Abstract
The lecticans are a family of chondroitin sulfate proteoglycans including aggrecan, versican, neurocan, and brevican. The C-terminal globular domains of lecticans are structurally related to selectins, consisting of a C-type lectin domain flanked by epidermal growth factor and complement regulatory protein domains. The C-type lectin domain of versican has been shown to bind tenascin-R, an extracellular matrix protein specifically expressed in the nervous system, and the interaction was presumed to be mediated by a carbohydrate–protein interaction. In this paper, we show that the C-type lectin domain of brevican, another lectican that is specifically expressed in the nervous system, also binds tenascin-R. Surprisingly, this interaction is mediated by a protein–protein interaction through the fibronectin type III domains 3–5 of tenascin-R, independent of any carbohydrates or sulfated amino acids. The lectin domains of versican and other lecticans also bind the same domain of tenascin-R by protein–protein interactions. Surface plasmon resonance analysis revealed that brevican lectin has at least a 10-fold higher affinity than the other lectican lectins. Tenascin-R is coprecipitated with brevican from adult rat brain extracts, suggesting that tenascin-R and brevican form complexes in vivo. These results demonstrate that the C-type lectin domain can interact with fibronectin type III domains through protein–protein interactions, and suggest that brevican is a physiological tenascin-R ligand in the adult brain.
The lecticans are a family of chondroitin sulfate proteoglycans (reviewed in ref. 1), encompassing aggrecan, versican, neurocan, and brevican (2–5). The core proteins of these proteoglycans share structural domains consisting of a central glycosaminoglycan carrying region and N-terminal and C-terminal globular domains. The C-terminal globular domain of lecticans consists of one or two epidermal growth factor (EGF) domains, a C-type lectin domain, and a complement regulatory protein domain. These are the same structural elements as seen in selectins, which mediate leukocyte–endothelium adhesion through various carbohydrate ligands (6, 7). The structural similarities between selectins and the C-terminal globular domains of lecticans suggested that the lecticans also bind carbohydrate ligands. Earlier studies have shown that the C-type lectin domain of aggrecan and versican bind simple carbohydrates as well as heparin/heparan sulfate (8–11).
Previous studies (11) have demonstrated that recombinant versican lectin domain binds tenascin-R, a member of tenascin family glycoproteins expressed specifically in the nervous system (12). This interaction is calcium-dependent, as expected of a carbohydrate–protein interaction mediated by a C-type lectin domain (11). This observation suggested that other members of the lectican family, notably brevican and neurocan, which are specifically expressed in the nervous system (4, 13), may also interact with tenascin-R. In this paper, we report that all lecticans do bind tenascin-R, but surprisingly the interaction is mediated by the fibronectin type III (FNIII) domains of the protein backbone of tenascin-R independent of any carbohydrates. Results of surface plasmon resonance analysis and coprecipitation experiments suggest that brevican is a physiological tenascin-R ligand in the adult brain.
EXPERIMENTAL PROCEDURES
Recombinant Lectin Domains.
The NotI–SalI fragment of the versican lectin domain construct (11) and a synthetic fragment encoding a hexahistidine (his)-tag flanked by SalI and XbaI sites were ligated into pcDNA3. For the construction of his-tagged expression vector for other lecticans, the fragment encoding the lectin domain of versican was excised, leaving the his-tag in the vector. cDNAs for the lectin domains of other lecticans were amplified from rat cartilage (for aggrecan) or rat brain (for brevican and neurocan) cDNA by PCR based on published sequence (2, 4, 14) and ligated into this vector. For production of his-tagged recombinant lectin domains of aggrecan and neurocan, human 293 cells were cotransfected with respective expression vectors and pSV2-pac, and spent media were collected from stable transfectants. For lectin domains of versican and brevican, for which stable 293 cell transfectants producing high levels of recombinant proteins were not isolated, the expression cassette for each lectin domain was subcloned into pCEP4 (Invitrogen), and transfected into 293–Epstein–Barr virus-encoded nuclear antigen cells (Invitrogen). The recombinant lectin domains with their C-terminal his-tag were purified from conditioned media on a Ni-NTA (nickel-nitrotriacetic acid) agarose (Qiagen, Chatsworth, CA) column. These recombinant proteins are referred to as brevican rCLD (recombinant C-type lectin domain), versican rCLD, etc. in this paper. For the surface plasmon resonance analysis, recombinant rCLDs were further purified on a Mono Q column. Biotinylation was performed as described (11).
