INTRODUCTION
Acquired immune responses are dynamic processes dependent upon the co-ordinated interaction of antigen-trapping accessory cells and lymphocytes of the immune system in specialized secondary lymphoid tissues that include lymph nodes, Peyer’s patches and spleen. The function of the immune system is to clear infections or neoplastic changes locally wherever they appear while avoiding autoimmune responses and non-specific damage of unaffected tissues. Mature lymphocytes with a broad range of potential antigen specificities that are not autoimmune are generated in the primary lymphoid tissues. Many, if not all, of these circulate between the blood and the tissues by way of the lymph nodes. The majority of small lymphocytes recirculate between the blood and secondary lymphoid tissue either directly through post-capillary venules or indirectly via tissues such as the skin and the gut mucosa.1–5 Blood CD4+ and CD8+ T cells recirculate efficiently, whereas a subset of B cells and γδ T cells do not.6–8 T and B cells (particularly CD4+ T cells) from peripheral lymph nodes and from gut-associated lymphoid tissue tend to recirculate to the tissue of origin,3–5,9 although this tissue-specific recirculation is by no means absolute. Recirculating lymphocytes can, therefore, monitor tissues for the presence of foreign antigens and enhance the recruitment of antigen-specific cells to the local lymph node. This tissue-specific immune surveillance by lymphocytes is facilitated by a network of lymph nodes distributed strategically around the body and connected by a system of lymphatic vessels.
The purpose of this article is to review recent advances in our understanding of the dynamics of local immune cell and cytokine responses in the two strikingly different compartments of afferent and efferent lymph that, respectively, enter and exit peripheral lymph nodes. The emphasis will be on studies in afferent and efferent lymph entering and exiting skin-draining lymph nodes in ruminants where lymphatic cannulation is relatively easy to perform.
AFFERENT AND EFFERENT LYMPH COMPARTMENTS
Afferent lymphatics are blind-ended vessels that arise in and permeate the tissues and selectively convey lymphocytes and antigen-presenting accessory cells into the subcapsular and medullary sinuses of lymph nodes. Efferent lymphatics (usually only one or two for each node) carry lymphocytes and lymph plasma out of lymph nodes ultimately into the major efferent ducts and then back to the blood. Efferent lymph from regional lymph nodes may pass through one or more lymph nodes downstream before reaching a major efferent duct. Under physiological conditions, there are profound differences between afferent and efferent lymph flow rates, cell content and activation status of the cells (Fig. 1, Table 1)
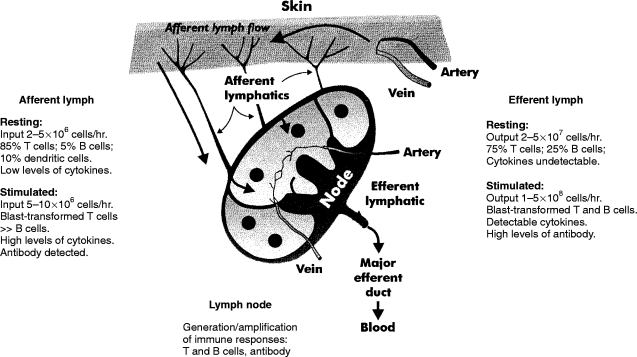
Figure 1.
Flow rates and cellular composition of lymph around single skin-draining lymph nodes of adult sheep. Afferent lymph contains antigen-presenting dendritic cells, T cells and a small proportion of B cells. Lymph flow and cell output rates are low. Efferent lymph contains >98% lymphocytes with a higher proportion of B cells and much higher flow and cell output rates than in afferent lymph.2–5 Greater than 90% of efferent lymphocytes are derived from the blood via high endothelial venules (HEV). L-selectin on lymphocytes is associated with adhesion to peripheral lymph node HEV. For the details of the cytokines studied to date, see Fig. 2 and the text.
Table 1.
The distribution of lymphocyte subsets in the lymph, blood, lymph nodes and skin of sheep
The physiological distribution of lymphocyte subsets in the skin, blood, afferent lymph, efferent lymph and lymph node of young adult sheep (aged 6 months to 3 years). Data from the literature and the authors' own laboratoroes (n > 10). Blank spaces = not determined. CD4+ T cells predominate proportionately over other lymphocyte subsets in both afferent and efferent lymph, whereas B cells are enriched in efferent lymph and lymph nodes compared to the skin and afferent lymph. γδ receptor+ T cells (γδ T cells) show a different distribution from the αβ T cells. As a proportion of other lymphocytes they are concentrated in the blood and tissues compared to within lymph nodes or in efferent lymph. T cells expressing the activation markers IL-2Rα (CD25) and MHC class-II DR and DQ are enriched proportionately in afferent lymph compared to efferent lymph and blood.
Afferent lymph
Analysis of afferent lymph gives an accurate picture of immune and inflammatory responses in the tissues as they occur. Afferent lymph contains antigen-presenting dendritic cells (ALDC), T cells (principally CD4+ T cells) and a small proportion of B cells, all migrating with a low cell output from the tissues into the local lymph node3,5,10–12 (Fig. 1, Table 1). This is consistent with continuous immune surveillance, involving dendritic cells and CD4+ T cells in particular, but with a lesser involvement of B cells. γδ receptor+ T cells (γδ T cells) are prevalent in the blood of ruminants11,12 and are the most abundant lymphocytes in normal ruminant skin (Table 1). However, they comprise a small population in afferent lymph compared to αβ T cells (Table 1) which suggests that they reside in the tissues longer than αβ T and B cells, and raises the possibility that a proportion of them may die there unless stimulated. This would be consistent with their envisaged role as surveillance cells for epithelial surfaces, with a lesser tendency to recirculate than αβ T cells.12
Afferent lymph T cells exhibit an activated phenotype compared to efferent lymph cells, with a higher proportion of T cells expressing interleukin (IL)-2Rα (CD25) and major histocompatibility complex (MHC) class II DQ antigens (Table 1). CD4+ T cells in ‘resting’ afferent lymph also express mRNA for the cytokines IL-2, IL-4, IL-6, IL-10 and interferon-γ (IFN-γ) (Fig. 3 and unpublished). This activated phenotype may be important for the migration of the T cells through the tissues and into afferent lymph. Afferent lymph plasma under physiological conditions contains the cytokines IL-1β, tumour necrosis factor-α (TNF-α), granulocyte–macrophage colony-stimulating factor (GM-CSF) and IL-8,13,14 and other biological activities generated in the tissue of origin.3,13–15 These cytokines can potentially influence cellular responses within the lymph node. Afferent lymph is important in maintaining lymph node integrity and architecture,16 and the cytokines/mediators and/or the afferent cells that they support are candidates for this function. These include the cytokines TNF-α and lymphotoxin (TNF-β) which are associated with the development and maintenance of lymph nodes and lymphoid architecture.17–19
Figure 3.

