Abstract
Recent studies of corticofugal modulation of auditory information processing indicate that cortical neurons mediate both a highly focused positive feedback to subcortical neurons “matched” in tuning to a particular acoustic parameter and a widespread lateral inhibition to “unmatched” subcortical neurons. This cortical function for the adjustment and improvement of subcortical information processing is called egocentric selection. Egocentric selection enhances the neural representation of frequently occurring signals in the central auditory system. For our present studies performed with the big brown bat (Eptesicus fuscus), we hypothesized that egocentric selection adjusts the frequency map of the inferior colliculus (IC) according to auditory experience based on associative learning. To test this hypothesis, we delivered acoustic stimuli paired with electric leg stimulation to the bat, because such paired stimuli allowed the animal to learn that the acoustic stimulus was behaviorally important and to make behavioral and neural adjustments based on the acquired importance of the acoustic stimulus. We found that acoustic stimulation alone evokes a change in the frequency map of the IC; that this change in the IC becomes greater when the acoustic stimulation is made behaviorally relevant by pairing it with electrical stimulation; that the collicular change is mediated by the corticofugal system; and that the IC itself can sustain the change evoked by the corticofugal system for some time. Our data support the hypothesis.
Keywords: descending system/hearing/learning/plasticity/tonotopic map
The response properties of neurons in the subcortical auditory nuclei, as well as in the auditory cortex (AC) can be changed by associating an acoustic stimulus (conditioning stimulus: CS) with an electric foot stimulus (unconditioned stimulus). After conditioning, the response of neurons in the AC (1–3) and the medial geniculate body (MGB) of the adult guinea pig (4, 5) increases to the frequency of a CS tone, but decreases to other frequencies, including the original best frequencies (BFs) of the neurons. These changes result in a shift in BF toward the frequency of the CS tone. The BF shift lasts at least 8 weeks in the AC (3), at least 1 hr in the dorsal division of the MGB (4), and less than 1 hr in the ventral division of the MGB (5). In the rat, the response of neurons in the inferior colliculus (IC) increases to frequencies used for a discrimination task (6), and 2-deoxyglucose uptake in the IC increases in the iso-BF band representing the frequency of the CS tone (7). All of these studies indicate that information processing in the central auditory system can be modified by auditory experience. However, the neural mechanisms underlying these changes have mostly remained unexplored.
Recent studies indicate that cortical neurons of the mustached bat (Pteronotus parnellii) mediate, via corticofugal projection, a highly focused positive feedback to subcortical neurons “matched” in tuning to a particular acoustic parameter and a widespread lateral inhibition to “unmatched” subcortical neurons. This cortical feedback results in the adjustment and improvement of subcortical signal processing (i.e., the adjustment and improvement of the cortical neurons’ own input). This function, named egocentric selection, enhances the neural representation of frequently occurring signals in the central auditory system (8–10). Egocentric selection shifts the BFs of collicular neurons not only toward the BF of electrically stimulated cortical neurons but also toward the frequency of a repetitively delivered acoustic stimulus (tone burst), resulting in local reorganization of the frequency map in the IC (11). It is likely that the changes observed in subcortical auditory nuclei after conditioning are partially, if not totally, mediated by the corticofugal system. Therefore, we hypothesized that the corticofugal system adjusts and improves subcortical information processing according to auditory experience based on associative learning.
To test our hypothesis, we delivered an acoustic stimulus paired with a mild electric stimulus of the leg of the big brown bat (Eptesicus fuscus). This classical conditioning paradigm permitted the animal to learn that the acoustic stimulus predicted the electrical stimulation and to make behavioral and neuronal adjustments based on the learned importance of the acoustic stimulus. We found that the frequency-response curves of neurons in the central nucleus of the IC shift toward the frequency of the acoustic stimulus, that the AC is necessary to evoke the shift, and that the IC sustains the shift for some time once it is evoked, without further input from the AC.
We studied the IC rather than the MGB for the following reasons. The central nucleus of the IC receives ascending inputs from 10 or more lower-order auditory nuclei and projects to the MGB and, to a lesser extent, to other divisions of the IC and the superior colliculus. In addition, the central nucleus of the IC receives the corticofugal projections and projects to the lower-order auditory nuclei. The central nucleus of the IC is an important site where ascending auditory signals are processed, and the processing is modulated by descending corticofugal fibers (12). Despite this complexity of connections, the central nucleus of the IC has a single, simple cochleotopic frequency map (13).
MATERIALS AND METHODS
Preparation.
Experiments were performed with 26 adult big brown bats, E. fuscus, whose auditory system is basically the same as that of other mammalian species (13–15). Procedures for animal preparation, acoustic stimulation, and recording of action potentials have been previously described (11, 16). The protocol for this research was approved by the animal studies committee of Washington University. Under neuroleptanalgesia (Innovar 4.0 mg/kg body weight), a 15-mm-long metal post was glued on the dorsal surface of the bat’s skull. Experiments for recording auditory responses from single or multiple neurons began 3–4 days after the surgery. The unanesthetized bat was placed in a styrofoam restraint suspended by an elastic band at the center of a soundproof room maintained at a temperature of about 31°C. The head was immobilized by fixing the metal post glued on the skull onto a metal rod with set screws, and it was adjusted to face directly at a loudspeaker located 74 cm away.
Acoustic and/or Electric Stimuli.
For the measurement of the frequency-tuning or frequency-response curves of collicular neurons, acoustic stimuli (20-ms tone bursts with a 0.5-ms rise-decay time) were delivered to the bat at a rate of 5/s. Their frequency and amplitude were manually varied. The frequency-tuning curve of single or multiple neurons first was measured audio-visually. Then, the tone bursts were computer-controlled. The sharpness of the manually measured tuning curve was used to determine the step size (0.2–0.5 kHz) of a computer-controlled frequency scan: the sharper the tuning curve, the smaller the step. The frequency scan usually consisted of 21 200-ms time blocks or 34 150-ms time blocks when a tuning curve was particularly wide. In each scan, a single tone burst was delivered at the beginning of each block, and the frequency of the tone burst was shifted in 20 or 33 steps across the BF of the neuron. The amplitude of the tone bursts in the scan was always set at 10 dB above the minimum threshold of the neuron, so as to easily detect a shift in BF. An identical frequency scan was delivered 50 or 100 times to obtain an array of poststimulus time (PST) histograms as a function of frequency. BFs and frequency-response curves were obtained by counting the total numbers of impulses discharged to 50 or 100 identical tone bursts.
