Abstract
The blastula Chordin- and Noggin-expressing (BCNE) center located in the dorsal animal region of the Xenopus blastula embryo contains both prospective anterior neuroectoderm and Spemann organizer precursor cells. Here we show that, contrary to previous reports, the canonical Wnt target homeobox genes, Double knockdown of these genes using antisense morpholinos in Xenopus laevis blocked head formation, reduced the expression of the other BCNE center genes, upregulated Bmp4 expression, and nullified hyperdorsalization by lithium chloride. Moreover, gain- and loss-of-function experiments showed that Siamois and Twin expression is repressed by the vegetal transcription factor VegT. We propose that VegT expression causes maternal β-Catenin signals to restrict Siamois and Twin expression to the BCNE region. A two-step inhibition of BMP signals by Siamois and Twin – first by transcriptional repression of Bmp4 and then by activation of the expression of the BMP inhibitors Chordin and Noggin – in the BCNE center is required for head formation.
Keywords: Cell signaling, Spemann organizer, Siamois, Twin, Chordin, Noggin, BMP, Wnt, Xenopus
1. Introduction
Head formation is of considerable interest in developmental biology (reviewed in De Robertis and Kuroda, 2004; Niehrs, 2004; Stern, 2005). In vertebrate embryos, the establishment of the anteroposterior (A-P) axis has been most fully studied in amphibians, and embryological and molecular biological approaches have led to the identification of many molecules that control A-P patterning. Several lines of evidence indicate that head formation in Xenopus is controlled by a maternal nuclear β-Catenin signal located in the dorsal side of the blastula embryo. However, at later stages of development zygotic Wnt/β-Catenin signals have the opposite effect, ventralizing the embryo (Christian and Moon, 1993). The late Wnt/β-Catenin effects are probablymediated by the increased duration of BMP signals caused by inhibition of phosphorylation of Smad1/5/8 by GSK3 (Fuentealba et al., 2007). The early effects, however, are mediated by the well-known β-Catenin/Tcf3 (T-Cell Factor-3, a transcriptional repressor) pathway (Schneider et al., 1996; Houston et al., 2002; De Robertis and Kuroda, 2004). In the early blastula embryo, overexpression of Wnt ligands, their downstream signaling components, or their target genes, on the ventral side of the early embryo results in axis duplication, including perfect secondary head structures.
The nuclear localization of β-Catenin on the dorsal side extends broadly from the bottom (vegetal) to the top (animal) pole of the blastula in Xenopus (Schneider et al., 1996). The egg cytoplasm is heterogeneous, and when zygotic gene transcription starts at the mid-blastula transition the Nieuwkoop signaling center is formed in the dorsal-vegetal region (Fig. 1A). Nieuwkoop center cells express Xenopus Nodal-related (Xnr1, 2, 4, 5, and 6), TGFβ-superfamily factors that have potent mesoderm-inducing activity (Agius et al., 2000; Takahashi et al., 2000). High levels of Xnrs emanating from the Nieuwkoop center induce the Spemann organizer in overlying cells at gastrula, while the Nieuwkoop center cells themselves go on to form anterior endoderm. The β-Catenin signal induces the expression of BMP antagonists such as Chordin and Noggin in the BCNE (blastula Chordin- and Noggin-expressing) center, which has only limited overlap with the Nieuwkoop center (Fig. 1A; see also Fig. 1E in Kuroda et al., 2004). Blastula expression of Chordin and Noggin in the cellular precursors of the brain itself causes the anterior neuroectoderm to be predisposed to neural induction by the underlying endomesoderm and explains the enigmatic phenomenon of planar neural induction in dorsal Keller explants in Xenopus (Ruiz i Altaba, 1993; Kuroda et al., 2004).
Fig. 1.

Siamois and Twin are regulated by β-Catenin. (A) The positioning of two dorsal signaling centers at blastula. The BCNE center expresses Chordin (Chd), Noggin (Nog), Siamois (Sia), Twin (Twn), Xenopus nodal-related 3 (Xnr3), FoxA4a/Pintallavis/HNF3β, Anti-dorsalizing morphogenetic protein (ADMP). D, dorsal; V, ventral. (B) The effect of β-Cat-MO, demonstrating that BCNE center genes require β-Catenin. (C–F) Blocking of Xnr inductive events by CerS mRNA injection inhibits Chordin expression at gastrula but not at blastula (when the BCNE is active), while Siamois and Twin are independent of Xnrs. Ornithine decarboxylase (ODC) was used as loading control.
In addition to Chordin and Noggin, other genes are expressed in a localized fashion in the BCNE center: the homeobox genes Siamois and Twin, the BMP antagonist Xnr3, the winged-helix gene FoxA4a/pintallavis/HNF3β, and the BMP-like gene Admp (Fig. 1A, Kuroda et al., 2004; Wessely et al., 2004; Reversade and De Robertis, 2005). The homeobox genes Siamois and Twin are expressed immediately after the mid-blastula transition and are direct targets of the early β-Catenin/Wnt signal with potent dorsalizing activity (Lemaire et al., 1995; Carnac et al., 1996; Laurent et al., 1997). According to their high similarity in amino acids sequence (88% in the homeodomain) and perfectly overlapping expression pattern these genes may have redundant functions during Xenopus embryogenesis or act synergistically, possibly as heterodimers, to regulate gene transcription (Laurent et al., 1997).
In the present study, we have analyzed the function of Siamois and Twin using morpholino antisense oligomers (MO) (Heasman, 2002). The loss-of-function phenotype for Siamois has been previously investigated using dominant negative forms containing an engrailed repression domain (Fan and Sokol, 1997; Kessler, 1997). However, it is important to study gene function in a true loss-of-function situation by MO knockdown rather than in a dominant-negative approach, since microinjection of dominant-negative forms could affect other regions where the desired genes being investigated are not expressed, and these constructs persist much longer time in the embryo than the wild-type products. We report here that the Siamois and Twin in combination are key regulators of all genes expressed in the BCNE center, and are required for head formation and planar neural induction. Furthermore, we show that the vegetal expression of the maternal T-box transcription factor VegT (Zhang et al., 1998) is the key component that restricts the expression region of Siamois and Twin to the dorsal-animal BCNE center, thereby generating the distinction between the BCNE and Nieuwkoop signaling centers in the animal and vegetal hemispheres.
