Abstract
Early nutrition shapes life history. Parents should, therefore, provide a diet that will optimize the nutrient intake of their offspring. In a number of passerines, there is an often observed, but unexplained, peak in spider provisioning during chick development. We show that the proportion of spiders in the diet of nestling blue tits, Cyanistes caeruleus, varies significantly with the age of chicks but is unrelated to the timing of breeding or spider availability. Moreover, this parental prey selection supplies nestlings with high levels of taurine particularly at younger ages. This amino acid is known to be both vital and limiting for mammalian development and consequently found in high concentrations in placenta and milk. Based on the known roles of taurine in mammalian brain development and function, we then asked whether by supplying taurine-rich spiders, avian parents influence the stress responsiveness and cognitive function of their offspring. To test this, we provided wild blue tit nestlings with either a taurine supplement or control treatment once daily from the ages of 2–14 days. Then pairs of size- and sex-matched siblings were brought into captivity for behavioural testing. We found that juveniles that had received additional taurine as neonates took significantly greater risks when investigating novel objects than controls. Taurine birds were also more successful at a spatial learning task than controls. Additionally, those individuals that succeeded at a spatial learning task had shown intermediate levels of risk taking. Non-learners were generally very risk-averse controls. Early diet therefore has downstream impacts on behavioural characteristics that could affect fitness via foraging and competitive performance. Fine-scale prey selection is a mechanism by which parents can manipulate the behavioural phenotype of offspring.
Keywords: neonatal nutrition, taurine, cognition, blue tits, personality
1. Introduction
Early nutrition has been shown to have long-term effects on individual fitness (Metcalfe & Monaghan 2003). Offspring diet, in many studies, is generally measured in terms of the total amount of food or energy received from parents. There is, however, now growing interest in how specific nutrients, which might only be abundant in a limited range of foods, underpin key life-history trade-offs (e.g. McGraw 2005). For many passerines, lepidopteran larvae comprise a significant component of the chicks' diet (Betts 1955), but spiders also form an important and apparently high-quality component of the diet (Magrath et al. 2004). Several studies of parids (Cowie & Hinsley 1988; Grundel & Dahlsten 1991; Naef-Daenzer et al. 2000) report a peak in spider provisioning during early stages of chick development (approx. 5–6 days old). This phenomenon, while strongly correlated with the age of the nestlings, seems to occur irrespective of season or habitat, indicating that it is not related to variations in natural spider availability. This leads to the question of whether avian parents seek out spiders, to provide a particular nutrient required for early development.
Spiders are nutritionally similar to caterpillars (S.L. Ramsay & K.E. Arnold 2005, unpublished data), except that spiders contain 40–100 times the level of taurine compared with caterpillars (Ramsay & Houston 2003). Interestingly, there is a large literature on the roles of taurine in mammalian development and physiology. Taurine (a free sulphur amino acid) cannot be made by peri- and neonatal mammals, so is found in high concentrations in placenta and milk (Sturman 1993). In mammals, it has multiple vital roles in early development (Aerts & Assche 2002), being required for normal growth as well as the development of brain and visual systems. Taurine deficiency results in abnormal development of the cerebellum and visual cortex regions in cats, and impaired motor function and IQ in children (Sturman 1993). More work is needed to know whether taurine is limiting for non-mammalian vertebrates, translating into functional changes in behaviour and cognition in wild animals.
Another major role of taurine is in modulating biochemical changes in the brain associated with anxiety (Lapin 2003) and stress (Lehmann et al. 1984), acting as an inhibitor of hypothalamic–pituitary–adrenal axis (HPA) activity (Engelmann et al. 2003). The HPA axis is activated in response to perturbations in the animal's environment resulting in a cascade of physiological and behavioural changes known as the stress response. Stress responsiveness varies enormously within a population, with some individuals for example, being consistently prone to taking risks when faced with a stressor such as a novel object (Sih et al. 2004). Severe malnutrition during the neonatal period is a known environmental source of variation in such behaviours (Tonkiss & Smart 1983; Carere et al. 2005). In contrast, we investigated whether relatively subtle adjustments in parental prey choices could have consequences for an individual's responses to novel and stressful experiences at maturity.
