Abstract
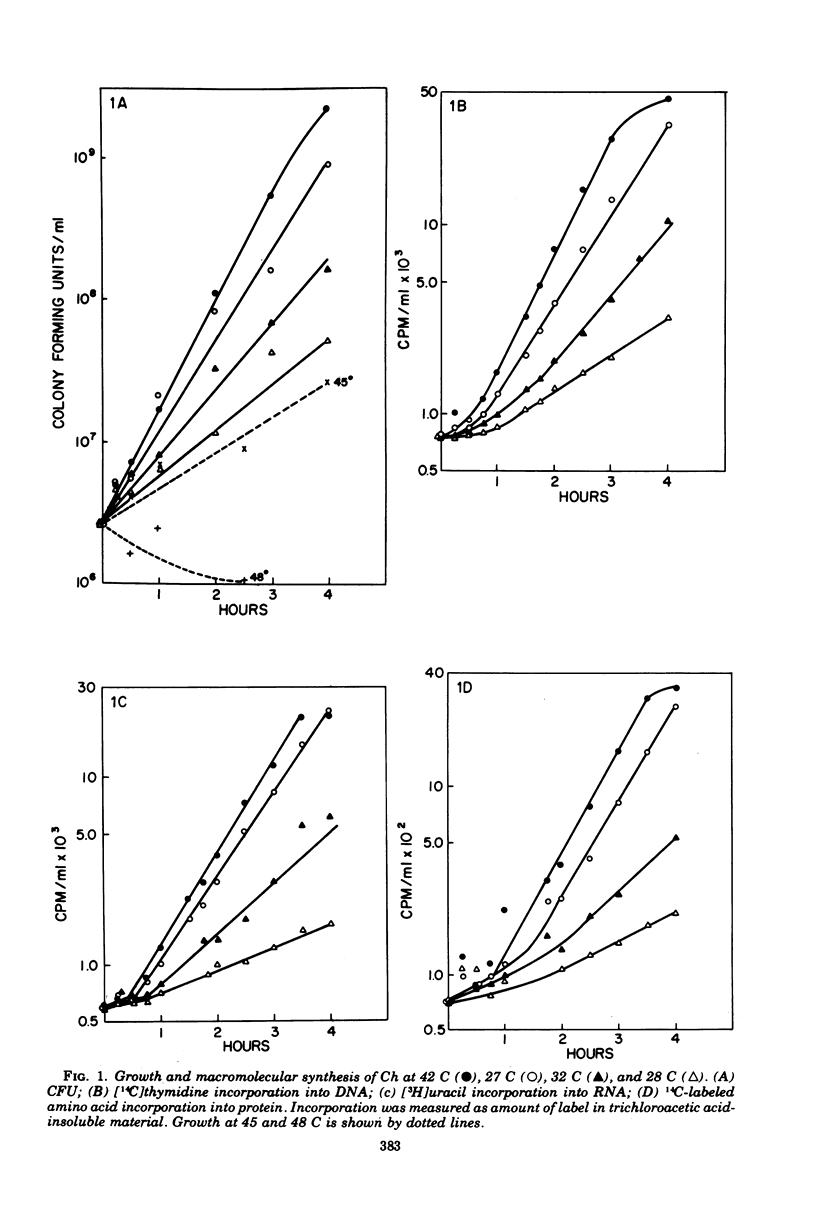
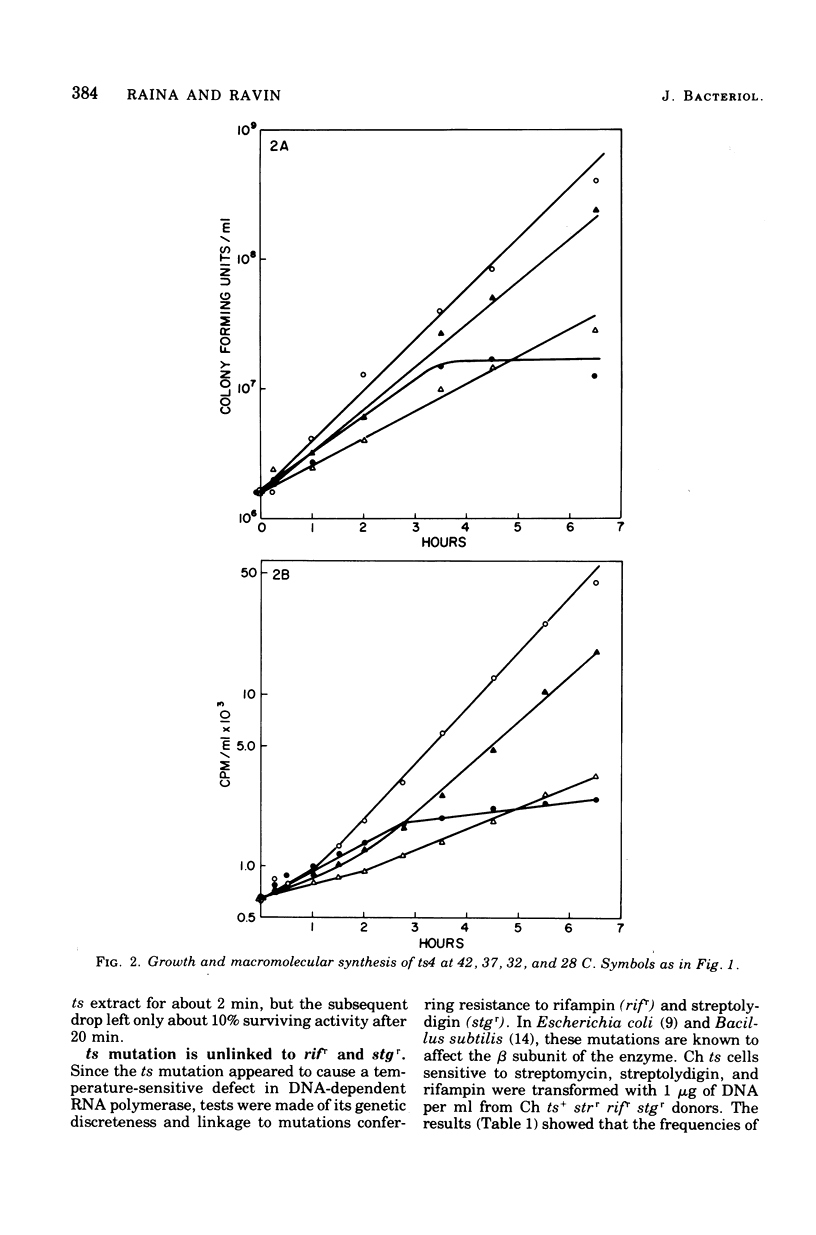
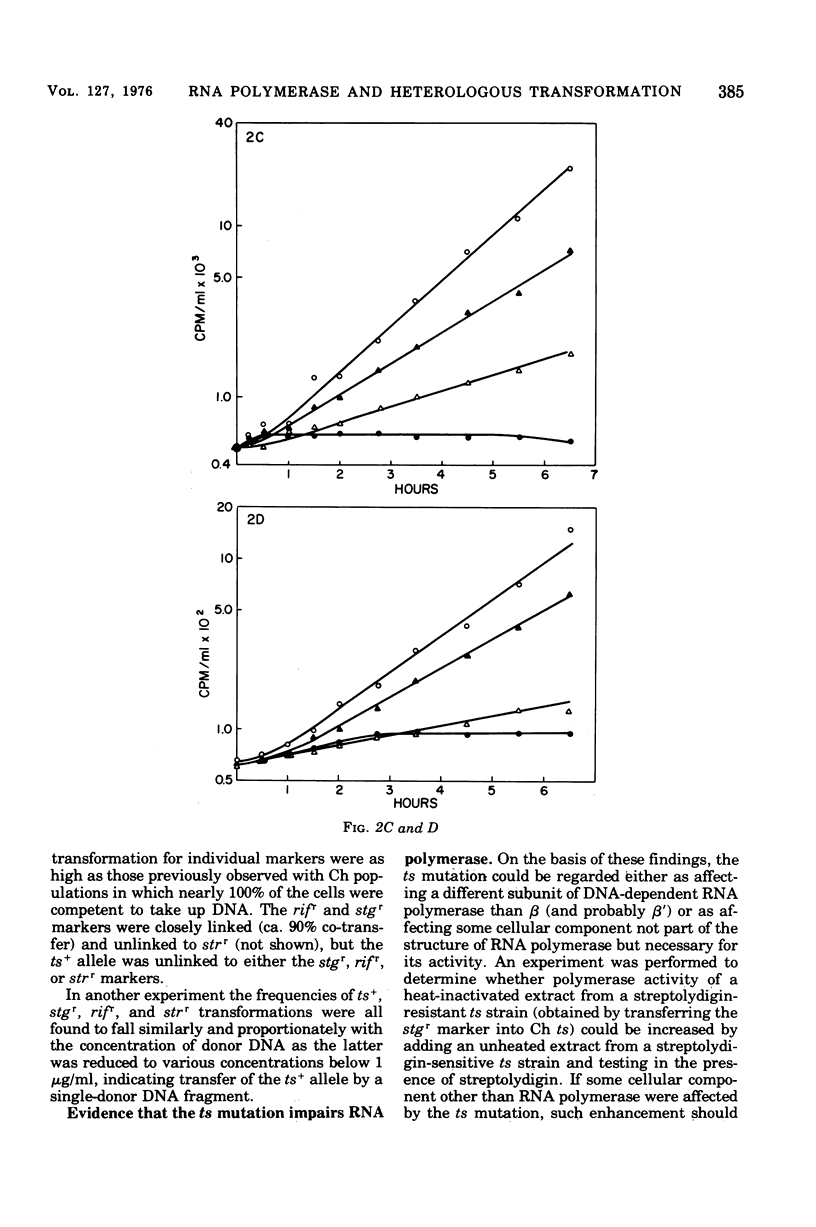
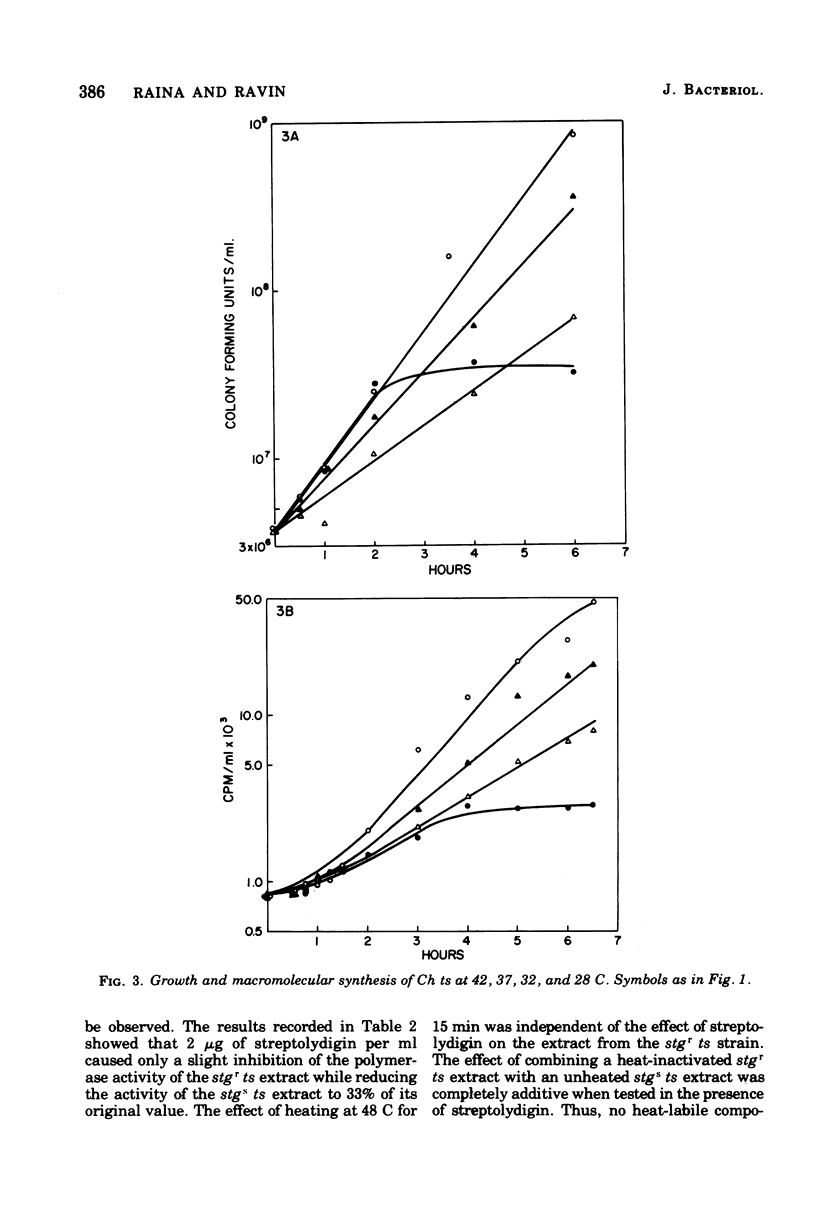
We have induced with nitrosoguanidine in Streptococcus sanguis a mutation conferring inability to grow and synthesize ribonucleic acid (RNA) at 42 C, the optimal temperature for growth and RNA synthesis in the parental strain. The mutation (ts) is transferable via transforming deoxyribonucleic acid (DNA) and is replaceable by its wild-type allele with fairly high efficiency in transformation reactions. The ts mutation is unlinked to the sites of mutation conferring resistance of rifampin (rifr) and streptolydigin (stgr), known to affect the beta subunit of DNA-dependent RNA polymerase. Extracts from strains carrying the ts mutation are more sensitive to elevated temperatures than are parental extracts when assayed for DNA-dependent RNA polymerase. The conclusion that the mutation causes a temperature-sensitive defect in some component of this enzyme (other than beta) is supported by the finding that the polymerase activity of a heat-inactivated ts stgr extract cannot be increased by addition