Abstract
The O antigen is both a major structural outer membrane component and the dominant epitope of most gram-negative bacteria. Pseudomonas aeruginosa 1244 produces a type IV pilus and covalently links an O-antigen repeating unit to each pilin monomer. Here we show that immunization of mice with pure pilin from strain 1244 by use of either the mouse respiratory model or the thermal injury model resulted in protection from challenge with a pilus-null O-antigen-producing 1244 mutant. These results provide evidence that the pilin glycan stimulates a protective response that targets the O antigen, suggesting that this system could be used as the basis for the development of a variety of bioconjugate vaccines protective against gram-negative bacteria.
With the widespread occurrence of antibiotic resistance among many bacterial pathogens (29, 33, 47), it is increasingly necessary to turn to vaccination to combat these disease-causing organisms. An important strategy for vaccine design involves the synthesis of glycoconjugates, which are produced by chemically cross-linking bacterial surface polysaccharides to carrier proteins (2, 23). Saccharides linked to proteins invoke T-cell-dependent immunity (4), which involves a memory response, avidity maturation, and immunoglobulin class switching (46). Conversely, bacterial polysaccharides alone are usually associated with a T-cell-independent response, which does not involve the induction of immunological memory (27). However promising this technology is, expansion of the glycoconjugate vaccine technology has been hampered by the expense of purifying and characterizing surface polysaccharides of bacterial pathogens, the selection of an effective carrier protein, and maintenance of the quality and stability of cross-linked products (23).
Several antibacterial glycoconjugate vaccines are commercially available (23). Haemophilus influenzae type b glycoconjugate vaccines (39), in which the surface oligosaccharide of this bacterium was conjugated to either diphtheria toxoid (3), tetanus toxoid (22), or a meningococcal outer membrane protein (15), were the first to be licensed (23). Currently, numerous investigations are under way to test and develop polysaccharide-protein conjugate vaccines against bacterial pathogens, including Francisella tularensis (9), Escherichia coli O157 (24), Shigella sonnei (36), and the opportunistic pathogen Pseudomonas aeruginosa (11).
The O antigen is the dominant surface polysaccharide of the lipopolysaccharide (LPS) molecule of most gram-negative bacteria, and consequently, antibodies directed against this cellular component have been shown to protect against infection (9, 10, 13, 14, 17). Interestingly, P. aeruginosa 1244 covalently attaches a single serotype O7 O-antigen repeating unit to Ser148 of the pilin (8), with no alternate glycoforms or evidence of nonglycosylated forms of this protein (6). Pilin (encoded by the pilA gene) is the monomeric subunit of the type IV pilus, an immunogenic bacterial surface appendage utilized for adhesion and surface motility (26). P. aeruginosa 1244 pilin glycosylation is mediated by the oligosaccharyltransferase PilO (5, 12). This type of O-linked, PilO-mediated pilin glycosylation is common among P. aeruginosa strains (7, 25, 41). Because the P. aeruginosa 1244 protein glycosylation machinery covalently links an O-antigen subunit and pilin, studies have suggested the possibility for exploitation of this system to biologically produce glycoconjugate vaccines (8, 20, 21).
Metabolically, the pilin glycan originates in the O-antigen-biosynthetic pathway (12). In accordance, expression of exogenous O-antigen gene clusters in P. aeruginosa 1244 allowed pilin to be glycosylated with the heterologous O-antigen repeating unit (12). Furthermore, expression of plasmid-borne pilAO1244 in nonserotype O7 P. aeruginosa strains resulted in pilin glycosylation, in which the glycan consisted of the host's O subunit (12). These experiments indicated that all nine P. aeruginosa O subunits tested and the E. coli O157 O subunit could serve as the 1244 pilin glycan (12). As a variety of O subunits could be conjugated to pilin, this indicated that the PilO glycan specificity was low, an extremely promising phenomenon in terms of exploiting the 1244 glycosylation machinery for the generation of glycoconjugate vaccines. Moreover, recent studies defined the substrate specificity of the 1244 glycosylation reaction (20, 21). Those studies indicated that PilO recognizes the sugar at the reducing end of the O-antigen repeating unit precursor (21) and that successful transfer of this carbohydrate is contingent upon the positioning of Ser at the pilin C terminus, as this configuration is essential for recognition by the glycosylation machinery (20). While no other specific pilin recognition features were present, the charge of the pilin surface must be compatible with the glycosylation apparatus (20). Application of these data may allow the use of O subunits of diverse gram-negative bacteria to be covalently linked to a variety of engineered proteins, including toxoids, likely expanding the potential breadth of protection. However, it is first necessary to test if the P. aeruginosa 1244 glycosylated pilin can provide O-antigen-specific protection, which is the objective of this study.
The current investigation shows that a bacterial protein glycosylation system is capable of biologically producing efficacious glycoconjugate vaccines. The work presented here demonstrates that the pilin glycan is a major immunogenic epitope, directing the production of antibodies against O antigen composed of analogous subunits. Vaccination with the 1244 pilin provided significant protection, with immunological specificity for the O polysaccharide, in two separate infection models. These results suggest that the pilin glycosylation system of P. aeruginosa 1244 may be useful for the biological production of anti-O-antigen glycoconjugate vaccines against a variety of harmful gram-negative bacterial pathogens.
MATERIALS AND METHODS
Animals.
The mice and rabbit strains used in this study are listed in Table 1. Immunizations involving female New Zealand White rabbits were conducted by Covance Research (Denver, PA). All animal experiments complied with federal and institutional specifications regarding the use of animals in research.
TABLE 1.
Animal strains, bacterial strains, and plasmid used in this study
| Organism or plasmid | Descriptiona | Source or reference |
|---|---|---|
| Animals | ||
| Mice | ||
| ICR | Age, 9-16 wk | Charles River Laboratories, Wilmington, MA |
| C3H/HeJ | Age, 9-16 wk | Jackson Laboratories, Bar Harbor, ME |
| BALB/c | Female; age, 6-8 wk | Hilltop Lab Animals, Scottdale, PA |
| CF-1 | Female; wt, 22 to 25 g | Carworth Farms, New York, NY |
| New Zealand White rabbits | Female, adult | Covance Research, Denver, PA |
| Bacterial strains | ||
| P. aeruginosa | ||
| 1244 | Wild type, IATS O7 | 35 |
| 1244N3 | 1244 rpoN (Tcr) | 34 |
| 1244.47 | 1244 pilA (Hgr) | 20 |
| PAK | Wild type, IATS O6 | 7 |
| 9D2 | Wild type, IATS O6 | 7 |
| K. pneumoniae B5055 | Serotype O1:K2 | 19 |
| Plasmid pPAC46 | pMMB66EH with P. aeruginosa 1244 pilAO Apr Cbr | 5 |
IATS, International Antigenic Typing System.