In this study, using wild blue tits (Cyanistes caeruleus) as a model, we ask whether passerine parents are selectively feeding spiders to their offspring at certain stages of development. We also investigate whether the rate of spider provisioning is related to the natural incidence of spiders relative to the number of caterpillars, and how this varies with date and habitat. Next, we ask whether this parental prey selection could be fulfilling a neonatal need for taurine. In mammals, taurine is required for early development, certain aspects of cognitive function and also modulating HPA activity. Based on these known functions of taurine in mammals, we can predict a period of high taurine need when the chicks are about 5 days old, the age at which other studies have reported a peak in spider provisioning. This is when blue tits are growing very fast. They are also starting to need to process spatial information because they are beginning to open their eyes and generally be more aware of their surroundings. Thus, we address whether experimentally supplementing wild blue tit nestlings with taurine: (i) impacts upon juveniles' risk-taking behaviour, (ii) affects spatial learning and memory, and (iii) interacts with an individual's propensity for risk taking to influence spatial learning ability. Our manipulation was within the natural variation in taurine intake, so relatively subtle, thus simulating a nestling diet rich in spiders.
2. Material and methods
A nest-box breeding population of blue tits in oak-dominated woodland around Loch Lomond, Scotland was studied from April to June 2004 and 2005. As blue tits tend to forage at the tips of tree branches, arboreal invertebrates were sampled throughout the period of chick rearing. In order to assess the availability of different prey items in the environment, once a week arboreal invertebrates were collected from oak trees in six zones of the study site differing markedly in the proportions of oak trees and other aspects of the vegetation and topography. A rope was thrown over a large branch and used to shake the branch for 2 min. Invertebrates were collected from sheets spread out on the ground below. Although this method cannot be used to accurately quantify differences between zones, the range of taxa and the ratios between spiders and other invertebrates should be representative of the arboreal prey available to blue tits at that point in the season. Invertebrates were collected in tubes and frozen as soon as possible to kill and store them. Later they were counted and weighed. The proportion of spiders present in the area was calculated as the number of individual spiders collected/total number of individual invertebrates collected.
To compare the number and type of prey items brought to chicks aged 5 and 11 days, video recordings from 18 families were analysed. When a brood was 4 and 10 days old, we put a small infrared video camera in the nest-box opposite the entrance hole, so we could observe the adults entering. A timer was set so that the camera started recording at 06.00 the following morning, i.e. days 5 and 11, for a duration of 4 hours. From the video tapes we noted each time an adult entered the box and the type of prey being carried: caterpillar; spider; or ‘other type of invertebrate’. The proportion of spiders provided to chicks was calculated as the number of spiders brought during a 4 hour period/total number of food items provided. All proportions were arcsine square-root transformed prior to analysis. Using the data on prey intake rates, estimates of the prey mass (S.L. Ramsay & K.E. Arnold, unpublished data) and the known concentrations of taurine in different types of prey (Ramsay & Houston 2003), we then calculated the approximate hourly intake of taurine per chick in each brood adjusted for mean body mass.
(a) Dietary manipulation
At the age of 2 days, each nestling was weighed and uniquely marked by colouring patches of down using non-toxic pens. Using our calculations of hourly taurine intake, we then estimated that the daily taurine intake per chick ranged from 0.2 to 2.0 mg with a mean of approximately 1.0 mg per nestling d−1 varying slightly with the age of the chicks. The taurine-supplemented group in this experiment received an extra 0.5 mg per chick d−1 of taurine in 0.05 ml of gelatine from the age of 2 to 6 days and 1 mg per chick d−1 of taurine in 0.1 ml of gelatine from the age of 7 to 14 days, around twice the normal average taurine levels, but within the natural range. The manipulation lasted longer than the natural period of peak spider intake; since rates of development can vary widely in the wild depending on food availability, and also there are few data on the timing of development of key organs and tissues in wild passerines.
The carrier medium of leaf gelatine was dissolved in boiled water and allowed to cool. While the gelatine was still liquid, powdered taurine (Sigma-Aldrich T-8691) was mixed in and transferred into sterile 1 ml syringes (without needles) for feeding. Gelatine contains some protein and fat but has relatively low taurine levels. Control nestlings received the same amount of plain gelatine. Gelatine was used as a carrier medium rather than just water because it was easier to feed a chick an exact amount of gelatine. In addition, we did not want to fill the chicks' crops without providing some nutritional content, as this might have lowered their overall energetic or nutritional intake. All nestlings were weighed and measured daily prior to hand-feeding to monitor morphological development. To control for inter-nest variation in natural taurine intake, half the nestlings within each nest were randomly assigned to the taurine group, with the remainder as controls.
