Abstract
Dendritic cells (DC) have recently been shown to play an important role in B-cell function. We have previously shown that DC can capture and retain unprocessed antigen in vitro and in vivo, and can transfer this antigen to naive B cells to initiate antigen-specific antibody responses. We also demonstrated that DC were providing B cells with isotype-switch signals independent of T cells but that T-cell help was essential for antibody production. In this study, using B cells and DC from wild type (WT) and CD40 knockout (CD40KO) mice we show that DC initiate proliferation of B cells independently of CD40, because WT or CD40KO DC could induce proliferation of WT or CD40KO B cells, but proliferation was greater in the absence of CD40. DC also provide B cells with survival signals as WT DC improved viability of B cells after a 5-day culture but survival was reduced in the absence of CD40 expression.
Studies by our group have shown that dendritic cells (DC) can retain and transport intact antigen (Ag), and that both in vivo and in vitro DC can transfer retained Ag to naive B cells. 1 This has also been shown for a novel population of DC-related cells. 2 In addition DC can give cell-bound signal(s) to B cells that are required for subsequent class switching. 1 This DC–B-cell interaction occurs in a primary antibody (Ab) response and is independent of any T-cell intervention, although T-cell help is essential for Ab synthesis. Similarly, human DC can influence Ab synthesis and proliferation of tonsillar B cells. 3–7 As we have shown that murine B cells express CD40L,8 and our (unpublished) and other studies have shown that DC can express CD40L,9 here we investigate the effects of murine DC on naïve murine B cells.
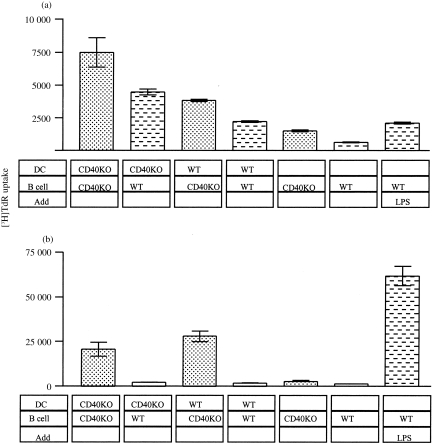
To determine whether CD40 has a role in DC–B-cell interaction, DC and B cells from wild type (WT) and CD40 knockout (CD40KO) mice10 were cultured over several days in replicate wells. At several time points, uptake of 3H-thymidine ([3H]TdR) was measured as an indication of B-cell proliferation. Figure 1 illustrates how WT and CD40KO B cells show low proliferation in the absence of any stimulation, although the growth of the latter is always marginally higher. However, in the first 24 hr, WT DC initiate proliferation of WT B cells to the same levels as lipopolysaccharide (LPS) (Fig. 1a). In the absence of CD40 expression on DC or B cells, proliferation is up to 5·5-fold higher. After 3 days, proliferation of WT B cells is reduced to 5% of LPS-induced proliferation while the proliferation of CD40KO B cells is 33–47%.
Figure 1.
Proliferation of Wild type (WT) and CD40 knockout (CD40KO) B cells cultured with WT and CD40KO DC for (a) 24 hr and (b) 3 days. Purified, irradiated DC14 and purified B cells8 were cultured at a ratio of 10 B cells to 1 DC. The cells were cultured in 96-well, round-bottomed plates for the required period, 1 μCi [3H]TdR added and cells harvested after 18 hr.
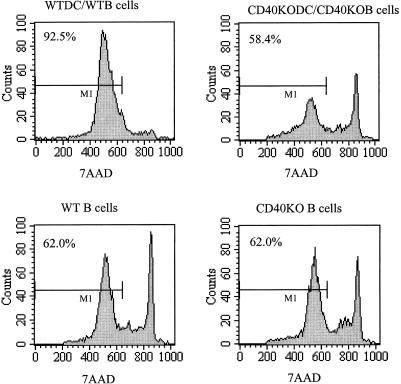
During these studies we also noticed that the viability of B cells was higher when cultured with DC. To investigate the role of CD40 in B-cell survival, purified DC and B cells from WT or CD40KO mice were cultured together for 5 days and the proportion of viable cells was assessed by the proportion of cells that excluded 7AAD (Fig. 2). Approximately 62% of unstimulated CD40KO and WT B cells are viable after 5 days in culture. While WTDC improved viability of B cells to greater than 92%, only 58% of B cells were viable following culture with CD40KO DC.
