Abstract
The effectiveness and potential immunosuppressive effects of anti-inflammatory glucocorticoids in the lungs of severe acute respiratory syndrome (SARS) patients are undefined. We treated porcine respiratory coronavirus (PRCV)-infected conventional pigs with the corticosteroid dexamethasone (DEX) as a model for SARS. Innate and Th1 cytokines in bronchoalveolar lavage (BAL) and serum were elevated in PRCV-infected pigs compared to controls, but were decreased after DEX treatment in the PRCV-infected, DEX-treated (PRCV/DEX) pigs. Although decreased in BAL, Th2 cytokine levels were higher in serum after DEX treatment. Levels of the proinflammatory cytokine interleukin-6 in BAL and serum were decreased in PRCV/DEX pigs early but increased later compared to those in phosphate-buffered saline-treated, PRCV-infected pigs, corresponding to a similar trend for lung lesions. PRCV infection increased T-cell frequencies in BAL, but DEX treatment of PRCV-infected pigs reduced frequencies of T cells; interestingly B and SWC3a+ (monocytes/macrophages/granulocytes) cell frequencies were increased. DEX reduced numbers of PRCV-stimulated Th1 gamma interferon-secreting cells in spleen, tracheobroncheolar lymph nodes, and blood. Our findings suggest that future glucocorticoid treatment of SARS patients should be reconsidered in the context of potential local immunosuppression of immune responses in lung and systemic Th1 cytokine-biased suppression.
The severe acute respiratory syndrome (SARS) global epidemic in 2003 to 2004 was caused by a newly emerged SARS coronavirus (SARS-CoV), likely of animal origin (19, 29, 42, 43). The SARS-CoV caused a severe and often fatal (10% mortality rate) atypical pneumonia, frequently accompanied by diarrhea in infected patients (12, 34, 35). The high public health impact of SARS has been attributed to a lack of information on control of the causative viral pathogen as well as a lack of effective treatments. A combination of the antiviral drug ribavirin and/or corticosteroids was used extensively in treating SARS patients, especially in hospitals in Hong Kong, China, and Canada (17, 58). In these hospitals, corticosteroids (e.g., prednisone or methylprednisolone) were used at high doses (1 to 10 mg/kg body weight per day) for a period spanning 1 to 3 weeks under a step-down course or pulsed intravenous regimen (3, 27, 46). The use of corticosteroids to treat SARS patients is controversial and remains under debate, with both beneficial and detrimental effects of this treatment being reported (3, 13, 17, 30, 31). Because corticosteroids are immunosuppressive, they may enhance local viral replication, thereby increasing lung lesions, and increase or prolong shedding of SARS-CoV (30). It is also suggested that ribavirin and corticosteroid treatments possibly contribute to extrapulmonary viral spread, leading to the multiorgan failure seen in some SARS patients (17).
Cytokines produced during a viral or bacterial infection are potent immunomodulatory molecules that act as mediators of inflammation and the immune response. They are key regulators in governing host defense against pathogens. Type I interferons (IFNs) including IFN-α and IFN-β are key components of innate immunity and are induced rapidly as early responses to viral infection (28, 40). Proinflammatory cytokines such as tumor necrosis factor alpha (TNF-α), interleukin-1 (IL-1), IL-6, and IL-8 are also produced early in the infection, triggering the production of Th1 cytokines such as IFN-γ and IL-2 involved in cellular immune responses. The Th2 cytokines such as IL-4, IL-5, and IL-10 evoke humoral immune responses. IFN-γ and IL-4 are mutually antagonistic and play an essential role in regulation of immune reactions, with IFN-γ supporting development of Th1 immunity, whereas IL-4 inhibits Th1 cell differentiation and promotes Th2 cell differentiation (37).
Despite general controversy over corticosteroid treatments in SARS patients, the mechanisms underlying corticosteroid treatment in modulating inflammation and immune responses to CoV infections are undefined. Research, although limited, indicates that cytokines are modulated by cortocosteroid treatment. In humans and mice, corticosteroids inhibited mRNA expression of IL-6 (2), IFN-γ (20, 26), and IL-4 (9). Corticosteroid-mediated inhibition of cytokine expression is a reversible event requiring the continued presence of the corticosteroid. Whereas short-term high-dose corticosteroids may be administered without precipitating major side effects, prolonged use of corticosteroids is frequently associated with major adverse effects that can involve virtually any organ system (32, 47).
Our goal was to define if corticosteroid treatment of porcine respiratory CoV (PRCV)-infected pigs alters immunity and lung inflammatory responses at the cytokine level and how these changes correlate with lung lesions in the pigs. Pigs have previously been exploited as a model to investigate the impact of steroids on human metabolic disorders because they have a metabolic response to steroids similar to humans (60, 61). Existence of PRCV, a counterpart in pigs to human respiratory CoV infections, makes pigs a valuable model for investigating responses to respiratory CoV infections, including SARS in humans. PRCV emerged independently in Europe and the United States in the 1980s as a spike gene deletion mutant of the porcine enteric CoV, transmissible gastroenteritis virus (10, 43, 44, 63). Members of the CoV family are prone to frequent mutations and recombination events in their genomes during replication, leading to generation of new CoVs that can have altered pathogenicity, different tissue tropism, or ability to cross the host species barrier. This phenomenon is exemplified by the emergence of SARS-CoV, likely becoming adapted to humans from bats after genomic mutation and recombination events either directly or via intermediate hosts (civet cats) (21, 36). Similar to SARS-CoV, PRCV almost invariably causes extensive lung lesions with atypical interstitial pneumonia, although many infections are clinically mild or asymptomatic (1, 23). However, the severity of PRCV infections is influenced by other factors such as polymicrobial coinfections (53, 54). Because of the pathological similarities of PRCV to SARS-CoV (atypical pneumonia and lung lesions), PRCV infection of outbred pigs is a useful model to mimic SARS-CoV lung lesions in humans (also outbred populations) and to test our hypothesis that corticosteroids impact CoV-induced lung disease and cytokine responses, thereby enhancing the clinical severity of a frequently mild respiratory CoV infection (PRCV).
MATERIALS AND METHODS
Virus.
The ISU-1 strain of PRCV (PRCV-ISU-1) was passaged eight times in swine testicular cell cultures and plaque purified two times (22) before it was propagated for six additional passages in swine testicular cells in our laboratory according to slightly modified procedures described previously (45). A single aliquoted virus pool was used as the inoculum throughout the study.
Pigs.
Conventional, specific-pathogen-free piglets (n = 130) were weaned at 16 to 20 days of age and transported to animal facilities at Ohio Agricultural Research and Development Center. Piglets were bled on the day of arrival, and prebleed sera were tested to confirm absence of neutralizing antibody to PRCV by a fluorescent focus neutralization test (62). Pigs were allowed to acclimate for an additional week before initiation of experiments. Five sequential batches of pigs were obtained for five individual trials. Piglets were randomly assigned to one of the four treatment groups: phosphate-buffered saline treated, PRCV infected (PRCV/PBS; n = 41); dexamethasone (DEX) treated, PRCV infected (PRCV/DEX; n = 41); PBS treated, mock infected (Mock/PBS; n = 23); and DEX treated, mock infected (Mock/DEX; n = 25). The results presented represent a composite of the five individual experiments.
