl MünchTierärztl Wschr 1991;104:357–360. [PubMed]
- 12.Watney GCG, Norman WM, Schumacher JP. Evaluation of a Reflectance Pulse Oximeter. In: Equine Anesthesia. Am J Vet Res 1993;54:497–501. [PubMed]
- 13.Whitehair KJ, Watney GCG, Leith DE, DeBowes RM. Pulse Oximetry. In: Horses. Vet Surg 1990;19:243–248. [DOI] [PubMed]
- 14.Alef M, Oechtering G. Nichtinvasive Patientenüberwachung in der Tiermedizin: Pulsoximetrie und Kapnographie. Teil II: Kapnographie. Tierärztl Prax 1995;23:1–16. [PubMed]
- 15.Cribb PH. Capnographic monitoring during anesthesia with controlled ventilation in the horse. Vet Surg 1988;17:48–52. [DOI] [PubMed]
- 16.Otto K. Kapnographie — eine Methode zur Beurteilung der Ventilationslage während der Allgemeinanästhesie beim Pferd. J Vet Med 1986;33:609–616. [PubMed]
- 17. Bland MJ, Altmann DG. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986;1:307–310. [PubMed]
- 18.Ralston AC, Webb RK, Runciman WB. Potential errors in pulse oximetry. I. Pulse oximeter evaluation. Anaesthesia 1991;46:202–206. [DOI] [PubMed]
- 19.Smale K, Anderson LS, Butler PJ. An algorithm to describe the oxygen equilibrium curve for the Thoroughbred racehorse. Equine Vet J 1994;26:500–502. [DOI] [PubMed]
- 20.Erhardt W, Lendl C, Hipp R, Schindele M, Bluemel G. Die Pulsoximetrie — ein nichtinvasives Verfahren zur unmittelbaren und kontinuierlichen Ueberwachung von Sauerstoffsaettigung und Pulsfrequenz-Vergleichsstudien zur Blutgasanalyse und zum Haemoreflektometer an Hund, Schwein und Schaf. Berl MünchTierärztl Wschr 1989;102:289–292. [PubMed]
- 21.Sendak MJ, Harris AP, Donham RT. Accuracy of pulse oximetry during arterial hemoglobin desaturation in dogs. Anesthesiology 1988;68:111–114. [DOI] [PubMed]
- 22.Clayton DG, Webb RK, Ralston AC, Duthie D, Runciman W. A comparison of the performance of 20 pulse oximeters under conditions of poor perfusion. Anaesthesia 1991;46:3–10. [DOI] [PubMed]
- 23.Webb RK, Ralston AC, Runciman WB. Potential errors in pulse oximetry. II. Effects of changes in saturation and signal quality. Anaesthesia 1991;46:207–212. [DOI] [PubMed]
- 24.Littlejohn A. Acid-base and blood gas studies in horses. Res Vet Sci 1969;10:263–266. [PubMed]
- 25.Barker SJ, Tremper KK. Respiratory monitoring, blood-gas measurement, oximetry, and pulse oximetry. Anesthesiology 1992;5:816–825.
- 26.Good ML. Capnography: Uses, interpretations, and pitfalls. In: Barash PG, ed. ASA Refresher Courses in Anesthesiology 1990;18:12,175–193.
- 27. Piiper J. Blood-gas equilibrium of carbon dioxide in lungs: a continuing controversy. J Appl Physiol 1986;60:1–8. [DOI] [PubMed]
- 28.Fletcher R, Jonson B, Cumming G. The concept of death space with special reference to the single breath test for carbon dioxide. Brit J Anaesth 1981;53:77–88. [DOI] [PubMed]
- 29.Neto FJT, Luna APL, Massone F, et al. The effect of changing the mode of ventilation on the arterial-to-end-tidal CO2 difference and physiological dead space in laterally and dorsally recumbent horses during halothane anesthesia. Vet Surg 2000;29:200–205. [DOI] [PubMed]