Constitutive cytokine gene transcription by ‘resting’ afferent versus efferent CD4+ T cells. Lymph flow was allowed to settle for 10 days prior to harvesting the cells. 2×107 CD4+ T cells isolated from afferent or efferent lymph by magnetic activated cell sorting (MACS) (>94% purity) were incubated with 0·01mM of α-amatin to block mRNA translation. RNA from 1×107 CD4+ T cells was purified and subjected to reverse transcription–polymerase chain reaction (RT–PCR) using primers for APTase, ovine IL-4, IL-6, IL-10 and IFN-γ. Ten millilitre samples of amplified DNA were slot-blotted onto Hybond-N® using a Biodot® manifold and visualised by Southern hybridization with 32P-labelled cytokine-specific oligonucleotides (internal sequence). The results show that activated CD4+ T cells exhibiting type 1 (IFN-γ) and type 2 (IL-4, IL-6 and IL-10) cytokine expression are concentrated in afferent lymph.
Efferent lymph
Efferent lymph contains >99% lymphocytes with a high proportion of CD4+ T cells and B cells compared to CD8+ and γδ T cells. Lymph flow and cell output is high (Fig. 1, Table 1). Greater than 90% of lymphocytes in efferent lymph are derived from the blood via high endothelial venules (HEV) and less than 4% are generated within the node under normal physiological conditions.2 Greater than 90% of lymphocytes enter the lymph node by HEV as compared with <10% from afferent lymph,3–5 demonstrating that the predominant recirculation route in the periphery for small lymphocytes is from the blood to the lymph nodes. The majority of efferent lymphocytes differ from afferent lymphocytes in not having an activated phenotype (Table 1 and Fig. 2). Efferent lymph plasma drains from within the lymph node, but cytokine levels, with the exception of GM-CSF, are undetectable or very low.
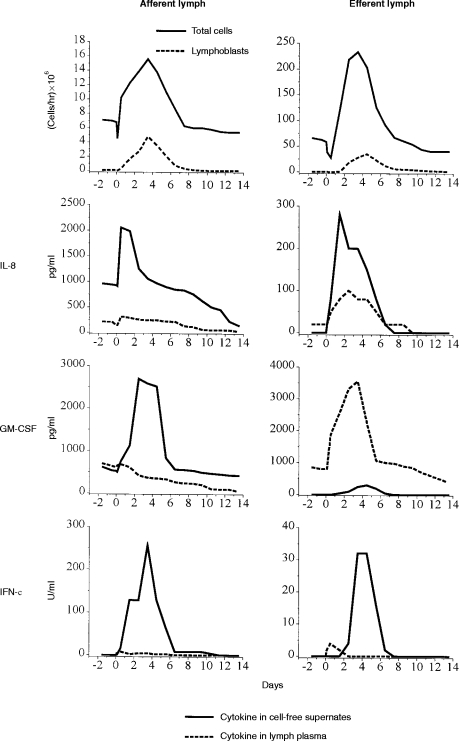
Figure 2.
Differential cytokine production in afferent and efferent lymph during an anamnestic immune response to 1 mg orf virus antigen given intra-dermally to previously infected sheep (refs 14, 21 and unpublished). Mean total and lymphoblast output for each group of five sheep is shown for comparison (top graphs). Lymph plasma contains activity generated in the skin (afferent lymph) or prefemoral lymph node (efferent lymph) as well as by cells in the lymph. Cytokine production by 4×106 lymph cells in 1 ml medium after culture for 18 hr in the absence of exogenous antigen was assayed in cell-free supernates (CFS) as a measure of the activation status of the cells following in vivo antigen challenge. Three examples of cytokines are shown. Note the difference in y-axis scales between afferent and efferent lymph. The chemokine IL-8 is produced within 12 hr of antigen challenge in both lymph compartments, most notably by afferent lymph cells. Low titres were detected in lymph plasma (there is no evidence for inhibitors in lymph plasma). GM-CSF is a haemopoietin active on macrophages, neutrophils and dendritic cells. Large quantities are produced within the lymph node and by afferent but not efferent lymph cells. This might indicate a consequence of accessory cell–lymphocyte interactions in afferent lymph and the lymph node. IFN-γ, like GM-CSF, is produced rapidly and at high titre by afferent lymph cells compared to efferent lymph cells where production is low and delayed by comparison. Production by cells correlated with the appearance of lymphoblasts. Differential and temporal appearance of the cytokines in the lymph compartments was similar in sheep reinfected with orf virus where CD4+/CD45RA− T lymphoblasts were the principal source of GM-CSF, IFN-γ (and IL-2).14,21 The results show a marked concentration of activated cytokine-producing cells in afferent lymph compared to efferent lymph during the anamnestic immune response, consistent with an activated effector/memory cell population in afferent lymph. A function for IL-8 (which precedes IL-1β detection14) in the initial phase of the anamnestic immune/inflammatory response is also implied.
Recent studies have shown that peripheral blood B cells in sheep consist of two subpopulations that differ in their capacity to recirculate through lymph.6–8 The recirculating subpopulation has the phenotype CD21+, CD11b−, L-selectin+ and is detected in afferent and efferent lymph and lymph nodes. The second subpopulation comprises ≈30–50% of blood-borne B cells, has the phenotype CD21−, CD11b+, L-selectin− and shows enhanced migration to the spleen. The significance of these phenotypes and their distribution is not known, but the presence of the two subpopulations may represent different stages of B cell activation or a division into ‘naïve’ and ‘memory’ cells as described for T cells.
