Abstract
We have previously reported that the T-cell receptor (TCR) γδ+ T cells increase in mice infected with an intracellular bacteria Listeria monocytogenes, and the cells predominantly express Vδ6 and Vγ1 genes. In this study, we used a monoclonal antibody (mAb) specific to TCR Vγ1 to estimate the frequency of Vγ1+ T cells and we discuss their significance in protection against L. monocytogenes. The spleen, liver and peritoneal exudate cells from mice intraperitoneally infected with L. monocytogenes were analysed by flow cytometry. In all the organs investigated, Vγ1+ cells increased predominantly among TCR γδ+ T cells at an early phase (day 5–7) of the infection. To elucidate the significance of the Vγ1+ T cells in the protection against L. monocytogenes, mice were depleted of TCR Vγ1+γδ T cells or all TCR γδ+ T cells by intraperitoneal inoculation of anti-Vγ1 mAb or anti-pan TCR γδ mAb, respectively, before infection with L. monocytogenes. The bacterial growth in the spleen and the liver examined on day 5 after the infection increased significantly by the depletion of TCR Vγ1+ T cells. The numbers of L. monocytogenes in TCR Vγ1+ T-cell-depleted mice were nearly the same as in mice depleted of all TCR γδ+ T cells. These results demonstrated that Vγ1+ T cells are the predominant population of γδ T cells in protection against L. monocytogenes at the early phase of the primary infection.
INTRODUCTION
Murine γδ T cells can be divided into three types based on the T-cell receptor (TCR) V gene repertoire and developmental pathway.1 The first type of γδ T cells matures in the fetal thymus, and is characterized by its selective homing to and residence in defined epithelial tissues. These cells rearrange TCR Vγ5, Vγ6 and Vδ1 genes with canonical junctional sequences, suggesting essentially monospecificity. The second type of γδ T cells develops in the adult thymus. Cells of this type preferentially use Vγ1 or Vγ4 and less frequently Vγ2 or Vγ7 and they exhibit great junctional diversity. These γδ T cells predominate in peripheral blood and conventional lymphoid organs and may occasionally localize to certain mucosal tissues. The third type of γδ T cells appears to develop extrathymically. These cells preferentially use Vγ7 and Vγ1 with high junctional diversity and localize not only in peripheral blood and lymphoid organs but also in liver and intestinal epithelium.
It has been reported that TCR γδ+ T cells increase in number and have an important role in protection against primary infection by an intracellular bacterial pathogen, Listeria monocytogenes at the early phase before mature T cells bearing TCR αβ appear.1–6 We have previously reported that the TCR γδ+ T cells of mice infected with L. monocytogenes predominantly express Vδ6 and Vγ1 genes as V-region genes.7,8 Since Vγ1+ Vδ6+ T-cell hybridomas have been reported to preferentially respond to mycobacterial antigen (Ag),9–13 and the γδ T cells obtained from L. monocytogenes-infected mice responded to mycobacterial Ag protein purified derivative (PPD),4 it is possible that mycobacterial Ag-reactive Vγ1+ Vδ6+ T cells have an important role in γδ T-cell-mediated protection against L. monocytogenes infection. However, it is not known whether Vγ1+ cells play a part in protection against L. monocytogenes. The antibodies specific for individual TCR V-regions will provide more definitive results on γδ T-cell subsets and their role in the immune response to L. monocytogenes.
In this report, we used a monoclonal antibody (mAb) 2·11 specific for TCR Vγ114 and demonstrated that at the early phase in primary infection by L. monocytogenes, Vγ1+ T cells expand dominantly among γδ T cells in the peritoneal cavity, spleen and liver, and have an important role in protection against L. monocytogenes.
MATERIALS AND METHODS
Mice and micro-organisms
Female C3H/He mice were obtained from Charles River Japan (Atsugi, Japan). Eight- to 10-week-old mice were used for the experiments. Listeria monocytogenes, strain EGD, was used in all experiments. Bacterial virulence was maintained by serial passages in BALB/c mice. Fresh isolates of L. monocytogenes were obtained from infected spleens, grown in tryptic soy broth (Difco, Detroit, MI), washed repeatedly, resuspended in phosphate-buffered saline (PBS) and stored at −70° in small aliquots until use.
