Abstract
The phenylurea herbicide diuron [N-(3,4-dichlorophenyl)-N,N-dimethylurea] is widely used in a broad range of herbicide formulations, and consequently, it is frequently detected as a major water contaminant in areas where there is extensive use. We constructed a linuron [N-(3,4-dichlorophenyl)-N-methoxy-N-methylurea]- and diuron-mineralizing two-member consortium by combining the cooperative degradation capacities of the diuron-degrading organism Arthrobacter globiformis strain D47 and the linuron-mineralizing organism Variovorax sp. strain SRS16. Neither of the strains mineralized diuron alone in a mineral medium, but combined, the two strains mineralized 31 to 62% of the added [ring-U-14C]diuron to 14CO2, depending on the initial diuron concentration and the cultivation conditions. The constructed consortium was used to initiate the degradation and mineralization of diuron in soil without natural attenuation potential. This approach led to the unexpected finding that Variovorax sp. strain SRS16 was able to mineralize diuron in a pure culture when it was supplemented with appropriate growth substrates, making this strain the first known bacterium capable of mineralizing diuron and representatives of both the N,N-dimethyl- and N-methoxy-N-methyl-substituted phenylurea herbicides. The ability of the coculture to mineralize microgram-per-liter levels of diuron was compared to the ability of strain SRS16 alone, which revealed the greater extent of mineralization by the two-member consortium (31 to 33% of the added [ring-U-14C]diuron was mineralized to 14CO2 when 15.5 to 38.9 μg liter−1 diuron was used). These results suggest that the consortium consisting of strains SRS16 and D47 could be a promising candidate for remediation of soil and water contaminated with diuron and linuron and their shared metabolite 3,4-dichloroaniline.
The phenylurea herbicides are an important group of pesticides that are used predominantly in either pre- or postemergence treatment of cotton, fruit, cereal, or other agricultural crops. One of the most widely used members of this group, diuron [N-(3,4-dichlorophenyl)-N,N-dimethylurea], is also used in many parts of the world as a broad-spectrum herbicide in urban and industrial areas, as an active ingredient in antifouling boat paints, and in algaecide formulations used in fountains and aquaculture. As a consequence, diuron has been detected in lakes, rivers, and groundwater (18, 23, 30, 44), in marine waters and sediments (43), and in rain collected at urban and rural sites (37). Diuron is generally found at trace concentrations (nanograms per liter to micrograms per liter), but higher concentrations (milligrams per liter) have been found in, for example, drainage water from agricultural soils (47, 51) and in rainfall runoff from fruit orchards (34). Besides diuron itself, the main metabolites 3,4-dichloroaniline (3,4-DCA), N-(3,4-dichlorophenyl)urea (DCPU), and N-(3,4-dichlorophenyl)-N-methylurea (DCPMU) have been detected in natural waters (9, 23, 30, 43, 44). These metabolites have been reported to be much more harmful to nontarget organisms than diuron itself (21, 34, 46). Consequently, diuron has been included on the European Commission's list of priority substances for European freshwater resources (19) and on the U.S. Environmental Protection Agency's Second Drinking Water Contaminant Candidate List (50).
Diuron has the unfortunate combination of being both mobile and slowly degraded in the environment. The low tendency of diuron to sorb to soil, combined with its relatively high water solubility, is the likely explanation for its potential for leaching into underlying or nearby water observed in several recent field experiments (22, 24). Biodegradation has been described as the primary mechanism for diuron dissipation in soils and waters (34, 39), and half-lives ranging from weeks to years have been measured and extrapolated from the results of laboratory experiments with different soils and waters (7, 13, 21, 32-34). Different metabolites resulting from partial degradation of diuron have been identified, and the initial degradation steps appear to follow the general pathways for N,N-dimethyl-substituted phenylureas involving either an initial N demethylation to DCPMU or a hydrolysis to 3,4-DCA (39). Another major metabolite that has been measured in field and laboratory experiments is DCPU (22, 24), which is produced from N demethylation of DCPMU (39). Mineralization of diuron in environmental samples has rarely been studied, and in one of the few cases where [ring-14C]diuron has been used to monitor degradation, mineralization half-lives of 444 days for previously untreated soil and 77 days for soil previously exposed to diuron were reported (13). In a similar study using [carbonyl-14C]diuron the workers determined extrapolated half-lives of up to 4,000 days using data obtained from degradation experiments with agricultural soil (33). Enhanced degradation has been observed in studies of agricultural soil samples previously exposed to diuron, indicating that microbial adaptation to diuron degradation is possible in soil (13, 35, 36).
Several diuron-degrading bacteria have been isolated from different agricultural soils (10, 13, 16, 42, 53) and river waters (1). Furthermore, it has been shown that different fungi, either obtained from various culture collections or freshly isolated from agricultural soils, are capable of degrading diuron (8, 20, 29, 52). However, neither the bacterial nor fungal diuron-degrading strains have been shown conclusively to mineralize the ring structure of diuron, and with the majority of the strains potentially harmful metabolites, such as 3,4-DCA, accumulate. The most efficient and well-characterized degraders of diuron described so far are two closely related Arthrobacter strains, strains D47 (48, 49) and N2 (46, 53), which were isolated from British and French agricultural soils previously treated with phenylurea herbicides. These strains are closely related to Arthrobacter globiformis, and they degrade broad spectra of phenylurea herbicides, including both N,N-dimethyl- and N-methoxy-N-methyl-substituted phenylureas, directly to the corresponding aniline metabolites without any further degradation (10, 45). 3,4-DCA is a common metabolite of linuron [N-(3,4-dichlorophenyl)-N-methoxy-N-methylurea] and diuron, and exchange of this metabolite between different members of linuron-mineralizing bacterial consortia has previously been suggested (6, 11, 17). 3,4-DCA has also been described as the only intermediate during the mineralization of linuron by Variovorax sp. strain SRS16 (40) (Fig. 1). Neither strain SRS16 nor any of the linuron-mineralizing consortia characterized so far have been shown to degrade diuron or other N,N-dimethyl-substituted phenylurea herbicides.
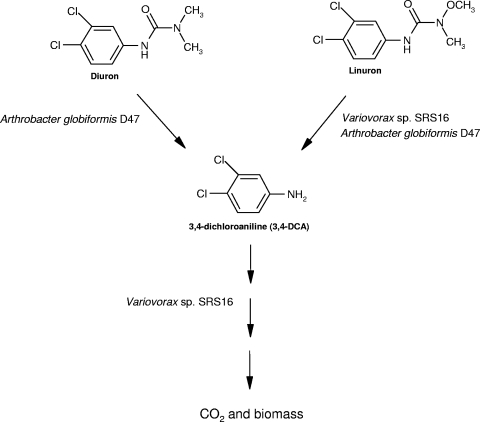
FIG. 1.