2. Results and discussion
2.1. Depletion of both Siamois and Twin causes anterior truncations
The expression of Siamois, Twin, Chordin, and Noggin requires maternal β-Catenin, as shown by microinjection of a MO that inhibits β-Catenin (β-Cat-MO) translation. β-Catenin depletion completely blocked the expression of these BCNE genes in the stage 9 blastula (Fig. 1B). It has been previously reported that Twin expression is blocked by a dominant-negative form of Smad4 (Nishita et al., 2000). We now investigated whether Siamois and Twin expression is regulated by Nodal. The mesoderm-inducing activity of endogenous Xenopus nodals (Xnrs) can be blocked by overexpression of CerS protein, the C-terminal portion of Cerberus (Bouwmeester et al., 1996; Piccolo et al., 1996; Ramis et al., 2007) (Fig. 1C–F). The expression of Chordin at gastrula stage is greatly inhibited by CerS, but the initial blastula expression is not. This indicates that the early expression of Chordin is regulated by β-Catenin, whereas Chordin expression at gastrula is driven by Xnrs (Fig. 1C) (Wessely et al., 2001). However, no differences were detected in Siamois and Twin expression in CerS injected embryos (Fig. 1D and E), indicating that Siamois and Twin are independent of Xnrs and are exclusively regulated by β-Catenin. In these RT-PCR analyses the house-keeping gene Ornithine decarboxylase (ODC) was used as an mRNA loading control (Fig. 1F). We conclude from these results that Siamois and Twin are under the control of the early β-Catenin signals, but not of Xnrs.
Loss-of-function studies were carried out with Siamois and Twin antisense morpholino oligomers (Sia-MO and Twn-MO) designed to hybridize to the translation initiation sites of these genes (Fig. 2A). There are only six base differences between these MOs, yet each MO effectively blocked only the translation of its own target gene (Fig. 2B). Thus, Sia-MO and Twn-MO injected on their own served as internal specificity controls. Sia-MO blocked the secondary axis inducing activity of ventrally injected Siamois mRNA (Fig. 2C and D), but Twn-MO did not (Fig. 2E), and vice-versa (data not shown).
Fig. 2.
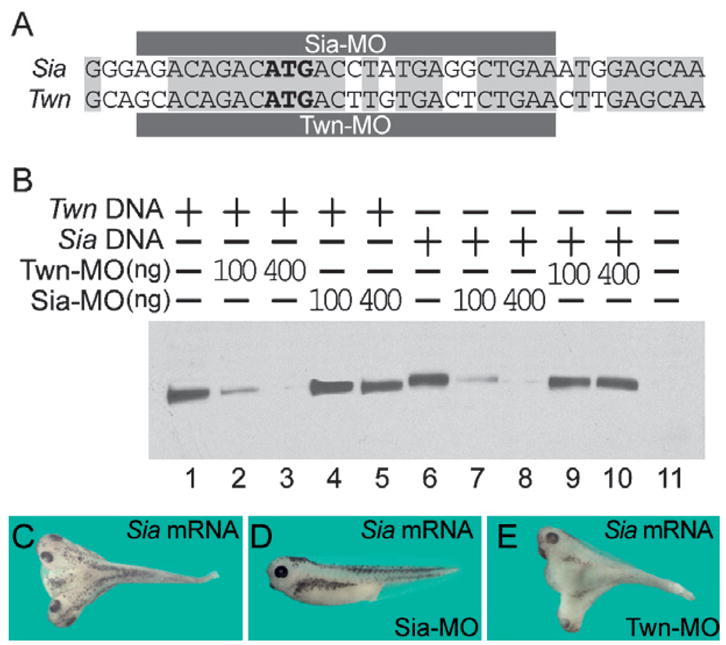
The function of Xenopus laevis Siamois and Twin is blocked by the antisense morpholino oligomer reagents designed in this study. (A) The MOs for Siamois and Twin target the translation initiation site. (B) In vitro translated Siamois and Twin protein were blocked only by each specific MO. (C) Secondary axes induced by ventral injection Siamois mRNA (5 pg); 77.3% (17/22) of embryos have secondary axes with formation of complete heads. (D) Secondary axisinducing activity by Siamois mRNA is blocked by Sia-MO (4 ng); 0% (0/25) of the embryos formed secondary axes. (E) Twn-MO (4 ng) did not block the effect of Siamois mRNA; 78.9% (15/19) of embryos had secondary axes with heads.
Single Sia-MO or Twn-MO injections were without phenotypic effect, except for slight reduction in eye size (Fig. 3A and B, compare to Fig. 3F). The Twn-MO small eye phenotype was over-rescued by a small amount (1 pg) of Siamois mRNA (Fig. 3D), and that of Sia-MO was rescued by 1 pg of Twin mRNA (Fig. 3E). Importantly, when both genes were depleted together, a severe head defect phenotype was observed with high penetrance (Fig. 3C). MOs for the BCNE center gene Chordin (Chd-MO) also have some head inhibitory activity (Oelgeschläger et al., 2003), but the effect of co-injection of Sia-MO and Twn-MO was much stronger (Fig. 3C). In histological sections, forebrain development, but not hindbrain or spinal cord, was severely impaired (Fig. 3G and H). In in situ hybridizations, the anterior brain marker Otx2 was blocked by co-injection of Sia-MO and Twn-MO (Fig. 3G–J), while the more posterior neural markers krox20 and HoxB9 continued to be expressed (data not shown). Notochord and heart formation still took place in embryos depleted for Siamois and Twin (Fig. 3G and H). We conclude that Siamois and Twin are required for anterior head formation in Xenopus development, in particular the forebrain.
Fig. 3.

Siamois and Twin are required for head formation. (A) Twn-MO injected embryo. Small eyeswere detected in 10.3% of injected embryos (3/29). (B) Sia-MO injected embryo. Small eyes were detected in 87.5% of embryos (42/48). (C) Twn-MO and Sia-MO co-injected embryos. 80% of embryos (28/35) lost head structures. (D) Co-injection of Twn-MO and Siamois mRNA rescued the Twn-MO phenotype to normal or mild dorsalization (big eyes in 42.9% (15/35) of the embryos). (E) Sia-MO and Twin mRNA coinjected embryo. Small eyes were detected in 14.8% of embryos (4/27). (F) Control uninjected sibling embryo for (A–E). (G) Sagittal histological section of the embryo shown in C. (H) Sagittal histological section of embryo shown in F. (I) Whole-mount in situ hybridization analysis of the anterior neural marker Otx2 in stage 26 embryo co-injected with Twin-MO and Siamois-MO. (J) Otx2 expression in control embryo at stage 26. fb, forebrain; mb, midbrain; hb, hindbrain; nt, neural tube; no, notochord; he, heart.
