Abstract
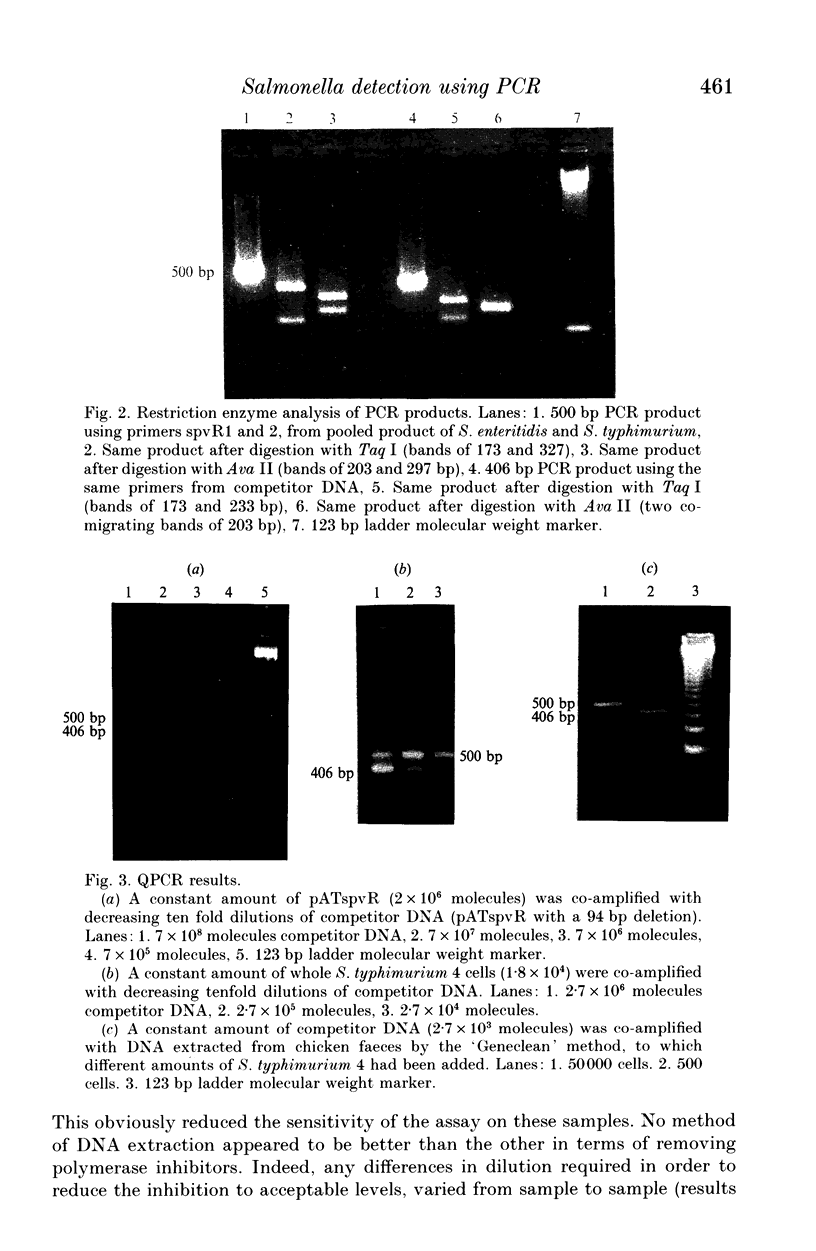
A quick, semi-quantitative method of detecting Salmonella species which contain the virulence plasmid has been developed using the polymerase chain reaction (PCR). A pair of primers have been synthesized encompassing a 500 bp fragment of the spvR virulence gene. Competitor DNA consisting of the spvR gene with a 94 bp deletion situated between the primer recognition sequences, was cloned into a plasmid vector. Co-amplification of the 'unknown' target salmonella DNA with known quantities of competitor DNA in the same reaction tube gave PCR products of 500 and 406 bp respectively. Visual assessment of the ratio of the two products on ethidium bromide stained agarose gels provided an estimate of the approximate number of salmonella cells present in avian faeces. The technique could be applied to detect quantifiably any non-host DNA in clinical samples if a suitable DNA sequence for primer construction is available.