Viral inoculation, DEX injection, and management of pigs.
The two PRCV pig groups were inoculated with PRCV via the intranasal (i.n.) and intratracheal (i.t.) routes using a total of 1 × 107 PFU of PRCV in 5 ml medium per pig, with 3 ml i.t. and 1 ml i.n. in each nostril. The i.t. inoculation was performed by sedating the pigs followed by an intubation procedure as an alternate to reported procedures (50). The anti-inflammatory corticosteroid DEX (VEDCO, St. Joseph, MO), was given daily by intramuscular injection at 2 mg/kg body weight (PRCV/DEX and Mock/DEX groups) from postinoculation days 1 to 6 (PID 1 to 6) to mimic the doses used for SARS patients (3, 27, 46). A shorter period of DEX administration (6 days from PID 1 to 6) was selected to reflect the shorter period (ranging from 1 to 3 weeks) of corticosteroid treatment used for some SARS patients. Uninfected control pigs received 5 ml of medium with or without injection with PBS (pH 7.4). Rectal temperatures, body weights, and respiration rates for all groups of pigs were recorded every other day. Pigs were maintained in accordance with the standards of the Institutional Laboratory Animal Care and Use Committee, The Ohio State University.
Evaluation of gross PRCV lung lesions.
Pigs from the four treatment groups were euthanized at PID 2, 4, 8, 10, and 21. Gross lesions were evaluated based on percentage of consolidation in each lung lobe of the six lobes. Final lung lesion scores were expressed as the total scores of all six lobes, with the full score being 10.875 if the lung was the most severely affected (100%).
Determination of cytokine concentrations in BAL fluids and serum by ELISA.
The bronchoalveolar lavage (BAL) fluids and blood were collected at euthanasia. The procedure for BAL extraction was modified from published methods (51, 52). At necropsy, either the left or right half of the lung of the euthanized pigs was lavaged with 50 ml of minimal essential medium supplemented with nonessential amino acids and antibiotic-antimycotic. Approximately 25 ml of BAL was recovered. The BAL was centrifuged at 800 × g for 10 min at 4°C to separate the BAL cells. Representative innate (IFN-α), proinflammatory (IL-6 and TNF-α), Th1 (IFN-γ), and Th2 (IL-4) cytokines were measured for serum and BAL fluids by enzyme-linked immunosorbent assay (ELISA) according to published procedures (4).
Isolation of MNC and cytokine ELISPOT assay.
For the isolation of mononuclear cells (MNC), the spleen, blood, and tracheobroncheolar lymph nodes (TBLN) were collected from euthanized pigs and processed for the enzyme-linked immunospot (ELISPOT) assay as previously described (49, 66).
Flow cytometric analysis (FACS) of CD3+, CD4+, CD8+, and CD21+ lymphocytes and SWC3a+ cells in BAL and PBMCs.
For each sample, 2 × 106 BAL cells or peripheral blood mononuclear cells (PBMCs) were stained. The cells were incubated with optimal dilutions of antibodies in staining buffer (PBS-0.02% sodium azide-0.5% bovine serum albumin) for 15 to 30 min at 4°C. Two washings with staining buffer were applied after each incubation. For CD4+ and CD8+ cell staining, the BAL cells were incubated with 1 μl of fluorescein isothiocyanate-conjugated mouse anti-pig CD4a or CD8a monoclonal antibodies (BD Biosciences). For CD3+ T cells, 1 μl of biotin-conjugated mouse anti-pig CD3ɛ (SouthernBiotech, Birmingham, AL) monoclonal antibody was used, followed by incubation with streptavidin-PerCP (peridinin-chlorophyll-protein complex) conjugate (BD Biosciences). The CD21+ B cells were stained with 1 μl of phycoerythrin-conjugated anti-human CD21 (BD Biosciences). Detection of pig CD21+ cells using this human clone is based on its cross-reaction with pig CD21 expressed on B cells (8). SWC3a is expressed on the cell membrane of monocytes, macrophages, and granulocytes (48). The SWC3a+ cells were stained with 1 μl of mouse anti-pig monocyte/granulocyte-R-phycoerythrin (RPE; SouthernBiotech, Birmingham, AL). Ten thousand BAL cells or PBMCs were analyzed of the 2 × 106 cells stained for each sample by fluorescence-activated cell sorting (FACS). Lymphocytes were defined by their light scatter characteristics (41). For discrimination of positive and negative populations, quadrant markers were set and these were controlled by nonstained samples and samples incubated only with isotype control antibodies. FACS data were analyzed using the Cellquest software (Becton Dickinson FACSCalibur, CA). The frequency of each individual type of lymphocyte or SWC3a+ cells was expressed as the percentage of these cells within the 10,000 BAL cells or PBMCs counted by FACS.
Data analysis.
Statistical analyses were performed for each PID among the four experimental groups using the nonparametric Kruskal-Wallis test. Statistical significance was assessed as P < 0.05.
RESULTS
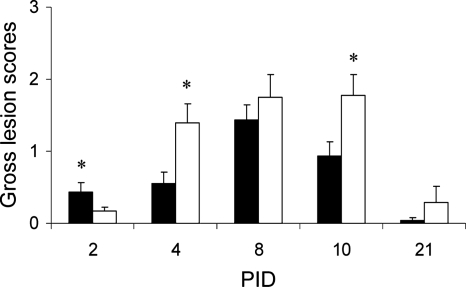
DEX treatment alleviated gross PRCV lung lesions at PID 2 but exacerbated lung lesions thereafter.
This study confirmed the subclinical nature of PRCV infection in pigs based on the dose (1 × 107 PFU) and strain (ISU-1) used (1, 23). Although PRCV-infected pigs treated with DEX remained clinically healthy, the DEX treatment precipitated mild fevers from PID 4 to 8, induced growth retardation, and enhanced respiratory rates. No macroscopic lesions were observed in organs (liver, spleen, kidney, etc.) other than the lungs. Typical gross lung lesions induced by PRCV infection were focal lobular and multifocal lobular consolidations on both dorsal and ventral sides across the entire lung but with more concentrated lesions seen in the apical lobes. Detailed clinical and histopathologic data are summarized in a separate article (25). Focal lobular consolidation was observed at PID 2 in lungs of both PRCV/PBS and PRCV/DEX pigs, but with lesions in PRCV/DEX pigs being significantly less severe than those in PRCV/PBS pigs, suggesting that DEX played a role in alleviating lung pathology induced by PRCV infection early at PID 2 (Fig. 1). Gross lesions peaked at PID 8 for the group infected with PRCV alone and then declined. However, lungs of the PRCV/DEX pigs had significantly more severe gross lesions than those of the PRCV/PBS pigs at PID 4 to 21, with peak lesions being sustained longer (PID 8 to 10), indicating that DEX treatment exacerbated PRCV-induced lung lesions from PID 4 to 21.
FIG. 1.
Mean gross lung lesion scores of PRCV/PBS and PRCV/DEX groups. Black bars, PRCV/PBS; white bars, PRCV/DEX. Asterisks represent significant differences by Kruskal-Wallis test. Pig numbers are as follows for each of the PRCV/PBS and PRCV/DEX groups: n = 8 on PID 2, 4, 8, and 10 and n = 9 on PID 21.
