Abstract
A juvenile male zebra finch, Taeniopygia guttata, kept singly with its father develops a fairly complete imitation of the father’s song. The imitation is less complete when other male siblings are present, possibly because as imitation commences, model abundance increases. Here we examine the consequences of allowing more or less access to a song model. Young males heard a brief song playback when they pecked at a key, but different males were allowed to hear different numbers of playbacks per day. Using an automated procedure that scored the similarity between model and pupil songs, we discovered that 40 playbacks of the song motif per day, lasting a total of 30 sec, resulted in a fairly complete imitation. More exposure led to less complete imitation. Vocal imitation often may reflect the interaction of diverse influences. Among these, we should now include the possible inhibitory effect of model overabundance, which may foster individual identity and explain the vocal diversity found in zebra finches and other songbirds.
A male zebra finch acquires its brief (0.5 to 1.5 sec), single-song motif by imitating song elements from other males. Model imitation occurs between posthatching days 30–60. Imitation is initially partial and variable, but with time it becomes more complete and stereotyped. Stable, adult song is reached by about 90 days. At that time the song motif is composed of dissimilar notes repeated in a set order. The boundaries between notes are defined by brief, silent intervals or by abrupt changes in frequency modulation. Chunks are clusters of notes preceded and followed by a silent interval. Songs occur in bouts. In a bout, which commences with several introductory notes, the song motif is repeated several times.
A juvenile zebra finch male kept singly with its father produces a close approximation of the father’s song. In contrast, incomplete imitations result when several male siblings of the same brood are reared together with their father. The greater the number of male siblings, the shorter the mean duration of their song motifs and the fewer the mean number of notes imitated (1). Late-hatched male siblings tend to develop their song early relative to their older brothers and produce the most complete imitations. Prior work (2, 3) has shown that each sibling can act as a tutor for other siblings. Therefore, partial imitation may reflect inhibition induced by an overabundance of similar song models; that is, the emerging songs of siblings that start imitating early and that sound like their father may reduce the tendency of other siblings to imitate similar sounds (1). However, this earlier experiment did not allow us to rule out other social variables that might affect song learning in clutches with variable numbers of male siblings.
The experiment we describe here was designed to circumvent the complexities of social interactions and test just for the effect of model abundance. Previous studies have shown that when a young bird induces the playback of a song model by pecking a key, this leads to reliable, if often partial, model imitation (4, 5). We used a similar approach to test whether narrowing a pupil’s opportunities to induce song playback resulted in more complete imitation.
Methods
Animals.
We examined song imitation in 28 young males that had been reared by their mothers and not exposed to adult males after their first week of posthatching life. Starting at the age of 30 days, each pupil was kept in social isolation and could induce song playbacks by pecking a key as described below.
Song Tutoring Apparatus.
Each bird was kept singly in a soundproof box (50 × 30 × 27 cm3) throughout the experiment. The box contained two keys, 1 inch above each of two perches. Keys were prepared from 2-g lever switches (Cherry Elect E22–85HX; Wallingford, CT). We glued a red, ½-inch round, plastic tiddlywinks piece to the end of the lever and, above this, attached a small piece of cuttlebone. By pecking either of the keys, the bird could induce song playbacks from a 11/4-inch samarium cobalt speaker (Intervox S125RL; Washington, DC) hidden inside a plastic model of an adult zebra finch male (6). Birds had free access to the keys and to the plastic male model throughout the experiment. All birds started pecking the key within 2–5 days of being placed in the training cage, at a posthatching age of 32–37 days. Key pecking persisted throughout the experiment.
Experimental Sessions.
Six birds had unrestricted access to the tutoring song, which they could trigger every time they pecked a key. The remaining birds had a morning and afternoon tutoring session each day (starting at 8 a.m., 30 min after lights-on, and at 5 p.m., respectively). During each session the bird was allowed to induce song playbacks by pecking a key until an upper bound of song playbacks was reached. Each playback consisted of two consecutive repetitions of a same motif, with a combined duration of 1.4 sec, and we limited the number of song playbacks that a pupil could induce per training session. When a pupil reached its allowed quota of playbacks, further key pecking did not induce more playbacks until the next session. Table 1 summarizes the experimental groups.