Constructed pathway for mineralization of diuron by the two-member consortium combining the cooperative degradation capacities of the diuron-degrading organism A. globiformis strain D47 and the linuron-mineralizing organism Variovorax sp. strain SRS16.
In the current study, we constructed a two-member linuron- and diuron-mineralizing consortium by combining the cooperative degradation capacities of the diuron-degrading organism A. globiformis strain D47 (10, 48) and the linuron-mineralizing organism Variovorax sp. strain SRS16 (40), as shown in Fig. 1. The objective of this approach was to obtain diuron-mineralizing bacterial cultures for use in bioremediation processes aimed at cleaning contaminated soils and water resources. The degradative capacity of the consortium was compared to that of each of the strains individually with regard to the ability to degrade and mineralize diuron at ecologically relevant concentrations in both liquid media and soil.
MATERIALS AND METHODS
Diuron and metabolites.
Analytical-grade diuron (CAS RN 330-54-01; purity, 99%; water solubility at 20°C, 42 mg liter−1), linuron (CAS RN 330-55-2; purity, 99.8%; water solubility at 24°C, 64 mg liter−1), 3,4-DCA (CAS RN 95-76-1; purity, 99.0%), DCPU (purity, 98.0%), and DCPMU (purity, 99.5%) were purchased from Dr. Ehrenstorfer GmbH (Augsburg, Germany). [phenyl-U-14C]linuron (16.24 mCi mmol−1; radiochemical purity, >98%) and [phenyl-U-14C]diuron (19.57 mCi mmol−1; radiochemical purity, >95%) were purchased from International Isotope (Munich, Germany). We identified the radiochemical impurities in the [ring-U-14C]diuron stock as 1% DCPMU and 4% DCPU by the thin-layer chromatography method described below. Radiochemical purity greater than 98% was verified for [ring-U-14C]linuron, but the nature of the 14C impurities was not clarified in this study. ring-U-14C-labeled 3,4-DCA was biosynthesized from [ring-U-14C]diuron by using the diuron-degrading organism Arthrobacter sp. strain D47 (48), and then the 14C-labeled metabolite was purified by extraction in analytical-grade dichloromethane (Sigma-Aldrich, Germany).
Bacterial strains and cultivation media.
The diuron-degrading organism A. globiformis D47 was kindly provided by A. Walker (48, 49). The linuron-mineralizing organism Variovorax sp. strain SRS16 was isolated and characterized in our laboratory previously (40), and it has been deposited in the Pasteur Collection under accession number CIP108393. Both strains were stored in 50% glycerol stock solutions at −80°C. Before each experiment the strains were thawed, and then D47 was grown in Luria-Bertani medium (LB) (Difco Detroit, MI) supplemented with 25 mg liter−1 diuron and strain SRS16 was grown in a mineral salt solution with 1 g liter−1 succinate as the carbon source and 25 to 50 mg liter−1 linuron as the sole nitrogen source, as described by Sørensen et al. (40). The strains were harvested in the late exponential growth phase and washed twice in a sterile phosphate buffer before initiation of the experiments. The initial cell densities of strains SRS16 and D47 in the degradation experiments were approximately 106 cells ml−1. Different variations of the mineral medium (MS) (38) were prepared. MSN contained added (NH4)2SO4 (238 mg liter−1) and KNO3 (50 mg liter−1) as nitrogen sources, and MSC contained succinate (0.5 g liter−1) and glucose (0.5 g liter−1) as carbon sources. MSCN contained a combination of the carbon and nitrogen sources in MSN and MSC. Complex growth medium was prepared by adding 1 g liter−1 LB (MS-LB), 1 g liter−1 Casamino Acids (MS-CAS) (Difco Laboratories), or 1 mg liter−1 yeast extract (Difco Laboratories) to MS. All liquid media had a final pH of 6.9 to 7.0.
Degradation and mineralization experiments.
For the degradation experiments with liquid media, diuron was added from stock solutions in acetone or acetonitrile to sterilized 100-ml glass flasks. The solvent was allowed to evaporate, and the media and bacterial strains were added to obtain a final volume of 25 ml. Quantification of the degradation and the amounts of metabolites in the liquid media was performed using a Hewlett-Packard series 1050 high-performance liquid chromatography (HPLC) system equipped with a UV detector (Phenomenex, Chire, United Kingdom) as described by Juhler et al. (27). Prior to the HPLC analysis, 750-μl subsamples of the media were filtered using a 17-mm 0.2-μm polytetafluoroethylene membrane syringe filter (Titan Filtration Systems, Sun SRI, Wilmington, NC), and the last 250 μl was collected for analysis. In the mineralization experiments, known amounts of 14C-labeled tracers (50,000 to 100,000 dpm) were added along with the unlabeled stock solutions, and each flask was equipped with a 10-ml glass tube containing 2.0 ml of 0.5 M NaOH to trap the 14CO2 resulting from mineralization. The NaOH was replaced at regular intervals in a laminar flow bench and mixed with 10 ml of Wallac OptiPhase HiSafe 3 scintillation cocktail (Turku, Finland), and the radioactivity was counted for 10 min with a Wallac 1409 liquid scintillation counter.
Soil microcosm experiment.
Subsoil obtained from the C horizon 200 to 230 cm below the surface at the Fladerne Creek agricultural site (31) was used for a mineralization experiment with diuron. Ten grams (wet weight) of soil was transferred into sterile 100-ml flasks, and an acetone stock solution containing 14C-labeled and unlabeled diuron was added to portions of the soil samples to obtain a final concentration of 2 mg kg−1 and a radioactivity of approximately 100,000 dpm per flask. The flasks were left open in a laminar flow bench for 1 h to allow the solvent to evaporate before the soil was mixed. The natural moisture of the soil samples was reestablished during addition of the strains suspended in sterile tap water. The final densities of strains D47 and SRS16 were approximately 1 × 108 cells g soil−1. The mineralization of [14C]diuron to 14CO2 was monitored by inserting a base trap as described above. At the end of the experiment, the 14C residue remaining in the soil was extracted by adding 10 ml methanol to each flask and shaking the flasks for 24 h. Samples (1 ml) of the methanol extracts were then centrifuged (1,200 × g, 10 min), and 20 μl of each supernatant was spotted on a Silica Gel 60 thin-layer chromatography plate (Merck, Darmstadt, Germany), eluted with a mixture of dichloromethane and methanol (95:5, vol/vol) in a CAMAG horizontal developing chamber (CAMAG, Berlin, Germany), and analyzed by autoradiography with a Cyclone scanner (Packard Instrument Company, Meriden, CT). The chromatograms were analyzed using the OptiQuant image analysis software (Packard Instrument Company) integrating the different bands, and the Rf values of diuron and its metabolites were as follows: DCPU, 0.21; DCPMU, 0.28; diuron, 0.36; and 3,4-DCA, 0.60. The 14C associated with the methanol-extractable humic fraction had an Rf of <0.01.
RESULTS
Degradation and mineralization of diuron by strains D47 and SRS16.