Figure 2.
Flow cytometry profiles of WT and CD40KO B cells either unstimulated or cultured with DC. Purified DC and B cells were cultured for 5 days and then treated with fluorescein isothiocyanate (FITC)–B220 and 7AAD to measure viability. The histograms show 7AAD exclusion of B220-expressing cells.
Dendritic cells have been shown to have a role in T-cell and B-cell functions, including proliferation and antibody isotype switching. 1,3–7 However, the molecules involved in these interactions are unknown. CD40 expression on DC and B cells is well established and it has been assumed that these cells then interact with CD40 ligand (CD40L) on activated T cells. However, we have shown that murine B cells express CD40L 8 as do murine DC (unpublished), human B cells, 11 and human DC. 9 This suggested the possibility of CD40L on DC, and B cells interacting with CD40 on B cells and DC, respectively. An earlier study found that human DC initiated proliferation of B cells, and it was proposed that CD40L on DC might provide B-cell CD40 with the proliferation signal. 6 Moreover, CD40 has been shown to have a role in B-cell survival12 but it was assumed to be via T cells.
Here we examined the role of DC and CD40 on B-cell proliferation and survival using DC and B cells from WT and CD40KO mice. We found that the culture of purified DC with purified B cells initiated proliferation of B cells as measured by [3H]TdR uptake. The levels of proliferation induced by WT DC were similar to LPS stimulation during the first 24 hr but dropped to 5% by day 3. Proliferation induced by DC is independent of CD40 on DC or B cells, as B cells from WT and CD40KO mice could be induced to proliferate by DC from WT and CD40KO mice. Surprisingly, day 3 proliferation was greatest in the absence of CD40 on DC and B cells, or when WT DC were cultured with CD40KO B cells. This probably reflects the proliferation of B cells that increases the initial ratio of 10 B cells to 1 DC. In this case, WT B cells could use neighbouring B-cell CD40 instead of DC CD40 to inhibit proliferation. This suggested that CD40 can down-regulate proliferation as in its absence proliferation increases. This observation that anti-CD40 signals to B cells inhibit proliferation is perhaps not surprising as it has been shown that anti-CD40 signals can arrest terminal B-cells differentiation and antibody secretion, which could not be overcome by cytokines or mitogens. 13
We also examined the role of CD40 in DC-mediated survival of B cells. Survival of WT DC/B cells was compared with CD40KO DC/B cells after a 5-day culture. We used CD40KO DC and B cells because CD40 is present on both cell types and in the absence on one cell type, CD40 on neighbouring cells may be used. While approximately 62% of unstimulated WT B cells are viable after 5 days in culture, viability is increased to greater than 92% by culture with DC. However, while 62% of unstimulated CD40KO B cells are viable, CD40KO DC do not improve viability. This shows that CD40 signalling is involved in B-cell survival. As we cultured CD40KODC with CD40KO B cells, we have not established whether DC provide CD40L or CD40 signals to B cells. We suggest that because CD40L is present on these DC, but B cells only express CD40L after encountering antigen,8 survival is provided by CD40L on DC to B-cell CD40.
Acknowledgments
We would like to thank Dr David Gray and Dr Elizabeth Adams for kindly providing several monoclonal antibodies. We also thank Dr Gray for providing the transgeneic animals. This work was funded by the Wellcome Trust, UK.