Kimpton and colleagues have shown that the recirculation of the different lymphocyte subsets (non-random T and B distribution) is the same in fetal lambs (which are immunologically virgin) as in adult animals. This demonstrates that the pathways of lymphocyte recirculation are imprinted during fetal life in the absence of antigen and remain stable through adult life.20
IMMUNE RESPONSES IN AFFERENT AND EFFERENT LYMPH COMPARTMENTS
During immune responses, profound changes take place in the cellular composition and activation status of the cells in afferent and efferent lymph. The gross changes are shown in Fig. 1, and examples of changes in cytokine production following antigen challenge are shown in Fig. 2.
Afferent lymph
Following antigen challenge or infection of the tissues, there is an early (and usually transient) inflammatory response during which neutrophils can be detected in the afferent lymph.3 In sheep cutaneously challenged with the larvae of Lucilia cuprina (the cause of blowfly strike), or the parapoxvirus orf virus, rapid increases in afferent lymph plasma levels of the pro-inflammatory cytokines IL-1β, TNF-α, and the chemokine IL-8 were detected concomitant with the appearance of neutrophils in the lymph.13,14 Following this phase there is an increase in the frequency of lymphoblasts, particularly CD4+ T-cell blasts, and in sheep reinfected with orf virus these cells are the principal source of the lymphokines IL-2, IFN-γ, and GM-CSF (ref. 14 and unpublished; Fig. 2). As CD4+ T cells are the principal source of regulatory cytokines in the antigen-specific phase of immune responses, this might have the effect of providing the appropriate tissue environment in which other lymphocytes and inflammatory cells could operate to clear antigen or infectious agents. In primary immune responses in afferent lymph, the appearance of lymphoblasts is delayed compared to anamnestic responses. Furthermore, antibodies are detected during anamnestic responses. In spite of all these changes, in most studies afferent lymph flow rate and cell output did not increase significantly during either primary or anamnestic immune responses. The exception to this is in sheep reinfected with orf virus14 or challenged with orf virus antigen (Fig. 2) where cell output can triple. Changes in dendritic cells are discussed below.
Efferent lymph
In efferent lymph, three phases in the lymph response to antigen or infection have been described.2–5 Firstly, there is often a ‘shut down’ of lymphocyte output, which lasts for up to 24 hr. Secondly, there is a subsequent rise in efferent lymphocyte output (‘recruitment phase’) associated with increased blood flow to the node and thought to represent an increased influx of lymphocytes across HEV for rapid screening of antigen on APC within the node. Efferent lymphoblasts appear after this phase. These are effector T and B cells and do not recirculate. The lymph node is the principal site of B-cell blast, plasma cell and antibody production, all of which are concentrated in efferent as opposed to afferent lymph. Thirdly there is the resolution phase, the onset of which is dependent upon the continuing presence of antigen/pathogen in the tissues and during which lymph flow rate and cellular composition return to pre-stimulation levels. Shutdown and recruitment are particularly marked during anamnestic immune responses and resolution is more rapid than in primary responses.
Following a first or second encounter with antigen or a pathogen, a sequential appearance of CD4+ T cells followed by CD8+ T cells and/or B cells has been described in efferent lymph.5,9,21,22 This may represent different transit times through the node where CD4+ T cells are selected most rapidly.5,22 This could be to expedite their antigen-specific activation in order to provide cognate help and IL-222 (or other lymphokines) for CD8+ T- and B-cell activation and subsequent migration. Lymphoblasts exit the node in the same phenotypic sequence, and in sheep reinfected with orf virus, CD4+ and CD8+ T-cell blasts are the source of the lymphokines IL-2, GM-CSF and IFN-γ.21 The production of lymphokines by efferent lymph cells (using a cell to cell comparison) is low compared to that of afferent lymph cells14,21 (Figs 2 and 3). This is probably because only a small number of efferent lymph T cells are activated by antigen and cytokines within the node. These are effector cells subsequently recruited to the tissues. A similar sequential output of lymphocytes and lymphoblasts has been described in afferent lymph during orf virus reinfection in sheep.14
Whether CD4+, CD8+, γδ T- or B-cell output predominates in either lymph compartment depends on the antigen or infectious agent, and demonstrates the usefulness of lymphatic cannulation for studying the type of immune response generated by different pathogens.5,9,23 For example, in sheep reinfected with orf virus, CD4+ T cells predominate at all stages of infection,14,21 whereas in sheep infected with Toxoplasma gondii CD8+ T cells predominate in the later stages of infection,24 and in sheep infected with Salmonella abortus ovis B cells predominate.25
NAIVE, MEMORY AND EFFECTOR CELLS
A seminal study in sheep showed that T cells that were naïve with respect to antigen encounter (measured by CD45RA expression and low levels of a range of adhesion molecules) predominated in efferent lymph.26 This indicated that they had recirculated from the blood into lymph nodes via HEV where antigen-reactive cells clonally expand in number. Memory T cells (lacking CD45RA expression and exhibiting high levels of a range of adhesion molecules) predominated in afferent lymph indicating that they had recirculated by way of the tissues where they can react rapidly to recall antigens precisely where they penetrate the body’s innate defences.26 Westerman and Pabst have argued that this scheme may be premature, based upon a critical evaluation of more recent data.27 In fact, adult sheep popliteal and prefemoral efferent lymph contain an ≈1:1 ratio of CD45R+ and CD45R− T cells21 and not the >90% CD45R+ dominance described previously26 (Table 1). This indicates that the putative ‘memory/effector’ phenotype cells can cross HEV into peripheral lymph nodes. A recent study in rats led to the same conclusion.28