(b) Molecular sex identification
At the age of 14 days, chicks were measured and blood sampled to provide DNA for molecular sex identification. DNA for the overnight sexing was extracted using a Qiagen DNeasy kit. Primers were P2/P8 (Griffiths et al. 1998). PCR amplification was carried out in a total volume of 10 μl. The final reaction conditions were as follows: primers P2 and P8 at 6 pmol, 200 μm of each dNTP, target DNA, 0.35 units Taq polymerase, 2.5 mM MgCl2, 50 mM KCl, 10 mM Tris–HCl (pH 8.8 at 25°C) and 0.1% Triton X-100 (the last four are in Promega Mg/Taq buffer). Thermal cycling was carried out in a Biometra UnoII: 94°/2 min, 30 cycles of (49°/40 s, 72°/40 s and 94°/30 s) 49°/1 min, 72°/5 min. PCR products were separated by electrophoresis on a 2% agarose gel stained with ethidium bromide.
(c) Risk-taking assessment
At the age of 15 days, one taurine-supplemented chick and one control chick from a total of 22 nests were brought into the aviaries at the University of Glasgow (N=41 in all experiments due to the ill health of three individuals). Dyads were matched for sex and size. Half the dyads consisted of sisters and the rest brothers. All offspring from then onwards were maintained on the same dietary regime, so the dietary manipulation ended when the birds were 14 days old. When nutritionally independent, birds were housed individually in similar visual environments.
Propensity for risk taking (also sometimes known as neophobia) was assessed by measuring the reactions of taurine and control birds to novel objects presented in the familiar environment of their home cage (Verbeek et al. 1994). Two novel object trials were performed. At the age of 39 days, a pink plastic frog glued to a perch was presented, and the following day at the age of 40 days, a large shiny metal nut (that screws onto a bolt, i.e. not a seed). These trials were carried out in the home cages of the birds between 11.00 and 13.00. Each sibling pair was tested consecutively on the same day and the order (taurine versus control sibling) was randomly decided by tossing a coin.
The food bowls were removed from the cage 60 min prior to the start of the trial. Then, 30 min before the trial, the water bowl was removed from the cage and a screen was placed 60 cm in front of the cage to conceal the observer and prevent other birds from seeing the novel object and the focal bird's reactions to it. Other forms of disturbance within the room were minimized. The novel object was placed in the far left-hand side of the cage and the bird was observed for 10 min through observation flaps in the screen. During the trial we recorded the time the bird took to land on the novel object perch, if it ever did, and scored the closest distance that the bird got to the novel object. As these two measures were correlated, we present only the time to approach the novel object. The results were averaged across the two trials prior to analysis. Birds that were relatively quick to approach both novel objects were defined as being risk prone, and vice versa for risk-averse birds.
(d) Spatial learning and memory tasks
Using methods established for testing spatial learning in small passerines (Hodgson & Healy 2005), individuals were trained to find food hidden within an array of 48 wells in a wooden tray. From the age of 45 days, each bird was trained to use the tray at its own pace. The bird first foraged for seeds in the tray and then learnt to pull back flaps of felt covering the wells in order to access the seeds. Gradually the ratio of rewarded to unrewarded wells was decreased. When the tray contained one rewarded and six empty holes all fully covered with a felt flap, and the bird was observed to keep removing the flaps until it found the food, testing could begin. Learning ability was tested as follows: following 60 min of food deprivation, the learning tray was placed in the cage. Of the 48 wells, seven were covered with beige felt flaps, only one of which hid food. The bird was allowed to investigate for up to 15 min or until it removed the flap covering food. Once the bird had found the food, it was allowed to eat one piece of sunflower seed from the well before the tray was removed. After 5 min, during which the flaps and food were replaced in their original positions, the tray was then returned to the cage. In phase two, a record was made of all incorrect flaps lifted and the time to locate the food again. Each bird repeated the trial once a day for 3 days with a different spatial arrangement of flaps and food each day. We defined two measures of learning: (i) general learning ability based on whether or not the bird ever learnt to remove flaps to find food in any phase of a trial, and (ii) spatial memory test. For this second tougher spatial memory test, a bird was defined as having succeeded in the spatial learning task if it learnt the location of the food, i.e. made three or fewer mistakes in phase two, in at least two of the three trials. Two measures of learning were used because there was a high degree of variability in learning performance, since some birds never learnt to use the tray, let alone to consistently locate the food using spatial cues.
