Abstract
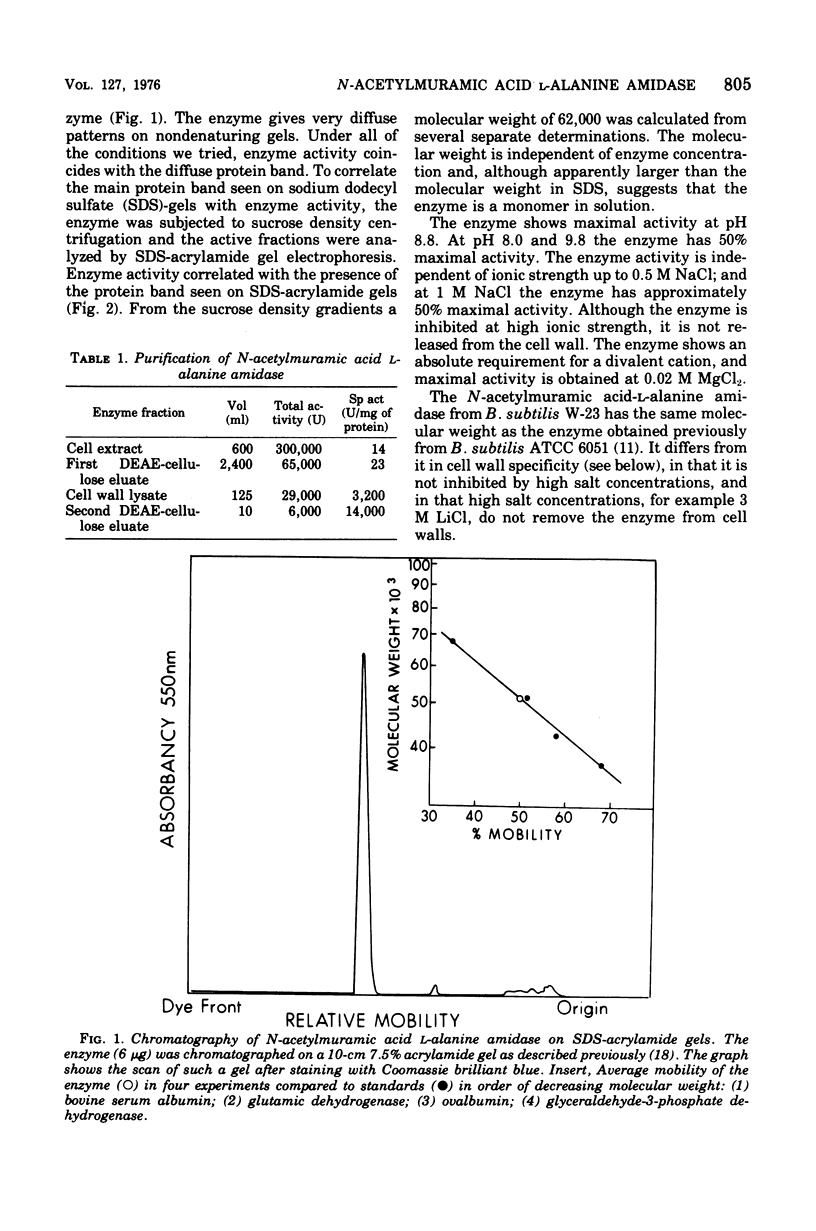
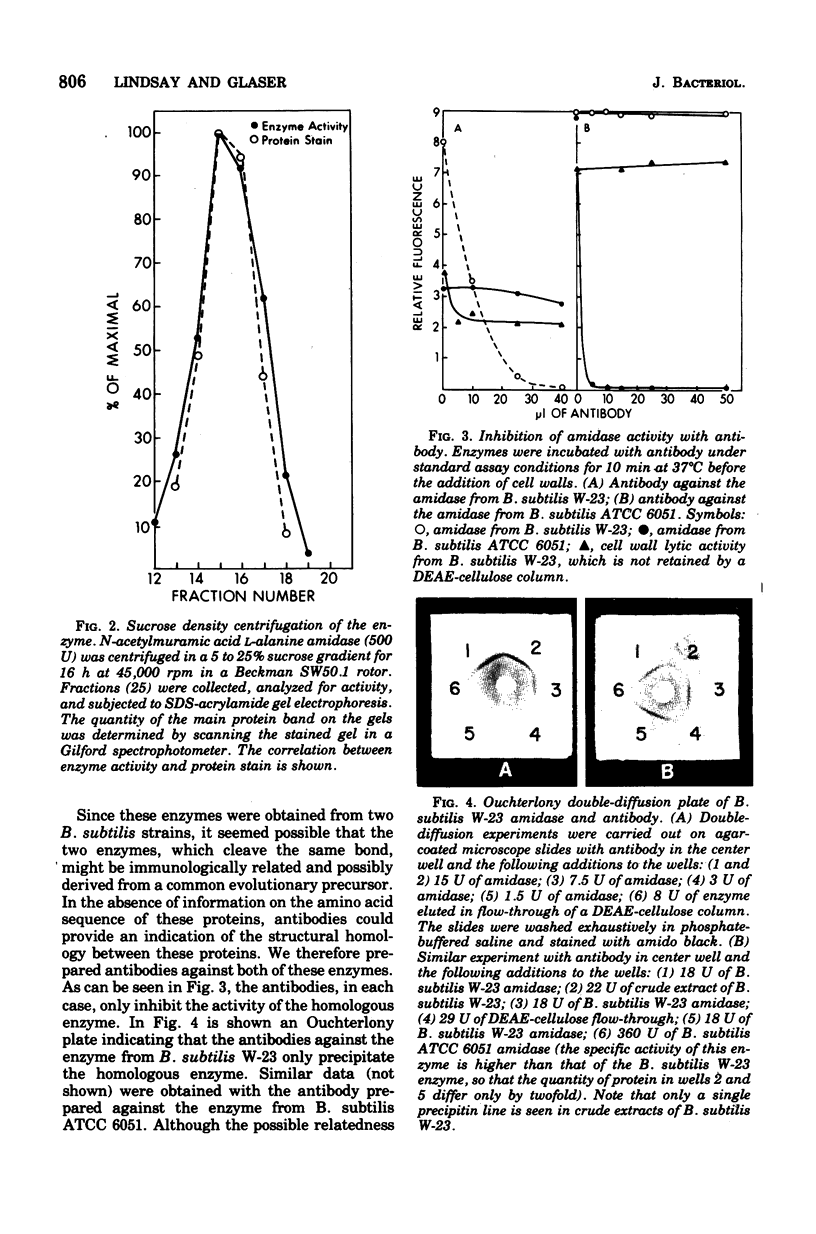
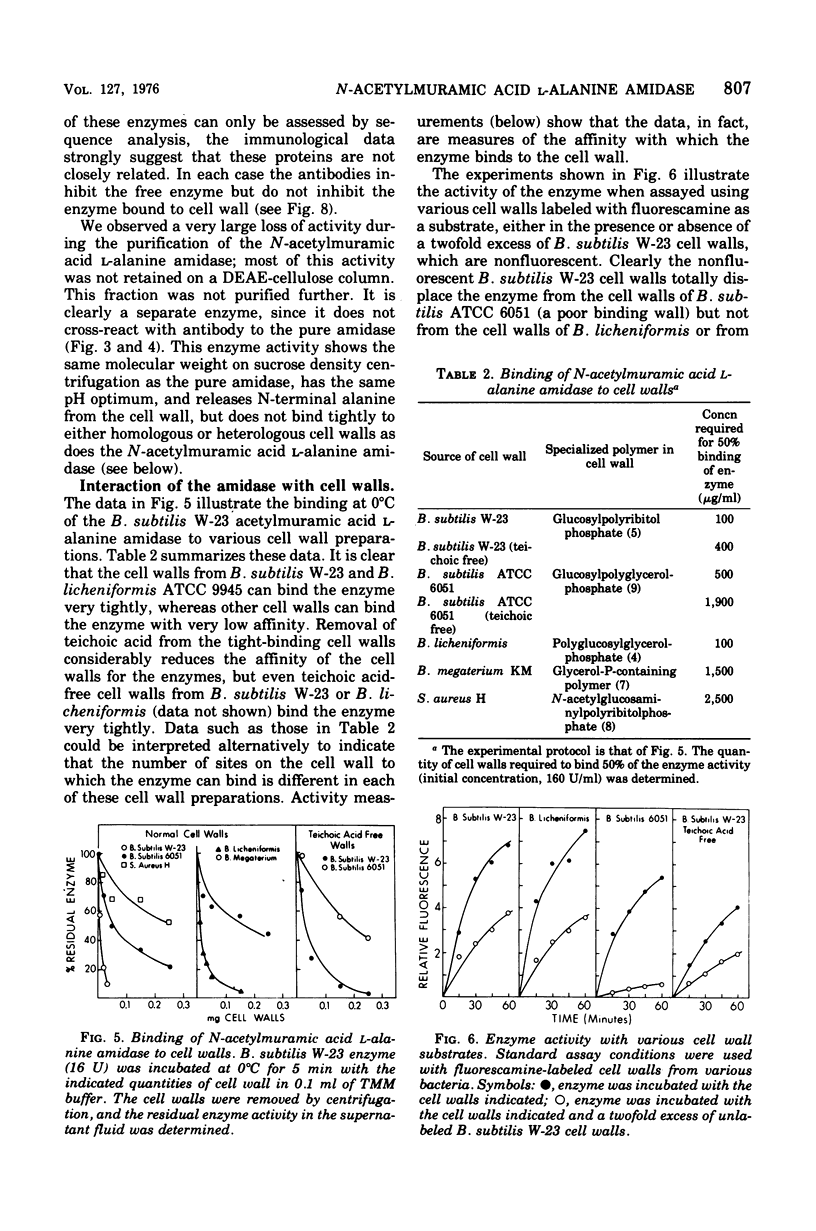
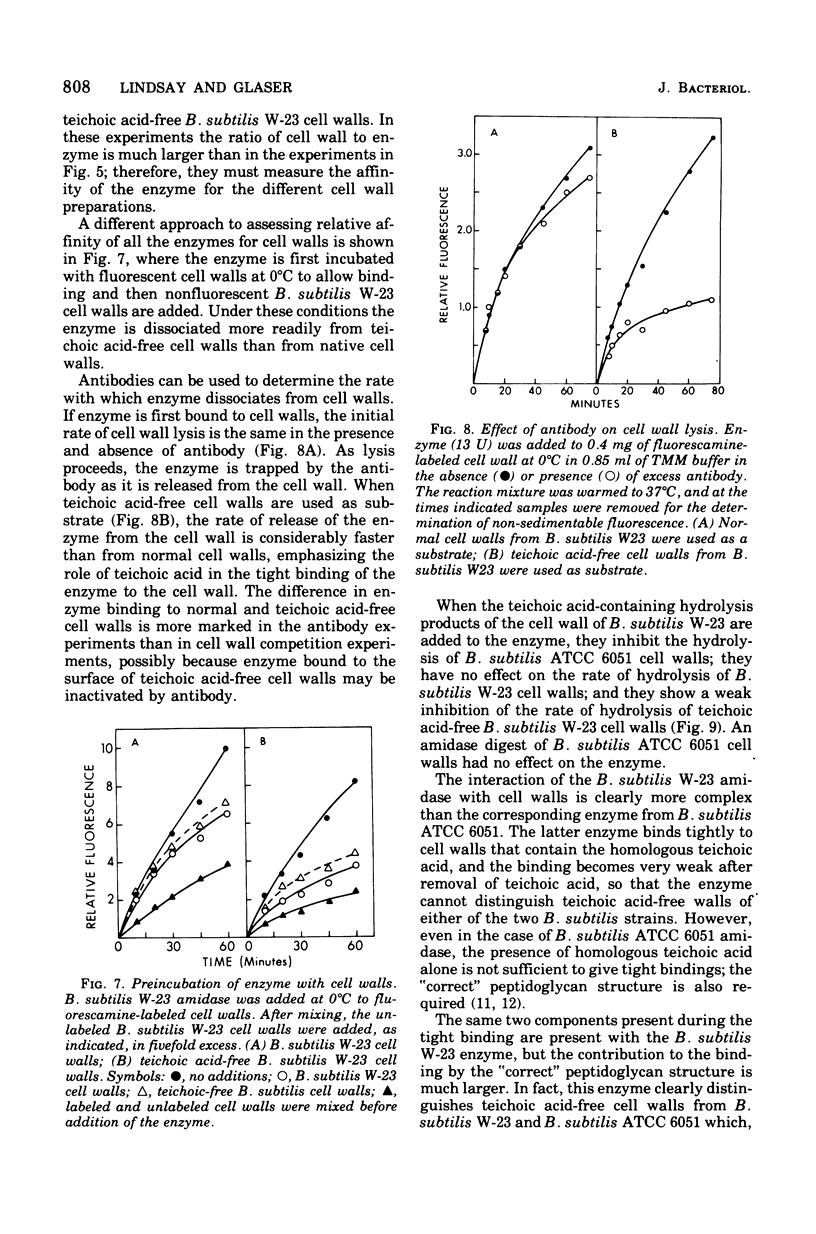
The N-acetylmuramic acid L-alanine amidase from Bacillus subtilis W-23 has been purified to apparent homogeneity. The enzyme is a monomer of molecular weight 51,000, which binds extremely tightly to homologous cell walls but not to heterologous cell walls, even of the closely related strain B. subtilis ATCC 6051. This difference in binding is only in part due to differences in teichoic acid between these two strains and to a large extent appears to represent differences in the arrangement of the peptidoglycan. A comparison of the amidase from B. subtilis W-23 and the enzyme previously purified from B. subtilis ATCC 6051 (Herbold and Glaser, 1975) shows that the two proteins, which cleave the same bond and are of the same size, do not cross-react immunologically and that the two enzymes are, therefore, not closely related in structure.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bracha R., Glaser L. In vitro system for the synthesis of teichoic acid linked to peptidoglycan. J Bacteriol. 