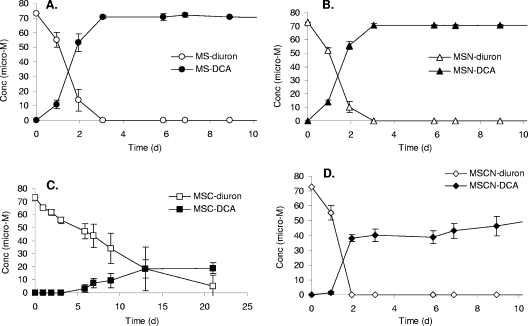
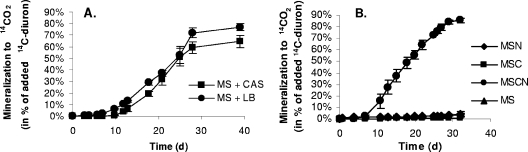
The degradation of diuron by A. globiformis D47 and the presence of the metabolites DCPMU, DCPU, and 3,4-DCA in different media were determined by HPLC analysis. Degradation of diuron by strain D47 to equivalent amounts of 3,4-DCA was measured in a mineral medium with diuron as the sole source of nitrogen and carbon (MS) (Fig. 2A) and in a medium with diuron as the sole source of carbon (MSN) (Fig. 2B). A reduced degradation rate and a lower concentration of 3,4-DCA were observed when diuron was the sole nitrogen source in a medium supplemented with succinate and glucose as carbon sources (MSC) (Fig. 2C). Rapid degradation of diuron was observed in a medium based on MSC supplemented with a mixture of nitrate and ammonium as nitrogen sources (MSCN), and lower levels of 3,4-DCA were again detected following diuron degradation (Fig. 2D). Due to the low level of 3,4-DCA produced compared to the extent of diuron degradation in MSC and MSCN (Fig. 2C and 2D), we examined the potential for mineralization of [14C]diuron to 14CO2 by strain D47 in all four mineral media, and no 14CO2 was detected in any of the preparations during 20 days of incubation (data not shown).
FIG. 2.
Degradation of diuron (open symbols) and production of 3,4-DCA (filled symbols) by A. globiformis strain D47 in MS (A), MSN (B), MSC (C), and MSCN (D). See Materials and Methods for details concerning the compositions of the four culture media. The initial cell density was 106 cells ml−1. The data are means (n = 3). The error bars indicate the standard deviations.
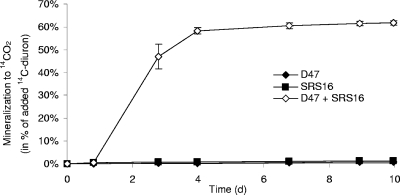
Neither strain D47 nor strain SRS16 was capable of performing extensive mineralization of diuron in pure culture in MS (Fig. 3), and only SRS16 mineralized 14C-labeled 3,4-DCA to 14CO2 (data not shown). Combining strains D47 and SRS16 resulted in rapid mineralization of diuron, and 61.8% ± 1.0% of the added [14C]diuron was metabolized to 14CO2 within 5 days (Fig. 3).
FIG. 3.
Mineralization of 14C-labeled diuron (14C-diuron) to 14CO2 by A. globiformis strain D47 (⧫) or Variovorax sp. strain SRS16 (▪) individually or by these two strains combined in a coculture (⋄) in liquid MS. The initial diuron concentration was 10 mg liter−1, and the initial cell density was approximately 106 cells ml−1. The data are means (n = 3). The error bars indicate the standard deviations, and some of the error bars are smaller than the symbols.
Degradation and mineralization of diuron in soil inoculated with strains D47 and SRS16.
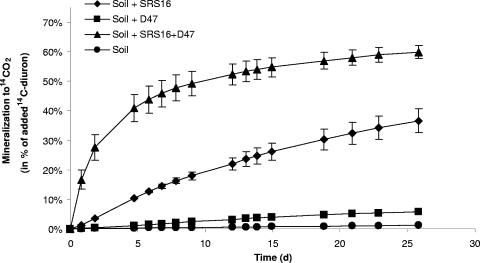
The effects on the degradation of diuron in soil (2 mg kg−1) after inoculation of strains D47 and SRS16 individually or in a coculture were determined using a microcosm setup that was incubated for 26 days (Fig. 4). At the end of the experiment, the distribution of the 14C initially added in the form of [14C]diuron was estimated (Table 1). Without any inoculum, 78.8% ± 0.9% of the added diuron remained in the soil, and only 1.2% of the added [14C]diuron was mineralized to 14CO2 during the experiment (Fig. 4 and Table 1). The diuron metabolites DCPMU and DCPU were both detected in soil extracts, but no 3,4-DCA was found in soil that was not inoculated (Table 1). Inoculation with strain D47 resulted in greater diuron degradation (16.6% ± 3.2% of the initial amount remained at the end of the experiment) and greater diuron mineralization (5.9% ± 0.5% of the added [14C]diuron was degraded to 14CO2 in the experiment). The metabolite 3,4-DCA was detected only in the samples inoculated with strain D47, and the amounts were equivalent to 3.9% ± 1.5% of the initially added 14C from [14C]diuron. The amount of 14C associated with the humic fraction and the amount constituting the nonextractable 14C residues were elevated following D47 inoculation compared to the other treatments (Table 1). As expected from the liquid culture experiments, inoculation with the coculture resulted in rapid diuron mineralization, and 59.9% ± 2.3% of the added [14C]diuron was metabolized to 14CO2 during the experiment (Fig. 4). Only 1.1% ± 0.6% of the added diuron and minor amounts of DCPMU and DCPU (0.2% ± 0.1% and 0.6% ± 0.2% of the added 14C) remained in the soil inoculated with the coculture (Table 1). Unexpectedly, inoculation with strain SRS16 alone resulted in increased diuron degradation in the soil; 23.1% ± 5.5% of the initial amount of diuron remained after 26 days, and 36.6% ± 4.1% of the added [14C]diuron was mineralized to 14CO2. The degradative activity of SRS16 with diuron was also shown by a comparison of the level of diuron remaining in soil inoculated with D47, in which 16.6% ± 3.2% of the initial amount was detected, with the level in soil treated with both D47 and SRS16, in which only 1.1% ± 0.6% of the initial amount of diuron was still present (Table 1). Based on these results, we studied the ability of Variovorax sp. strain SRS16 to mineralize diuron under different culture conditions in more detail.
FIG. 4.
Mineralization of 14C-labeled diuron (14C-diuron) in soil amended with diuron (2 mg kg−1) and inoculated with either A. globiformis strain D47 (▪), Variovorax sp. strain SRS16 (⧫), or both strains (▴). Natural soil was used as an uninoculated control (•). Final densities of approximately 108 cells g soil−1 were used for the strains. The data are means (n = 3). The error bars indicate the standard deviations, and some of the error bars are smaller than the symbols.