(e) Statistical analyses
Generalized linear mixed models (GLMMs) with either a normal (e.g. time to approach novel object) or binomial (e.g. learnt/did not learn task) error distribution were used to analyse most of the data (SAS v. 9). Family was entered as a random factor, to control for the non-independence of data from siblings.
3. Results
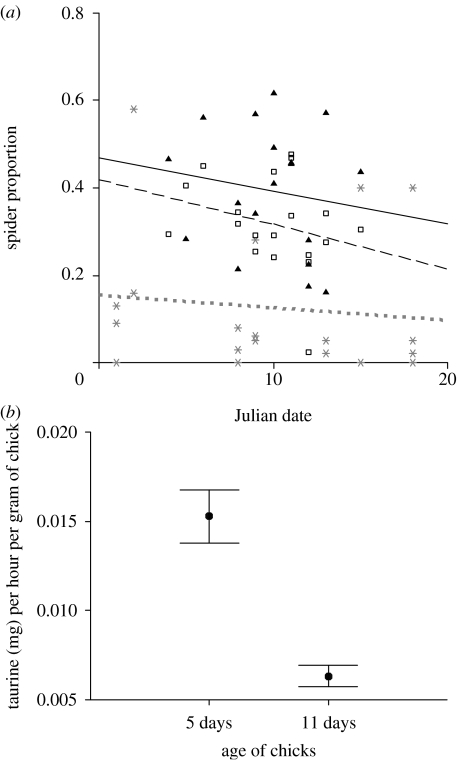
In our population of Scottish blue tits, 5-day-old broods received a significantly higher proportion of spiders in their diets (mean=0.39±0.14) than 11-day-old broods (mean=0.31±0.10; paired t=2.88, d.f.=17, p=0.01). The proportion of spiders in the diet while varying greatly with age of the nestlings was independent of natural spider availability (R2=0.02, d.f.=1,5, p>0.8; figure 1a). The proportions of spiders fed to broods (total number of spiders/total number of spiders, caterpillars and other invertebrates) from 06.00 to 10.00 did not vary significantly with date at the ages of 5 days (R2=0.02, d.f.=1,17, p>0.5) or 11 days (open squares; R2=0.08, d.f.=1,17, p>0.2). The proportion of spiders sampled from oak trees in the study site did not vary with date (R2=0.01, d.f.=1,17, p>0.6). As spiders were considerably less abundant than caterpillars (figure 1a) and had a mass of less than half that of caterpillars, we tested the idea that blue tit parents seek out spiders to provide a particular nutrient required for early development. We found that the age-specific differences in spider provisioning translated into a significantly higher taurine intake per hour (corrected for body size) for a chick aged 5 versus 11 days (paired t-test: t=4.83, d.f.=17, p<0.0001; figure 1b).
Figure 1.
Age-specific diet of nestlings. (a) The proportions of spiders fed to broods (total number of spiders/total number of spiders, caterpillars and other invertebrates) from 06.00 to 10.00 did not vary significantly with date at the ages of 5 (filled triangles, solid line) or 11 days (open squares, dashed line). The proportion of spiders sampled from oak trees in the study site did not vary with date (grey stars, dotted line). See text for further details. Day 1 (when the first brood reached the age of 5 days) was 13 May 2005. Displayed here are actual proportions rather than the arcsine square-root transformed proportions used in the statistical analysis. (b) Estimated hourly taurine intake per chick adjusted for age-specific mean body mass (mg h−1 g−1) was significantly higher for 5-day-old chicks than 11-day-old chicks (N=18 nests). Figure shows mean±s.e.m.
(a) Risk-taking assessment
Next, we tested whether taurine supplementation impacted upon juveniles' risk-taking behaviour when taken into captivity (N=41 chicks from 22 broods). Based on the mean time taken to approach the novel objects, taurine-treated birds were significantly more risk prone than controls (GLMM: F=8.00, d.f.=1,19.1, p=0.011), and heavier juveniles were more risk prone than lighter birds (GLMM: F1,31.3=25.29, p<0.0001). Again, when we investigated just main factors in the model explaining risk-taking behaviour, males (figure 2a) were in general more risk averse than females (figure 2b). Risk-taking behaviour was also affected by an interaction between treatment, sex and body mass (GLMM: F3,18=7.76, p=0.002), with a significant negative relationship between time to approach the novel object and body mass except among taurine-supplemented females (figure 2b). Dyads of siblings had been matched for size when they went into captivity, so at 39 days of age there was no significant difference between the treatment groups in body mass (GLMM: F1,20.6=1.86, p>0.1).