To evoke changes in the BFs of collicular neurons, i.e., in the frequency map of the IC, two types of acoustic stimuli were delivered. We named them repetitive acoustic stimuli (ASr) and a train of acoustic stimuli (ASt) for convenience. In ASr and ASt, short tone bursts were delivered at rates chosen to be consistent with the acoustic behavior of the bat; the echo-locating big brown bat emits short orientation sounds at ≈10/s in a search phase, 30–40/s at the middle of an approach phase, and 100–200/s in a terminal phase (17, 18). In ASr, the tone bursts at a given frequency between 21 and 55 kHz were 50 dB SPL (decibels in sound pressure level re. 20 μPa) and 20 ms long. They were delivered at a rate of 10/s for 30 min In ASt, the tone bursts at a given frequency also between 21 and 55 kHz were 50 dB SPL and 10 ms-long. The rate of the tone bursts within ASt was 33/s. ASt was 1.0 s long and was delivered every 30 s for 30 min (60 times in total).
The electric shock to the bat’s leg (ESl) was a 50-ms monophasic electric pulse and was delivered every 30 s for 30 min (60 times in total). ESl was either delivered alone or in a pair with ASt. When paired, it was delivered 1.0 s after or before ASt. Therefore, ASt + ESl was 1.0-s ASt + 1.0-s gap + 50-ms ESl, and ES1 + ASt was 50-ms ES1 + 1.0-s gap + 1.0-s ASt. One of these paired stimuli was delivered every 30 s for 30 min. The intensity of ESl was just above the threshold (0.15–0.57 mA) for eliciting a just-noticeable leg flexion, which was monitored with a strain gauge. ASt and ESl are a conditioning and an unconditioned stimulus, respectively.
There were four steps in our experiments: (step 1) to confirm the shift in BF, i.e., the shift in the frequency map observed in the central nucleus of the IC by Yan and Suga (11), we delivered ASr in the same way as in that research. The BFs of single or multiple IC neurons were measured before and after 30-min delivery of ASr. (Step 2) To examine whether auditory experience based on associative learning evokes a larger BF shift than that observed in step 1, the BFs of IC neurons were measured before and after 30-min delivery of ASt + ESl or ES1 + ASt. They also were measured before and after 30-min delivery of ASt or ESl. (Step 3) To examine whether BF shifts in the IC evoked by ASt + ESl are mediated by the corticofugal system, the corticofugal system was inactivated during ASt + ESl or immediately after collicular BF shifts were evoked by ASt + ESl by applying 0.4 μg of muscimol (an agonist of an inhibitory synaptic transmitter, γ-aminobutyric acid) to the primary AC (AI) 20 min before or 30–50 min after ASt + ESl. The method used to apply muscimol to the AC was the same as that in a previous study (19). (Step 4) To examine whether the primary somatosensory cortex (SI) influences the collicular BF shifts evoked by ASt + ESl, the SI was inactivated during ASt + ESl by applying 0.4 μg of muscimol to the SI 20 min before ASt + ESl. The AI and SI, respectively, were localized by identifying the frequency (15) and somatotopic (20) maps, and by recording neural responses to acoustic or touch stimuli before muscimol application.
Recording and Evaluation of Neural Responses.
To assess the BF shifts of collicular neurons (i.e., the change in the frequency map of the central nucleus of the IC) evoked by acoustic and/or electric stimulation, multi-unit mapping and single-unit recording experiments were performed before and after the stimulation. In the multi-unit mapping experiments, the BFs of multiple IC neurons (2–3 neuron clusters) first were recorded with a tungsten-wire microelectrode (≈7-μm tip diameter) at about 100-μm intervals along a dorsoventral electrode penetration. These recordings yielded a BF-depth curve in the control condition (i.e., the condition before the 30-min delivery of acoustic and/or electric stimuli). We then delivered either acoustic stimuli (ASr or ASt), electric stimuli (ESl), or both for 30 min. Within a half-minute thereafter, we started to remeasure the BF-depth curve. The curve was repeatedly measured while the electrode was first withdrawn near to the surface of the IC while remaining within the IC, and then reinserted. This procedure continued until the BF-depth curve returned to its control values (11). It took about 30 min to obtain a single BF-depth curve. Each multi-unit mapping experiment produced many data points indicating BF shifts.
In the single-unit experiments, responses of single neurons to tone bursts were recorded, and their BFs and frequency-response curves were measured before and after the stimulations described above. A recording electrode was not moved until all of the measurements on a given single neuron were completed. Each single-unit experiment produced just one datum indicating a BF shift.
The criteria used to define shift in the frequency-response curve (or BF) of a single neuron by acoustic and/or electrical stimulation (9, 11) are summarized below. If a shifted frequency-response curve did not recover by more than 50% before the disappearance of action potentials of the neuron, the data were excluded from our analysis. In a stable, long-lasting recording, all curves shifted by the stimulation recovered by more than 50%. This recovery itself helps to prove that the shift was significant. Furthermore, the shift was considered to be significant only if the shift in BF was accompanied by a shift in the same direction as that of the BF shift of more than 75% of the data points in the frequency-response curve. When a BF shift was small and its significance was not obvious, a weighted average frequency (i.e., BF) was calculated for the summed response to five consecutive frequency scans. Then the mean and SD of these weighted averages were computed, and a two-tailed, paired t test was used to determine whether or not the weighted average frequencies (BFs) obtained for control and stimulus conditions were significantly different for P < 0.01. The t test also was used to test the significance of difference between the data sets obtained before and after the acoustic and/or electrical stimulation.
RESULTS
BF Shifts in the IC Evoked by Acoustic Stimulation Paired with Electric Leg-Stimulation.