Bacterial strains and media.
The bacterial strains and plasmid used in this study are listed Table 1. For general culturing, the bacteria were grown aerobically at 37°C on LB agar plates or in LB broth shaken at 250 rpm. LB broth was used for cell growth for the preparation of strain 1244 pili glycosylated with the O7 repeating unit. For cell growth for the preparation of strain 1244 pili glycosylated with the O6 repeating unit, CAYE (2% agar, 0.75% Casamino Acids, 0.15% yeast extract) was used. These media contained carbenicillin (250 μg/ml) and/or tetracycline (50 μg/ml), as needed. When it was necessary, the medium was supplemented with 5 mM isopropyl-β-d-thiogalactopyranoside (IPTG).
Isolation of pili and purification of pilin.
The glycosylated 1244 pili used for vaccination were produced from P. aeruginosa 1244N3/pPAC46, as described previously (5, 12). Briefly, cell cultures were grown in LB broth supplemented with carbenicillin, tetracycline, and 5 mM IPTG at 37°C with shaking at 250 rpm for 14 h. The pili were isolated from the supernatant after centrifugation at 16,000 × g for 30 min at 4°C. The pili were purified by repeated precipitation in the presence of 0.5 M NaCl and 3% polyethylene glycol 8000 (40, 41). When it was required, traces of LPS were removed by first depolymerizing purified pili in 1.0% β-octylglucoside followed by gel filtration and chromatofocusing, as previously described in detail (8). No contaminating protein or LPS could be detected by polyacrylamide gel electrophoresis followed by silver staining or by Western blotting with an O-antigen-specific monoclonal antibody as a probe. The product of this procedure was referred to as chromatographically pure pilin (CPP). Samples of CPP bearing the glycan identical to the O7 LPS O-antigen subunit (CPPO7) were subjected to overnight dialysis against 6 liters H2O at 4°C, during which the subunits reaggregated into pilus-like, nonnative fibers (43). The LPS level of this preparation was measured by using the Limulus Gel-Clot test (Associates of Cape Cod). In these assays, in which strain 1244 LPS prepared by an established protocol (44) was used as a standard, a contamination level of less than 0.0004% (wt/wt) was detected. This material was lyophilized, resuspended in sterile phosphate-buffered saline (PBS), and filter sterilized. The bicinchoninic acid protein assay (Pierce) was used according to the manufacturer's protocol to determine the protein concentration.
P. aeruginosa 1244 pili glycosylated with the serotype O6 repeating unit were produced in two ways. In one, P. aeruginosa PAK, which produces O-antigen serotype O6 and a pilus which is antigenically distinct from that produced by strain 1244, was grown so that it contained pPAC46. This arrangement has previously been shown to produce 1244 pilin glycosylated with the O6 repeating unit (12). In the second situation, P. aeruginosa 9D2, which also produces O-antigen serotype O6, was also grown so that it contained pPAC46. Although this strain produces limited amounts of pili, the primary structure of the fibers produced is nearly identical to that of the fibers of strain 1244, differing only at residue 92 (7). This residue is situated in a region found to be poorly immunogenic (7, 8). Overexpression of pPAC46 resulted in large amounts of pilin glycosylated with the O6 repeating unit. For pilus production, a 5-ml LB culture inoculated with one of these strains containing pPAC46 was grown in the presence of carbenicillin at 37°C for 8 h. This suspension was used to inoculate aluminum foil-covered 68- by 28- by 3-cm metal pans containing 500 ml of solidified CAYE agar medium, carbenicillin, and IPTG, after which it was incubated at 37°C for 14 h. Cells were removed by scraping and were suspended in 50 ml of 40 mM sodium phosphate buffer, pH 7.2, per pan. This cell suspension was subjected to vigorous stirring for 30 min at room temperature to detach the pili and was then centrifuged at 16,000 × g for 30 min. Pili were isolated from the supernatant and purified by the procedure described above. CCP06 was produced as described above.
Protocols for determination of immunogenicity.
Two New Zealand White rabbits were injected intradermally in the back at multiple sites with a total of 250 μg of CPPO7 with Freund's complete adjuvant on day 1. On day 21, the rabbits received subcutaneous (s.c.) and intramuscular boosts containing 125 μg of CPPO7 with Freund's incomplete adjuvant (FIA). On day 42, the rabbits received s.c. boosts containing 125 μg of pilin administered in the rear flanks. On day 63, the rabbits were injected s.c. with 125 μg pilin with FIA. On day 84, the rabbits received s.c. dorsal injections containing 100 μg pilin and FIA. The final boost consisted of a s.c. injection in the neck region containing 100 μg pilin with FIA. On days 31, 52, 73, 94, and 115, sera were collected and analyzed for antibody production (data not shown). On day 118, approximately 50 ml of serum was collected in the terminal bleed and was used for subsequent assays. Adsorption of this serum was carried as follows. Six 82-mm nitrocellulose circles (Schleicher & Schuell) were incubated with stirring overnight at room temperature with 1.6 mg heterologously glycosylated (serotype O6 O-antigen subunit) 1244 pili (CPPO6) suspended in 15 ml deionized water. These circles were then treated with blocking buffer (12). Ten milliliters of serum recovered from one of the animals was incubated in succession overnight at room temperature with each of these circles.
To evaluate the induction of murine antibodies corresponding with acute-pneumonia protection studies, we used the following immunization schedule (first/second dose): intranasal (i.n.)/i.n. or i.n./s.c. At each immunization, a total dose of 5 μg of native 1244 pili diluted to the appropriate volume in sterile physiologic saline was administered to ICR mice. The time interval between the doses was 7 days. For i.n. immunization, the mice were anesthetized intraperitoneally with ketamine HCl (80 mg/kg of body weight; Aveco Co., Fort Dodge, IA) and xylazine HCl (8 mg/kg; Mobay Corporation, Shawnee, KA) prior to the instillation of the pili. The pili were delivered i.n. in a final volume of 25 μl by the use of individual sterile aerosol-resistant pipette tips for each mouse to prevent contamination. The final volume for s.c. injection was 100 μl. Control mice received sterile physiologic saline at exactly the same volume and by exactly the same route. At 3, 7, 10, and 14 days after the second dose of vaccine, the mice were killed by CO2 inhalation to obtain serum samples via cardiac puncture.