Figure 2.
Risk taking in juvenile blue tits depended upon body mass (age, 39 days), sex and taurine supplementation. Risk taking was measured on the y-axis from risk prone (short mean time to approach the novel objects) to risk averse (long time to approach). (a) males: heavier birds in both treatment groups were significantly more risk prone than lighter birds; however, (b) females: the negative relationship between body mass and risk taking was broken in the taurine group (filled squares, dashed line) but not in the control group (stars, solid line). This interaction remained significant even when a potential outlier (see arrow in b), a particularly heavy, shy female was removed (GLMM: F=4.41, d.f.=3,17.7, p=0.017). Family identity, entered as a random factor, contributed significantly to the full model (z=2.12, N=41, p=0.02), indicating that the variance in risk taking was greater between families than within families.
(b) Spatial learning and memory tasks
In our first general measure of learning ability, taurine-supplemented birds tended to be more likely to use the foraging tray and find food under flaps than controls (68 versus 58%; GLMM: deviance=33.05, F1,28.5=3.43, p=0.07). The sexes did not differ. In the second tougher task focusing on spatial memory and learning, the birds had to remember the spatial location of the food, and 5 min later find the food again with minimal mistakes in at least two out of the three trials. Many birds failed to do this in the majority of the trials but a significantly higher proportion of taurine-supplemented birds were consistently successful in the spatial learning task than their siblings in the control group (46 versus 35%; GLMM: deviance=38.38, F1,27.4=4.57, p=0.042). Taurine-supplemented females also showed a close to significant tendency for better spatial learning abilities than other birds (GLMM: F1,25.4=4.18, p=0.052). In addition, heavier birds (mass=11.05 g±0.13) were significantly more likely to learn to find food using spatial cues than lighter birds (mass=10.83 g±0.15; GLMM: F1,33.7=4.66, p=0.038). Thus, diet quality and quantity influenced spatial learning and memory abilities.
The propensity for risk taking also interacted with diet treatment to significantly affect both the likelihood of learning to use a novel foraging apparatus (GLMM: F1,26.2=6.36, p=0.018) and spatial learning ability (GLMM: F1,28.1=5.57, p=0.025; figure 3). Taurine birds that failed the general learning task were significantly more risk prone than successful birds (figure 3a), and risk-averse control birds consistently failed at both tasks (figure 3a,b).
Figure 3.
Propensity to take risks (mean time to approach a novel object) and nestling diet (taurine (filled circles) or control (crosses) treatment) interacted to affect (a) general (not found/found food) and (b) spatial learning abilities (not learnt/learnt location of food) of juvenile blue tits. The dashed line indicates the mean time to approach the novel objects of all birds in the colony (N=41). Relatively bold, risk-prone birds that had received a taurine supplement had poor general learning abilities and never found food under flaps, and shy, risk-averse controls failed the spatial learning tasks. Taurine birds with intermediate risk-taking propensities and relatively bold controls were significantly more likely to learn to successfully use spatial cues to find food. Family identity was entered into the GLMM as a random factor but was non-significant. Means±s.e.m. are shown.
4. Discussion
Blue tit parents varied the proportion of spiders provided to broods with the age of nestlings, but not in response to the timing of breeding or spider abundance. Indeed the rate at which spiders were provided to nestlings was much higher than the measured level in the oak trees, where the blue tits foraged. Consequently, the body size-corrected intake of taurine was significantly higher at 5 than at 11 days of age. These data suggest that parents were not simply collecting spiders at the rate that they were encountered in the environment. Despite weighing on average less than half that of a caterpillar at that time of year, it is possible that spiders were easier to find than caterpillars because they were darker in colour. Or it might have been that older chicks required larger prey than younger chicks, which explained the switch from spiders to caterpillars later in development. Another explanation is that blue tits were selecting spiders to feed offspring at certain stages of development, possibly owing to a requirement for taurine similar to that of neonatal mammals. Further research is needed to investigate why the nestling need for taurine is potentially so high around the age of 5 days. One possibility is linked with the known role of taurine in retinal and visual cortex development. In most mammals, the development of photoreceptor cells is predominantly postnatal and the concentration of taurine in photoreceptor cells has been shown to increase in parallel with cell maturation (Sturman 1993). The age of 5 days is when parid nestlings usually start to open their eyes, so this is probably a time of peak developmental activity in the visual system. It is also when chicks start moving around the nest, becoming more aware of the spatial layout of the nest cup and the positions of their siblings. Therefore, a second interlinked explanation is that around the age of 5 days, taurine is required for the development of brain regions associated with processing all this newly acquired information. This fits with the data from our study, which suggests that taurine is also used for the development of brain regions associated with responses to stress and spatial learning and memory.