TABLE 1.
Distribution of 14C residues from [14C]diuron in agricultural soil inoculated with Variovorax sp. strain SRS16 or A. globiformis D47 or with an SRS16-D47 coculture as estimated by thin-layer chromatography and autoradiographic quantification, including detection of the demethylated diuron metabolite DCPMU, the didemethylated metabolite DCPU, and 3,4-DCA
| Prepn |
14C recovery (%)a
|
||||||
|---|---|---|---|---|---|---|---|
| [14C]diuron | [14C]DCPMU | [14C]DCPU | [14C]3,4-DCA | 14CO2 | 14C-labeled humic fraction | Nonextractable 14C | |
| Soil + SRS16 | 23.1 (5.5) | 1.8 (0.5) | 1.5 (0.1) | 0.0 | 36.6 (4.1) | 5.4 (0.4) | 31.5 (1.5) |
| Soil + D47 | 16.6 (3.2) | 1.6 (0.1) | 1.7 (0.3) | 3.9 (1.5) | 5.9 (0.5) | 14.9 (0.8) | 55.4 (3.2) |
| Soil + SRS16 + D47 | 1.1 (0.6) | 0.2 (0.1) | 0.6 (0.2) | 0.0 | 59.9 (2.3) | 4.4 (0.5) | 33.7 (0.9) |
| Soil | 78.8 (0.9) | 3.0 (0.5) | 1.7 (0.2) | 0.0 | 1.2 (0.0) | 0.0 | 15.3 (1.2) |
The values are means and standard deviations (in parentheses) for triplicate experiments expressed as percentages of the initial amount of [14C]diuron.
Enhanced diuron mineralization by strain SRS16 in pure culture.
The ability of Variovorax sp. strain SRS16 to mineralize [14C]diuron to 14CO2 was initially tested using liquid media with and without different growth substrates. Representative mineralization curves are shown in Fig. 5A and 5B. The extents of mineralization in media supplemented with complex substrates were between 64.5 and 77.1% of the added [14C]diuron metabolized to 14CO2 within 30 days (Fig. 5A). Similar effects were observed when yeast extract was added (Table 2) and when mineral media amended with 0.1, 1.0, and 10.0% LB were used (data not shown). Stimulation of mineralization of diuron by strain SRS16 was also observed in the mineral medium MSCN supplemented with defined carbon and nitrogen sources (Fig. 5B). HPLC analysis of the liquid media supplemented with complex substrates (Fig. 5A) or MSCN (Fig. 5B) showed that there was no diuron, DCPMU, DCPU, or 3,4-DCA at the end of the mineralization experiments (data not shown). Clear differences in the growth of SRS16 in MSCN with and without diuron were observed. When SRS16 was grown in MSCN without diuron, the distribution of this strain in the liquid phase was homogeneous, which allowed, for example, representative sampling of the medium for measurement of growth. In contrast, when the strain was cultivated in MSCN with diuron (10 mg liter−1), it formed bright yellow biofilms on the sides of the cultivation flasks in a zone between the liquid and gas phases, and it subsequently produced flocs that eventually precipitated to the bottom of the flasks. Although the results were not as clear as in MSCN, this phenomenon was also observed in the media supplemented with complex substrates. No extensive mineralization above the level of 14C impurities was apparent without substrate addition (Fig. 5B and Table 2). Strain SRS16 uses linuron as a source of carbon and nitrogen (40), but this substrate appeared to be the only substrate that did not support extensive mineralization of diuron (Table 2). Previous results have shown that there is enhanced mineralization of linuron by SRS16 when succinate is used as an additional carbon source and linuron is the sole nitrogen source (40). The same phenomenon was not evident for diuron mineralization, and there was no indication that diuron was used as either a carbon source or a nitrogen source by SRS16 in media to which appropriate growth substrates were not added (Fig. 5B and Table 2). Strain SRS16 mineralized 65.1% ± 6.4% of the added [14C]linuron in MS, and in the presence of 1 mg liter−1 diuron the mineralization of linuron was slightly reduced (57.8% ± 0.7% of the added [14C]linuron was mineralized to 14CO2). Diuron was mineralized to a minor extent during linuron mineralization; 3.4% ± 1.4% of the added [14C]diuron was metabolized to 14CO2. Increasing the diuron concentration to 10 mg liter −1 inhibited the mineralization of linuron significantly, and only 10.3% ± 5.3% of the [14C]linuron was metabolized to 14CO2 (Table 2). During this mineralization of linuron, only 1.3% ± 0.1% of the added [14C]diuron was measured as 14CO2, which is below the level at which the occurrence of diuron mineralization can be determined conclusively.
FIG. 5.
Mineralization of 14C-labeled diuron (14C-diuron) to 14CO2 by Variovorax sp. strain SRS16 in MS supplemented with either Casamino Acids (CAS) or LB (A) or in MS, MSN, MSC, or MSCN (B). The initial diuron concentration was 10 mg liter−1, and the initial cell density was approximately 106 cells ml−1. The data are means (n = 3). The error bars indicate the standard deviations, and some of the error bars are smaller than the symbols.
TABLE 2.
Mineralization of diuron (1 mg liter−1) or linuron (10 mg liter−1) by Variovorax sp. strain SRS16 in liquid mineral medium (MS) following incubation for 30 days
| Compound(s) added to medium | % of initial [14C]diuron or [14C]linuron mineralized to 14CO2 |
|---|---|
| Mineralization of diuron | |
| [14C]diuron | 1.4 ± 0.8 |
| [14C]diuron + linuron | 3.4 ± 1.4 |
| [14C]diuron (10 mg liter−1) | 1.3 ± 0.2 |
| [14C]diuron (10 mg liter−1) + linuron | 1.3 ± 0.1 |
| [14C]diuron + succinate (1 mg liter−1) | 0.8 ± 0.1 |
| [14C]diuron + yeast extract (1 mg liter−1) | 65.3 ± 1.4 |
| Mineralization of linuron | |
| [14C]linuron | 65.1 ± 6.4 |
| [14C]linuron + diuron | 57.8 ± 0.7 |
| [14C]linuron + diuron (10 mg liter−1) | 10.3 ± 5.3 |
Mineralization of microgram-per-liter concentrations of diuron by a pure culture of SRS16 or the D47-SRS16 coculture.