1976 Mar;125(3):872–879. doi: 10.1128/jb.125.3.872-879.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown W. C., Fraser D. K., Young F. E. Problems in purification of a Bacillus subtilis autolytic enzyme caused by association with teichoic acid. Biochim Biophys Acta. 1970 Feb 11;198(2):308–315. doi: 10.1016/0005-2744(70)90063-x. [DOI] [PubMed] [Google Scholar]
- Burger M. M., Glaser L. The synthesis of teichoic acids. V. Polyglucosylglycerol phosphate and polygalactosylglycerol phosphate. J Biol Chem. 1966 Jan 25;241(2):494–506. [PubMed] [Google Scholar]
- Chin T., Burger M. M., Glaser L. Synthesis of teichoic acids. VI. The formation of multiple wall polymers in Bacillus subtilis W-23. Arch Biochem Biophys. 1966 Sep 26;116(1):358–367. doi: 10.1016/0003-9861(66)90042-7. [DOI] [PubMed] [Google Scholar]
- Fielder F., Glaser L. Assembly of bacterial cell walls. Biochim Biophys Acta. 1973 Dec 28;300(4):467–485. doi: 10.1016/0304-4157(73)90016-6. [DOI] [PubMed] [Google Scholar]
- GHUYSEN J. M. ALKALINE DEGRADATION OF THE PHOSPHOMUCOPOLYSACCHARIDE FROM CELL WALLS OF BACILLUS MEGATERIUM KM. Biochim Biophys Acta. 1964 Mar 2;83:132–134. doi: 10.1016/0926-6526(64)90063-1. [DOI] [PubMed] [Google Scholar]
- GLASER L., BURGER M. M. THE SYNTHESIS OF TEICHOIC ACIDS. 3. GLUCOSYLATION OF POLYGLYCEROPHOSPHATE. J Biol Chem. 1964 Oct;239:3187–3191. [PubMed] [Google Scholar]
- Ghuysen J. M. Use of bacteriolytic enzymes in determination of wall structure and their role in cell metabolism. Bacteriol Rev. 1968 Dec;32(4 Pt 2):425–464. [PMC free article] [PubMed] [Google Scholar]
- Heckels J. E., Archibald A. R., Baddiley J. Studies on the linkage between teichoic acid and peptidoglycan in a bacteriophage-resistant mutant of Staphylococcus aureus H. Biochem J. 1975 Sep;149(3):637–647. doi: 10.1042/bj1490637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herbold D. R., Glaser L. Bacillus subtilis N-acetylmuramic acid L-alanine amidase. J Biol Chem. 1975 Mar 10;250(5):1676–1682. [PubMed] [Google Scholar]
- Herbold D. R., Glaser L. Interaction of N-acetylmuramic acid L-alanine amidase with cell wall polymers. J Biol Chem. 1975 Sep 25;250(18):7231–7238. [PubMed] [Google Scholar]