2.2. Siamois and Twin function as key regulators of the BCNE center
The BCNE center is under the control of β-Catenin. Lithium chloride (LiCl) inhibits glycogen synthase kinase-3β (GSK-3), stabilizing β-Catenin and increasing its levels in the nucleus (Klein and Melton, 1996; Schneider et al., 1996). Embryos treated at the 32-cell stage with LiCl are dorso-anteriorized, resulting in radial head structures lacking trunk–tail structures (Kao et al., 1986) (Fig. 4A). Siamois mRNA overexpression in the whole embryo is sufficient to mimic the LiCl-dorsalized phenotype (Lemaire et al., 1995). To test whether Siamois and Twin mediate the LiCl phenotype, loss-of-function experiments were performed. Co-injection of Sia-MO and Twn-MO prior to LiCl treatment completely blocked the dorsalizing effects of LiCl (Fig. 4A–C). This indicates that Siamois and Twin constitute one of the main gene outputs acting downstream of the β-Catenin signal.
Fig. 4.
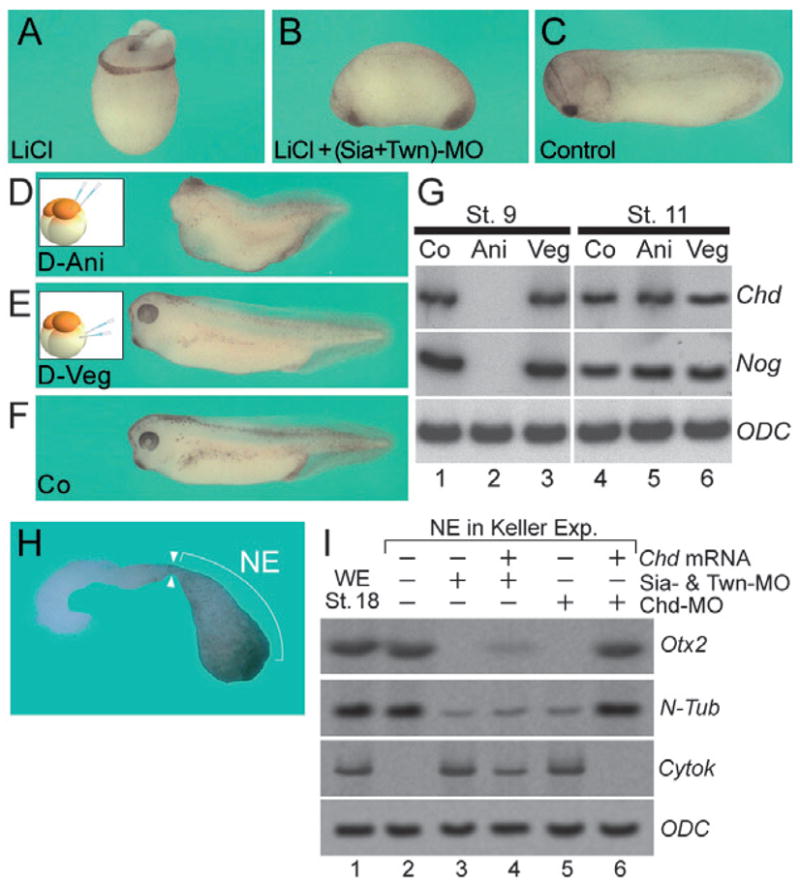
Dorsalization by LiCl and planar neural induction require Siamois and Twin. (A) Embryo treated with LiCl at the 32-cell stage. The average of dorso-anterior index (DAI); was 9.58 (Kao et al., 1986). (B) Embryo co-injected with Sia-MO and Twn-MO prior to LiCl treatment. The average of DAI was 5.38. (C) Control embryo for (A) and (B). In all cases 8 ng of MOs and 1 pg of mRNA were injected. (D) Phenotype of dorsal-animal injection of Sia-MO (total 4 ng) and Twn-MO (total 4 ng); 76.0% (19/25) of embryos lost head structures. (E) The phenotype of dorsal-vegetal injection of Sia-MO and Twn-MO. 7.7% (2/26) of embryos lost head structures. (F) Sibling control embryo at stage 38 for (A) and (B). (G) RT-PCR analyses of the effects of Sia-MO and Twn-MO on whole embryos at stage 9 and 11. (H) Neuroectoderm (NE) region of Keller explants used for molecular studies. (I) RT-PCR analyses of the effect of Sia-MO, Twn-MO, Chd-MO, and Chordin mRNA on the NE region shown in (H).
The entire BCNE center region is derived from dorsal-animal blastomeres at the 8-cell stage, whereas the Nieuwkoop center is derived from dorsal-vegetal blastomeres (Kuroda et al., 2004). To test whether Siamois and Twin also function as Nieuwkoop center genes, we injected both MOs into dorsal-animal or dorsal-vegetal blastomeres at the 8-cell stage and found that Siamois and Twin were required for head formation in the animal blastomeres, but not in ventral ones (Fig. 4D–F). At late blastula, stage 9, the expression levels of Chordin and Noggin were significantly reduced by dorsalanimal but not by dorsal-vegetal depletion of Siamois and Twin (Fig. 4G, lanes 1–3). At mid-gastrula, stage 11, the expression levels of Chordin and Noggin were not affected by loss-of-function of Siamois and Twin (Fig. 4G, lanes 4–6). These data are consistent with the view that at blastula Siamois and Twin function exclusively in the BCNE center, activating the expression of Chordin and Noggin. At gastrula stages, however, Chordin and Noggin are regulated by Nodal- related signals emanating from Spemann organizer mesoderm.