Birds that had received taurine supplementation as nestlings, mimicking a neonatal diet high in spiders, were more likely to be risk takers and had better general and spatial learning abilities than controls. This effect was independent of overall nutritional status because although heavier juveniles were also more risk prone and better learners, there were no significant body size differences between the treatments in our captive colony. In addition, we found that taurine supplementation broke down the relationship between body mass and risk taking in females. Thus, our data suggest that a relatively subtle adjustment in the quality of neonatal nutrition, within the natural range of variation in the wild, has permanent consequences for behavioural performance as adults. In common with most other studies of personality, a longitudinal study would need to be carried out to determine whether traits displayed at one life stage are consistent within individuals across different contexts and ages.
Individuals showing extremes of risk taking were least successful at learning the spatial foraging task. From the controls, it appeared that in a normal spectrum of behavioural types, non-learners were particularly risk-averse birds. During training, these shy individuals generally avoided the spatial learning tray, so never learnt to find the food (K. E. Arnold & S. L. Ramsay 2005, unpublished data). Supplementation with taurine seemed to result in birds being on average more risk prone, which presumably for inherently shy birds would have enhanced their learning performance. More work is needed to tease out whether taurine supplementation results in naturally risk-prone birds becoming too bold and/or proactive to successfully learn.
These interactions between taurine, stress responsiveness and spatial learning suggest a common underlying mechanism. In rodents, a proactive, risk-prone, coping style is associated with low HPA reactivity in response to stress and vice versa for reactive, risk-averse, animals (Koolhaas et al. 1999). Since taurine supplementation of wild birds resulted in more risk-prone individuals compared with the norm, taurine would seem to affect the development of brain regions involved in controlling the HPA axis. The hippocampus, with its high density of glucocorticoid receptors, is one of the key limbic structures known to be involved in the termination of HPA activity following stress (reviewed by Sapolsky et al. 2000). It is also the region of the brain involved in spatial learning and memory in birds and other taxa (Healy & Krebs 1993). Additionally, it is known that the in vitro electrophysiological activity of the adult hippocampus is enhanced by taurine supplementation of neonatal laboratory mice (Franconi et al. 2004). Our data suggest that taurine supplementation of outbred, wild, neonatal birds impacted upon hippocampal development and function, which, in turn, affected stress responsiveness and spatial learning.
Intriguingly, our results suggest that the development of female brains might be more sensitive to neonatal taurine intake than that of males. Taurine supplementation of females, but not males, broke the positive relationship between body mass and risk taking found among control birds. In addition, individuals in the taurine group were more likely to remember the spatial location of food with a significant female bias among these successful learners. This is in contrast to a number of correlational studies that have found males to have better spatial memories than females (but see review by Jones et al. 2003). Consequently, we show that a sex difference in neonatal diet has the potential to mediate or perhaps ameliorate sex differences in spatial learning and memory.
The provisioning of rare prey, providing important but potentially limiting nutrients, has been shown to have clear impacts on adult behaviour and cognition, probably via effects on neonatal hippocampal development. In adult birds, spatial learning ability and propensity to take risks would influence fitness via impacts on an individual's competitive ability and foraging success. Theory predicts that there are costs (e.g. energy and/or predation risk) and benefits (e.g. increase in competitive or foraging success) to being a risk taker or having high cognitive ability. As with other variable fitness-related traits, the relative pay-offs associated with one phenotype are environmentally or frequency dependent and, therefore potentially predictable by provisioning parents. Early nutrition mediated by parental prey selection is, therefore, a mechanism for manipulating not only physiological and life-history traits (Blount et al. 2003; Metcalfe & Monaghan 2003; Arnold et al. 2007), but also behavioural phenotype.