Full text
PDF








Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aguirre P. M., Cacho J. B., Folgueira L., López M., García J., Velasco A. C. Rapid fluorescence method for screening Salmonella spp. from enteric differential agars. J Clin Microbiol. 1990 Jan;28(1):148–149. doi: 10.1128/jcm.28.1.148-149.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allard A., Girones R., Juto P., Wadell G. Polymerase chain reaction for detection of adenoviruses in stool samples. J Clin Microbiol. 1990 Dec;28(12):2659–2667. doi: 10.1128/jcm.28.12.2659-2667.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Becker-André M., Hahlbrock K. Absolute mRNA quantification using the polymerase chain reaction (PCR). A novel approach by a PCR aided transcript titration assay (PATTY). Nucleic Acids Res. 1989 Nov 25;17(22):9437–9446. doi: 10.1093/nar/17.22.9437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bej A. K., Steffan R. J., DiCesare J., Haff L., Atlas R. M. Detection of coliform bacteria in water by polymerase chain reaction and gene probes. Appl Environ Microbiol. 1990 Feb;56(2):307–314. doi: 10.1128/aem.56.2.307-314.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi D., Tsang R. S., Ng M. H. Sandwich capture ELISA by a murine monoclonal antibody against a genus-specific LPS epitope for the detection of different common serotypes of salmonellas. J Appl Bacteriol. 1992 Feb;72(2):134–138. doi: 10.1111/j.1365-2672.1992.tb01814.x. [DOI] [PubMed] [Google Scholar]
- Davis G. R., Blumeyer K., DiMichele L. J., Whitfield K. M., Chappelle H., Riggs N., Ghosh S. S., Kao P. M., Fahy E., Kwoh D. Y. Detection of human immunodeficiency virus type 1 in AIDS patients using amplification-mediated hybridization analyses: reproducibility and quantitative limitations. J Infect Dis. 1990 Jul;162(1):13–20. doi: 10.1093/infdis/162.1.13. [DOI] [PubMed] [Google Scholar]
- Frankel G., Riley L., Giron J. A., Valmassoi J., Friedmann A., Strockbine N., Falkow S., Schoolnik G. K. Detection of Shigella in feces using DNA amplification. J Infect Dis. 1990 Jun;161(6):1252–1256. doi: 10.1093/infdis/161.6.1252. [DOI] [PubMed] [Google Scholar]
- Gilliland G., Perrin S., Blanchard K., Bunn H. F. Analysis of cytokine mRNA and DNA: detection and quantitation by competitive polymerase chain reaction. Proc Natl Acad Sci U S A. 1990 Apr;87(7):2725–2729. doi: 10.1073/pnas.87.7.2725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gulig P. A., Danbara H., Guiney D. G., Lax A. J., Norel F., Rhen M. Molecular analysis of spv virulence genes of the Salmonella virulence plasmids. Mol Microbiol. 1993 Mar;7(6):825–830. doi: 10.1111/j.1365-2958.1993.tb01172.x. [DOI] [PubMed] [Google Scholar]
- Holodniy M., Katzenstein D. A., Sengupta S., Wang A. M., Casipit C., Schwartz D. H., Konrad M., Groves E., Merigan T. C. Detection and quantification of human immunodeficiency virus RNA in patient serum by use of the polymerase chain reaction. J Infect Dis. 1991 Apr;163(4):862–866. doi: 10.1093/infdis/163.4.862. [DOI] [PubMed] [Google Scholar]