Peritoneal exudate cells (PEC)
PEC were obtained from mice by peritoneal lavage with Hanks' balanced salt solution (HBSS). The cells were collected by centrifugation at 120 g for 5 min, washed twice and resuspended in RPMI-1640 medium (Gibco Laboratories, Grand Island, NY) supplemented with 10% fetal calf serum (FCS; complete medium). PEC were plated on plastic plates at a concentration of 5×105 cells/ml and incubated for 1 hr in a CO2 incubator at 37° to obtain non-adherent cells.
Isolation of hepatic lymphocytes
Hepatic lymphocytes were prepared as described previously15,16 with minor modifications. Briefly, the liver was perfused with 10 ml of PBS via the portal vein before harvesting to reduce possible contamination with circulating lymphocytes. The liver was pressed through a 100-gauge stainless steel mesh in HBSS. The cell suspension was centrifuged through a 33–67·5% Percoll gradient. Cells at the interface were washed and used.
Antibodies and reagents
Anti-pan TCR γδ mAb (UC7-13D5, generously provided by Dr J. A. Bluestone, Chicago University, Chicago, IL) and anti-TCR Vγ1 mAb (2·11, generously provided by Dr P. Pereira, Institut Pasteur, Paris, France) were prepared from supernatants of hybridoma cells cultured in complete medium using HiTrap protein G column (Pharmingen, San Diego, CA). Phycoerythrin (PE)-conjugated anti-TCR γδ (Cδ) mAb (GL3) and allophycocyanin (APC)-conjugated anti-CD3ε mAb (145-2C11) was purchased from Pharmingen (San Diego, CA). Anti-TCR Vγ1 mAb was conjugated with fluorescein isothiocyanate (FITC) using a standard technique.
Flow cytometry
All samples were preincubated with anti-FcRγII/III antibody (2·4G2) to block FCRγII/III mediated binding of mAb. Cells were triple stained with FITC-conjugated anti-TCR Vγ1 mAb, PE-conjugated anti-TCR Cδ mAb, and APC-conjugated anti-CD3 mAb for 30 min at 4°. Cells were loaded on a flow cytometer (FACSCalibur, Becton Dickinson, Sunnyvale, CA). The data were analysed using CELL QUEST analysis software (Becton Dickinson). Percentages of TCR γδ+ cells or TCR Vγ1+ cells were determined as a fraction of all CD3+ cells. Percentages of TCR Vγ1+ cells were further calculated as a fraction of all CD3+ TCR γδ+ cells.
Determination of bacterial growth after intraperitoneal infections with L. monocytogenes
Mice were infected intraperitoneally (i.p.) with 1/10 lethal dose 50% (ld50) (1×103) of L. monocytogenes in 0·2 ml of PBS. In some experiments, mice were i.p. inoculated with 0·25 mg of anti-TCR γδ mAb or anti-TCR Vγ1 mAb in 0·2 ml of PBS 2 days before the infection with L. monocytogenes to eliminate all TCR γδ+ T cells or TCR Vγ1+ T cells, respectively. Mice were anaesthetized with ether and killed by cutting the cervical artery 5 days after the i.p. infection. Bacterial growth in the spleen and the liver were determined by plating 10-fold serial dilutions of organ homogenates on tryptic soy agar plates. The detection limit of this procedure was 103L. monocytogenes per organ. The numbers of colonies were counted after 24 hr of incubation at 37°.
Statistical analysis
Student's t-test was used for statistical analysis. A P value <0·05 was considered to indicate statistical significance.