of an unheated ts stgs extract, which is itself inactivated by streptolydigin. S. sanguis recipients carrying the ts mutation are highly transformable with heterospecific DNA, especially at the restrictive temperature.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BARKULIS S. S., SMITH C., BOLTRALIK J. J., HEYMANN H. STRUCTURE OF STREPTOCOCCAL CELL WALLS. IV. PURIFICATION AND PROPERTIES OF STREPTOCOCCAL PHAGE MURALYSIN. J Biol Chem. 1964 Dec;239:4027–4033. [PubMed] [Google Scholar]
- Biswas G. D., Ravin A. W. Heterospecific transformation of Pneumococcus and Streptococcus. IV. Variations in hybrid DNA produced by recombination. Mol Gen Genet. 1971;110(1):1–22. doi: 10.1007/BF00276040. [DOI] [PubMed] [Google Scholar]
- Burgess R. R. A new method for the large scale purification of Escherichia coli deoxyribonucleic acid-dependent ribonucleic acid polymerase. J Biol Chem. 1969 Nov 25;244(22):6160–6167. [PubMed] [Google Scholar]
- Burgess R. R., Travers A. A., Dunn J. J., Bautz E. K. Factor stimulating transcription by RNA polymerase. Nature. 1969 Jan 4;221(5175):43–46. doi: 10.1038/221043a0. [DOI] [PubMed] [Google Scholar]