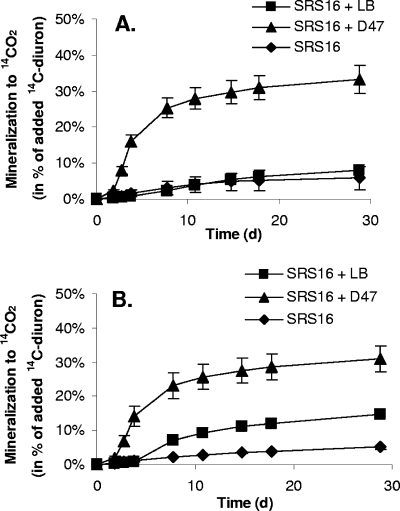
The mineralization activity of the coculture was compared to that of strain SRS16 in pure culture when preparations were challenged with microgram-per-liter concentrations of diuron (Fig. 6A and 6B). The coculture metabolized 33.3% ± 3.8% of the lowest [14C]diuron concentration tested (15.5 μg liter−1) to 14CO2 within 30 days (Fig. 6A). Increasing the diuron concentration to 38.9 μg liter−1 had no significant effect on the mineralization of diuron by the coculture, and 31.0% ± 3.7% of the diuron was metabolized to 14CO2 (Fig. 6B). Strain SRS16 mineralized only 8.0% ± 0.9% and 14.6% ± 0.9% of the added [14C]diuron to 14CO2 when it was grown in LB-supplemented medium containing 15.5 μg liter−1 diuron (Fig. 6A) and 38.9 μg liter−1 diuron (Fig. 6B), respectively. When the medium was not supplemented with LB, SRS16 mineralized 5.9% ± 3.2% of the added [14C]diuron to 14CO2 when the [14C]diuron concentration was 15.5 μg liter−1 (Fig. 6A) and 5.2% ± 0.7% when the [14C]diuron concentration was 38.9 μg liter−1 (Fig. 6B).
FIG. 6.
Mineralization of 15.5 μg liter−1 (A) or 38.9 μg liter−1 (B) 14C-labeled diuron (14C-diuron) to 14CO2 by Variovorax sp. strain SRS16 (⧫) in MS or in MS supplemented with LB (▪) or by Variovorax sp. strain SRS16 combined with A. globiformis strain D47 (▴) in MS. The initial cell density was approximately 106 cells ml−1 of each strain in pure culture or in the coculture.
DISCUSSION
Diuron and its metabolites are frequently encountered water contaminants in areas around the world where diuron is used extensively. Diuron and to an even greater extent its metabolites are suspected of having unwanted effects on nontarget organisms, and techniques for remediating contaminated water and soil are needed. With the aim of obtaining degradative microorganisms, in the last few years our laboratory has screened a range of agricultural soils from Denmark and the United Kingdom for diuron mineralization activity. In contrast to similar efforts with the related compounds isoproturon (4, 42) and linuron (40), we have not been able to pinpoint soils harboring diuron-mineralizing microorganisms. Reports of extensive mineralization of diuron in environmental samples are limited, and combined with the frequent detection of diuron and its residues in natural waters, this suggests that effective diuron-mineralizing microorganisms are rare in the environment. Based on the similarity between the degradation pathways for linuron and diuron, we initiated the current study by constructing a diuron-mineralizing consortium by combining the linuron-mineralizing organism Variovorax sp. strain SRS16 and the diuron-degrading organism A. globiformis strain D47.
The key enzymatic step in the mineralization of diuron in the present study was the hydrolysis of the amide bond, leading to the production of 3,4-DCA. Strain D47 performs this hydrolysis step very efficiently in soil (Table 1) and in different liquid media (Fig. 2A and 2B). The reason for the lack of equivalent amounts of 3,4-DCA following diuron degradation by D47 in MSC and MSCN (Fig. 2C and 2D) remains to be determined. No mineralization of the diuron ring structure by strain D47 was observed in any of the four media tested in our study. However, the possibility that there are unknown metabolites not detectable using our HPLC method that originate from incomplete degradation of 3,4-DCA in MSC and MSCN cannot be excluded. The metabolite 3,4-DCA is a problematic compound based on its greater potential toxic effect than diuron itself. Tixier et al. (45) used a Microtox test and found that the hydrolysis of diuron to 3,4-DCA increased the toxicity, expressed as the concentration having an effect on 50% of the population, from 68 ± 5 mg liter−1 for diuron to 0.48 ± 0.08 mg liter−1 for 3,4-DCA. In comparison, the corresponding values for the diuron metabolites DCPMU and DCPU were reported to be 18 ± 0.9 and 15 ± 1 mg liter−1, respectively (45). This strongly indicates that efforts aimed at remediation of contaminated soil and water should focus on the mineralization of diuron and avoid extensive accumulation of 3,4-DCA, DCPU, and DCPMU.
Our D47-SRS16 coculture mineralized diuron in a mineral medium due to the cooperative degradative capacities of A. globiformis strain D47 (which hydrolytically cleaved diuron to 3,4-DCA) and Variovorax sp. strain SRS16 (which completed the mineralization by metabolizing 14C-labeled 3,4-DCA to 14CO2). Similar cross-feeding with 3,4-DCA has been proposed by Dejonghe et al. (11) and Breugelmans et al. (6) for different multispecies linuron-mineralizing consortia enriched from various agricultural soils. 3,4-DCA-degrading bacteria without any degradation activity with diuron have been isolated from the linuron-mineralizing consortia, and one of the strains has recently been used by Bazot et al. (2) to prepare a diuron- and 3,4-DCA-degrading coculture by combining the strain with a diuron-degrading Arthrobacter sp. The Arthrobacter sp. used by Bazot et al. (2) performed a one-step degradation of diuron to 3,4-DCA similar to the degradation by A. globiformis strain D47. The ability of this constructed coculture to degrade diuron and 3,4-DCA was monitored using a sterile soil extract liquid medium (2). Although mineralization of diuron to CO2 was not measured in this study, the authors concluded that the degradation of 3,4-DCA was the limiting step in the mineralization of diuron by their coculture.
Augmentation with the D47-SRS16 coculture increased the degradation and mineralization of diuron in soil (Fig. 4), and the smallest amounts of residual diuron metabolites were observed following inoculation of the coculture (Table 1). In contrast, the natural potential for attenuation of diuron in the soil seemed to be minor, and the extent of mineralization measured was small. A. globiformis strain D47 has previously been shown to enhance the dissipation of diuron in augmented soils (10, 49), but these studies did not include detection of metabolites or mineralization. We found that besides stimulating the degradation of diuron in soil, strain D47 also increased both the mineralization of diuron and the amount of 14C in the humic and nonextractable soil fractions. The increased mineralization could have been due to the activity of an indigenous population of microorganisms capable of mineralization of 3,4-DCA, as the presence of 3,4-DCA degraders in agricultural soils without previous exposure to 3,4-DCA has been reported (12). Another possibility is that D47, under the influence of substrates naturally occurring in the soil, is capable of slowly mineralizing 3,4-DCA. The larger fractions of 14C immobilized in the soil inoculated with D47 were probably the result of the production of 3,4-DCA, followed by strong or even irreversible binding of this metabolite to organic soil components, as described previously by Bollag et al. (5) and recently by Albers et al. (C. N Albers, P. E. Hansen, G. T. Banta, and O. S. Jacobsen, unpublished results) for the same soil used in our study. Similar results have been described for the related herbicide isoproturon and the corresponding aniline metabolite, 4-isopropyl-aniline, by several authors who reported high levels of nonextractable residues following laboratory experiments with agricultural soils (3, 26).