Preparation of challenge.
Overnight broth cultures of P. aeruginosa 1244, Klebsiella pneumoniae B5055, or the 1244 isogenic pilA mutant, P. aeruginosa 1244.47, were used to streak a lawn on Trypticase soy agar plates, which were incubated at 37°C for 12 to 18 h. The cells were suspended in sterile PBS, and a spectrophotometer (A650) or a Klett colorimeter was used to estimate the cell density on the basis of the previously determined counts of CFU. These cells were serially diluted and plated to determine viable-cell counts, which were used to more accurately determine the inoculum density.
Acute-pneumonia model.
For the pilus-specific-protection studies, ICR mice were immunized by either the i.n./i.n. or the i.n./s.c. route with purified 1244 pili, as described above. Seven days after the second vaccination, anesthesia was administered and the mice received an i.n. challenge of a lethal dose (approximately 4.3 × 106 CFU) of P. aeruginosa 1244 in a final volume of 50 μl (25 μl per nostril). Morbidity, mortality, and body weight were monitored daily for 8 days. The control mice were vaccinated with sterile physiologic saline as described above. In order to determine persistence and dissemination, in a separate experiment immunized and challenged mice were killed at 4, 24, and 48 h after bacterial treatment. The presence of bacteria in bronchoalveolar lavage (BAL) fluid at 4 and 24 h postchallenge was determined by a single washing with sterile physiologic saline via a 25-gauge hypodermic needle inserted into the trachea. In addition, the lungs, livers, and spleens from the animals were excised aseptically, weighed, and homogenized at 4, 24, and 48 h. The CFU counts in BAL fluid and organ homogenates were determined by previously described methods (19). The K. pneumoniae B5055 control challenge employed the same procedure used for P. aeruginosa 1244 and used a dose of 1.4 × 104 CFU.
In the glycan-specific-protection studies, BALB/c mice were used, and the vaccine consisted of CPPO7. CPPO7 (9 μg in 20 μl PBS; 10 μl per nostril) was administered i.n./i.n., as described above. As a control, mice received CPPO6 (purified from P. aeruginosa 9D2/pPAK46) to which purified strain 1244 LPS, prepared as described above, was added to a level 0.0004% (wt/wt) of that of pilin. Seven days following the second vaccination, the mice were anesthetized and were challenged with 3.8 times the 50% lethal dose (LD50; 4.9 × 107 CFU) of strain 1244.47, which was administered i.n. in 20 μl PBS (10 μl per nostril). In a separate trial, mice immunized with CPPO6 were challenged with 2.6 times the LD50 (3.4 × 107 CFU) of strain 1244.47. The LD50 of strain 1244.47 was determined to be 1.3 × 107 CFU by using the probit analysis of StatPlus 2007 software (data not shown). Following the challenge, the mice were closely monitored for 96 h, during which time morbidity and mortality were recorded.
Burned-mouse model.
Tests with the burned-mouse model were conducted as described previously (28). On day 0, CPPO7 was diluted to a concentration of 40 μg/100 μl with saline and mixed 1:1 with FIA, resulting in a solution with a pilus concentration of 20 μg/100 μl. A total of 27 mice were immunized with 100 μl of this material via the s.c. route (in the back). A control group (containing the same number of animals) was treated in the same manner with the same solution, except that the pili were absent. This treatment was repeated on day 7. On day 14, the mice were subjected to a nonlethal thermal injury of 15% of the body surface. which caused host immunosuppression, and were challenged with a subeschar injection of approximately 5 × 107 CFU of P. aeruginosa 1244.47. Additionally, eight untreated mice were challenged in the same manner. Following the challenge, mice were observed for 5 days, during which time morbidity and mortality were recorded.
ELISA.
Antibody titers in serum and BAL fluid were determined by an enzyme-linked immunosorbent assay (ELISA) with 96-well plates coated with pili from either P. aeruginosa 1244 or strain PAK/pPAC46 at a concentration of 2 μg/ml (50 μl/well, with incubation overnight at 4°C) or P. aeruginosa 1244 LPS at a concentration of 1 μg/ml (100 μl/well, with incubation overnight at 4°C). After the wells were treated with blocking buffer and washed, samples were serially diluted (twofold) in blocking buffer and incubated overnight in duplicate at 4°C. Bound antibodies were detected by using goat anti-mouse immunoglobulin G (IgG), alkaline phosphatase-labeled goat anti-mouse IgM, goat anti-mouse-IgA, or alkaline phosphatase-labeled goat anti-rabbit secondary antibodies (Kirkegaard & Perry, Gaithersburg, MD) diluted appropriately in blocking buffer; and the plates were developed with p-nitrophenylphosphate (Sigma, St. Louis, MO) in diethanolamine buffer (pH 9.8). The reaction was stopped by adding 50 μl of 3 M NaOH to each well after 30 min. The ELISA titers were defined as the dilution which gave a change in the A405 of 0.200 at room temperature. The background A405 in all control wells was always less than 0.050.
Statistics.
Differences in survival between the groups were analyzed by the log rank test. For comparison of CFU counts, the two-tailed Mann-Whitney U test was used. The statistical analyses were done with either Statistica software (version 4.5) for Windows, StatSoft software, or GraphPad Prism software (version 4.02).
RESULTS
Pilus vaccine protection from lethal challenge.
It was important to establish a model to determine an effective route of immunization and an appropriate challenge with wild-type P. aeruginosa 1244 as a precedent for subsequent studies. As P. aeruginosa is a leading cause of nosocomial pneumonia (30), the murine acute-pneumonia model was used to determine the protective efficacy of a vaccine composed of strain 1244 pili. An i.n./i.n. or i.n./s.c. (first/second dose) immunization schedule was used, with 7 days between doses. At each immunization, the mice received 5 μg of native 1244 pili diluted in sterile physiologic saline. Serum from both i.n./i.n.- and i.n./s.c.-immunized mice contained high titers of LPS-specific antibodies (IgG and IgM) (Table 2). These results indicated that a specific immune response was well established at 1 week following the second vaccination, as evidenced by a high serum IgG titer with specificity for the serotype O7 O antigen (Table 2). If this response could sufficiently mediate clearance of the infection, then strain 1244 glycosylated pili may provide O-antigen-specific immunity (8).
TABLE 2.