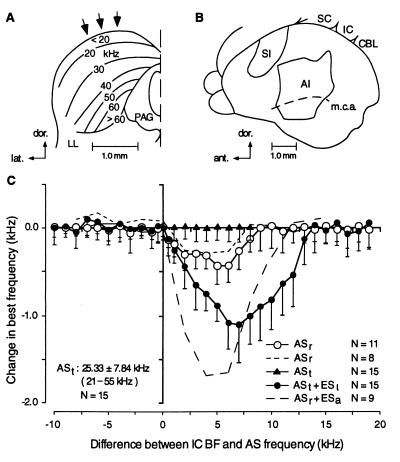
In dorsoventral electrode penetrations through the IC (Fig. 1A, arrows), BFs of neurons systematically increased, as previously reported by Casseday and Covey (13). The BF-depth curve remained highly consistent over multiple identical penetrations. That is, the BF measured at the same location stayed unchanged. However, when ASr was delivered for 30 min, the BF-depth curve (i.e., BFs) was lowered toward the ASr frequency, but only at locations tuned to frequencies within 10 kHz above the ASr frequency (Fig. 1C, ○), as previously shown by Yan and Suga (11) (Fig. 1C, short dashed line). For example, BF shift toward an ASr of 20 kHz occurred only in neurons with a BF between 20 and 30 kHz, whereas BF shift toward an ASr of 35 kHz occurred only in neurons with a BF between 35 and 45 kHz. The mean maximum shift was 0.43 ± 0.22 kHz, which occurred at 5.0 kHz above the ASr frequency.
Figure 1.
Shift in the BFs of neurons in the IC evoked by acoustic stimuli (AS) or AS paired with electric stimuli (ES). A and B show the left IC (frontal section) and the left cerebral cortex (dorsolateral surface view), respectively. The numbers and lines in A indicate iso-BF contour lines. The arrows in A indicate dorsoventral electrode penetrations. AI, primary auditory cortex; CBL, cerebellum; LL, lateral lemniscus; m.c.a., middle cerebral artery; PAG: periaqueductal gray; SC, superior colliculus. A and B are based on Dear et al. (15) and Casseday and Covey (13), respectively. (C) The amount of shift in BF as a function of the difference between the BFs of collicular neurons and the frequency of AS. ASr, delivered repetitively for 30 min. ASt, delivered as a 1.0-s train. ESa, focal electric stimulation of the AI. Each symbol and vertical bar indicate a mean and a SD, respectively. ASt alone evoked no BF shift, so that the data points are plotted only for 1–8 kHz differences between IC BF and AS frequency. N, the numbers of BF-depth curves used for averaging. The frequency of ASt ranged from 21 to 55 kHz (25.33 ± 7.84 kHz, n = 15). The short and long dashed curves were obtained by Yan and Suga (11).
The behavioral response to an electrical leg stimulation (ESl) was leg flexion and body movement. When ASt followed by ESl was delivered for 30 min, the animal showed the behavioral response to every ESl, and in addition, it showed the response to ASt in the last 10 min of the 30-min period. The animal apparently was conditioned, i.e., it learned the relationship between ASt and ESl. When ASt alone was delivered to the animal for 30 min, 2–3 hr after ASt + ESl, it did not evoke the behavioral response. This lack of behavioral response was probably because of the extinction of the conditioned response by repetitive delivery of tone bursts, which were used for the measurement of frequency-response curves during the 2- to 3-hr period.
When ASt + ESl was delivered for 30 min, the shift in the BF-depth curve became approximately three times larger than that produced by ASr alone, and the shift occurred within 15 kHz above the ASt frequency (Fig. 1C, •) and persisted 2–3 hr, even though the total number of tone bursts in 60 ASts was only 1,980, instead of 18,000 tone bursts in a single ASr, and each tone burst in ASt was only 10 ms, instead of 20 ms in ASr. The mean maximum shift for ASt + ES1 was −1.1 ± 0.44 kHz, which occurred at 7.0 kHz above the ASt frequency. Unlike ASt +ES1, ES1 + ASt delivered for 30 min did not evoke BF shift of collicular neurons (0.01 ± 0.2 kHz, n = 11, P > 0.5). Neither ASt nor ESl delivered alone for 30 min evoked a BF shift (Fig. 1C, ▴). That is, BF shift was −0.01 ± 0.18 kHz (n = 15, P > 0.5) for ASt alone and 0.02 ± 0.23 kHz (n = 15, P>0.5) for ES1 alone. Each mean ± SD of the above was calculated for collicular neurons with a BF of 6.0 kHz above ASt frequency, where the largest BF shift was expected (see Fig. 1C). Because ASr alone evoked a BF shift, ASt also should have evoked a BF shift, but it was perhaps too small to be detected.
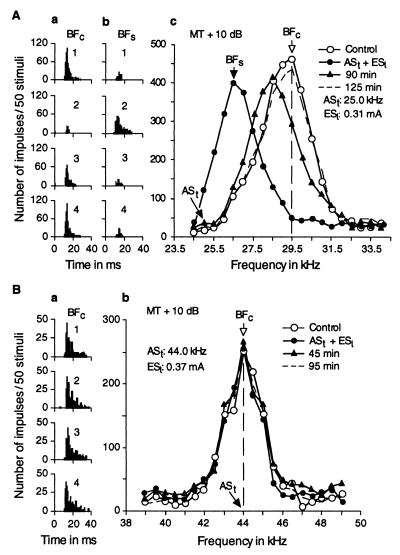
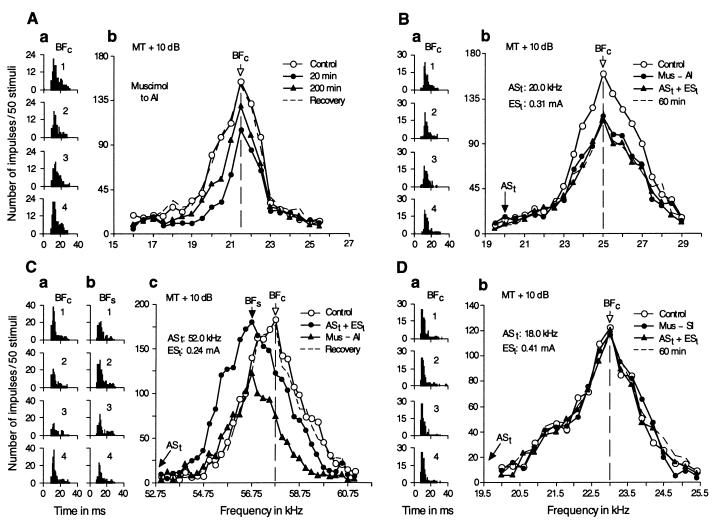
A BF shift always was associated with frequency-dependent changes in response magnitude. Before stimulation by ASt, the single IC neuron in Fig. 2A was tuned to 29.5 kHz (○ in Fig. 2Ac). When a 25.0-kHz tone burst was delivered in the ASt + ESl paradigm for 30 min, the neuron’s response to a 29.5-kHz tone burst decreased by 88%, whereas its response to a 26.5-kHz tone burst increased by 300% (2 in Fig. 2A a and b). In other words, the BF of this neuron shifted downward (Fig. 2Ac, •). About 1 hr after ASt + ESl, the frequency-response curve started to recover (Fig. 2Ac, ▴). The complete recovery of the curve occurred 125 min after ASt + ESl (dashed curve in Fig. 2Ac). The PST histograms in Fig. 2A a and b show the responses at the BFs in the control condition (BFc) and in the “shifted” condition (BFs) in four different situations corresponding to the four curves in Fig. 2Ac.