Strains belonging to the genus Variovorax have been found to degrade a broad range of different compounds, including the pesticides atrazine (15), 2,4-dichlorophenoxyacetic acid (28), carbofuran (14), linuron, and metobromuron (6, 17), and the current results expand this list. Unexpectedly, Variovorax sp. strain SRS16 stimulated both the degradation and the mineralization of diuron when it was inoculated into soil (Table 2), and we therefore chose to study its ability to mineralize diuron in simple liquid medium. A general feature of other known phenylurea degraders is their exclusive preference for either N,N-dimethyl-substituted phenylurea herbicides, such as diuron and isoproturon, or N-methoxy-N-methyl-substituted phenylurea herbicides (for example, linuron and metobromuron) (39). The only described exceptions to this generalization so far are the phenylurea-degrading organisms Arthrobacter sp. strains N2 and D47, which are capable of hydrolyzing both N,N-dimethyl- and N-methoxy-N-methyl-substituted phenylurea herbicides to the corresponding aniline metabolites (46, 48, 49, 53). However, none of the phenylurea-mineralizing strains or bacterial consortia described so far have been shown to mineralize representatives of both groups of phenylurea herbicides. In contrast to the conclusions reached by Breugelmans et al. (6) for linuron-degrading Variovorax spp. related to strain SRS16, our Variovorax strain is not restricted to the N-methoxy-N-methyl-substituted compounds linuron and metobromuron. Some of the linuron-mineralizing Variovorax spp. described by Breugelmans et al. (6) were obtained from the same agricultural soil as strain SRS16 by employing a similar enrichment strategy, and these Variovorax strains appear to be very similar taxonomically based on a comparison of 16S rRNA gene sequences. El-Fantroussi et al. (17) also reported no apparent or only very slow degradation of the N,N-dimethyl-substituted phenylurea herbicides diuron, chlorotoluron, and isoproturon by a linuron-mineralizing consortium in which a Variovorax strain closely related to strain SRS16 was the key degrader. Both Breugelmans et al. (6) and El-Fantroussi et al. (17) tested their linuron-mineralizing Variovorax isolates or Variovorax-containing consortia in liquid media containing diuron as a carbon and nitrogen source. Therefore, we cannot completely exclude the possibility that these workers might have overlooked an ability to degrade and mineralize diuron after inoculation into soil or stimulation after addition of growth substrates similar to what we here report for Variovorax sp. strain SRS16.
The low level of diuron mineralization that occurs during linuron mineralization by SRS16 (Table 2) suggests that there is a lack of a cometabolic type of diuron degradation, where the hydrolase involved in degrading linuron to 3,4-DCA also acts on diuron. This suggests that the first enzymes of the phenylurea herbicide degradation pathways may be specific for either N, N-dimethyl- or N-methoxy-N-methyl-substituted compounds, as previously reported for other degraders (6, 17). The stimulation of the mineralization of diuron by SRS16 during growth in various complex and defined media (Fig. 5A and 5B) suggests that a broad-spectrum hydrolase active during general growth is involved. This hypothesis and the possibility of stimulating other closely related linuron-mineralizing Variovorax spp. strains to mineralize diuron are currently being studied in our laboratory.
In line with other reports on toxic effects of diuron on different bacteria (2, 46), our results indicate that the linuron mineralization activity of Variovorax sp. strain SRS16 is significantly repressed by high diuron concentrations (Table 2). Growth in flocs and biofilms is a known response of bacterial populations to toxic compounds (25), and a toxic effect of diuron on SRS16 could explain the difference in growth patterns observed in the complex growth media with and without diuron, where the presence of diuron resulted in biofilm growth on the sides of the culture flasks or flocs in the media or in the bottom of the flasks. Lowering the diuron concentration to levels realistic for water contamination increased the extent of mineralization of diuron by strain SRS16 slightly when it was grown without strain D47 or an added growth substrate (Fig. 6). However, with the D47-SRS16 coculture and with SRS16 supplemented with growth substrates the extent of 14CO2 production from the mineralization of [14C]diuron was less than the extent observed with higher diuron concentrations. The same pattern was recently observed in a study focusing on nanomolar degradation and mineralization of the pesticide residue 2,6-dichlorobenzamide (BAM) by Aminobacter sp. strains (41). This study confirmed that there was extensive degradation of BAM despite the low extent of mineralization measured using 14CO2 from [ring-14C]BAM. It appears that the D47-SRS16 coculture mineralizes microgram-per-liter concentrations of diuron more efficiently than SRS16 supplemented with growth substrates, and further studies are needed to address the possibility of mineralization to an even greater extent with prolonged incubation periods and following additional optimization of the cultivation process.
In conclusion, a constructed coculture consisting of A. globiformis strain D47 and Variovorax sp. strain SRS16 appeared to be highly efficient in mineralizing realistic diuron concentrations in the range detected in different contaminated soil and water resources. The mineralization of both linuron and diuron suggests that this coculture could be ideal for remediation of soil and water affected by these herbicides, as well as their shared metabolite 3,4-DCA. Finally, Variovorax sp. strain SRS16 is the first described bacterium capable of mineralizing both an N,N-dimethyl-substituted phenylurea herbicide and an N-methoxy-N-methyl-substituted phenylurea herbicide and the first strain conclusively shown to mineralize the ring structure of diuron.
Acknowledgments
This work was supported by grant 26-04-0051 from the Danish Technical Research Council and by the Center for Environmental and Agricultural Microbiology, funded by the Willum Kann Rasmussen Foundation.
We thank Patricia Simpson for excellent help during preparation of the manuscript and the late Allan Walker, HRI-Warwick, for providing strain D47.
Footnotes
Published ahead of print on 22 February 2008.