RESULTS
Kinetics of appearance of TCR Vγ1+ T cells during L. monocytogenes infection
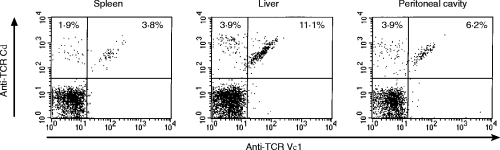
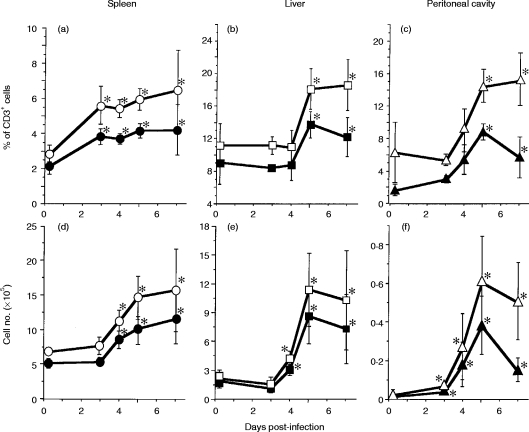
Figure 1 shows representative profiles of TCR γδ and TCR Vγ1 expression of lymphocytes from the spleen, liver and peritoneal cavity after i.p. infection with 1×103L. monocytogenes. The kinetics of percentages of TCR Vγ1+ or TCR γδ+ in CD3+ T cells is summarized in the upper panels of Fig. 2. Absolute numbers of the Vγ1+ and TCR γδ+ T cells were calculated and are shown in the lower panels of Fig. 2. In the spleen, the proportion of TCR γδ T cells among CD3+ cells and the number of TCR γδ T cells were significantly increased as early as on day 4 of i.p. infection with L. monocytogenes, and gradually increased thereafter (Fig. 2a and d). In the liver, the proportion of TCR γδ T cells among CD3+ cells was around 10% before infection, and it significantly increased to 20% on day 5 of the infection and maintained the same proportion thereafter (Fig. 2b). The number of TCR γδ T cells in the liver was around 2×105 before infection, and it increased significantly from day 4 to reach a maximum (approximately 10×105) on day 5 of the infection (Fig. 2e). The proportion of TCR γδ T cells among CD3+ cells was around 6% in the peritoneal cavity of naive mice, and it significantly increased to 15% on day 5 of the infection (Fig. 2c). The number of TCR γδ T cells was around 5×103 in the peritoneal cavity before infection, and it significantly increased from day 3, reaching a plateau of approximately 5×104–6×104 on day 5 after the infection, and remained nearly the same number during the experiments (Fig. 2f). All the data demonstrate that TCR γδ T cells increased in the spleen, liver and peritoneal cavity from day 3 to 4 of L. monocytogenes infection.
Figure 1.
TCR Vγ1 expression of TCR γδ T cells in the spleen cells, hepatic lymphocytes and PEC from L. monocytogenes-infected mice analysed by flow cytometry. Mice were infected i.p. with 1×103 of L. monocytogenes. On day 5, lymphocytes from the spleen, liver and peritoneal cavity were stained with FITC-conjugated anti-TCR Vγ1 mAb, PE-conjugated anti-TCR-Cδ mAb, and APC-conjugated anti-CD3 mAb and analysed by FACSCalibur. The analysis gate was set on CD3+ cells, and the profiles of TCR γδ and TCR Vγ1 expression in CD3+ cells are shown. The data shown are representative of data from individual analysis of 20 mice.
Figure 2.
Kinetics of TCR Vγ1+ T cells in the spleen, liver and peritoneal cavity after L. monocytogenes infection. Spleen cells, hepatic lymphocytes and plastic plate-non-adherent PEC were prepared as mentioned in the Materials and Methods from mice i.p. inoculated with 1×103L. monocytogenes at the indicated intervals after the infection, and examined as described in Fig. 1. Percentages of Cδ+ cells and Vγ1+ cells in the CD3+ population are demonstrated in the upper panels (a, b and c). The cell numbers of the T-cell subsets per mouse were further calculated based on the number of lymphocytes harvested per mouse and the percentage of TCR γδ+ or TCR Vγ1+ cells, and demonstrated in the lower panels (d, e and f). The data of spleen cells (a, d), hepatic lymphocytes (b, e) and PEC (c, f) are shown. Open symbols and closed symbols represent the data of TCR γδ+ T cells and TCR Vγ1+ T cells, respectively. The data shown are the summaries of data from individual analysis of 12 mice. *P <0·05 compared to the data on day 0.