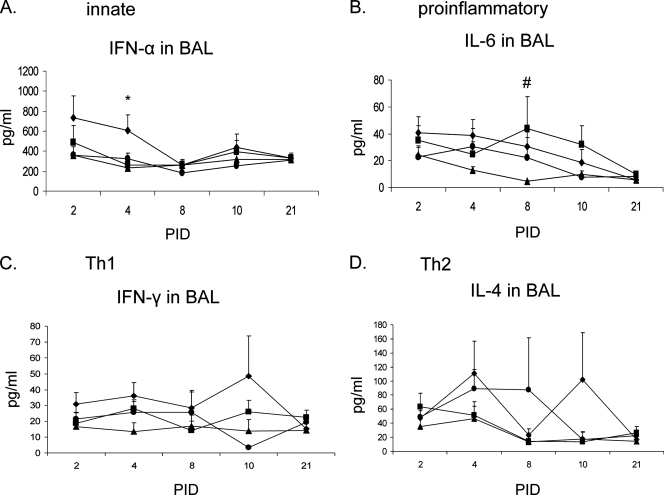
DEX downregulated the proinflammatory cytokine (IL-6) in BAL early (PID 2 and 4), but not later (PID 8, 10, and 21), and suppressed local innate (IFN-α), Th1 (IFN-γ), and Th2 (IL-4) cytokine levels in BAL.
We first assessed the cytokine levels in BAL fluids. Representative innate (IFN-α), proinflammatory (IL-6 and TNF-α), Th1 (IFN-γ), and Th2 (IL-4) cytokines were measured (Fig. 2). The PRCV infection alone (PRCV/PBS group) increased IFN-α in BAL from PID 2 to 10, with two- to threefold increases evident above the uninfected groups at PID 2 and 4 (Fig. 2A). The DEX treatment in the PRCV/DEX pigs reduced IFN-α (significant reduction at PID 4) to levels comparable to those in BAL of uninfected controls. Levels of TNF-α from most pigs were below the lowest detection limit of the ELISA (7.8 pg/ml), and therefore these data are not presented. Elevation of IL-6 was evident in the BAL of both PRCV-infected groups compared to controls (significantly elevated at PID 8) (Fig. 2B). Downregulation of IL-6 by DEX in the PRCV/DEX pigs was seen at the early stage (PID 2 to 4) compared to the higher concentrations of IL-6 induced in the PRCV/PBS pigs. However, the PRCV/DEX pigs had 1.2- to 1.8-fold-higher IL-6 levels than the PRCV/PBS pigs later at PIDs 8, 10, and 21. For IFN-γ and IL-4, the PRCV/PBS pigs had 1.5- to 5-fold higher levels of these cytokines in BAL from PID 2 to 10 (except IL-4 at PID 2 and 8) compared to PRCV/DEX and uninfected control pigs (Fig. 2C and D). In comparison, DEX treatment in the PRCV/DEX pigs resulted in decreased IFN-γ and IL-4 in BAL at most times to levels comparable to those of mock control pigs (Fig. 2C and D).
FIG. 2.
Cytokine levels in the BAL of euthanized pigs. Cytokine concentrations in pg/ml are expressed as means ± standard errors. Symbols: ⧫, PRCV/PBS; ▪, PRCV/DEX; ▴, Mock/PBS; •, Mock/DEX. Pig numbers are as follows for the PRCV/PBS, PRCV/DEX, Mock/PBS, and Mock/DEX groups, respectively: n = 8, 8, 4, and 5 on PID 2; n = 8, 8, 5, and 5 on PID 4; n = 8, 8, 5, and 5 on PID 8; n = 8, 8, 4, and 4 on PID 10; and n = 9, 9, 5, and 6 on PID 21. * and # denote statistically significant difference (P < 0.05) when comparing the PRCV/PBS versus PRCV/DEX groups and PRCV-infected versus uninfected control groups, respectively, by nonparametric Kruskal-Wallis test.
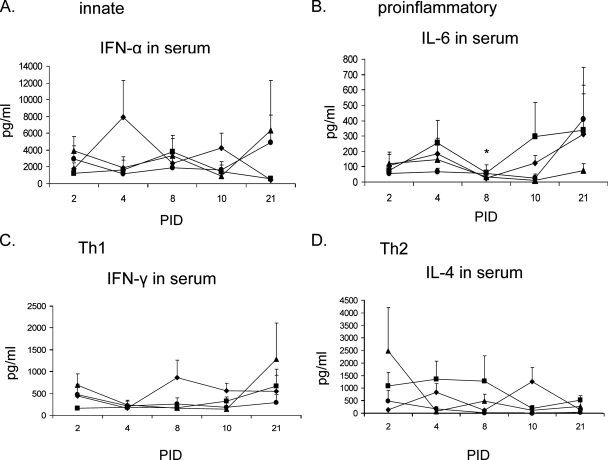
Similar trends were evident for IFN-α, IL-6, and IFN-γ in serum as in BAL, but unlike in BAL, DEX elevated IL-4 in serum of PRCV/DEX pigs.
To compare the effects of PRCV infection and DEX treatment on local cytokine levels in BAL to systemic cytokine levels in serum, cytokines were evaluated in the sera of the euthanized pigs (Fig. 3). The IFN-α in serum of PRCV/PBS pigs increased three- to fivefold compared to that in mock controls at PID 4 and 10 (Fig. 3A), but IFN-α was suppressed by DEX in the PRCV/DEX pigs at both PID. Similar to BAL, TNF-α in the serum was below the lowest detection limit of the ELISA (data not shown). After PRCV infection at PIDs 4 and 10, slightly increased IL-6 occurred in the sera of both PRCV-infected groups compared to the uninfected controls (Fig. 3B). However, like in BAL, serum IL-6 was slightly downregulated by DEX in the PRCV/DEX pigs at PID 2 but was increased 1.5- to 2-fold at PID 4 and 10 and significantly increased at PID 8 compared to the PRCV/PBS pigs. Unlike in BAL, no early IFN-γ responses occurred in serum of either of the PRCV-infected groups compared to mock controls, and although the responses of the PRCV/PBS pigs increased, peaking at PID 8 and 10, responses in the PRCV/DEX pigs remained low (Fig. 3C). It is noteworthy that the trend for serum IL-4 was almost opposite to that in BAL (Fig. 3D): whereas DEX treatment in the PRCV/DEX pigs decreased IL-4 in BAL, in serum IL-4 was increased (PID 2, 4, and 8) compared to that in the PRCV/PBS pigs. Differences in IL-4 concentrations between BAL and serum may possibly reflect different cell populations producing IL-4 locally in BAL versus systemically in serum and in response to the DEX-induced decreased Th1 responses in serum.
FIG. 3.
Cytokine levels in the serum of euthanized pigs. Cytokine concentrations in pg/ml are expressed as means ± standard error. Symbols: ⧫, PRCV/PBS; ▪, PRCV/DEX; ▴, Mock/PBS; •, Mock/DEX. Pig numbers are as follows for the PRCV/PBS, PRCV/DEX, Mock/PBS, and Mock/DEX groups, respectively: n = 8, 8, 4, and 5 on PID 2; n = 8, 8, 5, and 5 on PID 4; n = 8, 8, 5, and 5 on PID 8; n = 8, 8, 4, and 4 on PID 10; and n = 9, 9, 5, and 6 on PID 21. * denotes statistically significant difference (P < 0.05) between the PRCV/PBS and PRCV/DEX groups by nonparametric Kruskal-Wallis test.