Table 1.
Experimental groups
| No. of birds | Motifs per key peck | Motifs per session |
|---|---|---|
| 2 | 2 | 5 |
| 6 | 2 | 10 |
| 2 | 2 | 25 |
| 2 | 2 | 50 |
| 2 | 2 | 100 |
| 6 | 2 | UL |
| 4 | 20 | 50 |
| 2 | 20 | 250 |
| 2 | 20 | 500 |
The second column shows the number of consecutive song motif playbacks that occur each time a bird pecks on a key, as long as song quota has not been reached. The third column shows the maximal number of motif playbacks that a bird could induce per session. UL, unlimited.
System Configuration.
Twenty song tutoring boxes were controlled by a Power Macintosh 7200 computer running National Instruments ni-daq software (Austin, TX) and a program written by using Symantec’s C++ compiler (Cupertino, CA). Each key was connected through a 1K resistor to an input channel of a National Instruments PCI-96-DIO data acquisition card. Output channels of the same card controlled song output to each male model. Song playbacks delivered sound at a maximal amplitude of 85 dB measured 2 inches in front of the model. Key pecks were monitored at a frequency of 10 Hz, and the delay between a key peck and song playback was varied uniformly between 0.1 and 1.5 sec. Key pecking was monitored continuously from 7:30 a.m. to 7:30 p.m. (when lights were on).
Motif Duration.
The song motif of adult male zebra finches is stereotyped, but each particular rendering need not include all its sounds. We calculated the motif duration for each bird by using the median duration between the onset of two similar chunks during 10–15 sec of singing.
Measures of Song Similarity.
At the age of 100 days we digitally recorded the pupil’s female directed song as described in ref. 1. The song motif was identified by visual inspection, and its similarity to the song model was measured by using an automated procedure that analyzed features of the two sounds and scored their similarity (7). A detailed algorithm for the procedure will be published elsewhere (8), but here is a short summary of the computational stages: the procedure computes direct multitaper spectral estimates of the songs [time window = 7 ms; overlap = 1.4 ms; tapers = 2; time–bandwidth product (TW) = 1.6]. We followed the computational framework suggested by Ho et al. (7) to extract four features for every time window: pitch (the period of the sound), frequency modulation, Wiener entropy (a measure of randomness), and spectral continuity (the proportion of continuous vs. discontinuous frequency traces across time windows). To make the features additive we scaled their units according to their statistical distribution in a sample of 10 unrelated songs. The features are now normalized to units of their median absolute deviation from the average. This allowed us to transform the distance between the features of two sounds to a P value, namely, to the probability of obtaining such a distance or less by chance alone. The procedure examined the distances across features between all possible pairs of 50-ms intervals of the two songs, and pairs where P < 0.01 were categorized as similar. It then identified continuous sections of similarity between the two song motifs. In cases in which the projections of two sections of similarity on either the song model or on the pupil’s song overlapped, the procedure preferred sections of higher similarity and trimmed the overlapping parts of sections of lower similarity to give a unique similarity score.
The final similarity score has two components: the first is the proportion of time windows in the song model that had a similar version in the pupil’s song. The second is the magnitude of the similarity, that is, the actual P value for each pair of time windows that were categorized as similar. For the final score, P values were calculated again—not across intervals but separately for each pair of corresponding time windows (this fine-grain analysis extends the range of P values). Because the proportion of similar time windows and their corresponding P values are not independent measures, we combined them to a global similarity score. The global similarity score ranges between 0 and 100% and is defined as the sum of the complements of P values Σ(1 − P) across pairs of similar time windows, divided by the number of time windows in the song model. The higher the proportion of similar time windows, and the lower the distances across their features, the higher the global similarity score becomes. In all experimental groups we also scored song similarity by using the traditional eye-balling approach (1), and in all cases human and automated scores showed similar trends.