It is clear from studies in rodents that the division of naïve and memory cells based on the expression only of CD45R isoforms is inaccurate and does not reflect a dynamic change in expression of these molecules following activation.28–30 Furthermore, in antigen-naïve fetal lambs (where the placentation does not allow external antigens access to the fetus), both CD45RA+ and CD45RA− T cells were found in fetal blood and lymph, with enhanced migration of both T-cell subpopulations through the tissues and in afferent lymph compared to adult animals.20 This demonstrates that genuinely naïve T cells are either CD45RA+ or CD45RA− in the ovine fetus. L-selectin expression appears to be necessary for lymphocyte recirculation through peripheral lymphoid tissue,31 whereas other integrins are expressed by lymphocytes recirculating through other tissues, for example integrin β7 for intestinal tissues.32
The lack of CD45RA (sheep) or expression of CD45RO (cattle) in lymph may be more useful as markers of activated or effector T cells. Following orf virus antigen challenge or reinfection in sheep, CD4+ and CD8+, CD45RA− T cells were the principal source of IL-2, IFN-γ and GM-CSF in both afferent lymph (which does not contain CD45RA+ T cells) and efferent lymph14,21 (Fig. 2). In cattle, CD4+/CD45RO+ but not CD4+/CD45RO− T cells proliferate and produce multiple lymphokines rapidly after stimulation in culture.33
Orf virus-specific CD8+ cytotoxic T lymphocytes (CTL) that are directly cytotoxic for orf virus-infected autologous target cells (effector as opposed to precursor CTL) are also enriched in afferent lymph (and lack CD45RA) compared to efferent lymph.34 However, the proportion of cells expressing the CTL activation enzyme N-α-benzyloxy-carbonyl–lysine thiobenyl ester (BLT)-esterase is greatly reduced in afferent lymph compared to the infection skin site. This indicates that the activated cells are predominantly retained at the site of infection where they are required and do not migrate in large numbers to cause non-specific damage in adjacent tissues (ref. 34 and unpublished). Further work is required to define and map naïve, memory and effector cells as well as the function of the CD45R isoforms before the issue of naïve/memory T-cell phenotype and distribution can be resolved
DENDRITIC CELLS
Dendritic cells are potent antigen-presenting cells that initiate primary and anamnestic immune responses. Studies in mouse and man have shown that there is a maturational pathway for dendritic cells in the skin, from immature antigen-capturing epidermal Langerhans’ cells to cells which migrate and mature into potent antigen-presenting dendritic cells in the dermis and paracortical areas of lymph nodes.35–37
Skin-draining afferent lymph dendritic or veiled cells (ALDC) represent a migrating population of dermal dendritic cells (DDC) phenotypically similar, but not identical to epidermal Langerhans’ cells (LC) and lymph node DC.38–40 These migrating cells are not easily studied in mouse and man. ALDC transport antigen from the tissues to the paracortical areas of lymph nodes and are potent antigen-presenting cells for T cells in primary and anamnestic immune responses.38,39
Consistent with their antigen-presenting function, ruminant ALDC express high levels of MHC class I and II antigens, the co-stimulatory molecules CD80 and CD86 (B7.1 and B7.2), CD1, and a range of adhesion molecules including CD58 (leucocyte function-associated antigen-3; LFA-3), and CD44.38,39,41,42 The expression of CD58 and CD11a (LFA-1) on ruminant and canine ALDC is associated with DC–T-cell cluster formation that is an essential step in antigen presentation.43 A proportion of ovine ALDC are phagocytic in vivo,44 about 40% express functional FcγR-II45 and many bind immunoglobulin M (IgM).10 Interestingly, a proportion of ALDC also express CD21 (the CR2 receptor)42 which confers on these cells the ability to bind antigen-C3d complexes which have recently been shown to be effective in generating antibody responses via interaction with B cells.46 These phenotypes provide ALDC with several effective means of acquiring and presenting protein and glycolipid47 antigens to initiate both primary and anamnestic immune responses. A small proportion of the migrating ALDC are thought to survive as interdigitating cells (IDC) in the paracortical areas of lymph nodes where they can interact with T cells and initiate (and maintain) immune responses.38,39 The relationship between ALDC and follicular DC (FDC) in the follicles of lymph nodes is not known. ALDC transport antigen in a form suitable for stimulating T cells whereas FDC may trap lymph plasma-borne antigen for B-cell recognition.
In ruminants, as in mouse and man, the ALDC exhibit phenotypic and functional heterogeneity. In cattle, two populations of ALDC have been identified with differential expression of CD11a related to differences in antigen presentation or allo-stimulation of T cells.48,49
In different mammals, the cytokines TNF-α, GM-CSF, IL-1β, IL-4, IFN-γ and IL-10 in particular, amongst others, appear either to regulate Langerhans’ cell maturation or to control aspects of dendritic cell antigen uptake and presentation to T cells. For example, TNF-α supports the survival of dendritic cells.50 IL-1β and GM-CSF differentially stimulate the expression of certain dendritic cell adhesion and co-stimulatory molecules (e.g. CD80), whereas IL-10 and IFN-γ suppress CD80.51–53 However, the role of the different cytokines is difficult to determine in rodents and man, as the studies are complicated by the use of different dendritic cells at different stages of maturation derived from either haemopoietic precursors or isolated from the tissues. Cannulated afferent lymph on the other hand provides a source of genuine antigen-presenting dendritic cells for study, which are obtained without using enzymes or complicated cell separation and/or culture techniques.
In studies with canine ALDC, the pro-inflammatory cytokines IL-1β and TNF-α stimulated while the anti-inflammatory cytokines IL-10 and IL-1 receptor antagonist (IL-1RA) inhibited ALDC cluster formation.43 In sheep, TNF-α and Il-1β (and possibly GM-CSF) are constitutively expressed in the skin and afferent lymph13–15 and are therefore available to regulate dendritic cell migration, maturation and function following antigen capture. Ovine ALDC survive in culture with TNF-α and increase in number if GM-CSF is also present.54 IFN-γ inhibits this survival/growth. TNF-α and GM-CSF injection into ovine skin stimulates the recruitment of Langerhans’ cells and DDC.55 Taken together, these results demonstrate that the above cytokines are capable of regulating the migration, differentiation and accessory function of dendritic cells in the skin and afferent lymph. The availability of afferent lymph provides an opportunity to dissect the cytokine control of dendritic cell phenotypic and functional heterogeneity that is currently a difficult area of investigation in rodents and man.