Systemic antibody responses of mice specific for P. aeruginosa O7 LPS after immunization with strain 1244 pili
| Routea | Ig class | Mean ELISA antibody titer on the indicated day after the second immunizationb
|
|||
|---|---|---|---|---|---|
| 3 | 7 | 10 | 14 | ||
| i.n./i.n. | IgG | 112 | 79.4 | 58.9 | 234 |
| IgM | 479 | 269 | 91.2 | 93.3 | |
| IgA | ND | ND | ND | ND | |
| i.n./s.c. | IgG | 166 | 269 | 135 | 123 |
| IgM | 617 | 794 | 676 | 490 | |
| IgA | ND | ND | ND | ND | |
The routes denoted are for the first/second immunization, in which the time between each immunization was 7 days.
Mean ELISA antibody titer (dilution) which gave a change in the A405 of 0.200 after 30 min. Each value shown is the mean of the individual titers from three mice. ND, none detected, as these values were below the limits of detection (A405 = 0.050).
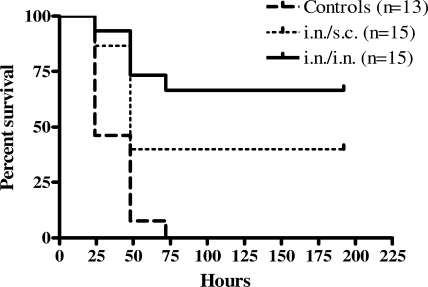
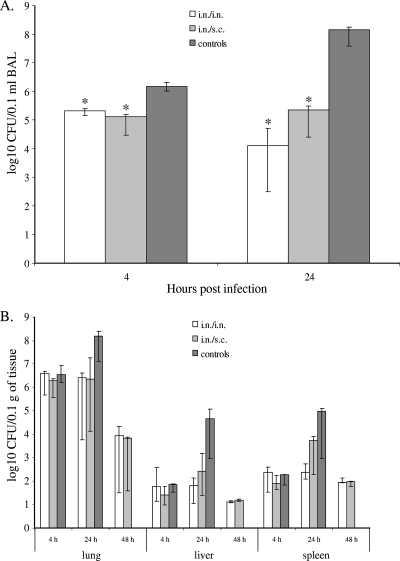
Because a specific immune response was established 1 week following the second pilus immunization (Table 2), the mice were challenged with a lethal dose of wild-type P. aeruginosa 1244 at this time point. The mice that received the pilus vaccine i.n./i.n. exhibited significant survival compared to the survival of the mice that received saline following an i.n. challenge of a lethal dose of strain 1244 (P < 0.0001) (Fig. 1). Although the mice that received the pilus vaccine i.n./s.c. showed reduced survival compared to the survival of the mice immunized with the pilus vaccine i.n/i.n., these mice still exhibited significantly increased survival compared to the survival of the saline-vaccinated mice (P = 0.0030) (Fig. 1). Compared to the saline-immunized controls, mice immunized with either regimen had significantly fewer bacteria in their BAL fluid at 4 h after infection (Fig. 2A). These data suggest that immunization with strain 1244 pili either prevents the adherence of the bacteria to epithelial surfaces or promotes the clearance of the bacteria from the airways. P. aeruginosa 1244 consistently disseminated from the site of infection, as evidenced by the culture of bacteria from homogenates of extrapulmonary organs, but only mice immunized twice i.n. were capable of lowering the numbers of bacterial CFU in the lungs as well as keeping bacterial multiplication in the liver and spleen at steady levels between 4 and 24 h after challenge (Fig. 2B). In addition, a significant decrease in the bacterial CFU from the BAL fluid over time (4 h versus 24 h after bacterial challenge) was observed only when the animals were immunized i.n./i.n. (Fig. 2A). Mice immunized i.n./s.c. and saline-immunized control mice showed increasing CFU counts in the lungs, liver, and spleen at 24 h after infection (Fig. 2B). At 48 h after infection, all saline-immunized mice were dead; however, the surviving mice immunized either i.n./i.n. or i.n/s.c. showed low CFU counts in all three organs investigated (Fig. 2B), indicating vaccine-induced clearance. Thus, survival correlated with decreasing bacterial counts both locally and systemically over the initial 48 h of infection.
FIG. 1.
Survival after P. aeruginosa 1244 challenge following immunization with pili in the acute-pneumonia model. Mice exhibited significantly increased survival when they were immunized i.n./i.n. or i.n./s.c. with strain 1244 pili compared to the survival of mice immunized with saline (control) following i.n. administration of a lethal dose of P. aeruginosa 1244. Results are shown as Kaplan-Meier survival curves, and differences in survival were calculated by log rank analysis. Results for i.n./i.n versus results for the control, P < 0.0001; results for i.n./s.c. versus results for the control, P = 0.0030; results for i.n./i.n. versus results for i.n./s.c., P = 0.1449.
FIG. 2.
P. aeruginosa 1244 growth in BAL fluid (A) or the lungs, livers, and spleens (B) of strain 1244 pilus-immunized and saline-immunized mice. The graphed values are medians, and error bars represent quartiles. Data are from two independent experiments (n = 4 mice per group per experiment). (A) *, P = 0.0062 for the counts in mice immunized i.n./i.n. and P = 0.0015 for the counts in mice immunized i.n./s.c. compared to the counts for the corresponding times postinfection of saline-immunized (control) animals (Mann-Whitney U test). (B) P. aeruginosa 1244 CFU counts from organ homogenates at 4, 24, or 48 h postinfection of mice immunized i.n./i.n., i.n./s.c., or with saline (control) are shown for all time points except for control mice at 48 h, as these mice were dead, making statistical analysis infeasible.
Analysis of vaccine specificity.
It was necessary to investigate if the immune response elicited by immunization with P. aeruginosa 1244 pili conferred specific protection or if the protection was due to a generalized inflammatory response caused by the administration of pili or trace LPS contamination. Therefore, by use of the respiratory model, mice that were immunized twice i.n. with strain 1244 pili were subsequently challenged with a heterologous bacterium (K. pneumoniae B5055) that did not produce pili and that was serologically distinct from strain 1244. Neither immunized nor control mice survived a lethal challenge with K. pneumoniae, indicating that vaccination with P. aeruginosa 1244 pili protects only against a homologous challenge (P = 0.0027 and P = 0.0009, respectively) (Table 3). These data suggest that the protection described here (Fig. 1 and 2) was a result of a specific immune response and was not due to nonspecific inflammation from the pilus vaccine. LPS contamination was not likely responsible for this protection, as a previous study showed that mice immunized s.c. with upwards of 200% of the contaminant level of strain 1244 LPS present in the current pilus dose did not induce a detectible anti-LPS response (8).
TABLE 3.