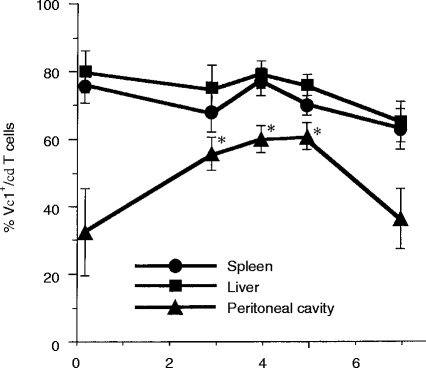
In the spleen (Fig. 2a and d) and the liver (Fig. 2b and e), TCR Vγ1+ T cells increased after the L. monocytogenes infection in parallel with the increase of TCR γδ T cells. The TCR Vγ1+ T cells represent 70–80% of all TCR γδ T cells in the spleen and liver of mice before the infection and the percentages are maintained during the infection (Fig. 3). In contrast, the proportion of TCR Vγ1+ PEC changed in the course of the infection. Before the infection, the proportion of TCR Vγ1+ T cells among TCR γδ T cells was approximately 30% in the peritoneal cavity, it significantly increased to around 60% on day 3 and then significantly decreased to 40% or less by day 7 (Fig. 3). All the data show that Vγ1+ T cells are the major population of γδ T cells induced by L. monocytogenes infection in the early stage, especially from day 3 to day 5 of the infection.
Figure 3.
Kinetics of percentage of TCR Vγ1+ T cells in total TCR γδ T cells in the spleen, liver and peritoneal cavity. Spleen cells, hepatic lymphocytes and plastic plate-non-adherent PEC were analysed as described in Fig. 2. Results are expressed as the percentages of TCR Vγ1+ T cells in the TCR γδ+ T-cell population. The data shown are the summaries of data from individual analysis of 12 mice. *P <0·05 compared to the percentage of Vγ1+ T cells on day 0.
Effect of in vivo depletion of TCR Vγ1+ T cells on the eradication of L. monocytogenes after infection
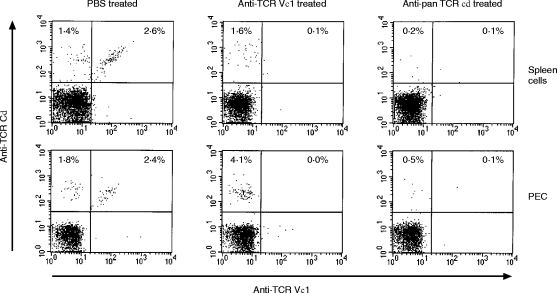
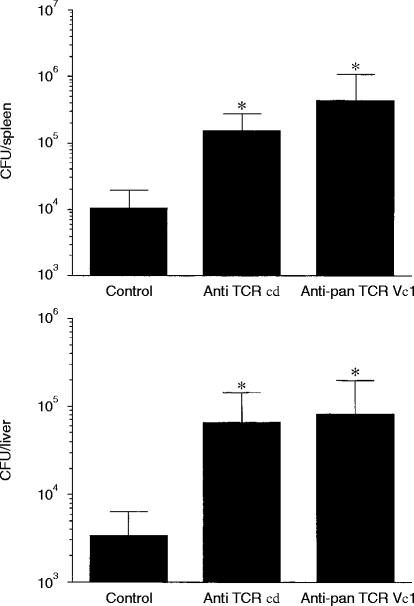
We have previously shown the involvement of TCR γδ T cells in anti-listerial protection by experiments in which mice were treated with anti-TCR γδ mAb. Here, to investigate the protective role of the TCR Vγ1+ T cells in listerial infection, TCR Vγ1+ T-cell-depleted mice or TCR γδ+ T-cell-depleted mice were prepared by in vivo administration of anti-TCR Vγ1 mAb or anti-TCR γδ mAb, respectively. We confirmed nearly complete depletion of TCR Vγ1+ T cells or TCR γδ+ T cells in the peritoneal cavity and spleen on day 7 of i.p. administration of 0·25 mg anti-TCR Vγ1 mAb or anti-TCR γδ mAb (Fig. 4). The TCR Vγ1+ T-cell-depleted mice showed significantly higher numbers of bacteria than control mice in both the spleen and liver on day 5 of infection with L. monocytogenes (Fig. 5). The bacterial burden in the spleen and liver of the TCR Vγ1+ T-cell-depleted mice was nearly the same as that of the TCR γδ T-cell-depleted mice (Fig. 5) which have been reported to show impaired early protection to L. monocytogenes infection.4 These results clearly demonstrated that TCR Vγ1+ T cells are the dominant population of TCR γδ T cells in protection against L. monocytogenes during the early phase of the primary infection.