DC may ‘select’ the T and B cells transported into the node via the afferent lymph. Several ligand–receptor interactions between DC and T cells for T-cell activation have been described: those mediating contact/adhesion/cluster formation, antigen–MHC–T-cell receptor (TCR)-mediated signalling and co-stimulation (e.g. CD28-CD80/86 interaction).56 In primary immune responses, DC that have captured antigen by phagocytosis (natural IgM and/or complement-mediated), macropinocytosis or via mannose receptors become activated, but productive stimulation of T cells in the tissues is unlikely because of the lack of antigen-specific T cells. However, ALDC will transport antigen (along with plasma-borne antigen) to the lymph node where large numbers of HEV-derived lymphocytes can be screened for low affinity antigen-binding cells, after which MHC–peptide–TCR recognition and co-stimulation can proceed followed by clonal expansion of antigen-specific T cells. In anamnestic responses, enhanced antigen-uptake (e.g. via FcγRII) by DC will facilitate interaction with memory CD4+ T cells leading to rapid production of T and B effector cells. Migration of ALDC with antigen, T cells and inflammatory cytokines to the node augments amplification of the appropriate anamnestic response that takes place by accelerated recruitment of antigen-specific lymphocytes from the blood. The quality and quantity of MHC molecules on ALDC and T cells39,57 may dictate whether co-stimulation proceeds. Optimum levels will stimulate a full effector T-cell response whereas low levels will lead to no co-stimulation and T-cell anergy and death in the lymph node. Recent studies in rodents suggest that differences in the duration of antigen stimulation may be required to stimulate naïve versus memory/effector T-cell proliferation, and may dictate whether the cells proliferate or die.58 Co-stimulation by the CD28 pathway shortens the time for T-cell activation, reduces the number of TCR molecules needed to be triggered by antigen, and prevents cell death or T-cell unresponsiveness.58,59 The lack of CD28 mRNA in bovine γδ T cells42 indicates different activation and signalling requirements for these cells compared to αβ T cells60,61 upon interaction with DC. Indeed, their TCR has certain similarities with immunoglobulin receptors12 suggesting that γδ T cells might respond to non-processed molecules and co-stimulation by IL-2.
Dendritic cells do not appear in efferent lymph and therefore must accumulate and die in the lymph node. FAS/FAS-ligand mediated DC apoptosis is a likely mechanism which has been shown in rodent and human DC which have not clustered with T cells62,63 and, therefore, have not functioned as antigen-presenting cells. DC that have presented antigen in the lymph node are also rapidly cleared.63 Furthermore, if DC express peptides of endogenous pathogens, they may be eliminated by cytotoxic T cell-induced apoptosis.64
CONCLUSION
Studies on immune responses in afferent and efferent lymph of lymph nodes draining the tissues have contributed to the concept that primary immune responses are generated in the lymph nodes with a delay before antigen-specific effector cells arrive at the affected tissue. Providing memory cells are still available, there is the possibility of an immediate but limited local anamnestic response in the tissues. This would involve memory cells recruited from the blood, supplemented shortly thereafter by a more dramatic tissue influx of antigen-specific lymphocytes from the lymph node which is the site of effector T- and B-cell amplification during anamnestic immune responses.
These studies also underline the importance of dendritic cells in immune surveillance of the tissues, initiation of primary immune responses, and interaction with putative effector T cells (CD4+ T cells in particular) upstream of the lymph node during anamnestic responses. The interaction of T cells with dendritic cells in the tissues as well as within lymph nodes may be important not only in generating immune responses but also in peripheral tolerance.35
There is no convincing division of type 1 and type 2 immune responses in ruminants, although Meeusen and colleagues have hypothesized that a type 2-inducing environment in the mucosal tissues and a type 1-inducing environment in the skin may direct tissue-specific migration of lymphocytes (gut versus skin).65 Recent studies in rodents and man locating cytokine production to cells in vivo and ex vivo indicate that T helper 1 (Th1) cells (that produce IL-2, IFN-γ, TNF-α, IL-12) are more frequently detected than Th2 cells (IL-4, IL-6, IL-10, IL-5) in the tissues, even in allergic individuals.66 In sheep, IFN-γ mRNA is detected more reproducibly than IL-4 mRNA in gastric efferent lymph T cells during nematode parasite challenge (C. McInnes, unpublished) and IgA (a mucosal type 2 antibody class) is detected in prefemoral efferent lymph as well as in skin washes.67,68 In addition, T cells migrating from the skin contain transcripts for IL-4, IL-6 and IL-10 as well as IFN-γ genes (Fig. 3).
Afferent lymph and the cells it contains may play a role along with endothelium-specific adhesion molecules in maintaining non-antigen-dependent tissue-biased lymphocyte recirculation as well as delivering antigen for primary and secondary immune responses and maintaining the integrity of lymph nodes. The presence of activated cells and cytokines in afferent lymph under physiological conditions would support this. The dendritic cell might ‘select’ lymphocytes for migration in afferent lymph through clustering as well as directing the type of immune response generated in the tissues. The expression of adhesion receptors on tissue endothelium allows the recruitment of lymphocytes expressing appropriate tissue-specific ligand molecules in response to chemotactic cytokines.1,69,70 Dendritic cells that are phenotypically and biochemically adapted to the tissues in which they reside may present different types of antigens to the recruited T cells. For example FcεR+ DC could be specialised for allergen detection. These can interact with, and influence, the effector phenotype of T cells through the expression of distinct co-stimulatory molecules and cytokines (IL-10 versus IL-12 for example). This does not exclude the involvement of other antigen-presenting cells during anamnestic responses.
There are many immunological hypotheses that can be tested experimentally in the outbred species amenable to lymphatic cannulation, several of which provide excellent models for human organ-specific disease.
Acknowledgments
The authors are currently sponsored by the Scottish Office Agriculture, Environment and Fisheries Department (DMH); the Wellcome Trust (HRPM) and the BBSRC (JH). Thanks to Wayne Hein, Chris Howard, Paul Wood and Jim Rothel for useful discussions.