References
- 1.Wykes M, Pombo A, Jenkins C, MacPherson GG. Dendritic cells interact directly with naive B lymphocytes to transfer antigen and initiate class switching in a primary T-dependent response. J Immunol. 1998a;161:1313. [PubMed] [Google Scholar]
- 2.Berney C, Herren S, Power CA, Gordon S, Martinez-pomares L, Kosco-vilbois MH. A member of the dendritic cell family that enters B cell follicles and stimulates primary antibody responses identified by a mannose receptor fusion protein. J Exp Med. 1999;190:851. doi: 10.1084/jem.190.6.851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Fayette J, Dubois B, Vandenabeele S, et al. Human dendritic cells skew isotype switching of CD40-activated naive B cells towards IgA1 and IgA2. J Exp Med. 1997;185:1909. doi: 10.1084/jem.185.11.1909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fayette J, Dubois B, Caux C, Banchereau J, Briere F. Human dendritic cells can drive CD40-activated sIgD+ B cells to mount mucosal-type humoral response. Adv Exp Med Biol. 1995;378:401. doi: 10.1007/978-1-4615-1971-3_90. [DOI] [PubMed] [Google Scholar]
- 5.Dubois B, Massacrier C, Vanbervliet B, et al. Critical role of IL-12 in dendritic cell-induced differentiation of naive B lymphocytes. J Immunol. 1998;161:2223. [PubMed] [Google Scholar]
- 6.Dubois B, Vanbervliet B, Fayette J, et al. Dendritic cells enhance growth and differentiation of CD40-activated B lymphocytes [see comments] J Exp Med. 1997;185:941. doi: 10.1084/jem.185.5.941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bjorck P, Flores Romo L, Liu YJ. Human interdigitating dendritic cells directly stimulate CD40-activated naive B cells. Eur J Immunol. 1997;27:1266. doi: 10.1002/eji.1830270531. [DOI] [PubMed] [Google Scholar]
- 8.Wykes M, Poudrier J, Lindstedt R, Gray D. Regulation of cytoplasmic, surface and soluble forms of CD40 ligand in mouse B cells. Eur J Immunol. 1998;28:548. doi: 10.1002/(SICI)1521-4141(199802)28:02<548::AID-IMMU548>3.0.CO;2-2. [DOI] [PubMed] [Google Scholar]
- 9.Pinchuk LM, Klaus SJ, Magaletti DM, Pinchuk GV, Norsen JP, Clark EA. Functional CD40 ligand expressed by human blood dendritic cells is up-regulated by CD40 ligation. J Immunol. 1996;157:4363. [PubMed] [Google Scholar]
- 10.Kawabe T, Naka T, Yoshida K, et al. The immune responses in CD40-deficient mice: impaired immunoglobulin class switching and germinal center formation. Immunity. 1994;3:167. doi: 10.1016/1074-7613(94)90095-7. [DOI] [PubMed] [Google Scholar]
- 11.Grammer AC, Bergman MC, Miura Y, Fujita K, Davis LS, Lipsky PE. The CD40 ligand expressed by human B cells costimulates B cell responses. J Immunol. 1995;154:4996. [PubMed] [Google Scholar]
- 12.Gray D, Siepmann K, van Essen D, et al. B–T lymphocyte interactions in the generation and survival of memory cells. Immunol Rev. 1996;150:45. doi: 10.1111/j.1600-065x.1996.tb00695.x. [DOI] [PubMed] [Google Scholar]
- 13.Randall TD, Heath AW, Santos Argumedo L, Howard MC, Weissman IL, Lund FE. Arrest of B lymphocyte terminal differentiation by CD40 signaling: mechanism for lack of antibody-secreting cells in germinal centers. Immunity. 1998;8:733. doi: 10.1016/s1074-7613(00)80578-6. [DOI] [PubMed] [Google Scholar]
- 14.Steinman RM, Cohn ZA. Identification of a novel cell type in peripheral lymphoid organs of mice Morphology, quantitation; tissue distribution. J Exp Med. 1973;137:1142. doi: 10.1084/jem.137.5.1142. [DOI] [PMC free article] [PubMed] [Google Scholar]