Lower numbers of Th1 (IFN-γ) cytokine-secreting cells were detected both systemically and locally in PRCV/DEX pigs than in PRCV/PBS pigs.
To determine the responsiveness of activated antigen-specific effector or memory Th1 or Th2 lymphocytes in secreting cytokines upon in vitro restimulation with PRCV antigen, MNC were purified from spleen, blood, and TBLN, and a cytokine ELISPOT assay was used to quantitate Th1 (IFN-γ) and Th2 (IL-4) cytokine-secreting cells (CSC) after in vitro stimulation of MNC with purified PRCV antigen (Fig. 4). Uninfected control pigs had few (less than 50 CSC per 5 × 105 MNC) or no PRCV-stimulated IFN-γ CSC. The PRCV/PBS-infected pigs showed a general trend of increasing numbers of IFN-γ CSC in spleen, blood, and TBLN early from PID 2 to 10, after which MNC secreting IFN-γ either plateaued (blood and TBLN) or declined (spleen) (PID 10 to 21) (Fig. 4). The DEX treatment in the PRCV/DEX pigs resulted in significantly decreased numbers of IFN-γ CSC in spleen (PID 4, 8, and 10), TBLN (PID 10), and blood (PID 4) compared to the PRCV/PBS pigs. The reduced numbers of IFN-γ CSC in the PRCV/DEX pigs compared to the PRCV/PBS pigs was consistent with the lower concentrations of IFN-γ in serum and BAL of the PRCV/DEX pigs. The numbers of IL-4 CSC were low (less than 20 CSC per 5 × 105 MNC) in the PRCV-infected pigs, making it difficult to evaluate the potential effects of DEX treatment on IL-4 CSC (data not shown).
FIG. 4.
IFN-γ CSC in spleen, TBLN, and blood of euthanized pigs. Symbols: ⧫, PRCV/PBS; ▪, PRCV/DEX; ▴ (triangles), Mock/PBS; •, Mock/DEX. Pig numbers are as follows for the PRCV/PBS, PRCV/DEX, Mock/PBS, and Mock/DEX groups, respectively: n = 8, 8, 4, and 5 on PID 2; n = 8, 8, 5, and 5 on PID 4; n = 8, 8, 5, and 5 on PID 8; n = 8, 8, 4, and 4 on PID 10; and n = 9, 9, 5, and 6 on PID 21. * denotes statistically significant difference (P < 0.05) between the PRCV/PBS and PRCV/DEX groups by nonparametric Kruskal-Wallis test.
DEX decreased PRCV-induced CD3+, CD4+, and CD8+ lymphocyte frequencies but increased CD21+ lymphocyte and SWC3a+ cell frequencies in BAL.
The recovered cells from the BAL fluids were stained with antibodies against surface markers of CD3+, CD4+, CD8+, and CD21+ lymphocytes or monocytes, macrophages, and granulocytes (SWC3a+) followed by FACS to determine the effects of PRCV infection and DEX treatment on frequencies of these cell types in lung (Fig. 5). The trend was for higher (1.2 to 20 times) CD3+, CD4+, and CD8+ cell frequencies in the PRCV/PBS pigs compared to the uninfected controls (except PID 21) and for lower (1.3 to 4 times) CD3+, CD4+, and CD8+ cell frequencies in the PRCV/DEX compared to PRCV/PBS pigs at PID 2, 8, and 10 (Fig. 5A, B, and C). However, frequencies of CD21+ cells in BAL were significantly increased in the PRCV/DEX and Mock/DEX pigs by DEX treatment independent of PRCV infection (except PID 10 and 21) (Fig. 5D). The PRCV infection alone in PRCV/PBS pigs caused a slight decrease in SWC3a+ cell frequencies in BAL compared to the uninfected controls (except PID 21), and DEX treatment of PRCV infected pigs increased frequencies of these cells at PID 2, 4, and 10 (Fig. 5E). These results suggest that PRCV infection increased CD3+, CD4+, and CD8+ lymphocyte frequencies in the lung, but DEX treatment of PRCV-infected pigs reduced these lymphocyte frequencies, although interestingly it increased the frequencies of the CD21+ and SWC3a+ cells in the lung.
FIG. 5.
Frequencies of CD3+, CD4+, CD8+, and CD21+ lymphocytes and SWC3a+ cells in total BAL cells of euthanized pigs. Bars: black, PRCV/PBS; white, PRCV/DEX; dashed, Mock/PBS; dotted, Mock/DEX. Pig numbers are as follows for the PRCV/PBS, PRCV/DEX, Mock/PBS, and Mock/DEX groups, respectively: n = 8, 8, 4, and 5 on PID 2; n = 8, 8, 5, and 5 on PID 4; n = 8, 8, 5, and 5 on PID 8; n = 8, 8, 4, and 4 on PID 10; and n = 9, 9, 5, and 6 on PID 21. * and # denote statistically significant difference (P < 0.05) when comparing the PRCV/PBS versus PRCV/DEX groups and Mock/PBS versus Mock/DEX groups, respectively, by nonparametric Kruskal-Wallis test.
Staining of PBMCs revealed no obvious trends for CD3+, CD4+, and CD8+ lymphocytes, CD21+ lymphocytes, and SWC3a+ cells in PBMCs after PRCV infection or DEX treatment (data not shown).
DISCUSSION
In this study, we assessed the impact of the corticosteroid DEX on PRCV lung lesions, immune responses, and the possible roles of proinflammatory, Th1, and Th2 cytokines in mediating PRCV pathogenesis and disease. Although chemokines are of interest in their potential role in mediating PRCV pathogenesis, they were not assayed in this study due to the lack of antibody reagents and specific tests for detection of porcine chemokines. The PRCV-seronegative outbred conventional pigs were chosen in this study to better mimic the diverse physiologic and immunological responses likely to occur in humans (also an outbred population) exposed to a new respiratory viral pathogen such as SARS-CoV.
Our data indicated that PRCV infection alone (PRCV/PBS) generally increased IFN-α, IFN-γ, and IL-4 in BAL and in serum in the early to middle phase of the infection (PID 2 to 10). The elevation of IFN-α is consistent with previous reports that indicated that PRCV is a good inducer of IFN-α (5, 6, 55). The PRCV infection elevated the proinflammatory cytokine IL-6 in BAL and in serum at PID 4 and 10 of PRCV/PBS pigs compared to uninfected controls. In this aspect, infection with PRCV resembles that with SARS-CoV, which also upregulated IL-6 in SARS patients (24) and in a murine macrophage SARS-CoV infection model (59). The PRCV-induced increases in Th1 and proinflammatory cytokines have some similarities to cytokine profiles observed in SARS patients where elevation of IFN-γ and IL-6 in the blood of SARS patients was observed (24, 64). The similarities in cytokine profiles between PRCV infection in pigs and SARS patients further support PRCV as a potential model for respiratory infections specifically targeting the lung and inducing atypical pneumonia resembling SARS lesions. Although PRCV infection in pigs is a valuable model for studying the respiratory phase of CoV infections such as SARS, there are also differences between PRCV and SARS-CoV pathogenesis. For instance, PRCV infection causes an acute self-limiting respiratory disease that is restricted to the lungs and has little or no enteric component. In contrast, some SARS cases manifested systemic multiorgan infection and an enteric infection phase with viable SARS-CoV shedding in stool (38, 65). So, PRCV infection of pigs may not reflect the potential effect of corticosteroids on these systemic or enteric aspects of SARS. However, whether these multiorgan effects may have been precipitated or enhanced by prolonged or high-dose corticosteroid or ribavirin use is uncertain (17).