Results
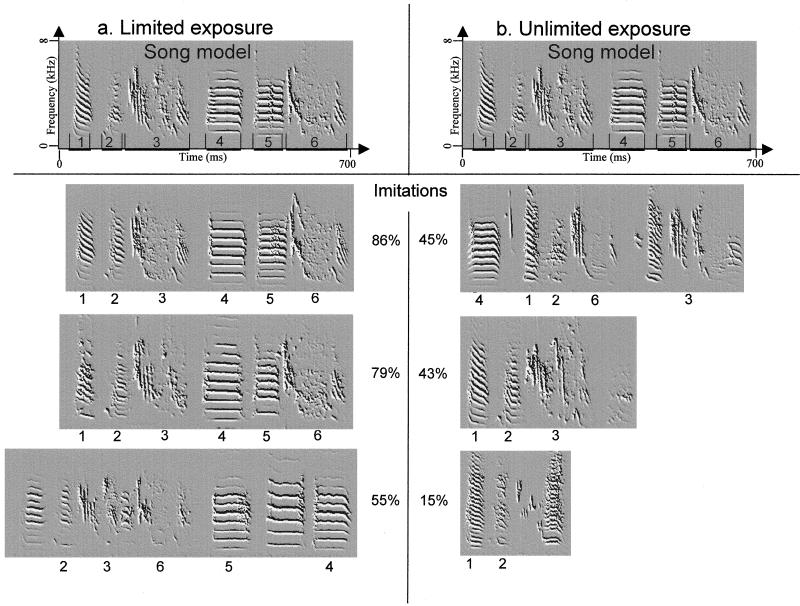
First we examined the similarity between the song model and the songs of pupils that could induce only up to 40 song motifs per day. These results were compared with those of pupils that received unlimited exposure to a song model. Fig. 1 provides examples illustrating that song similarity was relatively high in pupils that received limited exposure to the song model [similarity = 70% ± 5 (SE), n = 6; actual average exposure = 30 ± 4 presentations per day]. Song similarity decreased with unlimited exposure to the same model (similarity = 33% ± 4, n = 6; Mann–Whitney U test, U = 0, P = 0.004; actual exposure = 621 ± 164 presentations per day).
Figure 1.
Song imitation in birds that had (a) limited (40 motifs per day) and (b) unlimited exposure to a song model. We show the song of the birds that had the highest, median, and lowest similarity scores for each group. Spectral derivatives are used to give a sharp, visual representation of song, with frequency on the y axis and time on the x axis. The chunks of the song model are denoted by numerals above the time axis, and chunks of a pupil’s song that showed similarity to the corresponding chunk in the model song are denoted by the same numeral. The overall imitation accuracy across sound features is presented at the center.
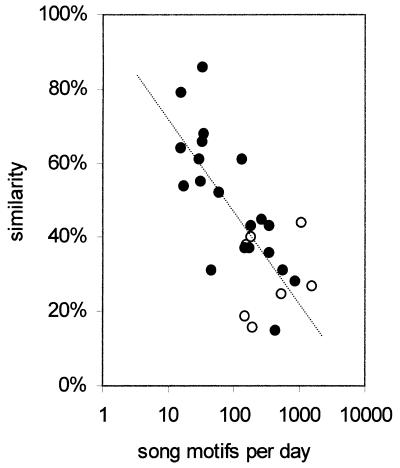
We next examined systematically the effect of a wide range of song model exposures as shown in Fig. 2. The resulting regression curve showed a negative correlation between the number of times the song model was heard per day and the similarity between the pupil and model songs. This regression was fairly linear when presented on a logarithmic scale (r = −0.75, P = 0.0002). Remarkably, even 2 min of song exposure per day (≈160 repetitions of the motif) reduced imitation. Because the actual number of key pecks determined exposure to the song model, we wondered whether changing the ratio of motif repetitions per key peck would affect the completeness of imitation. We therefore examined in seven birds the effect of increasing the number of motif playbacks per key peck to 20 (≈15 sec of continuous playback; open circles in Fig. 2). As shown, this manipulation did not induce any additional major effect and song similarity can be predicted based on exposure alone.
Figure 2.
Similarity to song model as a function of exposure to it. Solid circles represent birds that heard a same motif repeated twice when they pecked a key, whereas open circles represent birds that heard the same motif repeated 20 times each time they pecked a key; the horizontal axis shows a logarithmic scale of the number of times the song motif was heard per day, and the vertical axis represents the similarity between the pupil and model songs.