Vaccine specificity
| Immunizationa | No. of surviving mice/no. of total mice after challenge with:
|
|
|---|---|---|
| P. aeruginosa 1244 | K. pneumoniae | |
| P. aeruginosa 1244 pili | 4/4 | 0/8 |
| Saline | 0/6 | 0/6 |
Mice were immunized i.n./i.n.
Immunogenicity of the pilin glycan.
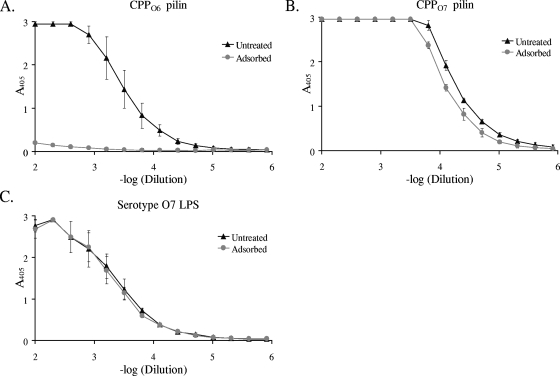
To better ascertain the immunogenicity of the pilin glycan, it was necessary to remove traces of LPS contamination. This was accomplished by depolymerization of the pili in the presence of β-octylglucoside, followed by chromatographic purification of the pilin produced (8). Detergent was removed upon dialysis of the purified pilin, which allowed reaggregation of the pilin subunits into nonnative pilus-like fibers (43). The Limulus assay showed that following this chromatographic purification, reaggregated pilin was contaminated with LPS to a level of less than 0.0004% (wt/wt). This substance, referred to as CPPO7, was used to immunize rabbits. One serum sample that was produced was adsorbed with CPPO6 in order to remove pilin protein-specific antibodies and retain those that were glycan specific. Adsorbed sera were unresponsive to CPPO6, while untreated sera had high titers of antibodies specific for these pili, suggesting that the adsorption had successfully removed pilin protein-specific antibodies (Fig. 3A). The sera that were adsorbed with CPPO6 had slightly lower titers of antibodies specific for CPPO7 than untreated serum (Fig. 3B). This difference was probably due to the absence of a pilin protein-specific response. Recognition of glycosylated pili by a serum sample that lacked a pilin protein-specific response suggested that the strong antipilus reaction was directed to the pilin glycan. Both the adsorbed and the untreated sera exhibited equal responses to serotype O7 LPS, showing that animals immunized with CPPO7 are capable of eliciting a strong anti-LPS response (Fig. 3C). Notably, the dominant antibody type produced by these rabbits was IgG, as determined by ELISA. Here, a 4-log-unit difference between the IgG and the IgM titers was seen (E. Jewell and P. Castric, unpublished observations), suggesting the involvement of T cells in the antiglycan response. These results confirm that CPPO7 is capable of stimulating a specific B-cell response against both the pilin glycan and the structurally similar O-antigen repeating unit of LPS (8) and suggest that the pilin glycan is a major pilin epitope.
FIG. 3.
ELISAs with sera from rabbits that had been immunized with CPPO7. Untreated or CPPO6-adsorbed rabbit serum was used as the primary antibody, and CPPO7 (A), CPPO6 (B), or serotype O7 LPS (C) was used as the antigen. Error bars represent standard deviations.
Immunization with CPPO7 followed by a lethal P. aeruginosa 1244 pilA-null challenge.
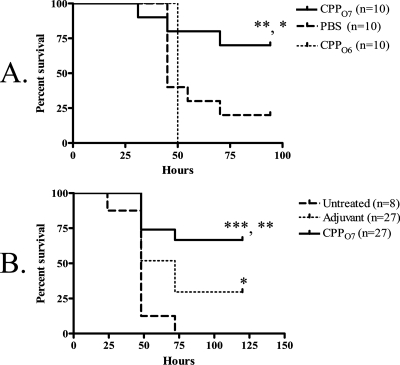
To determine the capacity of O-antigen-specific protection generated by a vaccine composed of CPPO7, we again employed the acute-pneumonia murine model but with a pilA-negative strain (P. aeruginosa 1244.47) as a challenge. This bacterial strain is an isogenic mutant of strain P. aeruginosa 1244 that does not produce pilin (20). However, strain 1244.47 produced wild-type levels of O7 LPS, as determined by Western blot analysis with a serotype O7-specific monoclonal antibody as a probe (data not shown). Mice receiving an i.n./i.n. administration of 9 μg CPPO7 exhibited significant survival compared to the survival of PBS-immunized mice (P = 0.0375) (Fig. 4A) following an i.n. challenge with a lethal dose of strain 1244.47, suggesting that this vaccine provides O-antigen-specific mucosal protection. Since CPPO7 may have contained a very small amount of remnant LPS following purification, the possibility remained that this material could have provided the protection seen here, as a previous study showed that intraperitoneal injection of 50 ng P. aeruginosa LPS could completely protect against a lethal challenge (32). To ensure that contaminant LPS did not provide the protection seen in the current study, mice were immunized i.n./i.n. with 9 μg CPPO6 that had been supplemented with serotype O7 LPS at an amount equivalent to that in the CPPO7 vaccine. This LPS-supplemented CPPO6 was therefore identical to the CPPO7 in both protein primary structure and maximum potential serotype O7 LPS contamination and differed only by the moieties of the glycan. Following an i.n. challenge with a lethal dose of strain 1244.47, the mice that were immunized with CPPO6 had significantly reduced survival compared to that for the mice immunized with CPPO7 (P = 0.0080) (Fig. 4A). These data indicate that the potentially minute levels of LPS present in CPPO7 were not responsible for the protection observed (Fig. 4A). Moreover, this control confirmed that the general inflammation generated by the pilin vaccine was not conferring protection. Therefore, this mucosal protection was elicited by the O7 pilin glycan, which is identical in structure to the O-antigen subunit of the challenge strain. Notably, isolated bacterial clones from tissues did not produce pilin, as determined by Western blot analysis with a pilin-specific monoclonal antibody as a probe, indicating that that this mutant did not revert to the wild type during infection (data not shown).
FIG. 4.