- Burgi A. W., Robinton J., Carlson C. L. Studies on the folded chromosome of Escherichia coli. Cold Spring Harb Symp Quant Biol. 1974;38:43–51. doi: 10.1101/sqb.1974.038.01.007. [DOI] [PubMed] [Google Scholar]
- Deddish P. A., Ravin A. W. Relation of macromolecular synthesis in streptococci to efficiency of transformation by markers of homospecific and heterospecific origin. J Bacteriol. 1974 Mar;117(3):1158–1170. doi: 10.1128/jb.117.3.1158-1170.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Errington L., Glass R. E., Hayward R. S., Scaife J. G. Structure and orientation of an RNA polymerase operon in Escherichia coli. Nature. 1974 Jun 7;249(457):519–522. doi: 10.1038/249519a0. [DOI] [PubMed] [Google Scholar]
- Hartmann G., Honikel K. O., Knüsel F., Nüesch J. The specific inhibition of the DNA-directed RNA synthesis by rifamycin. Biochim Biophys Acta. 1967;145(3):843–844. doi: 10.1016/0005-2787(67)90147-5. [DOI] [PubMed] [Google Scholar]
- Heil A., Zillig W. Reconstitution of bacterial DNA-dependent RNA-polymerase from isolated subunits as a tool for the elucidation of the role of the subunits in transcription. FEBS Lett. 1970 Dec;11(3):165–168. doi: 10.1016/0014-5793(70)80519-1. [DOI] [PubMed] [Google Scholar]