Brevican–Ig Chimera.
A cDNA fragment (0.9 kb) consisting of the EGF, lectin, and complement regulatory protein domains (ELC) of rat brevican (corresponding to nucleotides 1791–2669; ref. 14) was amplified by PCR and fused in-frame to another PCR fragment (0.2 kb) of brevican containing its signal peptide (nucleotides 1–182). The resultant 1.1-kb fragment was inserted into the BamHI site of pcDNAI/Fc (15), which contains human genomic DNA encoding the Fc region of IgG1. The plasmid was cotransfected with pSV2-dhfr into CHO/DG44 cells by the calcium phosphate method. The chimera was purified from serum-free culture supernatants of these cells by affinity chromatography on protein A-Sepharose.
Ligand Blotting, Immunoblotting, and Immunoprecipitation Assays.
Preparation of soluble and membrane fractions of adult rat brain and immunoblotting were performed as described (5). Ligand overlay assays with Ig chimeras and biotinylated recombinant lectin domains, as well as precipitation of tenascin-R with Ig chimera were performed according to Aspberg et al. (11). For immunoprecipitation with anti-brevican antibodies, soluble brain extracts were incubated with purified RB18 mouse monoclonal antibodies (28) and the immune complex was precipitated with protein G-Sepharose. Precipitated materials were resolved in SDS/PAGE and analyzed by the ligand overlay assay with brevican ELC chimera as described above. Enzymatic deglycosylation was performed as described previously (11).
Tunicamycin and Chlorate Treatment.
B9 cells (16) were maintained in Dulbecco’s modified minimal essential medium containing 10% fetal calf serum. Tunicamycin and chlorate treatments were performed according to Yasugi et al. (17) and Pouyani et al. (18), respectively. Effectiveness of tunicamycin treatment was monitored by the reduction of the apparent molecular weight of tenascin-R in SDS/PAGE. Inhibition of sulfation was monitored by anti-HNK-1 immunoblotting of tenascin-R secreted by B9 cells (the HNK-1 epitope includes sulfated glucuronic acid). The chlorate treatment almost completely abolished the reactivity of anti-HNK-1 antibody with tenascin-R, confirming the effectiveness of the treatment.
Glutathione S-Transferase (GST) Fusion Proteins.
GST fusion proteins consisting of various domains of rat tenascin-R, except for FN3–4, FN4–5, FN3, FN4, and FN5 fusion proteins, have been described (19). Constructs for FN3–4, FN4–5, FN3, FN4, and FN5 fusion proteins were similarly prepared in pGEX-KG by using the following primers: FN3 forward, 5′-CGAATTCCTGTCATTGACGGGC-3′; FN3 backward, 5′-CGTGTCGACTATGTTGTAAACTGGGTGC-3′; FN4 forward, 5′-CGAATTCCTGAAATTGATGCTCCC-3′; FN4 backward, 5′-CGTGTCGACTAAGTCCTGGCATTCAT-3′; FN5 forward, 5′-CGAATTCCTGAGCTTGACAGTCCC-3′; FN5 backward, 5′-CGTGTCGACTATGTGAAGGCATCCAC-3′. For FN3–4 and FN4–5 fusion proteins, primer combinations of FN3 forward/FN4 backward and FN4 forward/FN5 backward were used, respectively. Fusion proteins were expressed in Escherichia coli strain TG-1. For surface plasmon resonance analysis, FN3–5 fusion protein was isolated on glutathione-Sepharose and FN3–5 fragments were released from GST fusion partners by digestion with thrombin (19). FN3–5 fragments were further purified on a Mono Q column, followed by buffer change to 10 mM Hepes (pH 7.5)/0.15 M NaCl on a desalting column. The concentration of the purified FN3–5 fragment was determined using the calculated molar extinction coefficient of 29,870 M−1 cm−1 (20).
Affinity Measurement.