- Higgins M. L., Shockman G. D. Procaryotic cell division with respect to wall and membranes. CRC Crit Rev Microbiol. 1971 May;1(1):29–72. doi: 10.3109/10408417109104477. [DOI] [PubMed] [Google Scholar]
- Hughes R. C., Pavlik J. G., Rogers H. J., Tanner P. J. Organization of polymers in the cell walls of some bacilli. Nature. 1968 Aug 10;219(5154):642–644. doi: 10.1038/219642a0. [DOI] [PubMed] [Google Scholar]
- Hughes R. C., Tanner P. J. The action of dilute alkali on some bacterial cell walls. Biochem Biophys Res Commun. 1968 Oct 10;33(1):22–28. doi: 10.1016/0006-291x(68)90248-9. [DOI] [PubMed] [Google Scholar]
- Höltje J. V., Tomasz A. Specific recognition of choline residues in the cell wall teichoic acid by the N-acetylmuramyl-L-alanine amidase of Pneumococcus. J Biol Chem. 1975 Aug 10;250(15):6072–6076. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- MARTIN R. G., AMES B. N. A method for determining the sedimentation behavior of enzymes: application to protein mixtures. J Biol Chem. 1961 May;236:1372–1379. [PubMed] [Google Scholar]
- Mauck J., Chan L., Glaser L. Turnover of the cell wall of Gram-positive bacteria. J Biol Chem. 1971 Mar 25;246(6):1820–1827. [PubMed] [Google Scholar]
- Mintz G., Herbold D. R., Glaser L. A fluorescent assay for bacterial cell wall lytic enzymes. Anal Biochem. 1975 May 26;66(1):272–278. doi: 10.1016/0003-2697(75)90746-0. [DOI] [PubMed] [Google Scholar]
- Shockman G. D., Pooley H. M., Thompson J. S. Autolytic enzyme system of Streptococcus faecalis. 3. Localization of the autolysin at the sites of cell wall synthesis. J Bacteriol. 1967 Nov;94(5):1525–1530. doi: 10.1128/jb.94.5.1525-1530.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shockman G. D., Thompson J. S., Conover M. J. The autolytic enzyme system of Streptococcus faecalis. II. Partial characterization of the autolysin and its substrate. Biochemistry. 1967 Apr;6(4):1054–1065. doi: 10.1021/bi00856a014. [DOI] [PubMed] [Google Scholar]
- Tomasz A., Westphal M. Abnormal autolytic enzyme in a pneumococus with altered teichoic acid composition. Proc Natl Acad Sci U S A. 1971 Nov;68(11):2627–2630. doi: 10.1073/pnas.68.11.2627. [DOI] [PMC free article] [PubMed] [Google Scholar]