We reported previously that planar neural induction in Keller dorsal gastrula explants requires Chordin (Kuroda et al., 2004). We now asked whether the expression of Siamois and Twin in the prospective neuroectoderm at blastula is responsible for the “planar” neural induction signals. To investigate this, we examined gene expressions in the neuroectodermal (NE) region in Keller sandwiches (Fig. 4H) (Keller and Danilchik, 1988; Kuroda et al., 2004). The neuroectodermal region of control Keller sandwiches expressed the anterior neural marker Otx2 and the neuronal marker N-tubulin (Fig. 4I, lane 2), whereas the expression of Otx2 was blocked by co-injection of Sia-MO and Twn-MO (Fig. 4I, lane 3), just as in the case of Chd-MO (Fig. 4I, lane 5). These results show that the planar neural induction observed in Keller explants requires Siamois and Twin. Microinjection of Chordin mRNA partially rescued the anti-neural effect of Sia-MO and Twn- MO, and completely the inhibition of planar induction by Chd-MO (Fig. 4I, lanes 4 and 6). We conclude from these experiments that planar neural induction requires Siamois and Twin. As described earlier, this neural induction also has a complete requirement for the expression of their down-stream target Chordin (Kuroda et al., 2004).
In addition to Siamois and Twin, the BCNE center expresses Chordin, Noggin, Xnr3, FoxA4a/pintallavis/HNF3β, and Admp (Wessely et al., 2004; Reversade and De Robertis, 2005). To investigate the relationship between these other BCNE center genes and Siamois and Twin, we examined their transcription levels at blastula stage 9. Single MOs had very weak effects, although Sia-MO-injected embryos had a slightly stronger phenotype than Twin-morphants (Fig. 5A, lanes 1–3). However, all BCNE center genes, except for Siamois and Twin, were inhibited by co-injection of Sia-MO and Twn-MO (Fig. 5A, lanes 1 and 4). In particular, Chordin, Noggin, and FoxA4a/Pintallavis/HNF3β expression became undetectable (Fig. 5A, lane 4 and Fig. 5B and C). The Xnr3 promoter region has target binding sequences for LEF/TCF and for homeodomain transcription factors (McKendry et al., 1997). Thus, the residual expression observed in the case of Xnr3 and Admp might be explained by a feed-forward loop in which these genes are directly regulated by β-Catenin, as well as by Siamois and Twin. These results indicate that the Siamois and Twin transcriptional activators are central players in the regulation of BCNE center gene expression. They are required for the expression of Chordin, Noggin and FoxA4a/Pintallavis/HNF3β, but not for their own expression, which depends entirely on the β-Catenin signal.
Fig. 5.
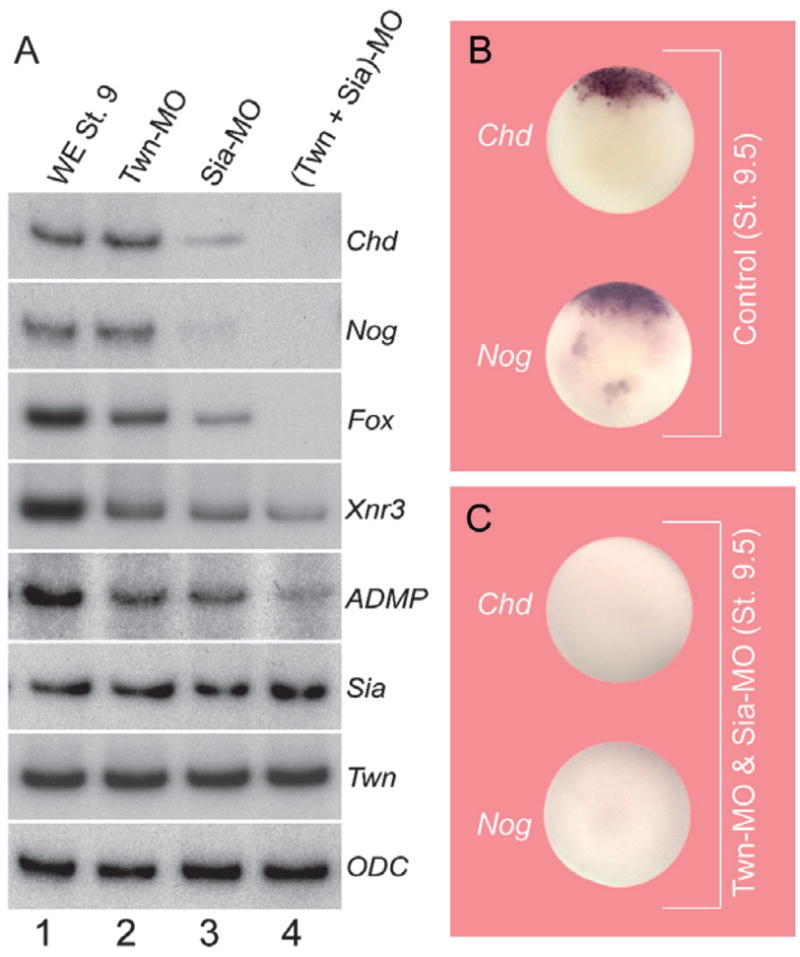
Siamois and Twin are key regulators of the transcription of BCNE center genes. (A) RT-PCR analyses of the effects of Sia-MO, Twn-MO, or both on whole embryos. (B) Chordin and Noggin expression at blastula in the animal cap and marginal zone (BCNE) of normal embryos. (C) Co-injection of Twn-MO and Sia-MO eliminates Chordin and Noggin expression. Embryos are shown in animal view.
2.3. Double inhibition of BMP by Siamois and Twin by transcriptional repression and BMP antagonists
The traditional embryological view held that neural tissue forms during gastrulation, when zygotic signals from dorsal mesoderm antagonize BMPs in the overlying dorsal ectoderm (Kuroda et al., 2004). Recent studies, however, have shown that the early β-Catenin is sufficient to cause neural tissue formation in the ectoderm in the absence of underlying mesoderm, explaining earlier observations of planar neural induction in Xenopus (Ruiz i Altaba, 1993). Some studies have previously shown that β-Catenin inhibits BMP transcription (Baker et al., 1999; Leung et al., 2003). On the other hand, we reported that nuclear β-Catenin causes the expression of Chordin and Noggin in the BCNE center and that these BMP antagonists are required for neuralization (Wessely et al., 2001; Kuroda et al., 2004). In an attempt to reconcile these two lines of evidence, we now investigated the function of Siamois and Twin in this process.