The Mono Q-purified rCLDs were diluted with 10 mM sodium acetate (pH 4.0) and coupled on a BIAcore CM5 sensorchip. Different levels of immobilization of the lectin domains were tested, and conditions that gave responses ranging from 50–600 resonance units (RU) with no detectable mass transport rate limitations were chosen. The residuals of the curve fittings were less than 0.4 RU, which is the noise level of the instrument. For affinity measurements, Mono Q-purified FN3–5 fragments in binding buffer (10 mM Hepes/0.15 M NaCl/1 mM CaCl2/0.005% P20) were injected on the coated sensorchip surfaces at 35 μl/min, and the binding and release of FN3–5 fragments were measured in a BIAcore 2000 instrument (Pharmacia). The sensorchip surfaces were regenerated through injection of a 200 μl pulse of 20 mM EDTA in the binding buffer between each experiment. Control experiments were performed in binding buffer containing 5 mM EDTA instead of CaCl2. Association (ka) and dissociation (kd) rate constants were determined using the homogenous kinetic models (A + B = AB and AB = A + B, respectively) in the biaevaluation 2.1 program, and the steady-state dissociation constants (KD) were calculated from these parameters.
RESULTS
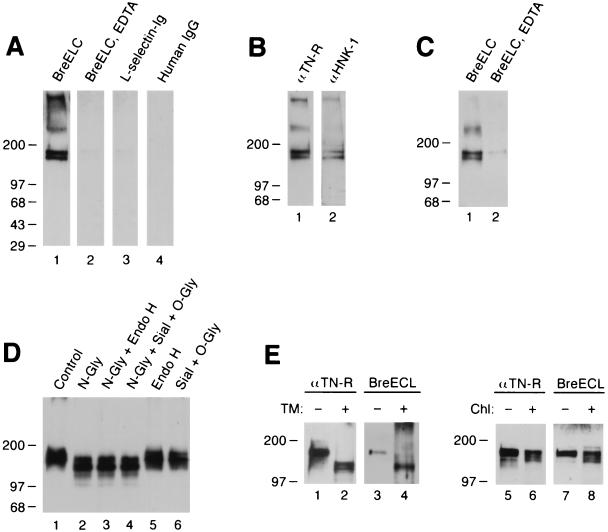
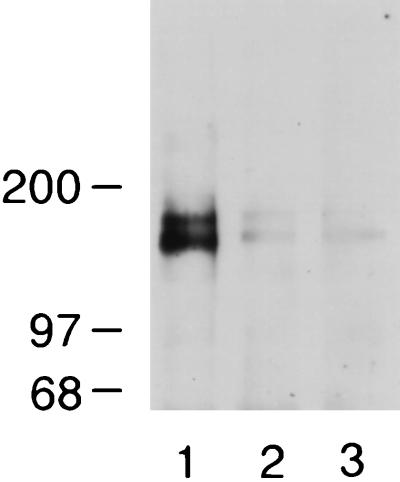
To determine whether brevican might share the tenascin-R binding property of versican, we first tested the binding of an IgG chimera, which encompasses the entire C-terminal globular domain of brevican (“brevican ELC chimera”). This chimeric molecule includes the C-type lectin-like domain as well as flanking EGF and complement regulatory protein-like domains. The brevican ELC chimera bound to two bands of 160 and 180 kDa in both soluble (Fig. 1A) and membrane (not shown) fractions of adult rat brain. The binding is inhibited in the presence of EDTA (lane 2). A similar IgG chimera of l-selectin and human IgG showed no binding to the 160/180 kDa bands (lanes 3 and 4). The identity of the 160/180 kDa band as tenascin-R was demonstrated by immunoblotting with anti-tenascin-R and anti-HNK-1 monoclonal antibodies (tenascin-R contains HNK-1 carbohydrates; ref. 22) (Fig. 1B). As a further confirmation, adult brain extracts were precipitated with the brevican ELC chimera and the precipitated materials were probed with anti-tenascin-R antibodies. Anti-tenascin-R monoclonal antibodies detected the 160/180 kDa bands in the precipitated materials when the precipitation was performed in the presence of Ca2+, but not when it was performed in the presence of EDTA (Fig. 1C). These results show that brevican ELC chimera binds tenascin-R in a calcium dependent manner.
Figure 1.