Figure 2.
Changes or lack of changes in the responses and frequency-response curves of two single collicular neurons evoked by ASt + ESl. The responses and frequency-response curves were obtained with tone bursts fixed at 10 dB above the minimum threshold of a given neuron. (A) The frequency of ASt was 25.0 kHz, and the BF of the neuron was 29.5 kHz. (B) The frequency of ASt was 44.0 kHz, which was the same as the BF of the neuron. The data obtained before, immediately after, and 90 and 125 min (A) or 45 and 95 min (B) after ASt + ESl are represented by open circles, filled circles, filled triangles and dashed lines, respectively. BFc and BFs are indicated by arrows. The PST histograms in A a and b show the changes in response at the BFc and BFs evoked by ASt + ESl. The PST histograms in Ba show no change in response at the BFc, which was not shifted by ASt + ESl. In these PST histograms, 1–4, respectively, correspond to the four conditions for the frequency-response curves: control, ASt + ESl, 90 or 45 min after and 125 or 95 min after ASt + ESl.
IC neurons with the same BF as the ASt frequency showed no change in frequency-response curve or response magnitude for ASt + ESl (Fig. 2B). All collicular neurons with BF lower than or more than 15 kHz higher than the ASt frequency also showed no change in frequency-response curve or response magnitude.
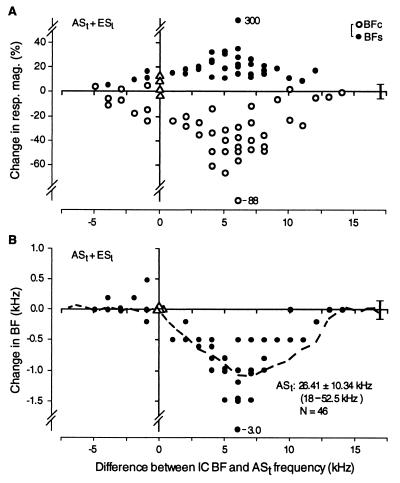
Because the decrease in response at BFc, the increase in response at BFs, and the BF shift differed quantitatively from neuron to neuron, all the data obtained from 46 single collicular neurons are presented in Fig. 3. Thirty of 32 neurons with BF between the ASt frequency and 14 kHz above it showed changes in response magnitude (decrease at BFc and increase at BFs) and downward shift in BF toward the ASt frequency. The amount of decrease in response at BFc was about two times larger than the amount of increase in response at BFs (Fig. 3A, ○ vs. •). Four neurons with a BF that was the same as the ASt frequency showed no BF shift, but two of them showed a small amount of increase in response only at the BFc (Fig. 3, ▵). Three of four neurons with a BF between the ASt frequency and 2.5 kHz below it showed a decrease in response to BFc and an increase in response to BFs. Their BFs were shifted either away or toward the ASt frequency. These changes were small, but statistically significant. As observed with multi-unit recording, the maximum BF shift was observed for single neurons with a BF that was 5–7 kHz higher than the ASt frequency. These single-unit data showed no significant difference from the multi-unit data (P = 0.50). The largest BF shift observed was 3.0 kHz (Fig. 3B). Because BF shifts of almost all collicular neurons were downward toward the ASt frequency, the shifts resulted in an over-representation of the frequency of ASt and a related under-representation of frequencies between 8 and 13 kHz above the ASt frequency.
Figure 3.
Changes in response magnitude (A) and BF (B) of 46 single collicular neurons as a function of difference in their BFs and the ASt frequency. (A) Almost all open circles show reduced responses to a tone burst at the original BF in the control condition (BFc), and all filled circles show increased responses to a tone burst at the BF shifted by ASt + ESl (BFs). (B) The filled circles indicate that almost all neurons with a BF higher than the ASt frequency shifted their BFs downward toward the ASt frequency. The dashed curve in B shows the BF shift observed in the multi-unit mapping experiments shown in Fig. 1C. The triangles indicate the data obtained from neurons whose BFs were the same as the ASt frequency. The vertical lines on the right indicate ± one SD for variations in response magnitude or BF in the control condition.
Effect of Cortical Inactivation on BF Shifts in the IC.
Because focal electrical stimulation of the AC evokes BF shifts (long dashed line in Fig. 1C) (11) similar to those evoked by ASt + ESl, we hypothesized that the changes evoked by ASt + ESl were mediated by the corticofugal system. To test this hypothesis, cortical inactivation experiments were performed with muscimol, which is a potent agonist of γ-aminobutyric acid (GABA), an inhibitory synaptic transmitter.