Small dorsal and ventral ectodermal explantswere excised from embryos injected with CerS mRNA to prevent mesoderm formation (Fig. 6A). CerS, which consists of the cystine knot of Cerberus, is an excellent reagent for this type of experiment because it blocks specifically the activity of all mesoderm-inducing Nodal-related genes (Piccolo et al., 1996; Agius et al., 2000). Double depletion of Siamois and Twin caused Bmp4 to be expressed in the dorsal animal cap fragments (Fig. 6B, compare lanes 2 and 4), and injection of Siamois mRNA inhibited Bmp4 expression in ventral animal cap fragments (Fig. 6B, lanes 5 and 7). Depletion of Chordin and Noggin increased expression of the BMP target Vent1 dorsally (Fig. 6B, lanes 2 and 3), and Chordin mRNA inhibited Vent1 but did not affect Bmp4 transcription (Fig. 6B; lanes 5 and 6). At the gastrula stage, the opposing homeobox genes Goosecoid and Vent1/2 have been reported to be mutually antagonistic in dorsal–ventral patterning (Sander et al., 2007). We believe that Siamois and Twin act on Vent1 at the blastula stage, before the later gastrula regulatory event of the mutual repression by goosecoid and Vent1 takes place.
Fig. 6.

Two-step inhibition of BMP4 by Siamois and Twin. (A) Experimental procedure. (B) RT-PCR analyses for the effects of Siamois, Twin, and the BMP antagonists Chordin and Noggin on Bmp4 and its target gene Vent1. The nodal-related target gene Mix1 was used to indicate that CerS was effective in blocking mesoderm and endoderm formation in these experiments. (C) Diagram showing a two-step model for the regulation of BMP4 via Siamois and Twin. BMP signaling is blocked in two steps (1) transcriptional repression and (2) BMP antagonists such as Chordin and Noggin which are induced by Siamois and Twin.
These results suggest to us a model in which Siamois and Twin first downregulate endogenous Bmp4 mRNA transcription at the blastula dorsal animal region in a BMP antagonist-independent manner (as proposed by Baker et al., 1999; Leung et al., 2003). In a second step, Chordin and Noggin produced in the BCNE center inhibit any residual BMPs. In this two-step model, depicted in Fig. 6C, BMP signals are cleared out in the BCNE center by transcriptional repression and also by BMP antagonists, both dependent on Siamois and Twin expression.
Since Siamois and Twin are transcriptional activators (Kessler, 1997), how can they repress transcription of Bmp4? In zebrafish, the homeobox gene bozozok induced by the Wnt/β-Catenin signal encodes a transcriptional repressor and is able to directly inhibit Bmp2b transcription (Leung et al., 2003). A distinct possibility is that Xenopus might have an as yet undiscovered bozozok-like transcriptional repressor homologue induced by Siamois and Twin that negatively regulates the transcription levels of Bmp4, explaining arrow number 1 in Fig. 6C.
2.4. VegT defines the border between the BCNE center and the Nieuwkoop center
What determines the molecular differences between the BCNE and Nieuwkoop centers? To address this, we used a VegT-MO (Heasman et al., 2001) against the maternal vegetally-expressed T-box transcription factor VegT (Zhang et al., 1998). This morpholino does not inhibit translation of the zygotic alternatively spliced VegT transcript designated as Antipodean (Stennard et al., 1999). We noted that VegT-MO-injected embryos always had big heads and cement glands, which are typical signs of low-BMP (Fig. 7A and B). This is what one might expect from an expansion of the BCNE center.
Fig. 7.

The BCNE center is restricted to the dorsal-animal region by the maternally localized VegT transcription factor. (A) The phenotype of VegT-MO. White arrowhead indicates enlarged cement gland. (B) Control embryo for (A). (C) RT-PCR analyses of the effects of Wnt8 and VegT mRNA for animal caps. 1, 4, or 16 pg of Wnt8 mRNA and 100 pg of VegT mRNA were injected. (D) Chordin expression in control embryo at stage 9. White and black arrowheads indicate the animal and vegetal poles, respectively. (E) Siamois expression in control embryo. (F) Chordin expression of VegT-MO (16 ng) injected embryo. (G) Siamois expression of VegT-MO injected embryo. Note the expansion of Chordin and Siamois expression towards the vegetal side upon inhibition of VegT. (H) Real-time PCR analyses of Chordin mRNA expression levels from stage 8 to 12. (I) Siamois mRNA expression levels in the same samples. (J) Model of the regulation of the two blastula signaling centers. The border between the BCNE and Nieuwkoop centers is generated by repression by two maternally provided transcription factors: B1-type Sox (animal; Zhang et al., 2004) and VegT (vegetal).
To investigate whether VegT controls BCNE center gene expression, we injected both Wnt8mRNAto activate β-Catenin (Christian and Moon, 1993) together with VegT mRNA. This combination recreates dorsal-vegetal conditions, mimicking a Nieuwkoop center in animal caps. In the conventional view currently held by the Xenopus field, Siamois and Twin are expressed in the Nieuwkoop center (Lemaire et al., 1995; Laurent et al., 1997). Therefore, the expectation would be that animal caps co-injected with Wnt8 and VegT mRNAs would express large amounts of Siamois and Twin. Surprisingly, BCNE center genes such as Chordin, Noggin, Siamois, and Twin, which are strongly induced by increasing doses of Wnt8 mRNA (Fig. 7C, lanes 2–5), were not upregulated in the presence of VegT mRNA (Fig. 7C, lanes 6–8). In contrast, the Nieuwkoop center genes Xnr2 and Xnr6 were induced by VegT mRNA (Fig. 7C, lanes 6–9). These data show that VegT mRNA inhibits the induction of BCNE center genes caused by Wnt8/β-Catenin (Fig. 7C).
Loss-of-function experiments support the view that VegT serves to exclude the BCNE from the vegetal pole. The Chordin and Siamois expressing region at the BCNE center was greatly expanded towards the vegetal side by depletion of VegT (Fig. 7D–G). As shown earlier, this early expression of Chordin requires Siamois and Twin (Fig. 5A–C). This result is somewhat conflicting with a previous report by Houston et al. (2002) in which the inhibition of VegT reduced the level of Chordin and Goosecoid, while we now report an increase. Therefore, we examined by real-time PCR analysis the level of expression of the mRNAs for Chordin and Siamois over the time course from stages 8 to 12 (Fig. 7H and I). The expression of Chordin and Siamois in VegT-MO-injected embryos was increased to approximately twice of control levels at stage 9. However, at stage 12 Chordin expression was greatly reduced by VegT-MO, while Siamois expression was not changed at stage 12 (Fig. 7H and I). One important experimental difference is that Houston et al. (2002) used antisense DNA oligo depletion in oocytes, while we used an antisense morpholino injected into cleavage stage embryos. It has been observed previously that these two loss-of-function approaches result in similar, yet not always identical phenotypes (Heasman et al., 2001). In addition, the timing of the samples may have had slight differences (Fig. 7H and I). It should also be mentioned that previous reports have shown that Chordin expression is activated by β-Catenin in animal but not vegetal cells (Darras et al., 1997).