REFERENCES
- 1.Batisson, I., S. Pesce, P. Besse-Hoggan, M. Sancelme, and J. Bohatier. 2007. Isolation and characterization of diuron-degrading bacteria from lotic surface water. Microb. Ecol. 54:761-770. [DOI] [PubMed] [Google Scholar]
- 2.Bazot, S., P. Bois, C. Joyeux, and T. Lebeau. 2007. Mineralization of diuron [3-(3,4-dichlorophenyl)-1,1-dimethylurea] by co-immobilized Arthrobacter sp. and Delftia acidovorans. Biotechnol. Lett. 29:749-754. [DOI] [PubMed] [Google Scholar]
- 3.Bending, G. D., E. Shaw, and A. Walker. 2001. Spatial heterogeneity in the metabolism and dynamics of isoproturon degrading microbial communities in soil. Biol. Fertil. Soils 33:484-489. [Google Scholar]
- 4.Bending, G. D., S. D. Lincoln, S. R. Sørensen, J. A. W. Morgan, J. Aamand, and A. Walker. 2003. In-field spatial variability in the degradation of the phenyl-urea herbicide isoproturon is the result of interactions between degradative Sphingomonas spp. and soil pH. Appl. Environ. Microbiol. 69:827-834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bollag, J. M., C. J. Myers, and R. D. Minard. 1992. Biological and chemical interactions of pesticides with soil organic matter. Sci. Total Environ. 123-124:205-217. [DOI] [PubMed]
- 6.Breugelmans, P., P.-J. D'Huys, R. D. Mot, and D. Springael. 2007. Characterization of novel linuron-mineralizing bacterial consortia enriched from long-term linuron-treated agricultural soils. FEMS Microbiol. Ecol. 62:374-385. [DOI] [PubMed] [Google Scholar]
- 7.Cabrera, A., L. Cox, P. Velarde, W. C. Koskinen, and J. Cornejo. 2007. Fate of diuron and terbuthylazine in soils amended with two-phase olive oil mill waste. J. Agric. Food Chem. 55:4828-4834. [DOI] [PubMed] [Google Scholar]
- 8.Castillo, M. A., N. Felis, P. Aragon, G. Cuesta, and C. Sabater. 2006. Biodegradation of the herbicide diuron by streptomycetes isolated from soil. Int. Biodeterior. Biodegrad. 58:196-202. [Google Scholar]
- 9.Claver, A., P. Ormad, L. Rodríguez, and J. L. Ovelleiro. 2006. Study of the presence of pesticides in surface waters in the Ebro River basin (Spain). Chemosphere 64:1437-1443. [DOI] [PubMed] [Google Scholar]
- 10.Cullington, J. E., and A. Walker. 1999. Rapid biodegradation of diuron and other phenylurea herbicides by a soil. Soil Biol. Biochem. 31:677-686. [Google Scholar]
- 11.Dejonghe, W., E. Berteloot, J. Goris, N. Boon, K. Crul, S. Maertens, M. Höfte, P. D. Vos, W. Verstrate, and E. M. Top. 2003. Synergistic degradation of linuron by a bacterial consortium and isolation of a single linuron-degrading Variovorax strain. Appl. Environ. Microbiol. 69:1532-1541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dejonghe, W., J. Goris, A. Dierickx, V. D. Dobbeleer, K. Crul, P. D. Vos, W. Verstraete, and E. M. Top. 2002. Diversity of 3-chloroaniline and 3,4-dichloroaniline degrading bacteria isolated from three different soils and involvement of their plasmids in chloroaniline degradation. FEMS Microbiol. Ecol. 42:315-325. [DOI] [PubMed] [Google Scholar]
- 13.Dellamatrice, P. M., and R. T. R. Monteiro. 2004. Isolation of diuron-degrading bacteria from treated soil. Braz. Arch. Biol. Technol. 47:999-1003. [Google Scholar]
- 14.Desaint, S., A. Hartmann, N. R. Parekh, and J. C. Fournier. 2000. Genetic diversity of carbonfuran-degrading soil bacteria. FEMS Microbiol. Ecol. 34:173-180. [DOI] [PubMed] [Google Scholar]
- 15.Devers, M., S. Henry, A. Hartmann, and F. Martin-Laurent. 2005. Horizontal gene transfer of atrazine-degrading genes (atz) from Agrobacterium tumefaciens St96-4 pADP::Tn5 to bacteria of maize-cultivated soil. Pest Manag. Sci. 61:870-880. [DOI] [PubMed] [Google Scholar]
- 16.El-Deeb, B. A., S. M. Sollan, A. M. Ali, and K. A. Ali. 2000. Detoxification of the herbicide diuron by Pseudomonoas sp. Folia Microbiol. 45:211-216. [DOI] [PubMed] [Google Scholar]
- 17.El-Fantroussi, S., W. Verstraete, and E. M. Top. 2000. Enrichment and molecular characterization of a bacterial culture that degrades methoxy-methyl urea herbicides and their aniline derivatives. Appl. Environ. Microbiol. 66:5110-5115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Eriksson, E., A. Baun, P. S. Mikkelsen, and A. Ledin. 2007. Risk assessment of xenobiotics in stormwater discharged to Harrestrup Å, Denmark. Desalination 215:187-197. [Google Scholar]
- 19.European Commission. 2000. Directive 2000/60/EC of the European Parliament and of the council of 23 October 2000. Establishing a framework for Community action in the field of water policy. http://www.bmu.de/files/pdfs/allgemein/application/pdf/water_framework_directive.pdf.
- 20.Fratila-Apachitei, L. E., J. A. Hirst, M. A. Siebel, and H. J. Gijzen. 1999. Diuron degradation by Phanerochaete chrysosporium BKM-F-1767 in synthetic and natural media. Biotechnol. Lett. 21:147-154. [Google Scholar]
- 21.Giacomazzi, S., and N. Cochet. 2004. Environmental impact of diuron transformation: a review. Chemosphere 56:1021-1032. [DOI] [PubMed] [Google Scholar]
- 22.Gooddy, D. C., P. J. Chilton, and I. Harrison. 2002. A field study to assess the degradation and transport of diuron and its metabolites in a calcareous soil. Sci. Total Environ. 297:67-83. [DOI] [PubMed] [Google Scholar]
- 23.Green, P. G., and T. M. Young. 2006. Loading of the herbicide diuron into the California water system. Environ. Eng. Sci. 23:545-551. [Google Scholar]
- 24.Guzzella, L., E. Capri, A. D. Corcia, A. B. Carracciolo, and G. Giuliano. 2006. Fate of diuron and linuron in a field lysimeter experiment. J. Environ. Qual. 35:312-323. [DOI] [PubMed] [Google Scholar]
- 25.Hall-Stoodley, L., J. W. Costerton, and P. Stoodley. 2004. Bacterial biofilms: from the natural environment to infectious diseases. Nat. Rev. Microbiol. 2:95-108. [DOI] [PubMed] [Google Scholar]
- 26.Johannesen, H., S. R. Sørensen, and J. Aamand. 2003. Mineralization of soil-aged isoproturon and isoproturon metabolites by Sphingomonas sp. strain SRS2. J. Environ. Qual. 32:1250-1257. [DOI] [PubMed] [Google Scholar]
- 27.Juhler, R. K., S. R. Sørensen, and L. Larsen. 2001. Analysing transformation products of herbicide residues in environmental samples. Water Res. 35:1371-1378. [DOI] [PubMed] [Google Scholar]