References
- 1.Butcher EC, Picker LJ. Lymphocyte homing and homeostasis. Science. 1996;272:60. doi: 10.1126/science.272.5258.60. [DOI] [PubMed] [Google Scholar]
- 2.Hall JG, Morris B. The origin of the cells in the efferent lymph from a single lymph node. J Exp Med. 1965;121:901. doi: 10.1084/jem.121.6.901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hay JB, Cahill RNP. Lymphocyte migration patterns in sheep. In: Hay JB, editor. Animal Models of Immunological Processes. London: Academic Press; 1982. p. 97. [Google Scholar]
- 4.Miyasaka M, Trnka Z. Lymphocyte migration and differentiation in a large animal model: the sheep. Immunol Rev. 1986;91:87. doi: 10.1111/j.1600-065x.1986.tb01485.x. [DOI] [PubMed] [Google Scholar]
- 5.Kimpton WG, Washington EA, Cahill RNP. Lymphocyte recirculation and homing. In: Goddeeris BM, Morrison WI, editors. Cell-Mediated Immunity in Ruminants. Boca Raton: CRC Press; 1994. p. 109. [Google Scholar]
- 6.Young AS, Marston WL, Dessing, M, Dudler L, Hein WR. Distinct recirculating and non-recirculating B-lymphocyte pools in the peripheral blood are defined by co-ordinated expression of CD21 and L-selectin. Blood. 1997;90:4865. [PubMed] [Google Scholar]
- 7.Andrade WN, Johnston MG, Hay JB. The relationship of blood lymphocytes to the recirculating lymphocyte pool. Blood. 1998;91:1653. [PubMed] [Google Scholar]
- 8.Gupta VK, McConnell I, Dalziel RG, Hopkins J. Two B cell subpopulations have distinct recirculation characteristics. Eur J Immunol. 1998;28:1597. doi: 10.1002/(SICI)1521-4141(199805)28:05<1597::AID-IMMU1597>3.0.CO;2-Y. [DOI] [PubMed] [Google Scholar]
- 9.Mackay CR, Marston WL, Dudler L, Spertini O, Tedder TF, Hein WR. Tissue-specific migration pathways by phenotypically distinct subpopulations of memory T cells. Eur J Immunol. 1992;22:887. doi: 10.1002/eji.1830220402. [DOI] [PubMed] [Google Scholar]
- 10.Miller HRP, Adams EP. Reassortment of lymphocytes in lymph from normal and allografted sheep. Am J Path. 1977;87:59. [PMC free article] [PubMed] [Google Scholar]
- 11.Mackay CR, Kimpton WG, Brandon MR, Cahill RNP. Lymphocyte subsets show marked differences in their distribution between blood and the afferent and efferent lymph of peripheral lymph nodes. J Exp Med. 1988;167:1755. doi: 10.1084/jem.167.6.1755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hein WR. Gamma delta T cells in ruminants. In: Goddeeris BM, Morrison WI, editors. Cell-Mediated Immunity in Ruminants. Boca Raton: CRC Press; 1994. p. 19. [Google Scholar]
- 13.Egan PJ, Kimpton W, Seow H-F, Bowles VM, Brandon MR, Nash AD. Inflammation-induced changes in the phenotype and cytokine profile of cells migrating through skin and afferent lymph. Immunology. 1996;89:539. doi: 10.1046/j.1365-2567.1996.d01-776.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Haig DM, Deane D, Percival A, et al. The cytokine response of afferent lymph following orf virus infection of sheep. Vet Dermatol. 1996;7:11. doi: 10.1111/j.1365-3164.1996.tb00221.x. [DOI] [PubMed] [Google Scholar]
- 15.Haig DM, McInnes CJ, Hutchison G, Seow H-F, Reid HW. Cyclosporin-A abrogates the acquired immunity to cutaneous reinfection with the parapoxvirus orf virus. Immunology. 1996;89:524. doi: 10.1046/j.1365-2567.1996.940967.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Drayson MT, Ford WI. Afferent lymph and lymph borne cells: their influence on lymph node function. Immunobiology. 1984;168:362. doi: 10.1016/S0171-2985(84)80123-0. [DOI] [PubMed] [Google Scholar]
- 17.De Togni P, Goellner J, Ruddle NH, et al. Abnormal development of peripheral lymphoid organs in mice deficient in lymphotoxin. Science. 1994;264:703. doi: 10.1126/science.8171322. [DOI] [PubMed] [Google Scholar]
- 18.Pasparakis M, Alexopoulou L, Episkopou VS, Kollias G. Immune and inflammatory responses in TNF-α-deficient mice: a critical requirement for TNF-α in the formation of primary B cell follicles, follicular dendritic cell networks and germinal centres, and in the maturation of the humoral immune response. J Exp Med. 1996;184:1397. doi: 10.1084/jem.184.4.1397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mebius RE, Rennert P, Weissman IL. Developing lymph nodes collect CD4+ CD3− LTβ+ cells that can differentiate into antigen presenting cells, NK cells and follicular cells but not T or B cells. Immunity. 1997;7:493. doi: 10.1016/s1074-7613(00)80371-4. [DOI] [PubMed] [Google Scholar]
- 20.Kimpton WG, Washington EA, Cahill RNP. Virgin αβ and γδ T cells recirculate extensively through peripheral tissues and skin during normal development of the fetal immune system. Int Immunol. 1995;7:1567. doi: 10.1093/intimm/7.10.1567. [DOI] [PubMed] [Google Scholar]
- 21.Haig DM, Deane DL, Myatt N, et al. The activation status of ovine CD45R+ and CD45R− efferent lymph T cells after orf virus reinfection. J Comp Pathol. 1996;115:163. doi: 10.1016/s0021-9975(96)80038-7. [DOI] [PubMed] [Google Scholar]
- 22.Bujdoso R, Young P, Hopkins J, Allen D, McConell I. Non-random migration of CD4 and CD8 T cells: changes in the CD4: CD8 ratio and interleukin-2 responsiveness of efferent lymph cells following in vivo antigen challenge. Eur J Immunol. 1989;19:1779. doi: 10.1002/eji.1830191003. [DOI] [PubMed] [Google Scholar]
- 23.Bird P, Blacklaws B, Reyburn HT, et al. Early events in immune evasion by the lentivirus Maedi-Visna occurring within infected lymphoid tissue. J Virol. 1993;67:5187. doi: 10.1128/jvi.67.9.5187-5197.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Innes EA, Panton WR, Sanderson A, et al. Induction of CD4+ and CD8+ T cell responses in efferent lymph responding to Toxoplasma gondii infection: analysis of phenotype and function. Parasite Immunol. 1995;17:151. doi: 10.1111/j.1365-3024.1995.tb01017.x. [DOI] [PubMed] [Google Scholar]