Survival after P. aeruginosa 1244.47 challenge following immunization with CPPO7 in the acute-pneumonia model (A) and burned-mouse model (B). Results are shown as Kaplan-Meier survival curves, and differences in survival were calculated by log rank analysis. (A) Mice were immunized i.n./i.n. with either CPPO7, PBS, or CPPO6. Mice immunized i.n./i.n. with CPPO7 exhibited significantly increased survival compared to the survival for mice immunized with PBS or CPPO6 following a lethal dose of the pilA mutant, strain 1244.47. *, P = 0.0375 for the results for mice immunized with CPPO7 versus the results for mice treated with PBS; **, P = 0.0080 for the results for mice immunized with CPPO7 versus the results for mice immunized with CPPO6. (B) Mice were immunized s.c. with either FIA mixed with CPPO7 or FIA alone or were untreated. Significant survival for mice that received the pilus vaccine compared to the survival for adjuvant-treated mice (**, P = 0.0087) or untreated mice (***, P = 0.0001) was observed.
To determine if the vaccination with CPPO7 could provide protective systemic immunity, we employed the burned-mouse model. Mice that were immunized twice with a mixture of 40 μg CPPO7 and FIA exhibited significant survival compared to that for the mice that received the control (P = 0.0042) (Fig. 4B) following a subeschar challenge with a lethal dose of strain 1244.47, indicating vaccine-induced, O-antigen-specific systemic protection. Because the challenge strain lacked pili but expressed the same O antigen as strain 1244, it is proposed that immunization with CPPO7 can provide O-antigen-specific protection and that the pilin glycan is a protective epitope. It should be noted that significant survival was observed in adjuvant-treated mice compared to the survival of the untreated mice (Fig. 4B). This is likely due to an inflammatory response and the attenuation of strain 1244.47 due to the absence of pili. However, as the survival of the mice that received the pilus vaccine was significantly higher than that of the adjuvant-treated mice, this suggests that this additional protection was due to specific immunity.
DISCUSSION
Like P. aeruginosa 1244, other bacteria such as Campylobacter jejuni and Neisseria gonorrhoeae possess molecular mechanisms that mediate the covalent linkage of surface oligosaccharides to proteins (1, 16, 42). Because of this peculiar phenomenon, all of these systems have been proposed to have the potential for use in the generation of bioconjugate vaccines (1, 8, 16). The current study was a necessary first step toward the assessment of the potential of a bacterial glycosylation system to generate an effective anti-O-antigen glycoconjugate vaccine. This work established that immunization with strain 1244 pilin, a glycoprotein in which the carbohydrate portion is an O subunit, provides O-antigen-specific protection against a P. aeruginosa challenge. By using both the P. aeruginosa acute-pneumonia and the thermal-injury models, we present evidence suggesting that glycosylated pili provide O-antigen-specific protection via the mucosal and systemic routes of immunity (Fig. 4). The results presented here are important because they show that a biological glycosylation system can be used to produce protective glycoconjugate vaccines that target the O antigen of a gram-negative bacterial pathogen. This work provides a foundation for the development of the 1244 pilin glycosylation technology to produce vaccines against many gram-negative bacterial pathogens. Future studies should focus on expanding the application of this technology by testing the protective efficacies of vaccines composed of strain 1244 pilin glycosylated with O subunits from highly virulent bacteria, such as E. coli O157. Fortuitously, O-antigen-biosynthetic genes are clustered on bacterial chromosomes (38), allowing ease of cloning (18). As shown previously, when P. aeruginosa 1244 expresses the E. coli O157 O-antigen biosynthesis genes in trans, the pili from the strain produced are decorated with the O157 O subunit (12). The present study would suggest that O157-glycosylated pilin should protect against challenge; however, this remains to be determined.
In current chemically cross-linked glycoconjugate vaccines, two purification steps must be performed, including isolation of an appropriate carrier protein, in addition to refinement of the bacterial surface polysaccharide. For gram-negative bacteria, this involves separation of the LPS O antigen from the lipid A portion. Following purification, these molecules are subjected to coupling chemistry. Altogether, these procedures are time-consuming and expensive. However, the production of large amounts of glycosylated pili can be accomplished quickly, is relatively inexpensive, and requires only common laboratory procedures. Moreover, it is well known that pili are immunogenic in humans, and their ability to generate protective immunity is documented (31). The immunogenicity of pili is partly due to the polymeric display of subunits in the pilus fiber (37). The arrangement of the glycan in a polymeric form both in native pilus fibers and in reaggregated pilin subunits (43) might promote expedient B-cell proliferation due to the proximity of adjacent glycan epitopes. Previous data from our laboratory indicated that the pilin glycan is exposed along the surface of the pilus fiber and can easily be accessed by antibodies (41) and, presumably, by B-cell membrane-bound Ig. If glycan recognition by membrane-bound Ig molecules leads to the internalization and major histocompatibility complex class II presentation of pilin peptides, this may stimulate the involvement of helper T cells, resulting in the proliferation and differentiation of the B cells producing glycan- and O-antigen-specific Ig. Additional subsequent work should explore the use of a mucosal adjuvant, such as the oligonucleotide CpG1826 (48), as well as the effect of additional boosts, in an effort to increase glycan-specific antibody production and protection. It will also be important to discern which antibody types are essential for the success of this vaccine.
Future studies should focus on optimizing this biological system for the production of glycosylated pili to be used as vaccines. For instance, although P. aeruginosa 1244 produces large yields of pili, the efficiency of pilus production may be increased by mutating pilT (45). As PilT is a pilus motor protein that mediates pilus retraction, mutational inactivation of this gene results in hyperpiliation (45). Additionally, a previous study has defined the pilin substrate of the 1244 glycosylation reaction, in which a C-terminal Ser or Thr and a compatible pilin surface charge are important (20). A gene encoding a normally nonglycosylated pilin (from P. aeruginosa PA103) was mutated to contain these characteristic structures and was made capable of glycosylation by the 1244 machinery (20). Although the current study essentially showed that pilin is an effective carrier protein for a glycoconjugate vaccine, other carriers, such as toxoids, may invoke a better response. It is therefore important to test the capacity of this glycosylation system to modify nonpilin proteins for use as vaccines. A recent study revealed that the glycan substrate recognition features lay within the reducing-end moiety of the O repeat (β-d-FucNAc) and that carbohydrates in this O-subunit position from many gram-negative bacteria are structurally similar (21). However, it is essential that more O-antigen-biosynthetic clusters be cloned and their products tested for compatibility with the 1244 glycosylation machinery to maximize its vaccine production potential.
Acknowledgments
This work was supported by a grant from the NIH to P.C. (grant AI054929).
We thank Antonio DiGiandomenico and Joanna Goldberg for instruction on animal models and for insightful discussion. In addition, we thank Liang Yuan, and Kathleen C. Glazer for technical assistance with the acute-pneumonia model.
Footnotes
Published ahead of print on 13 February 2008.