The DEX treatment of PRCV/DEX pigs suppressed early local IL-6 levels in BAL and systemic IL-6 levels in serum (PID 2 or 4), but led to increased IL-6 concentrations in BAL and serum later (PID 8, 10, and 21). Thus, the data suggest that DEX may decrease PRCV-induced inflammation (IL-6) at an early infection stage (PID 2 to 4), but not later. Similarly, elevation of proinflammatory gene expression in blood was found in the acute phase of SARS, but this increase was reduced together with decreases of antiviral cytokines such as IFN-α after corticosteroid treatment (11). This trend for IL-6 in the BAL corresponded to the results for gross lesions in the lungs of the PRCV/PBS and PRCV/DEX pigs that indicated that at PID 2, the lesion score of the PRCV/DEX pigs was significantly lower than that of the PRCV/PBS pigs and higher thereafter. Our results suggest that the DEX-induced reduction of IL-6 at early PID (PID 2) might have played a role in the delayed onset of PRCV lung lesions, and increases of IL-6 at later PIDs may contribute to the severity of the lung lesions, thus mimicking the delayed onset of disease severity seen in SARS patients treated with corticosteroids (31). A role for IL-6 in progression of lung pathology of SARS patients was suggested by the observation that serum IL-6 levels correlated most strongly with radiographic lung lesion scores of SARS patients (14). The overproduction of specific proinflammatory cytokines such as TNF-α and IL-6 is regarded as a hallmark of some viral infections (33), and the levels of TNF-α and IL-6 were also found to correlate positively with disease severity in swine viral pneumonias (56, 57).
The DEX treatment in the PRCV/DEX pigs caused a pronounced suppression of local innate (IFN-α), Th1 (IFN-γ), and Th2 (IL-4) cytokines in BAL and IFN-γ CSC in spleen, blood, and the lymph node draining the lung (TBLN). The early and often persisting (PID 4 to 10) decreased IFN-α, IFN-γ, and IL-4 in BAL of the PRCV/DEX pigs corresponded with increased gross lung lesions from PID 4 to 10 in this group compared to the PRCV/PBS pigs, suggesting a role for these cytokines in controlling PRCV replication in the lungs and therefore pathogenicity. Similarly, Cameron et al. (11) suggested that dysregulation of innate and adaptive immune responses due to loss of homeostasis of type I (IFN-α) and type II (IFN-γ) cytokines led to a failure of SARS-CoV clearance from the lungs in severe SARS patients at crisis, whereas resolution of IFN and IFN-stimulated genes in nonsevere SARS patients was associated with recovery. At PID 21 (15 days after cessation of DEX treatment), however, levels of cytokines in BAL were similar among the four experimental pig groups and the effects of DEX on serum cytokines, IFN-γ CSC, and percentages of lymphocytes and monocytes/macrophages at this PID were lower than at earlier PID, consistent with the finding that corticosteroid-mediated inhibition of cytokine expression is a reversible event requiring the continued presence of the corticosteroids (26).
The results also showed a trend for reduced numbers of CD3+, CD4+, and CD8+ lymphocytes in BAL of PRCV/DEX pigs compared to PRCV/PBS pigs, consistent with the decreased levels of Th1 (IFN-γ) and Th2 (IL-4) cytokines in BAL. In contrast, there was not a consistent decrease of SWC3a+ cells in BAL after DEX treatment in PRCV/DEX pigs (Fig. 5E), but instead, a slight increase was observed on PID 2, 4, and 10. The more severe lung pathology in the PRCV/DEX pigs may be associated with the increased SWC3a+ cells in lung, as seen in SARS patients, with severe immunopathological damage induced by stimulated (or possibly infected) macrophages in lungs. Immunohistochemical staining of PRCV-infected pig lung tissues occasionally revealed PRCV antigens in interstitial macrophages and in alveolar macrophage-like mononuclear cells within the thickened alveolar septa (25). These findings are consistent with observations by Cox et al., who reported PRCV antigens in alveolar macrophages (15, 16). It was speculated that macrophages play a role in the deteriorating respiratory condition of SARS patients (34, 38). However, a consistent reduction of CD3+, CD4+, and CD8+ cells in blood was not observed in PRCV/DEX pigs compared to PRCV/PBS pigs (data not shown). The discrepancy in the patterns of CD3+, CD4+, CD8+, and CD21+ lymphocytes and SWC3a+ cells in BAL versus PBMCs indicates that the composition of the BAL cells does not simply reflect cell recruitment from the blood, but PRCV infection and DEX treatment likely compromised migration of these cells into the lungs from other lymphoid tissues as well. Thus, in human respiratory infections, monitoring of total immune cell populations or cytokines (see below) in PBMCs or serum may not as accurately reflect those in the lung as those measured in BAL.
Although a generally similar pattern of cytokine responses including IFN-α, IL-6, and IFN-γ were observed in serum as in BAL, differences existed between the local BAL and systemic serum cytokine levels. Unlike in BAL, no early serum IFN-γ responses occurred in either of the PRCV-infected groups (Fig. 3C). The differences likely reflect the different cell types involved in local versus systemic cytokine secretion as well as the localized nature of PRCV infection in lungs. It is particularly noteworthy that the trend for serum IL-4 was almost opposite to that in BAL. Although DEX treatment in the PRCV/DEX pigs decreased local IL-4 in BAL, being also associated with reduced numbers of CD3+, CD4+, and CD8+ cells in BAL, in serum, IL-4 was increased (PID 2, 4, and 8) compared to that in the PRCV/PBS pigs, which suggests that DEX treatment in the PRCV/DEX pigs led to increased systemic Th2 (serum IL-4) responses in the face of the pronounced Th1 (serum IFN-γ) downregulation, supporting the notion that IFN-γ and IL-4 are mutually antagonistic and that IL-4 inhibits Th1 cell differentiation and promotes Th2 cell differentiation (7, 18, 37).
Because of the variability in responses encountered in outbred pigs, which reflects similar response variations among the human population, we used a large number of pigs (a total of 130) in a series of five pig batches in this study in an attempt to assess if statistically significant differences occurred among the treatment groups. Although a trend for the experimental parameters assessed was reproducible among the different pig batches, statistically significant differences, however, were only achieved in some assays at certain PID after the corresponding data were combined. Variation in biological parameters of outbred animals as models for humans is unavoidable and yet more accurately reflects the nature of biological differences in human populations in contrast to more uniform responses of inbred laboratory animal models.
