Abstract
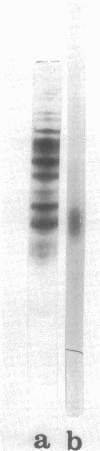
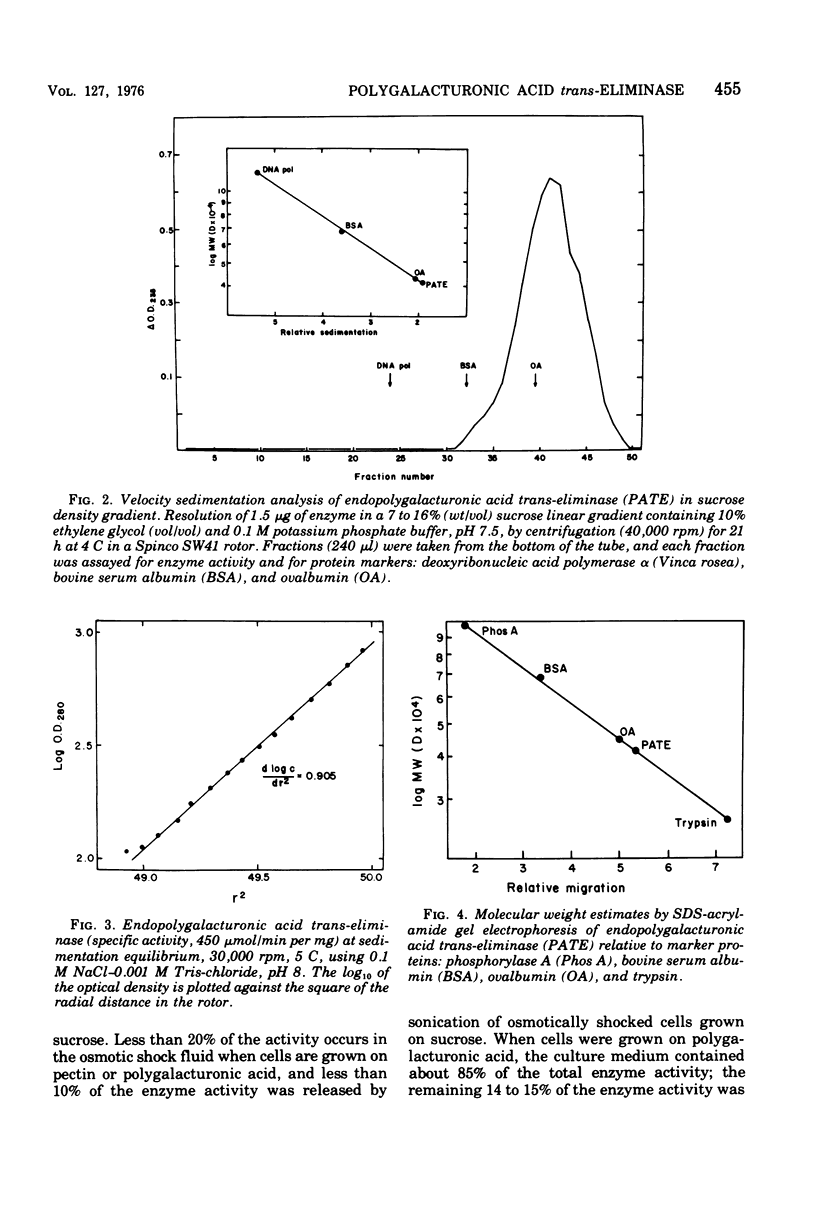
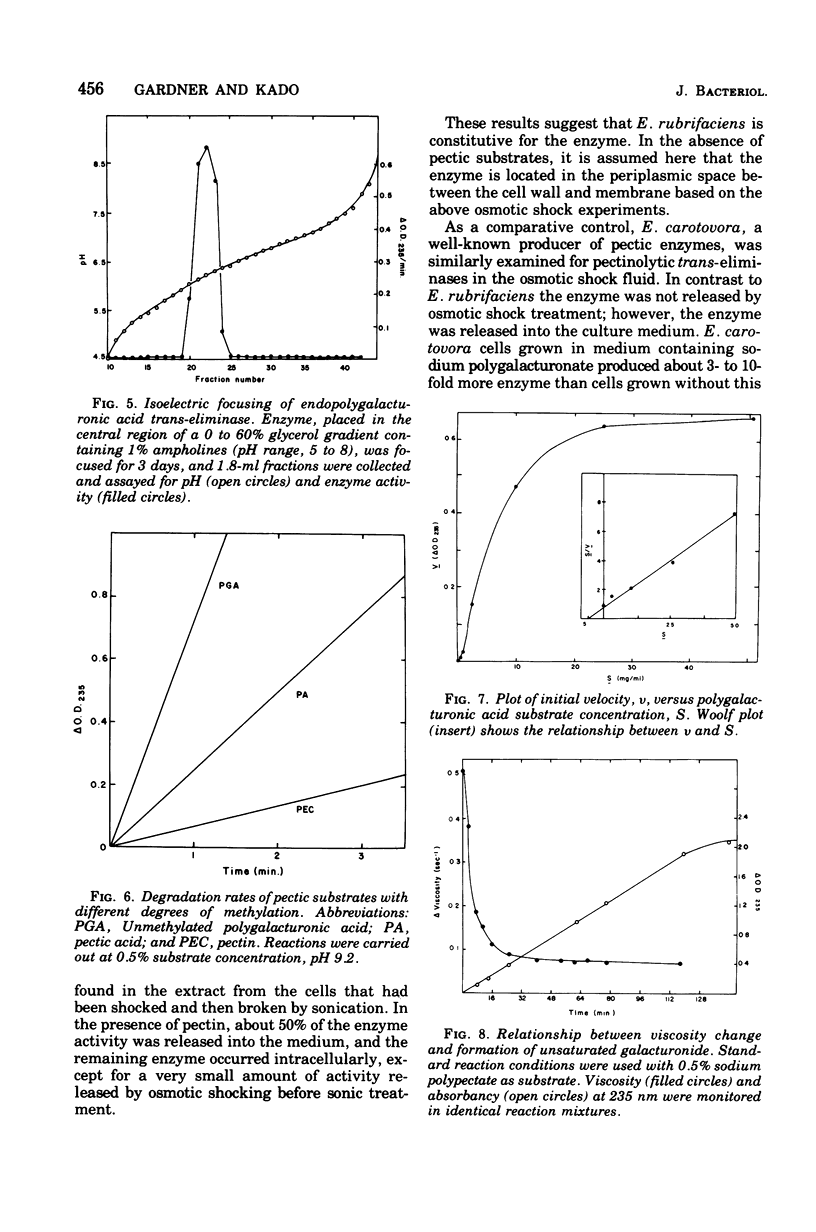
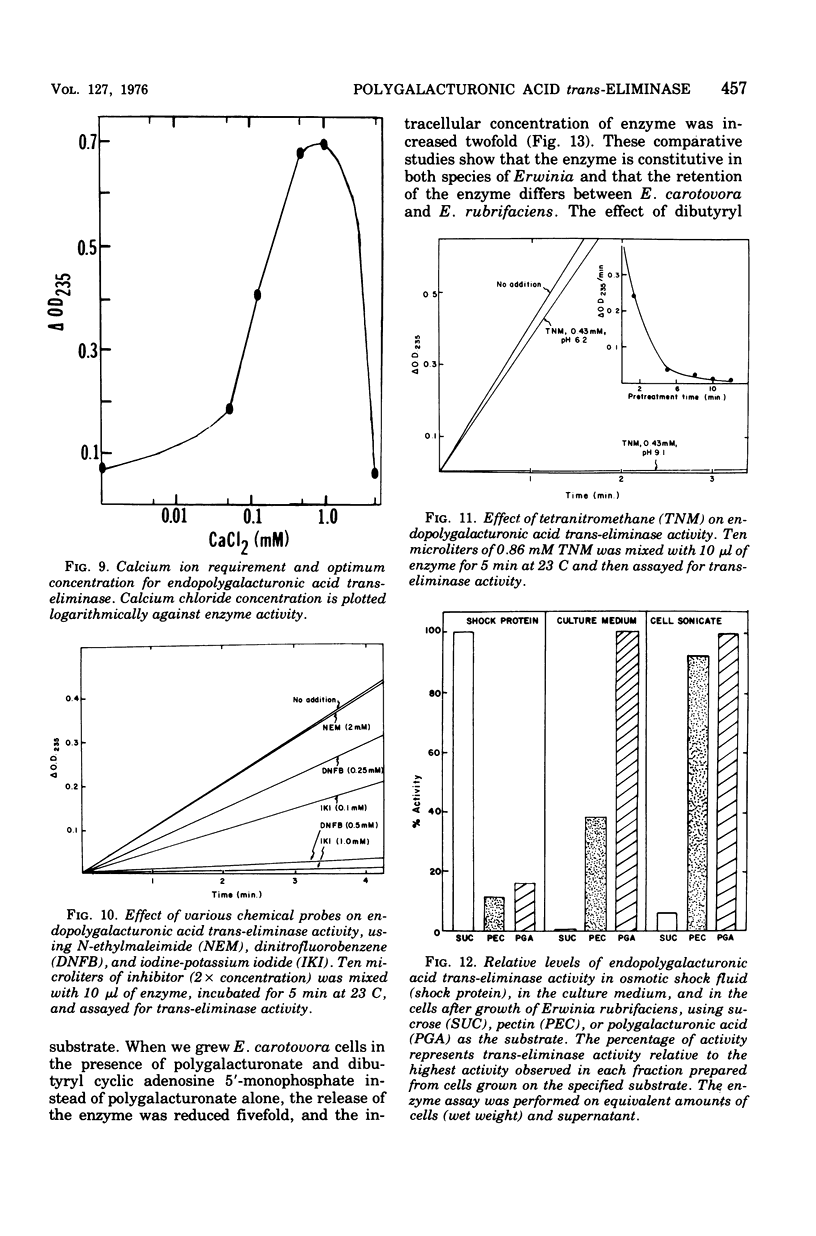
An endopolygalacturonic acid trans-eliminase (EC 4.2.2.2), released by osmotic shock of Erwinia rubrifaciens cells, has been purified to near homogeneity (3, 100-fold) by column chromatography on diethylaminoethyl-cellulose, phosphocellulose, and hydroxyapatite-cellulose followed by isoelectric focusing. It has a molecular weight of 41,000, s20,w of 3.09S, an isoelectric point of pH 6.25, pH optimum of 9.5, and a temperature optimum of 37 C and requires Ca2+ with an optimum concentration of 0.5 to 1.0 mM. Mg2+ could not substitute for Ca2+. Tyrosinyl residues seem essential for enzyme catalysis based on rapid inactivation by tetranitromethane. The enzyme prefers unmethylated polygalacturonic acid as the substrate, cleaving alpha-1,4-glycosidic linkages randomly to form unsaturated galacturonides at a Vmax of 1,166 mumol of product/min per mg of protein and a Km of 5 mg of polygalacturonic acid per ml. Over 90% of the enzyme activity is released from osmotically shocked E. rubrifaciens cells. Unlike E. rubrifaciens, trans-eliminase is not released from Erwinia carotovora cells by osmotic shock treatment, but enzyme activity is detected in the culture medium. The release of the enzyme is reduced fivefold by the