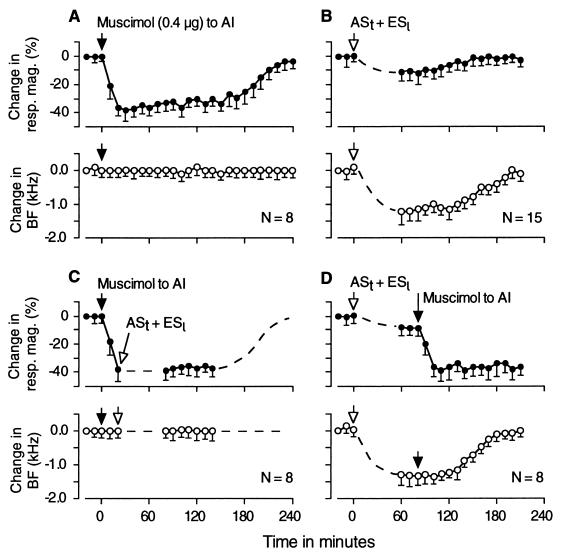
When 0.4 μg of muscimol was applied to the large region of the primary AC (Fig. 1B) containing matched neurons, which had the same BF as that of given collicular neurons, the magnitude of responses of the collicular neurons to tone bursts decreased by 29–51% (38 ± 8.1%, n = 8) over 2–3 hr, but their BFs showed no change (0.00 ± 0.21 kHz, n = 8). Fig. 4A shows the mean time course of the change in response magnitude evoked by muscimol in eight neurons. The large effect of cortical inactivation on collicular responses indicates that the corticofugal system amplifies collicular responses to single tone bursts 1.6 times on the average. Similar data to the above also has been obtained in the mustached bat, in which cortical inactivation by muscimol reduced collicular responses to single tone bursts by 34 ± 7.8%. That is, corticofugal amplification was 1.5 times (19).
Figure 4.
Mean time courses of changes in response magnitude (•) and BF (○) observed in collicular neurons. (A and B) The effect of 0.4 μg of muscimol applied to the AI and the effect of ASt + ESl on 8 and 15 collicular neurons, respectively. Muscimol evoked a reduction of response, but not a BF shift. ASt + ESl evoked a reduction in both response and BF. A response magnitude was measured at the BF in the control or shifted condition shown by ○ in the lower graph. (C) Muscimol first was applied to the AI, and then, during AI inactivation, ASt + ESl were delivered to the bat. These stimuli failed to evoke changes in the IC. (D) ASt + ESl were delivered to the bat, and, after the changes in the IC were evoked by these stimuli, muscimol was applied to the AI. The BF shift evoked by ASt + ESl stayed unaffected. The arrows indicate the time when muscimol or ASt + ESl was delivered. (C and D) Eight collicular neurons were studied. Each filled or opened circle and vertical bar indicates a mean and a SD, respectively.
The mean time courses of changes in response magnitude and BF evoked by ASt + ESl in 15 collicular neurons are shown in Fig. 4B. The maximum reductions in response (10 ± 7.2%) and BF (1.1 ± 0.44 kHz) were obtained by the end of ASt + ESl. Fig. 4 A and B is plotted to be used as the reference for evaluating the data presented in Fig. 4 C and D.
To examine the role of the AC in the collicular changes evoked by ASt + ESl, muscimol was applied to the primary AC, and then, 20 min later, ASt + ESl was delivered to the bat. That is, ASt + ESl was delivered during cortical inactivation. As expected, the collicular response to ASt was reduced significantly (39 ± 9.3%, n = 8). Interestingly, however, neither a further reduction in response nor a BF shift was evoked in the IC by ASt + ESl (Fig. 4C). When ASt + ESl was first delivered to the bat, and then 30–50 min later, muscimol was applied to the primary AC, muscimol evoked an additional reduction in response (32.1 ± 8.1%, n = 8) to that evoked by ASt + ESl (7.9 ± 6.2%, n = 8), but affected little both the amount (1.3 ± 0.32 kHz, n = 8) and the time course of the BF shift evoked by ASt + ESl (Fig. 4D).
The results shown in Fig. 4 are substantiated with PST histograms and frequency-response curves of single collicular neurons in Fig. 5. The neuron in Fig. 5A was tuned to 21.5 kHz (BFc). Application of muscimol to the primary AC reduced the response of this neuron at its BFc by 36%, but evoked no BF shift. The effect of muscimol lasted for nearly 4 hr. Fig. 5B shows the data obtained from a collicular neuron tuned to 25.0 kHz. When muscimol was applied to the primary AC and then, 20 min later (during muscimol inactivation), ASt + ESl was delivered to the bat, the response of this neuron was affected by muscimol, but not by ASt + ESl. Fig. 5C shows the data obtained from a collicular neuron when ASt + ESl was delivered to the bat and then, 30 min later (during the change evoked by ASt + ESl), muscimol was applied to the primary AC. ASt + ESl shifted the frequency-response curve of this neuron toward the ASt frequency along the frequency axis. The BFs was 56.75 kHz instead of 57.75 kHz (BFc). The subsequent application of muscimol reduced the curve toward the baseline without changing BFs. Complete recovery from the effects of ASt + ESl and muscimol occurred 250 min after ASt + ESl.
Figure 5.
Effects of cortical inactivation on auditory responses and frequency-response curves of four collicular neurons. (A) The effects of 0.4 μg of muscimol applied to the AI on a single collicular neuron. (B) Muscimol was applied to the AI immediately before ASt + ESl. That is, ASt + ESl were delivered to the bat during the AI inactivation. (C) Muscimol was applied to the AI immediately after the collicular changes were evoked by ASt + ESl. (D) Muscimol was applied to the SI immediately before ASt+ ESl. That is, ASt+ ESl were delivered to the bat during the SI inactivation. The frequency-response curves and PST histograms were obtained with tone bursts fixed at 10 dB above the minimum threshold of a given neuron. The PST histograms show the change or lack of change in the response magnitude at the BFc or BFs condition. 1–4, respectively, correspond to the four conditions in which the frequency-response curves were obtained (see Fig. 2 legend).
Because ESl excites the somatosensory system, muscimol was applied to the SI (Fig. 1B), and then, 20 min later (during muscimol inactivation), ASt + ESl was delivered to the bat. Unlike muscimol applied to the primary AC, muscimol applied to the SI did not reduce the auditory responses of the eight collicular neurons studied at all, but did abolish both the reduction in auditory response and BF shift of collicular neurons, which otherwise would be evoked by ASt + ESl (Fig. 5D).