Figure 4.
Depletion of TCR Vγ1+ T cells or TCR γδ+ T cells after administration of anti-TCR Vγ1 or anti-pan TCR γδ mAb. Mice were i.p. inoculated with anti-TCR Vγ1 mAb 2·11 or anti-pan TCR γδ mAb UC-7 2 days before infection with 1×103L. monocytogenes. PEC and spleen cells were obtained on day 7 after mAb administration and stained with FITC-conjugated anti-TCR Vγ1 mAb, PE-conjugated anti-TCR γδ mAb and APC-conjugated anti-CD3 mAb as described in the Materials and Methods. All panels show the profile of TCR γδ and Vγ1 expression in CD3+ cells. The data shown are representatives of data from individual analysis of 10 mice.
Figure 5.
Effects of administration of anti-TCR Vγ1 or anti-pan TCR γδ mAb on elimination of bacteria from the spleen and liver. Mice were treated with 0·25 mg of anti-TCR Vγ1 or anti-pan TCR γδ mAb on day −2 and i.p. infected with 1×103L. monocytogenes on day 0. The numbers of L. monocytogenes in the spleen and liver were determined on day 5 of the infection. Values are means±SD for groups of four mice. Two independent analyses showed nearly the same result. The results shown are representatives of the analysis. *P <0·05 compared to control.
DISCUSSION
We have previously shown the early appearance of γδ T cells preceding αβ T cells17 and the protective roles of the γδ T cells in vivo in listerial infection in mice.4 We have also reported that the TCR γδ T cells of mice infected with L. monocytogenes predominantly express Vγ1 and Vδ6 genes by reverse transcription–polymerase chain reaction analysis.7 However, it is not known whether Vγ1+ cells play a part in protection against L. monocytogenes. In this study, we used a mAb specific to TCR Vγ114 to analyse the frequency of Vγ1+ T cells directly and its significance in protection against L. monocytogenes. The analyses demonstrated that Vγ1+ T cells are the dominant population of γδ T cells in protection against L. monocytogenes at an early phase of the primary infection.
Appearance of γδ T cells with restricted V-region repertoire has been reported in several infections in mice. The present report demonstrated that Vγ1+γδ T cells increased in the spleen, liver and peritoneal cavity of mice after i.p. L. monocytogenes infection. Belles et al. generated a mAb specific for TCR Vδ6·3 and showed that the Vδ6·3+ population represented around 40% of γδ T cells in the spleen and liver of L. monocytogenes-infected mice.18 These results are in agreement with previous reports showing that Vγ1 and Vδ6 genes are predominantly expressed by the γδ T cells induced by L. monocytogenes infection.7,8 Since preferential co-expression of Vγ1 and Vδ6 has been reported at clonal level,9,12,13 the γδ T cells in L. monocytogenes-infected mice may have restricted TCR V-region repertoire which co-express Vγ1 and Vδ6. Similar skewing of the TCR repertoire to Vγ1 and Vδ6 has been observed on γδ T cells induced by Salmonella choleraesuis19 and Toxoplasma gondii.20 The appearance of the γδ T cells with the same V-region repertoire in various infections suggest that the Vγ1/Vδ6+γδ T cells may recognize Ag which is shared by various pathogens or some self Ag which is induced by the infections as discussed below.