In summary, these findings suggest that whereas one to two doses of DEX in the acute phase of the infection may effectively alleviate early proinflammatory responses in respiratory CoV infections, DEX administration for a longer period (PID 1 to 6) may play a role in enhancing viral replication in lung by depressing the innate and early cytokine responses followed later by inflammation-mediated (IL-6, etc.) or virus-induced necrotic lung damage. Because of the disease similarities in the atypical interstitial pneumonia and cytokine profiles induced by PRCV and SARS-CoV infection in lungs and the physiological and anatomic similarities between pigs and humans, this study may have implications for corticosteroid treatment of SARS patients. In severe SARS patients, IFN-mediated immune response deficiencies developed as the illness progressed (11). Longer-term use of high-dose corticosteroids may further dampen local and systemic Th1 IFN-γ responses, as shown in this study, leading to enhanced viral replication that subsequently precipitates more severe pathology in the lungs. The SARS patients typically develop acute respiratory disease syndrome at a later time when specific antibodies are appearing. In this scenario, proinflammatory cytokines such as IL-6 may play a role in sustaining postvirus inflammation and increasing immunopathogenicity in SARS if the patients are treated longer term with corticosteroids (39).
Acknowledgments
We thank Marli Azevedo for technical assistance with the ELISPOT assay. For pig management, we thank Juliette Hanson, Todd Root, Greg Myers, and Peggy Lewis. We are grateful for the technical assistance of Hong Liu, Manlin Lou, Hong Tang, Steve Corman, Nick Brooks, and Aaron Higgins.
This work was supported by grant R01 AI060739 from the NIAID, NIH, to L. J. Saif. Salaries and research support were provided by state and federal funds provided to the Ohio Agricultural Research and Development Center, The Ohio State University.
Footnotes
Published ahead of print on 20 February 2008.
REFERENCES
- 1.Ahn, K., C. Chae, and C. H. Kweon. 1997. Immunohistochemical identification of porcine respiratory coronavirus antigen in the lung of conventional pigs. Vet. Pathol. 34167-169. [DOI] [PubMed] [Google Scholar]
- 2.Amano, Y., S. W. Lee, and A. C. Allison. 1993. Inhibition by glucocorticoids of the formation of interleukin-1 alpha, interleukin-1 beta, and interleukin-6: mediation by decreased mRNA stability. Mol. Pharmacol. 43176-182. [PubMed] [Google Scholar]
- 3.Auyeung, T. W., J. S. Lee, W. K. Lai, C. H. Choi, H. K. Lee, J. S. Lee, P. C. Li, K. H. Lok, Y. Y. Ng, W. M. Wong, and Y. M. Yeung. 2005. The use of corticosteroid as treatment in SARS was associated with adverse outcomes: a retrospective cohort study. J. Infect. 5198-102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Azevedo, M. S., L. Yuan, S. Pouly, A. M. Gonzales, K. I. Jeong, T. V. Nguyen, and L. J. Saif. 2006. Cytokine responses in gnotobiotic pigs after infection with virulent or attenuated human rotavirus. J. Virol. 80372-382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Baudoux, P., L. Besnardeau, C. Carrat, P. Rottier, B. Charley, and H. Laude. 1998. Interferon alpha inducing property of coronavirus particles and pseudoparticles. Adv. Exp. Med. Biol. 440377-386. [DOI] [PubMed] [Google Scholar]
- 6.Baudoux, P., C. Carrat, L. Besnardeau, B. Charley, and H. Laude. 1998. Coronavirus pseudoparticles formed with recombinant M and E proteins induce alpha interferon synthesis by leukocytes. J. Virol. 728636-8643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Blotta, M. H., R. H. DeKruyff, and D. T. Umetsu. 1997. Corticosteroids inhibit IL-12 production in human monocytes and enhance their capacity to induce IL-4 synthesis in CD4+ lymphocytes. J. Immunol. 1585589-5595. [PubMed] [Google Scholar]
- 8.Boersma, W. J., R. J. Zwart, J. Sinkora, Z. Rehakova, K. Haverson, and A. T. Bianchi. 2001. Summary of workshop findings for porcine B-cell markers. Vet. Immunol. Immunopathol. 8063-78. [DOI] [PubMed] [Google Scholar]
- 9.Byron, K. A., G. Varigos, and A. Wootton. 1992. Hydrocortisone inhibition of human interleukin-4. Immunology 77624-626. [PMC free article] [PubMed] [Google Scholar]
- 10.Callebaut, P., I. Correa, M. Pensaert, G. Jimenez, and L. Enjuanes. 1988. Antigenic differentiation between transmissible gastroenteritis virus of swine and a related porcine respiratory coronavirus. J. Gen. Virol. 691725-1730. [DOI] [PubMed] [Google Scholar]
- 11.Cameron, M. J., L. Ran, L. Xu, A. Danesh, J. F. Bermejo-Martin, C. M. Cameron, M. P. Muller, W. L. Gold, S. E. Richardson, S. M. Poutanen, B. M. Willey, M. E. DeVries, Y. Fang, C. Seneviratne, S. E. Bosinger, D. Persad, P. Wilkinson, L. D. Greller, R. Somogyi, A. Humar, S. Keshavjee, M. Louie, M. B. Loeb, J. Brunton, A. J. McGeer, Canadian SARS Research Network, and D. J. Kelvin. 2007. Interferon-mediated immunopathological events are associated with atypical innate and adaptive immune responses in patients with severe acute respiratory syndrome. J. Virol. 818692-8706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chan, H. L., S. K. Tsui, and J. J. Sung. 2003. Coronavirus in severe acute respiratory syndrome (SARS). Trends Mol. Med. 9323-325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Chen, R. C., X. P. Tang, S. Y. Tan, B. L. Liang, Z. Y. Wan, J. Q. Fang, and N. Zhong. 2006. Treatment of severe acute respiratory syndrome with glucosteroids: the Guangzhou experience. Chest 1291441-1452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chien, J. Y., P. R. Hsueh, W. C. Cheng, C. J. Yu, and P. C. Yang. 2006. Temporal changes in cytokine/chemokine profiles and pulmonary involvement in severe acute respiratory syndrome. Respirology 11715-722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cox, E., J. Hooyberghs, and M. B. Pensaert. 1990. Sites of replication of a porcine respiratory coronavirus related to transmissible gastroenteritis virus. Res. Vet. Sci. 48165-169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cox, E., M. Pensaert, J. Hooyberghs, and K. Van Deun. 1990. Sites of replication of a porcine respiratory coronavirus in 5-week-old pigs with or without maternal antibodies. Adv. Exp. Med. Biol. 276429-433. [DOI] [PubMed] [Google Scholar]