Young zebra finches reared in social isolation can improvise a song that, though atypical, includes many normal sounds. Despite this potential, partial imitation in the group that received unlimited exposure was not compensated by improvisation. The result was that the mean duration of the song motif of the birds that had limited and unlimited access to the song model was, respectively, 667 ± 46 ms and 386 ± 66 ms. This finding is similar to what we observed in cases of sibling inhibition in single family groups (1).
Interestingly, reduction in the completeness of imitation did not affect all parts of the model song to an equal extent. Fig. 3 shows that whereas some song chunks were copied by most pupils regardless of exposure, other chunks were rarely copied when exposure was high. Because all of our data were obtained from use of a single song model, we do not know whether the selective imitation of some chunks and not of others was related to the position of the chunks in the motif or to the structure of the sounds.
Figure 3.
Different chunks of the model song were imitated to different extents by juveniles that had limited (red line) or unlimited (blue line) access to the model. The horizontal lines correspond to the mean similarity score for each chunk for all the birds in that group. The vertical line offers a simple yardstick for each of these comparisons.
Discussion
The current results support the hypothesis—model overabundance restricts imitation—that we offered to explain our earlier observations in a one-family setup (1). However, the test of this hypothesis that we offer here occurs in the simplest possible context, one in which only one model is offered at different levels of abundance. We do not know, yet, what will happen when a pupil is exposed to several song models that differed in their abundance. Will the least-abundant model be imitated the most, or will overtraining with one song alter brain circuitry or endocrine state in a way that affects not just the imitation of the overabundant song, but also that of the less frequent one? It also would be interesting to know whether overexposure affects the initial attempts at imitation. Would the young birds go through a stage in which all components of the model song were present, later to be partially culled as overexposure persists? At another level, the results we report here do not address the possible influence of social variables on vocal learning. The pairing of a single juvenile with a live-in adult model yields very high imitation (1) and we do not know whether this is caused by social interactions that might, for example, override the effect of model abundance. However, it is clear that when social interactions are excluded, model abundance is, in zebra finches, a good predictor of completeness of vocal imitation.
Our findings help reconcile previous contradictory results: zebra finches exposed to the song of a male they could not see and with which they could not interact (9) or trained to hop on a perch to trigger song playbacks (10) showed poor or no imitation. However, some imitation was observed when zebra finches were allowed to peck at a key to induce song playback (4, 5). So, one might conclude, imitation is enhanced by providing the birds with the correct operant control. Nevertheless, another study has shown that the extent of model imitation was comparable (45%) in zebra finches that pecked at a key to hear a song and in yoked controls (5). We got a further improvement on imitation by combining key pecking as an operant with limited access to the model.
Our present results suggest that differences in amount of exposure to a song model help explain the diversity of outcomes listed above. Unpublished observations from our laboratory suggest that birds trained in operant key pecking induce fewer song playbacks than birds trained in operant perch hopping. This may explain the failure to induce imitation by using the latter protocol. When the operant method was held constant (key pecking), the extent of imitation was best explained by the number of songs heard per day.
Intriguingly, Houx and ten Cate (5) made an incidental observation that pointed to an inverse relation between model abundance and imitation. They noted that in the yoked controls there was a significant negative correlation between amount of exposure and song imitation (r = −0.84). However, they attributed little importance to this outcome, which, for reasons unknown, was not reproduced in the key-pecking group (r = −0.14) and emphasized, instead, that operant control was not necessary for imitation to occur. It is worth mentioning that Houx and ten Cate did not manipulate song model abundance, and it was only the yoked control birds that were exposed to an external constraint on the number of song playbacks they could have heard.
The existing literature on song learning is predominantly silent on the effects of model abundance on imitation. The reasons for this are probably to be found in the biases brought to this research. Investigators have, on several occasions, emphasized that very good imitation is possible even when a model was presented a small number of times (11–13). The emphasis was on how much exposure was enough for learning to occur (14). Moreover, observations on some natural song dialects emphasized the relative homogeneity of song within a dialect area (15–18). What had not been done before was to see whether songs heard each day a relatively few number of times and imitated in great detail might, on further exposure, be imitated less well. Our results indicate that, under the conditions of our experiments, such an effect occurs. We suggest that the “too much” factor might have to be considered as one of the variables that affects vocal learning.