Taken together, the results presented in this section suggest that Siamois and Twin are regulated differently from Nieuwkoop center genes, since VegT represses them. Recently, maternal B1-type Sox genes expressed in the animal cap have been shown to restrict Xnr expression, except for Xnr3, to the vegetal hemisphere (Zhang et al., 2004). Thus, it appears that the border between the BCNE and Nieuwkoop centers is established by the maternally encoded gene products VegT and B1-Sox. Fig. 7J shows a model in which VegT and B1-Sox are proposed to regulate the distinction between the BCNE and Nieuwkoop centers within the dorsal region of stabilized nuclear β-Catenin.
2.5. The making of a brain
The role of the prospective neuroectoderm in amphibian neural induction has been the subject of much debate (Spemann, 1938; Holtfreter et al., 1955; Nieuwkoop and Koster, 1995; Kuroda et al., 2004). It has long been known that dorsal animal cap explants respond much better than ventral ones to induction by dorsal organizer mesoderm and the mesoderm-inducer Activin (Sharpe et al., 1987; Sokol and Melton, 1991). In addition, dorsal-animal blastomeres at the 8-cell stage that have not received mesoderm induction signals, have a self-differentiation predisposition to become anterior neural tissue (Kinoshita et al., 1993).
The BCNE center is formed in cells that give rise to neuroectoderm, in particular brain, even in the absence of a mesodermal substratum. Extirpation and transplantation experiments have shown that the BCNE center is required for brain formation in the embryo (Kuroda et al., 2004). Recently, the requirement of Noggin expressed in the blastula animal cap ectoderm for neural differentiation has provided independent support for our proposal that the BCNE in prospective neuroectoderm plays a critical role in the differentiation of the central nervous system in Xenopus (Huang et al., 2007). The results of the present study show that all known BCNE center genes require the combined action of the homeobox genes Siamois and Twin. Thus, the process of brain induction can be traced back to the earliest stages of embryonic development, as is the case in other embryos such as the chick (reviewed in Stern, 2006; De Robertis and Kuroda, 2004). Neural induction is not an event that occurs at gastrula as believed classically (Spemann, 1938), but rather starts with the cortical rotation of the 1-cell zygote, which stabilizes nuclear β-Catenin in the dorsal side of the embryo. The prospective neuroectoderm is activated by β-Catenin and repressed by VegTon the vegetal side (Fig. 7J). β-Catenin activates expression of Siamois and Twin at mid-blastula, causing the repression of Bmp4 transcription (Baker et al., 1999; Leung et al., 2003), as well as the expression of the Bmp-antagonists Chordin and Noggin in the prospective neuroectoderm (Kuroda et al., 2004; Huang et al., 2007). Using morpholino knockdown we have demonstrated here that the Siamois and Twin homeobox proteins are required for the expression of BCNE center genes such as Chordin, Noggin, FoxA4a/pintallavis/HNF3β, Xnr3, and Admp, which in turn participate in the formation of the vertebrate brain. In addition, we showed that Siamois and Twin are also required for the dorsalizing effects of LiCl treatment of early Xenopus embryos. In conclusion, Siamois and Twin represent the main output of β-Catenin in the future neuroectoderm at the blastula stage, and these homeobox genes play a key role as determinants of brain formation in Xenopus.
3. Experimental procedures
3.1. Embryo manipulations
Xenopus laevis embryos were obtained by in vitro fertilization (Sive et al., 2000). Animal cap assays were performed in 1 × Steinberg’s solution (58 mM NaCl, 0.67mM KCl, 0.83 mM MgSO4, 0.34 mM Ca(NO3)2, 4.6 mM Tris–HCl, pH 7.4, 100 mg/L Kanamycin) at stage 9 (7 h after fertilization at 22 °C). PCR conditions and primers, as well the protocol for whole-mount in situ hybridization, are described in http://www.kurodalab.com.
3.2. RNA and morpholino injections
To generate synthetic mRNAs, pRN3-Siamois was linearized with Sfi1 and transcribed with T3 RNA polymerase (Lemaire et al., 1995), pST64T-Twin with Sal1 and SP6 polymerase (Laurent et al., 1997), andpCS2-Chd, pCS2-Wnt8, andpCS2-VegTwith NotI and SP6 RNA polymerase. MOs for Siamois, Twin, Chordin, Noggin, and VegTwere ordered from Gene Tools, LLC. They consisted of the following sequences; Sia-MO: 5′-TTCAGCCTCATAGGTCATGTCTGTC-3′ (this work), Twn-MO: 5′-TTCAGAGT CACAAGTCATGTCTGTG-3′ (this work), β-Cat-MO: 5′-TTTCAAC CGTTTCCAAAGAACCAGG-3′ (Kuroda et al., 2004), Chd-MO1: 5′-ACGTTGTGTGTGGTATAGTGAGGGT-3′ and Chd-MO2: 5′-ACAG GATTTTTGTGGTTGTCCCGAA-3′ (Oelgeschläger et al., 2003), Nog-MO: 5′-TCACAAGGCACTGGGAATGATCCAT-3′ (Kuroda et al., 2004), VegT-MO: 5′-CCCGACAGCAGTTTCTCATTCCAGC-3′ (Heasman et al., 2001). MOs were resuspended in sterile water to a concentration of 1 mM each and then further diluted to give working solutions.
3.3. In vitro translation
Siamois and Twin proteins were in vitro translated using TnT T3/SP6 Coupled Transcription/Translation System (Promega) in the presence of Sia-MO or Twn-MO and [35S]-methionine, according to the manufacturer’s instructions. Half of each reaction was analyzed by 8% SDS–PAGE and visualized by autoradiography.