- Khan G., Kangro H. O., Coates P. J., Heath R. B. Inhibitory effects of urine on the polymerase chain reaction for cytomegalovirus DNA. J Clin Pathol. 1991 May;44(5):360–365. doi: 10.1136/jcp.44.5.360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pullinger G. D., Baird G. D., Williamson C. M., Lax A. J. Nucleotide sequence of a plasmid gene involved in the virulence of salmonellas. Nucleic Acids Res. 1989 Oct 11;17(19):7983–7983. doi: 10.1093/nar/17.19.7983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruiz J., Sempere M. A., Varela M. C., Gomez J. Modification of the methodology of stool culture for Salmonella detection. J Clin Microbiol. 1992 Feb;30(2):525–526. doi: 10.1128/jcm.30.2.525-526.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siebert P. D., Larrick J. W. Competitive PCR. Nature. 1992 Oct 8;359(6395):557–558. doi: 10.1038/359557a0. [DOI] [PubMed] [Google Scholar]
- Stieger M., Démollière C., Ahlborn-Laake L., Mous J. Competitive polymerase chain reaction assay for quantitation of HIV-1 DNA and RNA. J Virol Methods. 1991 Sep-Oct;34(2):149–160. doi: 10.1016/0166-0934(91)90095-h. [DOI] [PubMed] [Google Scholar]
- Tinge S. A., Curtiss R., 3rd Isolation of the replication and partitioning regions of the Salmonella typhimurium virulence plasmid and stabilization of heterologous replicons. J Bacteriol. 1990 Sep;172(9):5266–5277. doi: 10.1128/jb.172.9.5266-5277.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsai Y. L., Olson B. H. Detection of low numbers of bacterial cells in soils and sediments by polymerase chain reaction. Appl Environ Microbiol. 1992 Feb;58(2):754–757. doi: 10.1128/aem.58.2.754-757.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ursi J. P., Ursi D., Ieven M., Pattyn S. R. Utility of an internal control for the polymerase chain reaction. Application to detection of Mycoplasma pneumoniae in clinical specimens. APMIS. 1992 Jul;100(7):635–639. doi: 10.1111/j.1699-0463.1992.tb03978.x. [DOI] [PubMed] [Google Scholar]
- Wang A. M., Doyle M. V., Mark D. F. Quantitation of mRNA by the polymerase chain reaction. Proc Natl Acad Sci U S A. 1989 Dec;86(24):9717–9721. doi: 10.1073/pnas.86.24.9717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang R. F., Cao W. W., Johnson M. G. Development of cell surface protein associated gene probe specific for Listeria monocytogenes and detection of the bacteria in food by PCR. Mol Cell Probes. 1992 Apr;6(2):119–129. doi: 10.1016/0890-8508(92)90056-4. [DOI] [PubMed] [Google Scholar]
- Wernars K., Delfgou E., Soentoro P. S., Notermans S. Successful approach for detection of low numbers of enterotoxigenic Escherichia coli in minced meat by using the polymerase chain reaction. Appl Environ Microbiol. 1991 Jul;57(7):1914–1919. doi: 10.1128/aem.57.7.1914-1919.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wernars K., Heuvelman C. J., Chakraborty T., Notermans S. H. Use of the polymerase chain reaction for direct detection of Listeria monocytogenes in soft cheese. J Appl Bacteriol. 1991 Feb;70(2):121–126. doi: 10.1111/j.1365-2672.1991.tb04437.x. [DOI] [PubMed] [Google Scholar]
- Widjojoatmodjo M. N., Fluit A. C., Torensma R., Keller B. H., Verhoef J. Evaluation of the magnetic immuno PCR assay for rapid detection of Salmonella. Eur J Clin Microbiol Infect Dis. 1991 Nov;10(11):935–938. doi: 10.1007/BF02005447. [DOI] [PubMed] [Google Scholar]
- Widjojoatmodjo M. N., Fluit A. C., Torensma R., Verdonk G. P., Verhoef J. The magnetic immuno polymerase chain reaction assay for direct detection of salmonellae in fecal samples. J Clin Microbiol. 1992 Dec;30(12):3195–3199. doi: 10.1128/jcm.30.12.3195-3199.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williamson C. M., Baird G. D., Manning E. J. A common virulence region on plasmids from eleven serotypes of Salmonella. J Gen Microbiol. 1988 Apr;134(4):975–982. doi: 10.1099/00221287-134-4-975. [DOI] [PubMed] [Google Scholar]