- 17.Cyranoski, D. 2003. Critics slam treatment for SARS as ineffective and perhaps dangerous. Nature 4234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.DeKruyff, R. H., Y. Fang, and D. T. Umetsu. 1998. Corticosteroids enhance the capacity of macrophages to induce Th2 cytokine synthesis in CD4+ lymphocytes by inhibiting IL-12 production. J. Immunol. 1602231-2237. [PubMed] [Google Scholar]
- 19.Drosten, C., S. Gunther, W. Preiser, S. van der Werf, H. R. Brodt, S. Becker, H. Rabenau, M. Panning, L. Kolesnikova, R. A. Fouchier, A. Berger, A. M. Burguiere, J. Cinatl, M. Eickmann, N. Escriou, K. Grywna, S. Kramme, J. C. Manuguerra, S. Muller, V. Rickerts, M. Sturmer, S. Vieth, H. D. Klenk, A. D. Osterhaus, H. Schmitz, and H. W. Doerr. 2003. Identification of a novel coronavirus in patients with severe acute respiratory syndrome. N. Engl. J. Med. 3481967-1976. [DOI] [PubMed] [Google Scholar]
- 20.Gessani, S., S. McCandless, and C. Baglioni. 1988. The glucocorticoid dexamethasone inhibits synthesis of interferon by decreasing the level of its mRNA. J. Biol. Chem. 2637454-7457. [PubMed] [Google Scholar]
- 21.Hampton, T. 2005. Bats may be SARS reservoir. JAMA 2942291. [DOI] [PubMed] [Google Scholar]
- 22.Hill, H., J. Biwer, R. Woods, and R. Wesley. 1990. Porcine respiratory coronavirus isolated from two US swine herds. Proc. Am. Assoc. of Swine Practitioners 21333-335. [Google Scholar]
- 23.Jabrane, A., C. Girard, and Y. Elazhary. 1994. Pathogenicity of porcine respiratory coronavirus isolated in Quebec. Can. Vet. J. 3586-92. [PMC free article] [PubMed] [Google Scholar]
- 24.Jiang, Y., J. Xu, C. Zhou, Z. Wu, S. Zhong, J. Liu, W. Luo, T. Chen, Q. Qin, and P. Deng. 2005. Characterization of cytokine/chemokine profiles of severe acute respiratory syndrome. Am. J. Respir. Crit. Care Med. 171850-857. [DOI] [PubMed] [Google Scholar]
- 25.Jung, K., K. P. Alekseev, X. Zhang, D.-S. Cheon, A. N. Vlasova, and L. J. Saif. 2007. Altered pathogenesis of porcine respiratory coronavirus in pigs due to immunosuppressive effects of dexamethasone: implications for corticosteroid use in treatment of severe acute respiratory syndrome coronavirus. J. Virol. 8113681-13693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kunicka, J. E., M. A. Talle, G. H. Denhardt, M. Brown, L. A. Prince, and G. Goldstein. 1993. Immunosuppression by glucocorticoids: inhibition of production of multiple lymphokines by in vivo administration of dexamethasone. Cell. Immunol. 14939-49. [DOI] [PubMed] [Google Scholar]
- 27.Lau, A. C., L. K. So, F. P. Miu, R. W. Yung, E. Poon, T. M. Cheung, and L. Y. Yam. 2004. Outcome of coronavirus-associated severe acute respiratory syndrome using a standard treatment protocol. Respirology 9173-183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Levy, D. E., I. Marie, and A. Prakash. 2003. Ringing the interferon alarm: differential regulation of gene expression at the interface between innate and adaptive immunity. Curr. Opin. Immunol. 1552-58. [DOI] [PubMed] [Google Scholar]
- 29.Li, W., Z. Shi, M. Yu, W. Ren, C. Smith, J. H. Epstein, H. Wang, G. Crameri, Z. Hu, H. Zhang, J. Zhang, J. McEachern, H. Field, P. Daszak, B. T. Eaton, S. Zhang, and L. F. Wang. 2005. Bats are natural reservoirs of SARS-like coronaviruses. Science 310676-679. [DOI] [PubMed] [Google Scholar]
- 30.Li, X. W., R. M. Jiang, and J. Z. Guo. 2003. Glucocorticoid in the treatment of severe acute respiratory syndrome patients: a preliminary report. Zhonghua Neike Zazhi 42378-381. (In Chinese.) [PubMed] [Google Scholar]
- 31.Meng, Q. H., P. L. Dong, Y. B. Guo, K. Zhang, L. C. Liang, W. Hou, and J. L. Dong. 2003. Use of glucocorticoid in treatment of severe acute respiratory syndrome cases. Zhonghua Yufang Yixue Zazhi 37233-235. (In Chinese.) [PubMed] [Google Scholar]
- 32.Min, D. I., and A. P. Monaco. 1991. Complications associated with immunosuppressive therapy and their management. Pharmacotherapy 11119S-125S. [PubMed] [Google Scholar]
- 33.Mogensen, T. H., and S. R. Paludan. 2001. Molecular pathways in virus-induced cytokine production. Microbiol. Mol. Biol Rev. 65131-150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Nicholls, J. M., L. L. Poon, K. C. Lee, W. F. Ng, S. T. Lai, C. Y. Leung, C. M. Chu, P. K. Hui, K. L. Mak, W. Lim, K. W. Yan, K. H. Chan, N. C. Tsang, Y. Guan, K. Y. Yuen, and J. S. Peiris. 2003. Lung pathology of fatal severe acute respiratory syndrome. Lancet 3611773-1778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Nie, Q. H., X. D. Luo, and W. L. Hui. 2003. Advances in clinical diagnosis and treatment of severe acute respiratory syndrome. World J. Gastroenterol. 91139-1143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Normile, D. 2005. Virology. Researchers tie deadly SARS virus to bats. Science 3092154-2155. [DOI] [PubMed] [Google Scholar]
- 37.Paludan, S. R. 1998. Interleukin-4 and interferon-gamma: the quintessence of a mutual antagonistic relationship. Scand. J. Immunol. 48459-468. [DOI] [PubMed] [Google Scholar]
- 38.Peiris, J. S., C. M. Chu, V. C. Cheng, K. S. Chan, I. F. Hung, L. L. Poon, K. I. Law, B. S. Tang, T. Y. Hon, C. S. Chan, K. H. Chan, J. S. Ng, B. J. Zheng, W. L. Ng, R. W. Lai, Y. Guan, and K. Y. Yuen. 2003. Clinical progression and viral load in a community outbreak of coronavirus-associated SARS pneumonia: a prospective study. Lancet 3611767-1772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Perlman, S., and A. A. Dandekar. 2005. Immunopathogenesis of coronavirus infections: implications for SARS. Nat. Rev. Immunol. 5917-927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Prakash, A., E. Smith, C. K. Lee, and D. E. Levy. 2005. Tissue-specific positive feedback requirements for production of type I interferon following virus infection. J. Biol. Chem. 28018651-18657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Robinson, J. P., Z. Darzynkiewicz, P. N. Dean, A. R. Hibbs, A. Orfao, P. S. Rabinovitch, and L. L. Wheeless. 1997. Current protocols in cytometry, vol I, suppl. 24. John Wiley & Sons, Inc., New York, NY.