Acknowledgments
All work followed ethical guidelines of the UK and experimental procedures were carried out under licence from the UK Home Office.
We thank Lindsay Henderson, Gabrielle Roy, Patrick White, Clare Toner and many others for their assistance in the field and aviary. Zoe Hodgson and Sue Healy kindly provided advice on spatial learning tests and Nicky Clayton guidance on hand-rearing passerines. Feedback was provided by R. Nager and P. Monaghan. Comments by John Quinn and an anonymous reviewer greatly improved the manuscript. This work was supported by the Leverhulme Trust, the Natural Environment Research Council, the Glasgow Natural History Society and the University of Glasgow. K.E.A. was funded by a Royal Society University Research Fellowship and C.D. by a Royal Society Summer Studentship.
References
- Aerts L, Assche F.A.V. Taurine and taurine-deficiency in the perinatal period. J. Perinat. Med. 2002;30:281–286. doi: 10.1515/JPM.2002.040. doi:10.1515/JPM.2002.040 [DOI] [PubMed] [Google Scholar]
- Arnold K.E, Blount J.D, Metcalfe N.B, Orr K.J, Adam A, Houston D, Monaghan P. Sex-specific differences in compensation for poor neonatal nutrition in the zebra finch Taeniopygia guttata. J. Avian Biol. 2007;38:356–366. doi:10.1111/j.0908-8857.2007.03818.x [Google Scholar]
- Betts M.M. The food of titmice in oak woodland. J. Anim. Ecol. 1955;24:282–323. doi:10.2307/1715 [Google Scholar]
- Blount J.D, Metcalfe N.B, Arnold K.E, Surai P.F, Devevey G.L, Monaghan P. Neonatal nutrition, adult antioxidant defences and sexual attractiveness in the zebra finch. Proc. R. Soc. B. 2003;270:1691–1696. doi: 10.1098/rspb.2003.2411. doi:10.1098/rspb.2003.2411 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carere C, Drent P.J, Koolhaas J.M, Groothuis T.G.G. Epigenetic effects on personality traits: early food provisioning and sibling competition. Behaviour. 2005;142:1329–1355. doi:10.1163/156853905774539328 [Google Scholar]
- Cowie R.J, Hinsley S.A. Feeding ecology of great tits (Parus major) and blue tits (Parus caeruleus), breeding in suburban gardens. J. Anim. Ecol. 1988;57:611–626. doi:10.2307/4928 [Google Scholar]
- Engelmann M, Landgraf R, Wotjak C.T. Taurine regulates corticotropin secretion at the level of the supraoptic nucleus during stress in rats. Neurosci. Lett. 2003;348:120–122. doi: 10.1016/s0304-3940(03)00741-9. doi:10.1016/S0304-3940(03)00741-9 [DOI] [PubMed] [Google Scholar]
- Franconi F, Diana G, Fortuna A, Galietta G, Trombetta G, Valentini G, Seghieri G, Loizzo A. Taurine administration during lactation modifies hippocampal CAl neurotransmission and behavioural programming in adult male mice. Brain Res. Bull. 2004;63:491–497. doi: 10.1016/j.brainresbull.2004.04.004. doi:10.1016/j.brainresbull.2004.04.004 [DOI] [PubMed] [Google Scholar]
- Griffiths R, Double M.C, Orr K, Dawson R.J.G. A DNA test to sex most birds. Mol. Ecol. 1998;7:1071–1075. doi: 10.1046/j.1365-294x.1998.00389.x. doi:10.1046/j.1365-294x.1998.00389.x [DOI] [PubMed] [Google Scholar]
- Grundel R, Dahlsten D.L. The feeding ecology of mountain chickadees (Parus gambeli)—patterns of arthropod prey delivery to nestling birds. Can. J. Zool. 1991;69:1793–1804. [Google Scholar]
- Healy S.D, Krebs J.R. Development of hippocampal specialisation in a food-storing bird. Behav. Brain Res. 1993;53:127–131. doi: 10.1016/s0166-4328(05)80272-4. doi:10.1016/S0166-4328(05)80272-4 [DOI] [PubMed] [Google Scholar]