Acknowledgments
We thank P. Lemaire, K.W.Y. Cho, R. Moon, C. Wylie, and T. Hayata for materials; V. Sander and members of the Kuroda laboratory for comments on the manuscript. This work was supported by the Research Reports of Uehara Memorial Foundation and the Sumitomo Foundation, NIH Grant HD21502-21, and the Howard Hughes Medical Institute.
References
- Agius E, Oelgeschläger M, Wessely O, Kemp C, De Robertis EM. Endodermal Nodal-related signals and mesoderm induction in Xenopus. Development. 2000;127:1173–1183. doi: 10.1242/dev.127.6.1173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baker JC, Beddington RS, Harland RM. Wnt signaling in Xenopus embryos inhibits bmp4 expression and activates neural development. Genes Dev. 1999;13:3149–3159. doi: 10.1101/gad.13.23.3149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bouwmeester T, Kim S, Sasai Y, Lu B, De Robertis EM. Cerberus is a head-inducing secreted factor expressed in the anterior endoderm of Spemann’s organizer. Nature. 1996;382:595–601. doi: 10.1038/382595a0. [DOI] [PubMed] [Google Scholar]
- Carnac G, Kodjabachian L, Gurdon JB, Lemaire P. The homeobox gene Siamois is a target of the Wnt dorsalisation pathway and triggers organiser activity in the absence of mesoderm. Development. 1996;122:3055–3065. doi: 10.1242/dev.122.10.3055. [DOI] [PubMed] [Google Scholar]
- Christian JL, Moon RT. Interactions between Xwnt-8 and Spemann organizer signaling pathways generate dorsoventral pattern in the embryonic mesoderm of Xenopus. Genes Dev. 1993;7:13–28. doi: 10.1101/gad.7.1.13. [DOI] [PubMed] [Google Scholar]
- Darras S, Marikawa Y, Elinson RP, Lemaire P. Animal and vegetal pole cells of early Xenopus embryos respond differently to maternal dorsal determinants: implications for the patterning of the organiser. Development. 1997;124:4275–4286. doi: 10.1242/dev.124.21.4275. [DOI] [PubMed] [Google Scholar]
- De Robertis EM, Kuroda H. Dorsal–ventral patterning and neural induction in Xenopus embryos. Annu Rev Cell Dev Biol. 2004;20:285–308. doi: 10.1146/annurev.cellbio.20.011403.154124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fan MJ, Sokol SY. A role for Siamois in Spemann organizer formation. Development. 1997;124:2581–2589. doi: 10.1242/dev.124.13.2581. [DOI] [PubMed] [Google Scholar]
- Fuentealba LC, Eivers E, Ikeda A, Hurtado C, Kuroda H, Pera EM, De Robertis EM. Integrating patterning signals: Wnt/GSK3 regulates the duration of the BMP/Smad1 signal. Cell. doi: 10.1016/j.cell.2007.09.027. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heasman J. Morpholino oligos: making sense of antisense? Dev Biol. 2002;243:209–214. doi: 10.1006/dbio.2001.0565. [DOI] [PubMed] [Google Scholar]
- Heasman J, Wessely O, Langland R, Craig EJ, Kessler DS. Vegetal localization of maternal mRNAs is disrupted by VegT depletion. Dev Biol. 2001;240:377–386. doi: 10.1006/dbio.2001.0495. [DOI] [PubMed] [Google Scholar]
- Holtfreter J, Hamburger V. Amphibians. In: Willer BH, et al., editors. Analysis of Development. Haffer; NY: 1955. pp. 230–296. [Google Scholar]
- Houston DW, Kofron M, Resnik E, Langland R, Destree O, Wylie C, Heasman J. Repression of organizer genes in dorsal and ventral Xenopus cells mediated by maternal XTcf3. Development. 2002;129:4015–4025. doi: 10.1242/dev.129.17.4015. [DOI] [PubMed] [Google Scholar]
- Huang S, Yan B, Sullivan SA, Moody SA. Noggin signaling from Xenopus animal blastomere lineages promotes a neural fate in neighboring vegetal blastomere lineages. Dev Dyn. 2007;236:171–183. doi: 10.1002/dvdy.20944. [DOI] [PubMed] [Google Scholar]
- Kao KR, Masui Y, Elinson RP. Lithium-induced respecification of pattern in Xenopus laevis embryos. Nature. 1986;322:371–373. doi: 10.1038/322371a0. [DOI] [PubMed] [Google Scholar]
- Keller R, Danilchik M. Regional expression, pattern and timing of convergence and extension during gastrulation of Xenopus laevis. Development. 1988;103:193–209. doi: 10.1242/dev.103.1.193. [DOI] [PubMed] [Google Scholar]
- Kessler DS. Siamois is required for formation of Spemann’s organizer. Proc Natl Acad Sci USA. 1997;94:13017–13022. doi: 10.1073/pnas.94.24.13017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kinoshita K, Bessho T, Asashima M. Competence prepattern in the animal hemisphere of the 8-cell-stage Xenopus embryo. Dev Biol. 1993;160:276–284. doi: 10.1006/dbio.1993.1305. [DOI] [PubMed] [Google Scholar]
- Klein PS, Melton DA. A molecular mechanism for the effect of lithium on development. Proc Natl Acad Sci USA. 1996;93:8455–8459. doi: 10.1073/pnas.93.16.8455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuroda H, Wessely O, De Robertis EM. Neural induction in Xenopus: requirement for ectodermal and endomesodermal signals via Chordin, Noggin, β-Catenin, and Cerberus. PLoS Biol. 2004;2:623–634. doi: 10.1371/journal.pbio.0020092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laurent MN, Blitz IL, Hashimoto C, Rothbacher U, Cho KW. The Xenopus homeobox gene twin mediates Wnt induction of goosecoid in establishment of Spemann’s organizer. Development. 1997;124:4905–4916. doi: 10.1242/dev.124.23.4905. [DOI] [PubMed] [Google Scholar]
- Lemaire P, Garrett N, Gurdon JB. Expression cloning of Siamois, a Xenopus homeobox gene expressed in dorsalvegetal cells of blastulae and able to induce a complete secondary axis. Cell. 1995;81:85–94. doi: 10.1016/0092-8674(95)90373-9. [DOI] [PubMed] [Google Scholar]