Heat-shock protein (hsp) 60 is a possible ligand of the Vγ1+γδ T cells induced by L. monocytogenes infection. O'Brien's group has reported that T-cell hybridomas expressing Vγ1 and Vδ6 preferentially respond to mycobacterial hsp 60 and its mammalian homologue.9,12,13 Since the γδ T cells in the early stage of L. monocytogenes infection consist of approximately 70% Vγ1+ T cells (Fig. 3) and 40–60% of Vδ6·3+ T cells,18 a population of the γδ T cells must express both Vγ1 and Vδ6. Therefore, it is quite possible that the γδ T cells induced by L. monocytogenes infection contain Vγ1/Vδ6+ T cells which recognize hsp 60. In agreement with that assumption, it was demonstrated that γδ T cells from the peritoneal cavity of mice infected i.p. with L. monocytogenes responded to mycobacterial hsp 60 and its peptide, which share amino acid sequence with mammalian hsp 60.4 All these results suggest that hsp 60 is a ligand of Vγ1+γδ T cells induced by L. monocytogenes infection. This is further supported by the observations that induction of Vγ1/Vδ6-expressing γδ T cells is correlated with hsp 60 expression by pathogens or infected host cells.7,19,20
It is also possible that the Vγ1+γδ T cells recognize some listerial peptide Ag. In the human system, it has been reported that peripheral γδ T cells increase in the course of listeriosis.21 Culture of human γδ T cells with proteinase K-sensitive L. monocytogenes Ag induced expansion of γδ T cells.22 Furthermore, incubation of γδ T cells with a listerial protein Ag listeriolysin O (LLO) and its oligopeptide induced proliferation of γδ T cells.23 Although our previous report demonstrated that the γδ T cells from L. monocytogenes-infected mice showed no response against a heat-killed L. monocytogenes preparation which activated conventional CD4+ TCR αβ T cells,4,7 some listerial Ag, such as LLO which is lost from the bacteria during heat treatment, is a ligand of the Vγ1+γδ T cells. To clarify Ag specificity of the L. monocytogenes-induced γδ T cells, we need further investigation.
This is the first report demonstrating that γδ T cells expressing a single V gene product exclusively participate in the protective response against infection. However, the mechanism of protection mediated by the Vγ1+γδ T cells is unclear. There are several possible mechanisms of the protection. It is possible that the γδ T cells participate in the protective response through secretion of cytokines which modulate macrophage functions such as interferon-γ and macrophage chemotactic factors.4 Alternatively, the γδ T cells could support eradication of bacteria by suppressing neutrophil abscess formation through unidentified molecular mechanism.5,24 Cytotoxic activity against infected cells could be another mechanism of γδ T-cell-mediated protection because γδ T cells are known to express cytotoxic molecules such as perforin.25 One, or a combination, of the mechanisms would be important in the Vγ1+γδ T-cell-mediated protection.
Roark et al. reported preferential appearance of canonical Vγ6 Vδ1-bearing γδ T cells in the liver during L. monocytogenes infection,26 which is inconsistent with our observations demonstrating predominance of the Vγ1-bearing γδ T cells. Although the cause of the difference is unclear at present, there are several possibilities. First, the different mouse strain used could have resulted in the different V-region repertoire of γδ T cells. We used Listeria-susceptible C3H/He (H-2k) mice but Roark et al. used Listeria-resistant C57BL/10 (H-2b) mice. However, different resistance to Listeria infection or H-2 may not be the major cause of the difference in the V repertoire of γδ T cells since our flow cytometric analysis demonstrated predominant appearance of Vγ1+γδ T cells in the liver of Listeria-resistant C57BL/6 (H-2b) mice after L. monocytogenes infection (T. Nakamura, data not shown). Another possibility is that the different method to detect the V-repertoire resulted in a different readout. We used freshly prepared cells for analysis while Roark et al. used γδ T-cell hybridomas to analyse the fine structure of γδ TCR. Unfortunately an antibody to TCR Vγ6 or Vδ1 is not available at present. Analysis of freshly prepared γδ T cells with such reagents may resolve the discrepancy between the two systems in the future.