These cortical inactivation experiments with muscimol indicate the following important facts. (i) The auditory corticofugal system evokes changes in both the auditory response and the frequency map of the IC, when the animal is exposed to an acoustic stimulus. (ii) The corticofugal system evokes larger changes in the IC when the acoustic stimulus becomes behaviorally relevant to the animal. (iii) The changes in the IC occur predominantly in neurons tuned to frequencies slightly higher than the frequency of the acoustic stimulus, resulting in an augmented representation of that sound in the IC. (iv) Once the changes in the IC are evoked by the corticofugal system, the changes persist for some time in the IC without further corticofugal input. In other words, the IC itself shows plasticity. (v) Subcortical interactions between the ascending auditory and somatosensory systems are not involved in the changes in the IC evoked by the paired acoustic and somatosensory stimuli. (vi) The SI is necessary for the association of somatosensory information with auditory information (see Discussion).
DISCUSSION
Corticofugal System and Plasticity in Central Sensory Systems.
The auditory, visual, and somatosensory systems have cochleotopic (frequency), retinotopic, and somatotopic maps in their central neural pathways, respectively. These topographic maps are modified by deprivation, injury and experience, even in adult animals (21–26). Such plasticity has been explained by changes in divergent and convergent projections of the ascending sensory system. The contribution of the massive corticofugal system to the plasticity of sensory systems has been given little consideration, although there has been a great deal of data demonstrating the corticofugal modulation of the responses of subcortical neurons to sensory stimuli.
In the visual system, studies have shown excitatory and inhibitory effects of the corticofugal projection on matched and unmatched thalamic neurons, respectively (27). The contribution of the corticofugal system to visual signal processing has been demonstrated in many experiments (e.g., refs. 28 and 29). However, modification of the retinotopic map (i.e., shift in receptive field) in the thalamus via the corticofugal system has not yet been shown.
In the somatosensory system, modification of the somatotopic map because of somatosensory experience is evident in the thalamus as well as in the cortex (30, 31). A treatment of the SI for several months with an NMDA receptor agonist induces a large change in the somatotopic map in the thalamus (32). This intriguing finding most likely indicates that the corticofugal system can modulate the subcortical somatotopic map.
In the auditory system, modification of the frequency and echo-delay map in the IC and/or MGB by the corticofugal system has been demonstrated in bats (8, 9, 11). In the present experiment, we have further demonstrated that the corticofugal system evokes large plastic changes in the IC during classical conditioning.
Relationships of Our Findings to Learning, Memory, and Attention.
In our present studies, a classical conditioning paradigm was used to evoke conditioned behavioral response to ASt, so that the animal learned the relationship between ASt and ESl and anticipated ESl following ASt. Because ASt + ESl was repeatedly delivered at a rate of 2/min for 30 min, the animal presumably paid attention to ASt once ASt + ESl began to be delivered to the animal. Changes in the BFs and auditory responses of collicular neurons (hereafter simply collicular changes) were not evoked by ASt alone, ESl alone, or ES1 + ASt, but by ASt + ESl. Therefore, we conclude that the collicular changes are the result of auditory experience based on associative learning and, perhaps, attention. In the mustached bat, it has been demonstrated that egocentric selection adjusts subcortical signal processing in the same way in both the frequency (9) and time domains (8). Therefore, it may also be concluded that the corticofugal modulation described above occurs not only for a frequency-domain analysis but also for a time-domain analysis.
In our present studies, we made ASt behaviorally relevant by pairing it with ESl, and found that ASt + ESl evoked collicular changes that were larger than those evoked by ASr. Therefore, we may conclude that corticofugal adjustment and improvement of subcortical signal processing is larger for behaviorally relevant sounds than for behaviorally irrelevant sounds.
As reviewed in the Introduction, neurons in the MGB and IC of the guinea pig and/or the rat change their responses to acoustic stimuli according to the frequency of a conditioned acoustic stimulus. These changes are similar to those observed in the present studies. Therefore, it is likely that the subcortical changes observed in the guinea pig and/or rat are evoked by the corticofugal system.
The BF shifts observed in the AC of the guinea pig by Weinberger et al. (3) are also similar to those evoked by the corticofugal system, except for the duration of the shift. In our present studies, we did not measure the cortical BF shifts evoked by ASt + ESl. However, it has been found that BF shifts basically the same as those reported in the present paper are evoked in both the IC (11) and the AC of the big brown bat by focal electric stimulation of the AC, and that the amount of BF shift is larger for the AC than for the IC (S. A. Chowdhury and N.S., unpublished work). It also has been found that in the mustached bat, cortical inactivation with muscimol reduces the responses of IC and MGB neurons to single tone bursts by 34 ± 7.8% and 60 ± 16%, respectively (19). This finding certainly indicates that the AC is influenced more than the IC by the corticofugal feedback loop. Therefore, the BF shifts observed in the AC of the guinea pig (3) presumably are influenced by the feedback loop consisting of the ascending and corticofugal (descending) systems.
In our current studies, the collicular changes are retained for up to 3 hr after the animal was exposed to ASt + ESl. During this 3-hr period, we repeatedly delivered tone bursts of different frequencies to measure frequency-response curves. These tone bursts may have shortened the duration of the changes, because of a phenomenon called extinction. The duration of the collicular changes observed probably would last longer than 3 hr if we could minimize the effect of extinction. It is also quite possible that the collicular changes would last longer if ASt + ESl were delivered for longer than 30 min. We also found that the collicular changes, once evoked by ASt + ESl, last for some time without a corticofugal input. This finding indicates that the IC itself has plasticity related to learning and memory.
Neural Pathways for Collicular Plasticity.
In our current studies, inactivation of either the AI or SI during ASt + ESl abolished the collicular changes, which otherwise would be evoked by ASt + ESl. On the other hand, once the collicular changes were evoked, inactivation of the AI did not abolish them. These data indicate that the plasticity of the subcortical sensory nuclei depends at least partly on neural mechanisms that have not been considered before. The mechanisms, we propose, are as follows. The AC first shows plastic changes. Then, subcortical auditory nuclei are changed by the corticofugal system. The subcortical changes are maintained for some time without descending cortical input. The AC is influenced by the subcortical changes, because the ascending and descending systems form a positive feedback loop. The changes mediated by this feedback loop are stabilized by an inhibitory mechanism yet to be explored. Suga et al. (33) hypothesized that the thalamic reticular nucleus is involved in this inhibitory mechanism.