- Leung T, Bischof J, Soll I, Niessing D, Zhang D, Ma J, Jackle H, Driever W. bozozok directly represses bmp2b transcription and mediates the earliest dorsoventral asymmetry of bmp2b expression in zebrafish. Development. 2003;130:3639–3649. doi: 10.1242/dev.00558. [DOI] [PubMed] [Google Scholar]
- McKendry R, Hsu SC, Harland RM, Grosschedl R. LEF-1/TCF proteins mediate wnt-inducible transcription from the Xenopus nodal-related 3 promoter. Dev Biol. 1997;192:420–431. doi: 10.1006/dbio.1997.8797. [DOI] [PubMed] [Google Scholar]
- Niehrs C. Regionally specific induction by the Spemann–Mangold organizer. Nat Rev Genet. 2004;5:425–434. doi: 10.1038/nrg1347. [DOI] [PubMed] [Google Scholar]
- Nieuwkoop PD, Koster K. Vertical versus planar induction in amphibian early development. Dev Growth Differ. 1995;37:653–668. doi: 10.1046/j.1440-169X.1995.t01-5-00004.x. [DOI] [PubMed] [Google Scholar]
- Nishita M, Hashimoto MK, Ogata S, Laurent MN, Ueno N, Shibuya H, Cho KW. Interaction between Wnt and TGF-beta signalling pathways during formation of Spemann’s organizer. Nature. 2000;403:781–785. doi: 10.1038/35001602. [DOI] [PubMed] [Google Scholar]
- Oelgeschläger M, Kuroda H, Reversade B, De Robertis EM. Chordin is required for the Spemann organizer transplantation phenomenon in Xenopus embryos. Dev Cell. 2003;4:219–230. doi: 10.1016/s1534-5807(02)00404-5. [DOI] [PubMed] [Google Scholar]
- Piccolo S, Agius E, Leyns L, Bhattacharyya S, Grunz H, Bouwmeester T, De Robertis EM. The head inducer Cerberus is a multifunctional antagonist of Nodal, BMP and Wnt signals. Nature. 1996;397:707–710. doi: 10.1038/17820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramis JM, Collart C, Smith JC. Xnrs and activin regulate distinct genes during Xenopus development: activin regulates cell division. PLoS ONE. 2007;2:e213. doi: 10.1371/journal.pone.0000213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reversade B, De Robertis EM. Regulation of ADMP and BMP2/4/7 at opposite embryonic poles generates a selfregulating morphogenetic field. Cell. 2005;123:1147–1160. doi: 10.1016/j.cell.2005.08.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruiz i Altaba A. Induction and axial patterning of the neural plate: planar and vertical signals. J Neurobiol. 1993;17:233–243. doi: 10.1002/neu.480241004. [DOI] [PubMed] [Google Scholar]
- Sander V, Reversade B, De Robertis EM. The opposing homeobox genes Goosecoid and Vent1/2 self-regulate Xenopus patterning. EMBO J. 2007;26:2955–2965. doi: 10.1038/sj.emboj.7601705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneider S, Steinbeisser H, Warga RM, Hausen P. β-Catenin translocation into nuclei demarcates the dorsalizing centers in frog and fish embryos. Mech Dev. 1996;57:191–198. doi: 10.1016/0925-4773(96)00546-1. [DOI] [PubMed] [Google Scholar]
- Sharpe CR, Fritz A, De Robertis EM, Gurdon JB. A homeobox-containing marker of posterior neural differentiation shows the importance of predetermination in neural induction. Cell. 1987;50:749–758. doi: 10.1016/0092-8674(87)90333-3. [DOI] [PubMed] [Google Scholar]
- Sive HL, Grainger RM, Harland RM. Early Development of Xenopus laevis. Cold Spring Harbor Laboratory Press; Cold Spring Harbor, NY: 2000. [Google Scholar]
- Sokol S, Melton DA. Pre-existent pattern in Xenopus animal pole cells revealed by induction with activin. Nature. 1991;351:409–411. doi: 10.1038/351409a0. [DOI] [PubMed] [Google Scholar]
- Spemann H. Embryonic Development and Induction. Yale University Press; New Heaven, CT: 1938. [Google Scholar]
- Stennard F, Zorn AM, Ryan K, Garrett N, Gurdon JB. Differential expression of VegT and Antipodean protein isoforms in Xenopus. Mech Dev. 1999;86:87–98. doi: 10.1016/s0925-4773(99)00119-7. [DOI] [PubMed] [Google Scholar]
- Stern CD. Neural induction: old problem, new findings, yet more questions. Development. 2005;132:2007–2021. doi: 10.1242/dev.01794. [DOI] [PubMed] [Google Scholar]
- Stern CD. Neural induction: 10 years on since the ‘default model’. Curr Opin Cell Biol. 2006;18:692–697. doi: 10.1016/j.ceb.2006.09.002. [DOI] [PubMed] [Google Scholar]
- Takahashi S, Yokota C, Takano K, Tanegashima K, Onuma Y, Goto J, Asashima M. Two novel nodal-related genes initiate early inductive events in Xenopus Nieuwkoop center. Development. 2000;127:5319–5329. doi: 10.1242/dev.127.24.5319. [DOI] [PubMed] [Google Scholar]
- Wessely O, Agius E, Oelgeschläger M, Pera EM, De Robertis EM. Neural induction in the absence of mesoderm: β-catenin-dependent expression of secreted BMP antagonists at the blastula stage in Xenopus. Dev Biol. 2001;234:161–173. doi: 10.1006/dbio.2001.0258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wessely O, Kim JI, Geissert D, Tran U, De Robertis EM. Analysis of Spemann organizer formation in Xenopus embryos by cDNA macroarrays. Dev Biol. 2004;269:552–566. doi: 10.1016/j.ydbio.2004.01.018. [DOI] [PubMed] [Google Scholar]
- Zhang J, Houston DW, King ML, Payne C, Wylie C, Heasman J. The role of maternal VegT in establishing the primary germ layers in Xenopus embryos. Cell. 1998;94:419–421. doi: 10.1016/s0092-8674(00)81592-5. [DOI] [PubMed] [Google Scholar]
- Zhang C, Basta T, Hernandez-Lagunas L, Simpson P, Stemple DL, Artinger KB, Klymkowsky MW. Repression of nodal expression by maternal B1-type SOXs regulates germ layer formation in Xenopus and zebrafish. Dev Biol. 2004;273:23–37. doi: 10.1016/j.ydbio.2004.05.019. [DOI] [PubMed] [Google Scholar]