- Iwakura Y., Ishihama A., Yura T. RNA polymerase mutants of Escherichia coli. Streptolydigin resistance and its relation to rifampicin resistance. Mol Gen Genet. 1973 Mar 1;121(2):181–196. doi: 10.1007/BF00277531. [DOI] [PubMed] [Google Scholar]
- Jaskunas S. R., Burgess R. R., Nomura M. Identification of a gene for the alpha-subunit of RNA polymerase at the str-spc region of the Escherichia coli chromosome. Proc Natl Acad Sci U S A. 1975 Dec;72(12):5036–5040. doi: 10.1073/pnas.72.12.5036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- KISSANE J. M., ROBINS E. The fluorometric measurement of deoxyribonucleic acid in animal tissues with special reference to the central nervous system. J Biol Chem. 1958 Jul;233(1):184–188. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Linn T., Losick R., Sonenshein A. L. Rifampin resistance mutation of Bacillus subtilis altering the electrophoretic mobility of the beta subunit of ribonucleic acid polymerase. J Bacteriol. 1975 Jun;122(3):1387–1390. doi: 10.1128/jb.122.3.1387-1390.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pettijohn D. E., Hecht R. RNA molecules bound to the folded bacterial genome stabilize DNA folds and segregate domains of supercoiling. Cold Spring Harb Symp Quant Biol. 1974;38:31–41. doi: 10.1101/sqb.1974.038.01.006. [DOI] [PubMed] [Google Scholar]
- ROTHEIM M. B., RAVIN A. W. The mapping of genetic loci affecting streptomycin resistance in Pneumococcus. Genetics. 1961 Dec;46:1619–1634. doi: 10.1093/genetics/46.12.1619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ravin A. W., Chakrabarti T. Genetic hybridization at the unlinked thy and str loci of Streptococcus. Genetics. 1975 Oct;81(2):223–241. doi: 10.1093/genetics/81.2.223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ravin A. W., Ma M. Specific effects of heating of transformable streptococci on their ability to discriminate between homospecific, heterospecific, and hybrid deoxyribonucleic acid. J Bacteriol. 1972 Feb;109(2):616–625. doi: 10.1128/jb.109.2.616-625.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simchen G., Piñon R., Salts Y. Sporulation in Saccharomyces cerevisiae: premeiotic DNA synthesis, readiness and commitment. Exp Cell Res. 1972 Nov;75(1):207–218. doi: 10.1016/0014-4827(72)90538-1. [DOI] [PubMed] [Google Scholar]