- 25.Gohin I, Olivier M, Lantier I, Pepin M, Lantier F. Analysis of the immune response in sheep efferent lymph during Salmonella abortusovis infection. Vet Immunol Immunopathol. 1997;60:111. doi: 10.1016/s0165-2427(97)00090-1. [DOI] [PubMed] [Google Scholar]
- 26.Mackay CR, Marston W, Dudler L. Naïve and memory T cells show distinct pathways of lymphocyte recirculation. J Exp Med. 1990;171:801. doi: 10.1084/jem.171.3.801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Westerman J, Pabst R. How organ-specific is the migration of ‘naïve’ and ‘memory’ T cells. Immunol Today. 1996;17:278. doi: 10.1016/0167-5699(96)80545-7. [DOI] [PubMed] [Google Scholar]
- 28.Sparshott SM, Bell EB. Lymphocyte trafficking: CD4 T cells with a ‘memory’ phenotype (CD45RC−) freely cross lymph node high endothelial venules in vivo. Immunology. 1998;93:447. doi: 10.1046/j.1365-2567.1998.00471.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bell EB, Sparshott SM. Interconversion of CD45R subsets of CD4 T cells in vivo. Nature. 1990;348:163. doi: 10.1038/348163a0. [DOI] [PubMed] [Google Scholar]
- 30.Sparshott SM, Bell EB. Membrane CD45R isoform exchange on CD4 T cells is rapid, frequent and dynamic in vivo. Eur J Immunol. 1994;24:2573. doi: 10.1002/eji.1830241102. [DOI] [PubMed] [Google Scholar]
- 31.Catalina MDS, Carroll MC, Arizpe H, Takashima A, Estess P, Siegelman MH. The route of antigen entry determines the requirement for L-selectin during immune responses. J Exp Med. 1996;184:2341. doi: 10.1084/jem.184.6.2341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Abitorabi MA, MacKay CR, Jerome EH, Osorio O, Butcher EC, Erle DJ. Differential expression of homing molecules on recirculating lymphocytes from sheep gut, peripheral and lung lymph. J Immunol. 1996;156:3111. [PubMed] [Google Scholar]
- 33.Bembridge GP, MacHugh ND, McKeever D, et al. CD45RO expression on bovine T-cells – relation to biological function. Immunology. 1995;86:537. [PMC free article] [PubMed] [Google Scholar]
- 34.Haig DM, Hutchison G, Thomson J, Yirrell D, Reid HW. Cytolytic activity and associated serine protease expression by skin and afferent lymph CD8+ T cells during orf virus reinfection. J Gen Virol. 1996;77:953. doi: 10.1099/0022-1317-77-5-953. [DOI] [PubMed] [Google Scholar]
- 35.Steinman RM, Pack M, Inaba K. Dendritic cells in the T cell areas of lymphoid organs. Immunol Rev. 1997;156:25. doi: 10.1111/j.1600-065x.1997.tb00956.x. [DOI] [PubMed] [Google Scholar]
- 36.Lappin MB, Kimber I, Norval M. The role of dendritic cells in cutaneous immunity. Arch Dermatol Res. 1996;288:109. doi: 10.1007/BF02505819. [DOI] [PubMed] [Google Scholar]
- 37.Knight SC, Stagg A, Hill S, Fryer P, Griffiths S. Development and function of dendritic cells in health and disease. J Invest Dermatol. 1992;99:335. doi: 10.1111/1523-1747.ep12668601. [DOI] [PubMed] [Google Scholar]
- 38.Bujdoso R, Hopkins J, Dutia BM, Young P, McConnell I. Characterisation of sheep afferent lymph dendritic cells and their role in antigen carriage. J Exp Med. 1989;170:1285. doi: 10.1084/jem.170.4.1285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hopkins J, Dutia BM, Bujdoso R, McConnell I. In vivo modulation of CD1 and MHC class II expression by sheep afferent lymph dendritic cells. J Exp Med. 1989;170:1303. doi: 10.1084/jem.170.4.1303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lear A, Hutchison G, Reid H, Norval M, Haig DM. Phenotypic characterisation of the dendritic cells accumulating in ovine dermis following primary and secondary orf virus infections. Eur J Dermatol. 1996;6:135. [Google Scholar]
- 41.McKeever DJ, Awino E, Morrison WI. Afferent lymph veiled cells prime CD4+ T cell responses in vivo. Eur J Immunol. 1992;22:3057. doi: 10.1002/eji.1830221205. [DOI] [PubMed] [Google Scholar]
- 42.Howard CJ, Sopp P, Brownlie J, Parsons KR, Kwong L-S, Collins RA. Afferent lymph veiled cells stimulate proliferative responses in allogeneic CD4+ and CD8+ T cells but not γδ TCR+ cells. Immunology. 1996;88:558. doi: 10.1046/j.1365-2567.1996.d01-680.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Galkowska H, Wojewodzka U, Olszewski WL. Cytokines and adherence molecules involved in spontaneous dendritic cell-lymphocyte clustering in skin afferent lymph. Scand J Immunol. 1995;42:324. doi: 10.1111/j.1365-3083.1995.tb03663.x. [DOI] [PubMed] [Google Scholar]
- 44.Hall JG, Robertson D. Phagocytosis, in vivo, of immune complexes by dendritic cells in the lymph of sheep. Int Arch Allergy Appl Immunol. 1984;73:155. doi: 10.1159/000233457. [DOI] [PubMed] [Google Scholar]
- 45.Coughlan SN, Harkiss GD, Dickson L, Hopkins J. Fcγ receptor expression on sheep afferent lymph dendritic cells and rapid modulation of cell surface phenotype following Fcγ receptor engagement in vitro and in vivo. Scand J Immunol. 1996;43:31. doi: 10.1046/j.1365-3083.1996.d01-11.x. [DOI] [PubMed] [Google Scholar]
- 46.Dempsey PW, Allison MED, Akkaraju CC, Goodnow DT, Fearon DT. C3d as a molecular adjuvant – bridging innate and acquired immunity. Science. 1996;271:348. doi: 10.1126/science.271.5247.348. [DOI] [PubMed] [Google Scholar]
- 47.Ernst WA, Mather J, Cho S, et al. Molecular interaction of CD1b with lipoglycan antigens. Immunity. 1998;8:331. doi: 10.1016/s1074-7613(00)80538-5. [DOI] [PubMed] [Google Scholar]