We do not know whether model abundance affects song learning in free-ranging zebra finches. In nature, zebra finches live in social groups where many potential song models are available to juveniles. Sons imitate song chunks from their father as well as from other tutors (19). This diversity of potential sources, combined with the phenomenon we describe here, may encourage each juvenile male to develop a song that is based on tradition, yet also fosters individual identity.
Apparently, the process of vocal imitation is more complex than a mere striving for a perfect match. The effect of model abundance on imitation supports the view, first suggested by Marler, that in some cases, at least, imitation shortfalls should not be characterized as “poor learning” (20, 21). Model overexposure may trigger processes of active winnowing that focus imitation on some features and reject others. The balance between processes of model acceptance and model rejection may explain the rich diversity of songs found in wild and captive populations of many songbirds.
Acknowledgments
We thank Peter Marler, David Vicario, Marcelo Magnasco, and Boris Shraiman for their useful comments. Timothy Gale, Leonard Hare, and Michael Chang helped with technical support. This work was supported by the National Institute of Mental Health, the Mary Flagler Cary Charitable Trust, the McKnight Foundation, and the generosity of Mr. Romie Shapiro and the late Mr. Herbert Singer.
References
- 1.Tchernichovski O, Nottebohm F. Proc Natl Acad Sci USA. 1998;95:8951–8956. doi: 10.1073/pnas.95.15.8951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Morrison R G. Ph.D. dissertation. New York: The Rockefeller University; 1991. [Google Scholar]
- 3.Volman S F, Khanna H. J Comp Psychol. 1995;109:211–221. doi: 10.1037/0735-7036.109.3.211. [DOI] [PubMed] [Google Scholar]
- 4.Adret P. Anim Behav. 1993;46:149–159. [Google Scholar]
- 5.Houx B B, ten Cate C. Anim Behav. 1999;57:837–845. doi: 10.1006/anbe.1998.1046. [DOI] [PubMed] [Google Scholar]
- 6.Tchernichovski O, Schwabl H, Nottebohm F. Anim Behav. 1998;55:1003–1010. doi: 10.1006/anbe.1997.0673. [DOI] [PubMed] [Google Scholar]
- 7.Ho C E, Pesaran B, Fee M S, Mitra P P. Proceedings of the Fifth Symposium of Neural Computation. San Diego: Univ. of California Press; 1998. pp. 76–83. [Google Scholar]
- 8.Tchernichovski, O. Nottebohm, F., Ho, C. E., Pesaran, B. & Mitra, P. P., Anim. Behav., in press. [DOI] [PubMed]
- 9.Eales L A. Anim Behav. 1989;37:507–508. [Google Scholar]
- 10.ten Cate C. Anim Behav. 1991;42:857–859. [Google Scholar]
- 11.Hultsch H, Todt D. Naturwissenschaftern. 1989;76:83–85. [Google Scholar]
- 12.Hultsch H. Neth J Zool. 1993;43:155–171. [Google Scholar]
- 13.Marler P, Peters S. Ethology. 1987;76:89–100. [Google Scholar]
- 14.Petrinovich L. J Comp Psychol. 1985;99:15–29. [PubMed] [Google Scholar]
- 15.Marler P, Tamura M. Science. 1964;146:1483–1486. doi: 10.1126/science.146.3650.1483. [DOI] [PubMed] [Google Scholar]
- 16.Nottebohm F. Condor. 1969;71:299–315. [Google Scholar]
- 17.Nottebohm F. Am Nat. 1975;109:605–624. [Google Scholar]
- 18.Baker M C, Cunningham M A. Behav Brain Sci. 1985;8:85–133. [Google Scholar]
- 19.Zann R. The Zebra Finch. London: Oxford Univ. Press; 1996. [Google Scholar]
- 20.Marler P. In: Proceedings of the 14th International Ornithological Congress. Snow D W, editor. Oxford: Blackwell Scientific; 1967. pp. 231–244. [Google Scholar]
- 21.Marler P, Mundinger P, Waser M S, Luthen A. Anim Behav. 1972;20:586–606. doi: 10.1016/s0003-3472(72)80024-1. [DOI] [PubMed] [Google Scholar]