- 42.Rota, P. A., M. S. Oberste, S. S. Monroe, W. A. Nix, R. Campagnoli, J. P. Icenogle, S. Penaranda, B. Bankamp, K. Maher, M. H. Chen, S. Tong, A. Tamin, L. Lowe, M. Frace, J. L. DeRisi, Q. Chen, D. Wang, D. D. Erdman, T. C. Peret, C. Burns, T. G. Ksiazek, P. E. Rollin, A. Sanchez, S. Liffick, B. Holloway, J. Limor, K. McCaustland, M. Olsen-Rasmussen, R. Fouchier, S. Gunther, A. D. Osterhaus, C. Drosten, M. A. Pallansch, L. J. Anderson, and W. J. Bellini. 2003. Characterization of a novel coronavirus associated with severe acute respiratory syndrome. Science 3001394-1399. [DOI] [PubMed] [Google Scholar]
- 43.Saif, L. J. 2004. Animal coronaviruses: what can they teach us about the severe acute respiratory syndrome? Rev. Sci. Technol. 23643-660. [DOI] [PubMed] [Google Scholar]
- 44.Sanchez, C. M., F. Gebauer, C. Sune, A. Mendez, J. Dopazo, and L. Enjuanes. 1992. Genetic evolution and tropism of transmissible gastroenteritis coronaviruses. Virology 19092-105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Simkins, R. A., P. A. Weilnau, J. Bias, and L. J. Saif. 1992. Antigenic variation among transmissible gastroenteritis virus (TGEV) and porcine respiratory coronavirus strains detected with monoclonal antibodies to the S protein of TGEV. Am. J. Vet. Res. 531253-1258. [PubMed] [Google Scholar]
- 46.So, L. K., A. C. Lau, L. Y. Yam, T. M. Cheung, E. Poon, R. W. Yung, and K. Y. Yuen. 2003. Development of a standard treatment protocol for severe acute respiratory syndrome. Lancet 3611615-1617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Talar-Williams, C., and M. C. Sneller. 1994. Complications of corticosteroid therapy. Eur. Arch. Oto-Rhino-Laryngol. 251131-136. [DOI] [PubMed] [Google Scholar]
- 48.Thacker, E., A. Summerfield, K. McCullough, A. Ezquerra, J. Dominguez, F. Alonso, J. Lunney, J. Sinkora, and K. Haverson. 2001. Summary of workshop findings for porcine myelomonocytic markers. Vet. Immunol. Immunopathol. 8093-109. [DOI] [PubMed] [Google Scholar]
- 49.VanCott, J. L., T. A. Brim, R. A. Simkins, and L. J. Saif. 1993. Isotype-specific antibody-secreting cells to transmissible gastroenteritis virus and porcine respiratory coronavirus in gut- and bronchus-associated lymphoid tissues of suckling pigs. J. Immunol. 1503990-4000. [PubMed] [Google Scholar]
- 50.Van Gucht, S., K. Atanasova, F. Barbe, E. Cox, M. Pensaert, and K. Van Reeth. 2006. Effect of porcine respiratory coronavirus infection on lipopolysaccharide recognition proteins and haptoglobin levels in the lungs. Microbes Infect. 81492-1501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Van Gucht, S., K. Van Reeth, and M. Pensaert. 2003. Interaction between porcine reproductive-respiratory syndrome virus and bacterial endotoxin in the lungs of pigs: potentiation of cytokine production and respiratory disease. J. Clin. Microbiol. 41960-966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Van Reeth, K., H. Nauwynck, and M. Pensaert. 1998. Bronchoalveolar interferon-alpha, tumor necrosis factor-alpha, interleukin-1, and inflammation during acute influenza in pigs: a possible model for humans? J. Infect. Dis. 1771076-1079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Van Reeth, K., H. Nauwynck, and M. Pensaert. 1996. Dual infections of feeder pigs with porcine reproductive and respiratory syndrome virus followed by porcine respiratory coronavirus or swine influenza virus: a clinical and virological study. Vet. Microbiol. 48325-335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Van Reeth, K., H. Nauwynck, and M. Pensaert. 2000. A potential role for tumour necrosis factor-alpha in synergy between porcine respiratory coronavirus and bacterial lipopolysaccharide in the induction of respiratory disease in pigs. J. Med. Microbiol. 49613-620. [DOI] [PubMed] [Google Scholar]
- 55.Van Reeth, K., and M. B. Pensaert. 1995. Production of interferon-alpha, tumor necrosis factor-alpha and interleukin-1 in the lungs of pigs infected with the porcine respiratory coronavirus, p.197-201. In Proceedings of the 3rd Congress of the European Society for Veterinary Virology, Interlaken, Switzerland.
- 56.Van Reeth, K., S. Van Gucht, and M. Pensaert. 2002. Correlations between lung proinflammatory cytokine levels, virus replication, and disease after swine influenza virus challenge of vaccination-immune pigs. Viral Immunol. 15583-594. [DOI] [PubMed] [Google Scholar]
- 57.Van Reeth, K., S. Van Gucht, and M. Pensaert. 2002. In vivo studies on cytokine involvement during acute viral respiratory disease of swine: troublesome but rewarding. Vet. Immunol. Immunopathol. 87161-168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Wang, H., Y. Ding, X. Li, L. Yang, W. Zhang, and W. Kang. 2003. Fatal aspergillosis in a patient with SARS who was treated with corticosteroids. N. Engl. J. Med. 349507-508. [DOI] [PubMed] [Google Scholar]
- 59.Wang, W., L. Ye, L. Ye, B. Li, B. Gao, Y. Zeng, L. Kong, X. Fang, H. Zheng, Z. Wu, and Y. She. 2007. Up-regulation of IL-6 and TNF-alpha induced by SARS-coronavirus spike protein in murine macrophages via NF-kappaB pathway. Virus Res. 1281-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Ward, W. E., S. M. Donovan, and S. A. Atkinson. 1998. Dexamethasone-induced abnormalities in growth and bone metabolism in piglets are partially attenuated by growth hormone with no synergistic effect of insulin-like growth factor-I. Pediatr. Res. 44215-221. [DOI] [PubMed] [Google Scholar]
- 61.Weiler, H. A., Z. Wang, and S. A. Atkinson. 1995. Dexamethasone treatment impairs calcium regulation and reduces bone mineralization in infant pigs. Am. J. Clin. Nutr. 61805-811. [DOI] [PubMed] [Google Scholar]
- 62.Welch, S. K., and L. J. Saif. 1988. Monoclonal antibodies to a virulent strain of transmissible gastroenteritis virus: comparison of reactivity with virulent and attenuated virus. Arch. Virol. 101221-235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wesley, R. D., R. D. Woods, and A. K. Cheung. 1991. Genetic analysis of porcine respiratory coronavirus, an attenuated variant of transmissible gastroenteritis virus. J. Virol. 653369-3373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Wong, C. K., C. W. Lam, A. K. Wu, W. K. Ip, N. L. Lee, I. H. Chan, L. C. Lit, D. S. Hui, M. H. Chan, S. S. Chung, and J. J. Sung. 2004. Plasma inflammatory cytokines and chemokines in severe acute respiratory syndrome. Clin. Exp. Immunol. 13695-103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Xu, D., Z. Zhang, L. Jin, F. Chu, Y. Mao, H. Wang, M. Liu, M. Wang, L. Zhang, G. F. Gao, and F. S. Wang. 2005. Persistent shedding of viable SARS-CoV in urine and stool of SARS patients during the convalescent phase. Eur. J. Clin. Microbiol. Infect. Dis. 24165-171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Yuan, L., L. A. Ward, B. I. Rosen, T. L. To, and L. J. Saif. 1996. Systematic and intestinal antibody-secreting cell responses and correlates of protective immunity to human rotavirus in a gnotobiotic pig model of disease. J. Virol. 703075-3083. [DOI] [PMC free article] [PubMed] [Google Scholar]