addition of dibutyryl cyclic adenosine 5'-monophosphate. The hypersensitive reaction in tobacco leaves was induced within 60 min after injection of less than 1 mug of purified E. rubrifaciens trans-eliminase. Single cells of tobacco in suspension culture are readily killed by the enzyme, whereas tobacco protoplasts remain unaffected when treated in the same manner. These results indicate that endopolygalacturonic acid trans-eliminase is a constitutive enzyme possibly located in the periplasmic space of the E. rubrifaciens cell and releases enzyme into the culture medium in the presence of substrate. The release of the enzyme in tobacco tissue and the trans-eliminative cleavage of plant cell wall components may be steps leading to hypersensitivity of the tobacco tissue.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Christen P., Riordan J. F. Syncatalytic modification of a functional tyrosyl residue in aspartate aminotransferase. Biochemistry. 1970 Jul 21;9(15):3025–3034. doi: 10.1021/bi00817a014. [DOI] [PubMed] [Google Scholar]
- Davé B. A., Vaughn R. H. Purification and properties of an polygalacturonic acid trans-eliminase produced by Bacillus pumilus. J Bacteriol. 1971 Oct;108(1):166–174. doi: 10.1128/jb.108.1.166-174.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuchs A. The trans-eliminative breakdown of Na-polygalacturonate by Pseudomonas fluorescens. Antonie Van Leeuwenhoek. 1965;31(3):323–340. doi: 10.1007/BF02045912. [DOI] [PubMed] [Google Scholar]