- 48.McKeever DJ, MacHugh ND, Goddeeris BM, Awino E, Morrison WI. Bovine afferent lymph veiled cells differ from blood monocytes in phenotype and accessory function. J Immunol. 1991;147:3703. [PubMed] [Google Scholar]
- 49.Howard CJ, Sopp P, Brownlie J, Kwong LS, Parsons KR, Taylor G. Identification of two distinct populations of dendritic cells in afferent lymph that vary in their ability to stimulate T cells. J Immunol. 1997;159:5372. [PubMed] [Google Scholar]
- 50.Koch F, Heufler C, Kampgen E, et al. Tumor necrosis factor-α maintains the viability of epidermal Langerhans cells in culture, but in contrast to granulocyte–macrophage colony-stimulating factor, without inducing their functional maturation. J Exp Med. 1990;171:159. doi: 10.1084/jem.171.1.159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ozawa H, Nakagawa S, Tagami H, Aiba S. Interleukin-1β and granulocyte–macrophage colony-stimulating factor mediate Langerhans cell maturation differently. J Invest Dermatol. 1996;106:441. doi: 10.1111/1523-1747.ep12343589. [DOI] [PubMed] [Google Scholar]
- 52.Ozawa H, Aiba S, Nakagawa S, Tagami H. Interferon-γ and interleukin-10 inhibit antigen presentation by Langerhans cells for T helper type-1 cells by suppressing their CD80 (B7.1) expression. Eur J Immunol. 1996;26:648. doi: 10.1002/eji.1830260321. [DOI] [PubMed] [Google Scholar]
- 53.Morel AS, Quaratino S, Douek DC, Londei M. Split activity of interleukin-10 on antigen capture and antigen presentation by human dendritic cells: definition of a maturation step. Eur J Immunol. 1997;27:26. doi: 10.1002/eji.1830270105. [DOI] [PubMed] [Google Scholar]
- 54.Haig DM, Percival A, Mitchell J, Green I, Sargan D. The survival and growth of ovine afferent lymph dendritic cells in culture depends on tumour necrosis factor-α and is enhanced by granulocyte–macrophage colony-stimulating factor but inhibited by interferon-γ. Vet Immunol Immunopathol. 1995;45:221. doi: 10.1016/0165-2427(94)05341-o. [DOI] [PubMed] [Google Scholar]
- 55.Haig DM, Hutchison G, Green I, Sargan D, Reid HW. The effect of intradermal injection of GM-CSF and TNF-α on the accumulation of dendritic cells in ovine skin. Vet Dermatol. 1995;6:221. doi: 10.1111/j.1365-3164.1995.tb00067.x. [DOI] [PubMed] [Google Scholar]
- 56.Janeway CA, Bottomly K. Signals and signs for lymphocyte responses. Cell. 1994;76:275. doi: 10.1016/0092-8674(94)90335-2. [DOI] [PubMed] [Google Scholar]
- 57.Hopkins J, McConnell I, Dalziel RG, Dutia B. Patterns of MHC class-II expression by T-cell subsets in different immunological compartments, 2. Altered expression and cell function following activation in vivo. Eur J Immunol. 1993;23:2889. doi: 10.1002/eji.1830231124. [DOI] [PubMed] [Google Scholar]
- 58.Iezzi G, Karjalainen K, Lanzavecchia A. The duration of antigenic stimulation determines the fate of naïve and effector cells. Immunity. 1998;8:89. doi: 10.1016/s1074-7613(00)80461-6. [DOI] [PubMed] [Google Scholar]
- 59.Bachman MF, McKall-Faienza K, Schmits R, et al. Distinct roles for LFA-1 and CD28 during activation of naïve T cells: adhesion versus co-stimulation. Immunity. 1997;7:549. doi: 10.1016/s1074-7613(00)80376-3. [DOI] [PubMed] [Google Scholar]
- 60.Collins RA, Sopp P, Gelder KI, Morrison WI, Howard CJ. Bovine gamma delta TCR+ lymphocytes are stimulated to proliferate by autologous Theileria annulata-infected cells in the presence of IL-2. Scand J Immunol. 1996;44:444. doi: 10.1046/j.1365-3083.1996.d01-332.x. [DOI] [PubMed] [Google Scholar]
- 61.Hanrahan CF, Kimpton WG, Howard CJ, et al. Cellular requirements for the activation and proliferation of ruminant gamma delta T cells. J Immunol. 1997;159:4287. [PubMed] [Google Scholar]
- 62.Koppi TA, Toughbement T, Lewinsohn DM, Lynch DH, Alderson MR. CD40 ligand inhibits FAS/CD95-mediated apoptosis of human blood-derived dendritic cells. Eur J Immunol. 1997;27:3161. doi: 10.1002/eji.1830271212. [DOI] [PubMed] [Google Scholar]
- 63.Ingulli E, Mondino A, Khoruts A, Jenkins MK. In vivo detection of dendritic cell antigen presentation to CD4+ T cells. J Exp Med. 1997;185:2133. doi: 10.1084/jem.185.12.2133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Knight SC, Askonas BA, Macatonia SE. Dendritic cells as targets for cytotoxic T lymphocytes. Adv Exp Med Biol. 1997;417:389. doi: 10.1007/978-1-4757-9966-8_63. [DOI] [PubMed] [Google Scholar]
- 65.Meeusen ENT, Premier RR, Brandon MR. Tissue-specific migration of lymphocytes: a key role for Th1 and Th2 cells? Immunol Today. 1996;17:421. doi: 10.1016/0167-5699(96)10055-4. [DOI] [PubMed] [Google Scholar]
- 66.Schauer U, Jung T, Krug N, Frew A. Measurement of intracellular cytokines. Immunol Today. 1996;17:305. doi: 10.1016/0167-5699(96)30020-0. [DOI] [PubMed] [Google Scholar]
- 67.Rothel J. Melbourne, Australia: University of Melbourne; 1996. The immune response to Taenia ovis infection in sheep. PhD thesis. [Google Scholar]
- 68.Lloyd DH, Jenkinson DM, Mabon RM. Location of immunoglobulins in the skin of cattle and sheep. Res Vet Sci. 1979;26:47. [PubMed] [Google Scholar]
- 69.Murphy WJ, Tian ZG, Asai O, et al. Chemokines and T-lymphocyte activation. 2. Facilitation of human T cell trafficking in severe combined immunodeficiency mice. J Immunol. 1996;156:2104. [PubMed] [Google Scholar]
- 70.Bleul CC, Wu LJ, Hoxie JA, Springer TA, MacKay CR. The HIV co-receptors CXCR4 and CCR5 are differentially expressed and regulated on human T lymphocytes. Proc Natl Acad Sci USA. 1997;94:1925. doi: 10.1073/pnas.94.5.1925. [DOI] [PMC free article] [PubMed] [Google Scholar]