The SI, not subcortical multisensory nuclei, plays an essential role in sending somatosensory information to the AC. There is no direct cortico-cortical connection between the SI and the AC. How is somatosensory information associated with auditory information? The amygdala of the limbic system and the cholinergic forebrain nuclei may be involved.
During learning of a behavioral task, cortical acetylcholine levels increase (34). Blockage of cortical cholinergic receptors (35) or lesion of a cholinergic nucleus (nucleus basalis) in the forebrain (36) prevents learning of a behavioral task. Lesions of the nucleus basalis prevent reorganization of the SI (37), whereas electrical stimulation of the nucleus paired with acoustic stimulation induces long-lasting plastic changes in the AC (38, 39). AS + ES results in increased response in both the nucleus basalis and the AC in rats (40). The nucleus basalis responds to stimuli associated with behavior (41). Cortical sensory areas as well as thalamic sensory nuclei send signals to the lateral nucleus of the amygdala, which interacts with the amygdala’s central nucleus interfaced with the motor system (42).
Based on these findings, LeDoux and Muller (42) proposed a model to explain how the brain forms memories about unpleasant experiences. This model contains a pathway from the sensory thalamus to the amygdala, then to the nucleus basalis, and then to the primary sensory cortex. This pathway, as well as the projection from the amygdala to the primary sensory cortex, modifies neural activity in the primary sensory cortex. They speculated that this pathway mediated cortical arousal and attention.
Weinberger (43) proposed a model similar to the model above to explain cortical frequency-tuning plasticity in the learning of a classical conditioning paradigm. An auditory signal (tone burst, conditioning stimulus) is sent to the primary AC through the ventral division of the medial geniculate body (MGBv) and also is sent to the magnocellular division of the MGB (MGBm) and the posterior intralaminar complex (PIN), both in the thalamus. A somatosensory signal (electric foot-shock, unconditioned stimulus) also is sent to the MGBm and PIN, where the somatosensory and auditory signals are associated with each other. The associated signal is sent up to the AI to modulate cortical frequency tuning. This associated signal also is sent to the amygdala, which, in turn, presumably projects to the nucleus basalis. Then, the nucleus basalis increases cortical acetylcholine levels, so that cortical modulation based on the signal from the MGBm and PIN is augmented.
Based on our findings and what has been known of the amygdala and the nucleus basalis, we propose the following model, which is somewhat different from Weinberger’s model. ASt and ESl excite the AI and SI, respectively. These sensory cortices send signals to the amygdala through the association cortex. When associative learning takes place in the amygdala, that is, when an animal is conditioned by ASt + ESl, the cholinergic nucleus in the forebrain is excited by ASt + ESl through the amygdala. Then, the acetylcholine level in the cortex increases and the ASt-related changes in the AC, which are highly specific to acoustic stimuli, are augmented. These changes are transmitted down to the subcortical auditory nuclei by the corticofugal system. As a result, processing of behaviorally relevant acoustic stimuli is adjusted and improved. Cortical changes, which are highly specific to acoustic stimuli, are based on egocentric selection mediated by the corticofugal (descending) system that forms a feedback loop with the ascending system.
Acknowledgments
We thank Drs. S. Dear, L. S. Green, M. Konishi, W. E. O’Neill, and N. M. Weinberger, Mr. N. Laleman, and Ms. J. F. Linden for their comments on the manuscript. This work has been supported by a research grant from the National Institute on Deafness and Other Communicative Disorders (DC 00175).
ABBREVIATIONS
- AC
auditory cortex
- AI
primary AC
- ASr
repetitive acoustic stimulus
- ASt
a train of acoustic stimuli
- BF
best frequency
- BFc
BF in the control condition
- BFs
BF in the shifted condition
- ESl
electric shock to the leg
- IC
inferior colliculus
- MGB
medial geniculate body
- PST
poststimulus time
- SI
primary somatosensory cortex
References
- 1. Diamond D M, Weinberger N M. Brain Res. 1986;372:357–360. doi: 10.1016/0006-8993(86)91144-3. [DOI] [PubMed] [Google Scholar]
- 2.Bakin J S, Weiberger N M. Brain Res. 1990;536:271–286. doi: 10.1016/0006-8993(90)90035-a. [DOI] [PubMed] [Google Scholar]
- 3.Weinberger N M, Javid R, Lepan B. Proc Natl Acad Sci USA. 1993;90:2394–2398. doi: 10.1073/pnas.90.6.2394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Edeline J M, Weinberger N M. Behav Neurosci. 1991;105:154–175. doi: 10.1037//0735-7044.105.1.154. [DOI] [PubMed] [Google Scholar]
- 5.Edeline J M, Weinberger N M. Behav Neurosci. 1991;5:618–639. doi: 10.1037//0735-7044.105.5.618. [DOI] [PubMed] [Google Scholar]