- Hodgson Z, Healy S. Preference for spatial cues in a non-storing songbird species. Anim. Cogn. 2005;8:211–214. doi: 10.1007/s10071-004-0249-4. doi:10.1007/s10071-004-0249-4 [DOI] [PubMed] [Google Scholar]
- Jones C.M, Braithwaite V.A, Healy S.D. The evolution of sex differences in spatial ability. Behav. Neurosci. 2003;117:403–411. doi: 10.1037/0735-7044.117.3.403. doi:10.1037/0735-7044.117.3.403 [DOI] [PubMed] [Google Scholar]
- Koolhaas J.M, Korte S.M, Boer S.F.D, Vegt B.J.V.D, Reenen C.G.V, Hopster H, Jong I.C.D, Ruis M.A.W, Blokhuis H.J. Coping styles in animals: current status in behavior and stress-physiology. Neurosci. Biobehav. Rev. 1999;23:925–935. doi: 10.1016/s0149-7634(99)00026-3. doi:10.1016/S0149-7634(99)00026-3 [DOI] [PubMed] [Google Scholar]
- Lapin I.P. Neurokynurenines (NEKY) as common neurochemical links of stress and anxiety. Adv. Exp. Med. Biol. 2003;527:121–125. doi: 10.1007/978-1-4615-0135-0_14. [DOI] [PubMed] [Google Scholar]
- Lehmann A, Hagberg H, Hamberger A. A role for taurine in the maintenance of homeostais in the central nervous system during hyperexcitation. Neurosci. Lett. 1984;52:341–346. doi: 10.1016/0304-3940(84)90185-x. doi:10.1016/0304-3940(84)90185-X [DOI] [PubMed] [Google Scholar]
- Magrath M.J.L, Lieshout E.V, Visser G.H, Komdeur J. Nutritional bias as a new mode of adjusting sex allocation. Proc. R. Soc. B. 2004;271(Suppl.):S347–S349. doi: 10.1098/rsbl.2004.0187. doi:10.1098/rsbl.2004.0187 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGraw K.J. The antioxidant function of many animal pigments: are there consistent health benefits of sexually selected colourants? Anim. Behav. 2005;69:757–764. doi:10.1016/j.anbehav.2004.06.022 [Google Scholar]
- Metcalfe N.B, Monaghan P. Growth versus lifespan: perspectives from evolutionary ecology. Exp. Gerontol. 2003;38:935–940. doi: 10.1016/s0531-5565(03)00159-1. doi:10.1016/S0531-5565(03)00159-1 [DOI] [PubMed] [Google Scholar]
- Naef-Daenzer L, Naef-Daenzer B, Nager R.G. Prey selection and foraging performance of breeding great tits Parus major in relation to food availability. J. Avian Biol. 2000;31:206–214. doi:10.1034/j.1600-048X.2000.310212.x [Google Scholar]
- Ramsay S.L, Houston D.C. Amino acid composition of some woodland arthropods and its implications for breeding tits and other passerines. Ibis. 2003;145:227–232. doi:10.1046/j.1474-919X.2003.00133.x [Google Scholar]
- Sapolsky R.M, Romero L.M, Munck A.U. How do glucocorticoids influence stress responses? Integrating permissive, suppressive, stimulatory, and preparative actions. Endocr. Rev. 2000;21:55–89. doi: 10.1210/edrv.21.1.0389. doi:10.1210/er.21.1.55 [DOI] [PubMed] [Google Scholar]
- Sih A, Bell A.M, Johnson J.C, Ziemba R.E. Behavioral syndromes: an integrative review. Q. Rev. Biol. 2004;79:241–277. doi: 10.1086/422893. doi:10.1086/422893 [DOI] [PubMed] [Google Scholar]
- Sturman J.A. Taurine in development. Physiol. Rev. 1993;73:119–147. doi: 10.1152/physrev.1993.73.1.119. [DOI] [PubMed] [Google Scholar]
- Tonkiss J, Smart J.L. Interactive effects of genotype and early life undernutrition on the development of behavior in rats. Dev. Psychobiol. 1983;16:287–301. doi: 10.1002/dev.420160404. doi:10.1002/dev.420160404 [DOI] [PubMed] [Google Scholar]
- Verbeek M.E.M, Drent P.J, Wiepkema P.R. Consistent individual differences in early exploratory behaviour of male great tits. Anim. Behav. 1994;48:1113–1121. doi:10.1006/anbe.1994.1344 [Google Scholar]