- 28.Kamagata, Y., R. R. Fulthrope, K. Takami, L. J. Forney, and J. M. Tiedje. 1997. Pristine environments harbor a new group of oligotrophic 2,4-dichlorophenoxyacetic acid-degrading bacteria. Appl. Environ. Microbiol. 63:2266-2272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Khadrani, A., F. Seigle-Murandi, R. Steiman, and T. Vroumsia. 1999. Degradation of three phenylurea herbicides (chlorotoluron, isoproturon and diuron) by micromycetes isolated from soil. Chemosphere 38:3041-3050. [DOI] [PubMed] [Google Scholar]
- 30.Lapworth, D. J., and D. C. Gooddy. 2006. Source and persistence of pesticides in a semi-confined chalk aquifer of southeast England. Environ. Pollut. 144:1031-1044. [DOI] [PubMed] [Google Scholar]
- 31.Larsen, L., S. R. Sørensen, and J. Aamand. 2000. Mecoprop, isoproturon, and atrazine in and above a sandy aquifer: vertical distribution of mineralization potential. Environ. Sci. Technol. 34:2426-2430. [Google Scholar]
- 32.Louchart, X., and M. Voltz. 2007. Aging effects on the availability of herbicides to runoff transfer. Environ. Sci. Technol. 41:1137-1144. [DOI] [PubMed] [Google Scholar]
- 33.Madhun, Y. A., and V. H. Freed. 1987. Degradation of the herbicides bromacil, diuron and chlortoluron in soil. Chemosphere 16:1003-1011. [Google Scholar]
- 34.Moncada, A. 2004. Environmental fate of diuron. Department of Pesticide Regulation Report, Sacramento, CA. http://www.cdpr.ca.gov/docs/emon/pubs/fatememo/diuron.pdf
- 35.Piutti, S., A. L. Marchand, B. Lagacherie, F. Martin-Laurent, and G. Soulas. 2002. Effect of cropping cycles and repeated herbicide applications on the degradation of diclofopmethyl, bentazone, diuron, isoproturon and pendimethalin in soil. Pest Manag. Sci. 58:303-312. [DOI] [PubMed] [Google Scholar]
- 36.Rouchaud, J., O. Neus, R. Bulcke, K. Cools, H. Eelen, and T. Dekker. 2000. Soil dissipation of diuron, chlorotoluron, simazine, propyzamide, and diflufenican herbicides after repeated applications in fruit tree orchards. Arch. Environ. Contam. Toxicol. 39:60-65. [DOI] [PubMed] [Google Scholar]
- 37.Scheyer, A., S. Morville, P. Mirabel, and M. Millet. 2007. Pesticides analysed in rainwater in Alsace region (eastern France): comparison between urban and rural sites. Atmos. Environ. 41:7241-7252. [Google Scholar]
- 38.Sørensen, S. R., and J. Aamand. 2003. Rapid mineralization of the herbicide isoproturon in soil from a previously treated Danish agricultural field. Pest Manag. Sci. 59:1118-1124. [DOI] [PubMed] [Google Scholar]
- 39.Sørensen, S. R., G. D. Bending, C. S. Jacobsen, A. Walker, and J. Aamand. 2003. Microbial degradation of isoproturon and related phenylurea herbicides in and below agricultural fields. FEMS Microbiol. Ecol. 45:1-11. [DOI] [PubMed] [Google Scholar]
- 40.Sørensen, S. R., J. Rasmussen, C. S. Jacobsen, O. S. Jacobsen, R. K. Juhler, and J. Aamand. 2005. Elucidating the key member of a linuron-mineralizing bacterial community by PCR and reverse transcription-PCR denaturing gradient gel electrophoresis 16S rRNA gene fingerprinting and cultivation. Appl. Environ. Microbiol. 71:4144-4148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Sørensen, S. R., M. S. Holtze, A. Simonsen, and J. Aamand. 2007. Degradation and mineralization of nanomolar concentrations of the herbicide dichlobenil and its persistent metabolite 2,6-dichlorobenzamide by Aminobacter spp. isolated from dichlorobenil-treated soils. Appl. Environ. Microbiol. 73:399-406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Sørensen, S. R., Z. Ronen, and J. Aamand. 2001. Isolation from agricultural soil and characterization of a Sphingomonas sp. able to mineralize the phenylurea herbicide isoproturon. Appl. Environ. Microbiol. 67:5403-5409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Thomas, K. V., M. McHugh, and M. Waldock. 2002. Antifouling paint booster biocides in UK coastal waters: input, occurrence and environmental fate. Sci. Total Environ. 293:117-127. [DOI] [PubMed] [Google Scholar]
- 44.Thurman, E. M., K. C. Bastian, and T. Mollhagen. 2000. Occurrence of cotton herbicides and insecticides in playa lakes of the high plains of west Texas. Sci. Total Environ. 248:189-200. [DOI] [PubMed] [Google Scholar]
- 45.Tixier, C., M. Sancelme, F. Bonnemoy, A. Cuer, and H. Veschambre. 2001. Degradation products of a phenylurea herbicide, diuron: synthesis, ecotoxicity, and biotransformation. Environ. Toxic Chem. 20:1381-1389. [DOI] [PubMed] [Google Scholar]
- 46.Tixier, C., M. Sancelme, S. Aït-Aïssa, P. Widehem, F. Bonnemoy, A. Cuer, N. Truffaut, and H. Veschambre. 2002. Biotransformation of phenylurea herbicides by a soil bacterial strain, Arthrobacter sp. N2: structure, ecotoxicity and fate of diuron metabolite with soil fungi. Chemosphere 46:519-526. [DOI] [PubMed] [Google Scholar]
- 47.Tran, A. T. K., R. V. Hyne, and P. Doble. 2007. Determination of commonly used polar herbicides in agricultural drainage waters in Australia. Chemosphere 67:944-953. [DOI] [PubMed] [Google Scholar]
- 48.Turnbull, G. A., J. E. Cullington, A. Walker, and J. A. W. Morgan. 2001. Identification and characterisation of a diuron-degrading bacterium. Biol. Fertil. Soils 33:472-476. [Google Scholar]
- 49.Turnbull, G. A., M. Ousley, A. Walker, E. Shaw, and J. A. W. Morgan. 2001. Degradation of substituted phenylurea herbicides by Arthrobacter globiformis strain D47 and characterization of a plasmid-associated hydrolase gene, puhA. Appl. Environ. Microbiol. 67:2270-2275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.U.S. Environmental Protection Agency. 2005. CCL 2. Second drinking water contaminant candidate list. U.S. Environmental Protection Agency, Washington, DC. http://www.epa.gov/safewater/ccl/ccl2.html.
- 51.Vega, A. B., A. G. Frenich, and J. L. M. Vidal. 2005. Monitoring of pesticides in agricultural water and soil samples from Andalusia by liquid chromatography coupled to mass spectrometry. Anal. Chim. Acta 538:117-127. [Google Scholar]
- 52.Weinberger, M., and J.-M. Bollag. 1972. Degradation of chlorbromuron and related compounds by the fungus Rhizoctonia solani. Appl. Microbiol. 24:750-754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Widehem, P., S. Aït-Aïssa, C. Tixier, M. Sancelme, H. Veschambre, and N. Truffaut. 2002. Isolation, characterization and diuron transformation capacities of a bacterial strain, Arthrobacter sp. N2. Chemosphere 46:527-534. [DOI] [PubMed] [Google Scholar]