REFERENCES
- 1.Aas, F. E., A. Vik, J. Vedde, M. Koomey, and W. Egge-Jacobsen. 2007. Neisseria gonorrhoeae O-linked pilin glycosylation: functional analyses define both the biosynthetic pathway and glycan structure. Mol. Microbiol. 65:607-624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ada, G., and D. Isaacs. 2003. Carbohydrate-protein conjugate vaccines. Clin. Microbiol. Infect. 9:79-85. [DOI] [PubMed] [Google Scholar]
- 3.Anderson, P., M. E. Pichichero, and R. A. Insel. 1985. Immunogens consisting of oligosaccharides from the capsule of Haemophilus influenzae type b coupled to diphtheria toxoid or the toxin protein CRM197. J. Clin. Investig. 76:52-59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Avery, O., and W. Goebel. 1931. Chemo-immunological studies on conjugated carbohydrate-proteins. V. The immunological specificity of an antigen prepared by combining the capsular polysaccharide of type III pneumococcus with foreign protein. J. Exp. Med. 54:437-447. [DOI] [PMC free article] [PubMed]
- 5.Castric, P. 1995. pilO, a gene required for glycosylation of Pseudomonas aeruginosa 1244 pilin. Microbiology 141:1247-1254. [DOI] [PubMed] [Google Scholar]
- 6.Castric, P., F. J. Cassels, and R. W. Carlson. 2001. Structural characterization of the Pseudomonas aeruginosa 1244 pilin glycan. J. Biol. Chem. 276:26479-26485. [DOI] [PubMed] [Google Scholar]
- 7.Castric, P. A., and C. D. Deal. 1994. Differentiation of Pseudomonas aeruginosa pili based on sequence and B-cell epitope analyses. Infect. Immun. 62:371-376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Comer, J. E., M. A. Marshall, V. J. Blanch, C. D. Deal, and P. Castric. 2002. Identification of the Pseudomonas aeruginosa 1244 pilin glycosylation site. Infect. Immun. 70:2837-2845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Conlan, J., H. Shen, A. Webb, and M. Perry. 2002. Mice vaccinated with the O-antigen of Francisella tularensis LVS lipopolysaccharide conjugated to bovine serum albumin develop varying degrees of protective immunity against systemic or aerosol challenge with virulent type A and type B strains of the pathogen. Vaccine 20:3465-3471. [DOI] [PubMed] [Google Scholar]
- 10.Coughlin, R., and W. Bogard, Jr. 1987. Immunoprotective murine monoclonal antibodies specific for the outer-core polysaccharide and for the O-antigen of Escherichia 0111:B4 lipopolysaccharide (LPS). J. Immunol. 139:557-561. [PubMed] [Google Scholar]
- 11.Cryz, S. J., A. Lang, J. Wedgewood, J. Que, E. Furer, and U. Schaad. 1997. Immunization of cystic fibrosis patients with a Pseudomonas aeruginosa O-polysaccharide-toxin A conjugate vaccine. Behring Inst. Mitt. 98:345-349. [PubMed] [Google Scholar]
- 12.DiGiandomenico, A., M. J. Matewish, A. Bisaillon, J. R. Stehle, J. S. Lam, and P. Castric. 2002. Glycosylation of Pseudomonas aeruginosa 1244 pilin: specificity of glycan substrate. Mol. Microbiol. 46:519-530. [DOI] [PubMed] [Google Scholar]
- 13.DiGiandomenico, A., J. Rao, and J. B. Goldberg. 2004. Oral vaccination of BALB/c mice with Salmonella enterica serovar Typhimurium expressing Pseudomonas aeruginosa O antigen promotes increased survival in an acute fatal pneumonia model. Infect. Immun. 72:7012-7021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.DiGiandomenico, A., J. Rao, K. Harcher, T. S. Zaidi, J. Gardner, A. N. Neely, G. B. Pier, and J. B. Goldberg. 2007. Intranasal immunization with heterologously expressed polysaccharide protects against multiple Pseudomonas aeruginosa infections. Proc. Natl. Acad. Sci. USA 104:4624-4629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Donnelly, J. J., R. R. Deck, and M. A. Liu. 1990. Immunogenicity of a Haemophilus influenzae polysaccharide-Neisseria meningitidis outer membrane protein complex conjugate vaccine. J. Immunol. 145:3071-3079. [PubMed] [Google Scholar]
- 16.Feldman, M., M. Wacker, M. Hernandez, P. Hitchen, C. Marolda, M. Kowarik, H. R. Morris, A. Dell, M. Valvano, and M. Aebi. 2005. Engineering N-linked protein glycosylation with diverse O antigen lipopolysaccharide structures in Escherichia coli. Proc. Natl. Acad. Sci. USA 102:3016-3021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Fulop, M., M. Mastroeni, M. Green, and R. Titball. 2001. Role of antibody to lipopolysaccharide in protection against low- and high-virulence strains of Francisella tularensis. Vaccine 19:4465-4472. [DOI] [PubMed] [Google Scholar]
- 18.Goldberg, J. B., K. Hatano, G. M. Meluleni, and G. B. Pier. 1992. Cloning and surface expression of Pseudomonas aeruginosa O antigen in Escherichia coli. Proc. Natl. Acad. Sci. USA 89:10716-10720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Held, T., M. Trautmann, M. Mielke, H. Neudeck, S. J. Cryz, Jr., and A. Cross. 1992. Monoclonal antibody against Klebsiella capsular polysaccharide reduces severity and hematogenic spread of experimental Klebsiella pneumoniae pneumonia. Infect. Immun. 60:1771-1778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Horzempa, J., J. E. Comer, S. Davis, and P. Castric. 2006. Glycosylation substrate specificity of Pseudomonas aeruginosa 1244 pilin. J. Biol. Chem. 281:1128-1136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Horzempa, J., C. R. Dean, J. B. Goldberg, and P. Castric. 2006. Pseudomonas aeruginosa 1244 pilin glycosylation: glycan substrate recognition. J. Bacteriol. 188:4244-4252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jennings, H. J., and C. Lugowski. 1981. Immunochemistry of groups A, B, and C meningococcal polysaccharide-tetanus toxoid conjugates. J. Immunol. 127:1011-1018. [PubMed] [Google Scholar]
- 23.Jones, C. 2005. Vaccines based on the cell surface of carbohydrates of pathogenic bacteria. An. Acad. Bras. Cienc. 77:293-324. [DOI] [PubMed] [Google Scholar]