- Heppel L. A. Selective release of enzymes from bacteria. Science. 1967 Jun 16;156(3781):1451–1455. doi: 10.1126/science.156.3781.1451. [DOI] [PubMed] [Google Scholar]
- Kado C. I., Heskett M. G. Selective media for isolation of Agrobacterium, Corynebacterium, Erwinia, Pseudomonas, and Xanthomonas. Phytopathology. 1970 Jun;60(6):969–976. doi: 10.1094/phyto-60-969. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lakso J. U., Starr M. P. Comparative injuriousness to plants of Erwinia spp. and other enterobacteria from plants and animals. J Appl Bacteriol. 1970 Dec;33(4):692–707. doi: 10.1111/j.1365-2672.1970.tb02252.x. [DOI] [PubMed] [Google Scholar]
- MACMILLAN J. D., VAUGHN R. H. PURIFICATION AND PROPERTIES OF A POLYGALACTURONIC ACID-TRANS-ELIMINASE PRODUCED BY CLOSTRIDIUM MULTIFERMENTANS. Biochemistry. 1964 Apr;3:564–572. doi: 10.1021/bi00892a016. [DOI] [PubMed] [Google Scholar]
- MARTIN R. G., AMES B. N. A method for determining the sedimentation behavior of enzymes: application to protein mixtures. J Biol Chem. 1961 May;236:1372–1379. [PubMed] [Google Scholar]
- Moran F., Nasuno S., Starr M. P. Extracellular and intracellular polygllacturonic acid trans-eliminases of Erwinia carotovora. Arch Biochem Biophys. 1968 Feb;123(2):298–306. doi: 10.1016/0003-9861(68)90138-0. [DOI] [PubMed] [Google Scholar]