- 6.Disterhoft J F, Stuart D K. Brain Res. 1977;130:315–333. doi: 10.1016/0006-8993(77)90278-5. [DOI] [PubMed] [Google Scholar]
- 7.Gonzalez-Lima F, Agudo J. NeuroReport. 1990;1:161–164. doi: 10.1097/00001756-199010000-00019. [DOI] [PubMed] [Google Scholar]
- 8.Yan J, Suga N. Science. 1996;273:1100–1103. doi: 10.1126/science.273.5278.1100. [DOI] [PubMed] [Google Scholar]
- 9.Zhang Y, Suga N, Yan J. Nature (London) 1997;387:900–903. doi: 10.1038/43180. [DOI] [PubMed] [Google Scholar]
- 10.Suga N, Yan J, Zhang Y. Trends Cognit Sci. 1997;1:13–20. doi: 10.1016/S1364-6613(97)01002-4. [DOI] [PubMed] [Google Scholar]
- 11.Yan W, Suga N. Nat Neurosci. 1998;1:54–58. doi: 10.1038/255. [DOI] [PubMed] [Google Scholar]
- 12.Oliver D L, Huerta M F. In: The Mammalian Auditory Pathway: Neuroanatomy. Webster D B, Popper A N, Fay R R, editors. New York: Springer; 1992. pp. 168–195. [Google Scholar]
- 13.Casseday J H, Covey E. J Comp Physiol. 1992;319:34–50. doi: 10.1002/cne.903190106. [DOI] [PubMed] [Google Scholar]
- 14.Jen P H, Sun X D, Lin P J. J Comp Physiol A. 1989;165:1–14. doi: 10.1007/BF00613794. [DOI] [PubMed] [Google Scholar]
- 15.Dear S P, Simmons J A, Fritz J. Nature (London) 1993;364:620–623. doi: 10.1038/364620a0. [DOI] [PubMed] [Google Scholar]
- 16.Suga N, O'Neill W E, Kujirai K, Manabe T. J Neurophysiol. 1983;49:1573–1626. doi: 10.1152/jn.1983.49.6.1573. [DOI] [PubMed] [Google Scholar]
- 17.Griffin D R. Symp Zool Soc London. 1962;7:61–72. [Google Scholar]
- 18.Simmons J A, Fenton M B, O’Farrel M J. Science. 1979;203:16–21. doi: 10.1126/science.758674. [DOI] [PubMed] [Google Scholar]
- 19.Zhang Y, Suga N. J Neurophysiol. 1997;78:3489–3492. doi: 10.1152/jn.1997.78.6.3489. [DOI] [PubMed] [Google Scholar]
- 20.Krubitzer L A, Calford M B. J Comp Neurol. 1992;317:1–30. doi: 10.1002/cne.903170102. [DOI] [PubMed] [Google Scholar]
- 21.Kaas J H, Krubitzer L A, Chino Y M, Langston A L, Polley H, Blair N. Science. 1990;248:229–231. doi: 10.1126/science.2326637. [DOI] [PubMed] [Google Scholar]
- 22.Irvine D R F, Rajan R. Clin Exp Pharmacol Physiol. 1996;23:939–947. doi: 10.1111/j.1440-1681.1996.tb01146.x. [DOI] [PubMed] [Google Scholar]
- 23.Merzenich M M, Nelson R J, Stryker M P, Cynader M S, Schoppmann A, Zook J M. J Comp Neurol. 1984;224:591–605. doi: 10.1002/cne.902240408. [DOI] [PubMed] [Google Scholar]
- 24.Recanzone G H, Schreiner C E, Merzenich M M. J Neurosci. 1993;13:87–103. doi: 10.1523/JNEUROSCI.13-01-00087.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Snyder R L, Rebscher S J, Cao K L, Leake P A, Kelly K. Hear Res. 1990;50:7–33. doi: 10.1016/0378-5955(90)90030-s. [DOI] [PubMed] [Google Scholar]
- 26.Weinberger N M. Annu Rev Neurosci. 1995;18:129–158. doi: 10.1146/annurev.ne.18.030195.001021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Tsumoto T, Creutzfeldt O D, Legendy C R. Exp Brain Res. 1978;32:345–364. doi: 10.1007/BF00238707. [DOI] [PubMed] [Google Scholar]
- 28.Sillito A M, Cudeiro J, Murphy P C. Exp Brain Res. 1993;93:6–16. doi: 10.1007/BF00227775. [DOI] [PubMed] [Google Scholar]
- 29.Sillito A M, Jones H E, Gerstein G L, West D C. Nature (London) 1994;369:479–482. doi: 10.1038/369479a0. [DOI] [PubMed] [Google Scholar]
- 30.Pollin B, Albe-Fessard P. Brain Res. 1979;173:431–449. doi: 10.1016/0006-8993(79)90240-3. [DOI] [PubMed] [Google Scholar]
- 31.Garraghty P E, Kaas J H. NeuroReport. 1991;2:747–750. doi: 10.1097/00001756-199112000-00004. [DOI] [PubMed] [Google Scholar]
- 32.Ergenzinger E R, Glasier M M, Hahm J O, Pons T P. Nat Neurosci. 1998;1:226–229. doi: 10.1038/673. [DOI] [PubMed] [Google Scholar]
- 33.Suga N, Butman J N, Teng H, Yan J, Olsen J F. In: Active Hearing. Flock A, Ottoson D, Ulfendahl M, editors. London: Elsevier; 1995. pp. 13–30. [Google Scholar]
- 34.Orsetti M, Casamenti F, Pepeu G. Brain Res. 1996;724:89–96. doi: 10.1016/0006-8993(96)00292-2. [DOI] [PubMed] [Google Scholar]
- 35.Leanza G, Muir J, Nilsson O G, Wiley R G, Dunnet S B, Bjorklund A. Eur J Neurosci. 1996;8:1535–1544. doi: 10.1111/j.1460-9568.1996.tb01616.x. [DOI] [PubMed] [Google Scholar]
- 36.Butt A E, Hodge G K. Behav Neurosci. 1995;109:699–713. doi: 10.1037//0735-7044.109.4.699. [DOI] [PubMed] [Google Scholar]
- 37.Webster H H, Hanisc U, Dykes R W, Biesold D. Somatosens Mot Res. 1991;8:327–346. doi: 10.3109/08990229109144756. [DOI] [PubMed] [Google Scholar]
- 38.Bjordahl T S, Dimyan M A, Weinberger N M. Behav Neurosci. 1998;112:467–479. doi: 10.1037//0735-7044.112.3.467. [DOI] [PubMed] [Google Scholar]
- 39.Kilgard M P, Merzenich M M. Science. 1998;279:1714–1718. doi: 10.1126/science.279.5357.1714. [DOI] [PubMed] [Google Scholar]
- 40.Maho C, Hars B, Edeline J M, Hennevin E. Psychobiology. 1995;23:10–25. [Google Scholar]
- 41.Richardson R T, DeLong M R. Adv Exp Med Biol. 1991;295:233–252. doi: 10.1007/978-1-4757-0145-6_12. [DOI] [PubMed] [Google Scholar]
- 42.LeDoux J E, Muller J. Philos Trans R Soc London B. 1997;352:1719–1726. doi: 10.1098/rstb.1997.0154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Weinberger, N. M. (1998) Neurobiol. Learning Memory 69, in press. [DOI] [PubMed]