Acknowledgments
We thank Dr P. Pereira and Dr J. A. Bluestone for kindly providing 2·11 and UC7-13D5 hybridoma, respectively.
Abbreviations
- Ag
antigen
- hsp
heat-shock protein
- IFN
interferon
- LLO
listeriolysin O
- mAb
monoclonal antibody
- PEC
peritoneal exudate cells
- TCR
T-cell receptor
REFERENCES
- 1.Haas W, Pereira P, Tonegawa S. Gamma/delta cells. Annu Rev Immunol. 1993;11:637. doi: 10.1146/annurev.iy.11.040193.003225. [DOI] [PubMed] [Google Scholar]
- 2.Chien YH, Jores R, Crowley MP. Recognition by γ/δ T cells. Annu Rev Immunol. 1996;14:511. doi: 10.1146/annurev.immunol.14.1.511. [DOI] [PubMed] [Google Scholar]
- 3.Kaufmann SH. γ/δ and other unconventional T lymphocytes: what do they see and what do they do? Proc Natl Acad Sci USA. 1996;93:2272. doi: 10.1073/pnas.93.6.2272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hiromatsu K, Yoshikai Y, Matsuzaki G, et al. A protective role of γ/δ T cells in primary infection with Listeria monocytogenes in mice. J Exp Med. 1992;175:49. doi: 10.1084/jem.175.1.49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mombaerts P, Arnoldi J, Russ F, Tonegawa S, Kaufmann SH. Different roles of αβ and γδ T cells in immunity against an intracellular bacterial pathogen. Nature. 1993;365:53. doi: 10.1038/365053a0. [DOI] [PubMed] [Google Scholar]
- 6.Skeen MJ, Ziegler HK. Induction of murine peritoneal γ/δ T cells and their role in resistance to bacterial infection. J Exp Med. 1993;178:971. doi: 10.1084/jem.178.3.971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Matsuzaki G, Hiromatsu K, Yoshikai Y, Muramori K, Nomoto K. Characterization of T-cell receptor γδ T cells appearing at the early phase of murine Listeria monocytogenes infection. Immunology. 1993;78:22. [PMC free article] [PubMed] [Google Scholar]
- 8.Fujise S, Matsuzaki G, Kishihara K, Kadena T, Molin T, Nomoto K. The role of p56lck in the development of γδ T cells and their function during an infection by Listeria monocytogenes. J Immunol. 1996;157:247. [PubMed] [Google Scholar]
- 9.O'Brien RL, Happ MP, Dallas A, Palmer E, Kubo R, Born WK. Stimulation of a major subset of lymphocytes expressing T cell receptor γδ by an antigen derived from Mycobacterium tuberculosis. Cell. 1989;57:667. doi: 10.1016/0092-8674(89)90135-9. [DOI] [PubMed] [Google Scholar]
- 10.Born WK, Hall L, Dallas A, et al. Recognition of a peptide antigen by heat shock-reactive γδ T lymphocytes. Science. 1990;249:67. doi: 10.1126/science.1695022. [DOI] [PubMed] [Google Scholar]
- 11.Happ MP, Kubo RT, Palmer E, Born WK, O'Brien RL. Limited receptor repertoire in a mycobacteria-reactive subset of γδ T lymphocytes. Nature. 1989;342:696. doi: 10.1038/342696a0. [DOI] [PubMed] [Google Scholar]
- 12.O'Brien RL, Fu YX, Cranfill R, et al. Heat shock protein Hsp 60-reactive γδ cells, a large diversified T lymphocyte subset with highly focused specificity. Proc Natl Acad Sci USA. 1992;89:4348. doi: 10.1073/pnas.89.10.4348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Roark CE, Vollmer MK, Cranfill RL, Carding SR, Born WK, O'Brien RL. Liver γδ T cells: TCR junctions reveal differences in heat shock protein-60-reactive cells in liver and spleen. J Immunol. 1993;150:4867. [PubMed] [Google Scholar]