- Moran F., Nasuno S., Starr M. P. Oligogalacturonide trans-eliminase of Erwinia carotovora. Arch Biochem Biophys. 1968 Jun;125(3):734–741. doi: 10.1016/0003-9861(68)90508-0. [DOI] [PubMed] [Google Scholar]
- Motoyoshi F., Bancroft J. B., Watts J. W., Burgess J. The infection of tobacco protoplasts with cowpea chlorotic mottle virus and its RNA. J Gen Virol. 1973 Aug;20(2):177–193. doi: 10.1099/0022-1317-20-2-177. [DOI] [PubMed] [Google Scholar]
- Nasuno S., Starr M. P. Polygalacturonic acid trans-eliminase of Xanthomonas campestris. Biochem J. 1967 Jul;104(1):178–185. doi: 10.1042/bj1040178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nossal N. G., Heppel L. A. The release of enzymes by osmotic shock from Escherichia coli in exponential phase. J Biol Chem. 1966 Jul 10;241(13):3055–3062. [PubMed] [Google Scholar]
- WEISSBACH A., HURWITZ J. The formation of 2-keto-3-deoxyheptonic acid in extracts of Escherichia coli B. I. Identification. J Biol Chem. 1959 Apr;234(4):705–709. [PubMed] [Google Scholar]
- Weber K., Osborn M. The reliability of molecular weight determinations by dodecyl sulfate-polyacrylamide gel electrophoresis. J Biol Chem. 1969 Aug 25;244(16):4406–4412. [PubMed] [Google Scholar]
- YPHANTIS D. A. EQUILIBRIUM ULTRACENTRIFUGATION OF DILUTE SOLUTIONS. Biochemistry. 1964 Mar;3:297–317. doi: 10.1021/bi00891a003. [DOI] [PubMed] [Google Scholar]