- 24.Konadu, E., J. Parke, Jr., H. Tran, D. Bryla, J. Robbins, and S. Szu. 1998. Investigational vaccine for Escherichia coli O157: phase 1 study of O157 O-specific polysaccharide-Pseudomonas aeruginosa recombinant exoprotein A conjugates in adults. J. Infect. Dis. 177:383-387. [DOI] [PubMed] [Google Scholar]
- 25.Kus, J., E. Tullis, D. Cvitkovitch, and L. L. Burrows. 2004. Significant differences in type IV pilin allele distribution among Pseudomonas aeruginosa isolates from cystic fibrosis (CF) versus non-CF patients. Microbiology 150:1315-1326. [DOI] [PubMed] [Google Scholar]
- 26.Mattick, J. S. 2002. Type IV pili and twitching motility. Annu. Rev. Microbiol. 56:289-314. [DOI] [PubMed] [Google Scholar]
- 27.Mond, J., A. Lees, and C. Snapper. 1995. T cell-independent antigens type 2. Annu. Rev. Immunol. 13:655-692. [DOI] [PubMed] [Google Scholar]
- 28.Neely, A., I. Holder, and G. Warden. 1999. Then and now: studies using a burned mouse model reflect trends in burn research over the past 25 years. Burns 25:603-609. [DOI] [PubMed] [Google Scholar]
- 29.Neu, H. C. 1992. The crisis in antibiotic resistance. Science 257:1064-1073. [DOI] [PubMed] [Google Scholar]
- 30.Obritsch, M., D. Fish, R. MacLean, and R. Jung. 2005. Nosocomial infections due to multidrug-resistant Pseudomonas aeruginosa: epidemiology and treatment options. Pharmacotherapy 25:1353-1364. [DOI] [PubMed] [Google Scholar]
- 31.Ohama, M., K. Hiramatsu, Y. Miyajima, K. Kishi, M. Nasu, and J. Kadota. 2006. Intratracheal immunization with pili protein protects against mortality associated with Pseudomonas aeruginosa pneumonia in mice. FEMS Immunol. Med. Microbiol. 47:107-115. [DOI] [PubMed] [Google Scholar]
- 32.Pier, G. B., H. F. Sidberry, and J. C. Sadoff. 1978. Protective immunity induced in mice by immunization with high-molecular-weight polysaccharide from Pseudomonas aeruginosa. Infect. Immun. 22:919-925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Pootoolal, J., J. Neu, and G. Wright. 2002. Glycopeptide antibiotic resistance. Annu. Rev. Pharmacol. Toxicol. 42:381-408. [DOI] [PubMed] [Google Scholar]
- 34.Ramphal, R., L. Koo, K. S. Ishimoto, P. A. Totten, J. C. Lara, and S. Lory. 1991. Adhesion of Pseudomonas aeruginosa pilin-deficient mutants to mucin. Infect. Immun. 59:1307-1311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ramphal, R., J. C. Sadoff, M. Pyle, and J. D. Silipigni. 1984. Role of pili in the adherence of Pseudomonas aeruginosa to injured tracheal epithelium. Infect. Immun. 44:38-40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Robin, G., Y. Keisari, R. Slepon, S. Ashkenazi, and D. Cohen. 1999. Quantitative analysis of IgG class and subclass and IgA serum response to Shigella sonnei and Shigella flexneri 2a polysaccharides following vaccination with Shigella conjugate vaccines. Vaccine 17:3109-3115. [DOI] [PubMed] [Google Scholar]
- 37.Schaffer, C., M. Graninger, and P. Messner. 2001. Prokaryotic glycosylation. Proteomics 1:248-261. [DOI] [PubMed] [Google Scholar]
- 38.Schnaitman, C. A., and J. D. Klena. 1993. Genetics of lipopolysaccharide biosynthesis in enteric bacteria. Microbiol. Rev. 57:655-682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Schneerson, R., O. Barrera, A. Sutton, and J. B. Robbins. 1980. Preparation, characterization, and immunogenicity of Haemophilus influenzae type b polysaccharide-protein conjugates. J. Exp. Med. 152:361-376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Silipigni-Fusco, J. 1987. Studies on the role of somatic pili as virulence and immunity factors in the pathogenicity of Pseudomonas aeruginosa. Ph.D. thesis. University of Pittsburgh, Pittsburgh, PA.
- 41.Smedley, J., III, E. Jewell, J. Roguskie, J. Horzempa, A. Syboldt, D. Beer Stolz, and P. Castric. 2005. Influence of pilin glycosylation on Pseudomonas aeruginosa 1244 pilus function. Infect. Immun. 73:7922-7931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Szymanski, C., and B. Wren. 2005. Protein glycosylation in bacterial mucosal pathogens. Nat. Rev. 3:225-237. [DOI] [PubMed] [Google Scholar]
- 43.Watts, T., D. Scraba, and W. Paranchych. 1982. Formation of 9-nm filaments from pilin monomers obtained by octyl-glucoside dissociation of Pseudomonas aeruginosa pili. J. Bacteriol. 151:1508-1513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Westphal, O., and K. Jann. 1965. Bacterial lipopolysaccharides. Extraction with phenol-water and further applications of the procedure, p. 83-91. In R. L. Whistler (ed.), Methods in carbohydrate chemistry, vol. 5. Academic Press, New York, NY. [Google Scholar]
- 45.Whitchurch, C. B., M. Hobbs, S. P. Livingston, V. Krishnapillai, and J. S. Mattick. 1991. Characterisation of a Pseudomonas aeruginosa twitching motility gene and evidence for a specialised protein export system widespread in eubacteria. Gene 101:33-44. [DOI] [PubMed] [Google Scholar]
- 46.Wuorimaa, T., R. Dagan, M. Vakevainen, F. Bailleux, R. Haikala, M. Yaich, J. Eskola, and H. Kayhty. 2001. Avidity and subclasses of IgG after immunization of infants with an 11-valent pneumococcal conjugate vaccine with or without aluminum adjuvant. J. Infect. Dis. 184:1211-1215. [DOI] [PubMed] [Google Scholar]
- 47.Yonath, A. 2005. Antibiotics targeting ribosomes: resistance, selectivity, synergism, and cellular regulation. Annu. Rev. Biochem. 74:649-679. [DOI] [PubMed] [Google Scholar]
- 48.Zuercher, A. W., M. P. Horn, H. Wu, Z. Song, C. J. Bundgaard, H. K. Johansen, N. Hoiby, P. Marcus, and A. B. Lang. 2006. Intranasal immunisation with conjugate vaccine protects mice from systemic and respiratory tract infection with Pseudomonas aeruginosa. Vaccine 24:4333-4342. [DOI] [PubMed] [Google Scholar]