- 14.Pereira P, Gerber D, Huang SY, Tonegawa S. Ontogenic development and tissue distribution of Vγ1-expressing γ/δ T lymphocytes in normal mice. J Exp Med. 1995;182:1921. doi: 10.1084/jem.182.6.1921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ohteki T, Seki S, Abo T, Kumagai K. Liver is a possible site for the proliferation of abnormal CD3+4−8− double-negative lymphocytes in autoimmune MRL-lpr/lpr mice. J Exp Med. 1990;172:7. doi: 10.1084/jem.172.1.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hiromatsu K, Matsuzaki G, Tauchi Y, Yoshikai Y, Nomoto K. Sequential analysis of T cells in the liver during murine listerial infection. J Immunol. 1992;149:568. [PubMed] [Google Scholar]
- 17.Ohga S, Yoshikai Y, Takeda Y, Hiromatsu K, Nomoto K. Sequential appearance of γ/δ-and α/β-bearing T cells in the peritoneal cavity during an i.p. infection with Listeria monocytogenes. Eur J Immunol. 1990;20:533. doi: 10.1002/eji.1830200311. [DOI] [PubMed] [Google Scholar]
- 18.Belles C, Kuhl AL, Donoghue AJ, et al. Bias in the γδ T cell response to Listeria monocytogenes. J Immunol. 1996;156:4280. [PubMed] [Google Scholar]
- 19.Emoto M, Naito T, Nakamura R, Yoshikai Y. Different appearance of γδ T cells during salmonellosis between Ityr and Itys mice. J Immunol. 1993;150:3411. [PubMed] [Google Scholar]
- 20.Hisaeda M, Sakai T, Nagasawa H, et al. Contribution of extrathymic γδ T cells to the expression of heat-shock protein and to protective immunity in mice infected with Toxoplasma gondii. Immunology. 1996;88:551. doi: 10.1046/j.1365-2567.1996.d01-694.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Jouen-Beades F, Paris E, Dieulois C, et al. In vivo and in vitro activation and expansion of γδ T cells during Listeria monocytogenes infection in humans. Infect Immun. 1997;65:4267. doi: 10.1128/iai.65.10.4267-4272.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Munk ME, Elser C, Kaufmann SHE. Human γ/δ T-cell response to Listeria monocytogenes protein components in vitro. Immunology. 1996;87:230. doi: 10.1046/j.1365-2567.1996.470549.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ying G, Ziegler HK, Safley SA, Niesel DW, Vaidya S, Klimpel GR. Human T-cell recognition of Listeria monocytogenes, recognition of listeriolysin O by TCR αβ+ and TCR γδ+ T cells. Infect Immun. 1995;63:2288. doi: 10.1128/iai.63.6.2288-2294.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Fu Y-X, Roark CE, Kelly K, et al. Immune protection and control of inflammatory tissue necrosis by γδ T cells. J Immunol. 1994;153:3101. [PubMed] [Google Scholar]
- 25.Nakata M, Smith MJ, Norihisa Y, et al. Constitutive expression of Pore-forming protein in peripheral blood γ/δ T cells: implication for their cytotoxic role in vivo. J Exp Med. 1990;172:1877. doi: 10.1084/jem.172.6.1877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Roark CE, Volluer MK, Campbell PA, Born WK, O'Brien RL. Response of a γδ+ T cell receptor invariant subset during bacterial infection. J Immunol. 1996;156:2214. [PubMed] [Google Scholar]