Brevican ELC chimera binds tenascin-R. (A) Brevican ELC chimera binds to 160/180 kDa bands in ligand blotting. Soluble extracts of rat brain were resolved in an 8–16% gradient SDS/PAGE under nonreducing conditions and transferred to nitrocellulose filters. Filters were probed in the absence (lanes 1, 3, and 4) or presence (lane 2) of 20 mM EDTA with brevican ELC chimera (lanes 1 and 2), l-selectin-Ig chimera (21) (lane 3), or human IgG (lane 4). (B) Bands at 160 and 180 kDa are recognized by anti-tenascin-R and anti-HNK-1 monoclonal antibodies. Soluble extracts from adult rat brain were probed in immunoblotting with anti-rat tenascin-R monoclonal antibody 596 (11) (lane 1) or anti-HNK-1 monoclonal antibody (lane 2). (C) Tenascin-R is precipitated with brevican ELC chimera. Soluble brain extracts were precipitated with brevican ELC chimera and protein A-agarose in the absence (lane 1) or presence (lane 2) of 20 mM EDTA, and the precipitated materials were probed by immunoblotting with anti-tenascin-R monoclonal antibody 596. (D) Enzymatic deglycosylation does not affect the binding of brevican ELC chimera to tenascin-R. Soluble brain extracts were treated without any enzyme (lane 1); with N-glycosidase F (lane 2); with N-glycosidase F and endo-β-acetylglucosaminidase H (lane 3); with N-glycosidase F, sialidase, and O-glycanase (lane 4); with endo-β-acetylglucosaminidase H (lane 5); or with sialidase and O-glycanase (lane 6). Treated samples were probed with brevican ELC chimera in ligand blotting. (E) Metabolic deglycosylation and desulfation do not affect the binding of brevican ELC chimera to tenascin-R. B9 cells, which secrete the 180 kDa form of tenascin-R, were treated with tunicamycin (lanes 1–4) or chlorate (lanes 5–8). Culture supernatants from the treated B9 cells were immunoblotted with anti-tenascin-R monoclonal antibody 596 (lanes 1, 2, 5, and 6) or brevican ELC chimera (lanes 3, 4, 7, and 8).
To examine if brevican ELC chimera binds carbohydrate determinants on tenascin-R, as might be expected because of its C-type lectin domain, we examined the effects of enzymatic and metabolic deglycosylation on the binding. For enzymatic treatment, brain extracts were treated with panels of glycosidases and tested in ligand blotting (Fig. 1D). For metabolic deglycosylation, rat glial B9 cells (16), which secrete the 180 kDa form of tenascin-R into culture supernatants, were treated with tunicamycin (Fig. 1E, lanes 1–4). All of these treatments, except for EndoH digestion, reduced the apparent molecular mass of tenascin-R, demonstrating the effectiveness of deglycosylation. None of these treatments, however, affected the binding of the chimera.
Ligand binding by l-selectin and p-selectin requires sulfation of carbohydrates and sulfated amino acids in the ligands (18, 23, 24). To investigate the possible role of sulfation in the brevican–tenascin-R interaction, B9 cells were treated with chlorate, a metabolic inhibitor of sulfation, and tenascin-R secreted from these cells were tested in ligand blotting. As shown in Fig. 1E, chlorate treatment did not affect the binding of the chimera to tenascin-R (lanes 5–8).
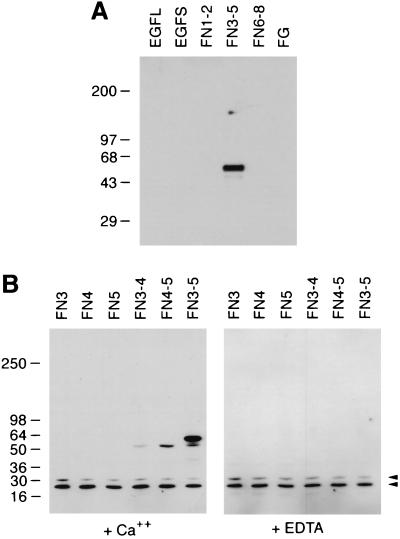
These negative results strongly suggested that the interaction of the brevican lectin and tenascin-R was not mediated by a carbohydrate–protein interaction, but by a protein–protein interaction. To demonstrate directly that this is the case, panels of fusion proteins consisting of different domains of tenascin-R were produced in E. coli. Bacterial fusion proteins are generally thought to contain no carbohydrates, or at least no carbohydrates recognizable by animal lectins. The brevican ELC chimera bound to a fusion protein consisting of FNIII domains 3–5 of tenascin-R (Fig. 2A). No binding to fusion proteins consisting of other tenascin-R domains was observed.
Figure 2.
Brevican lectin binds tenascin-R fragments produced as bacterial fusion proteins. (A) Fusion proteins representing various tenascin-R domains were probed with brevican ELC chimera by ligand blotting. EGFL, EGF repeats with cysteine-rich sequences; EGFS, EGF repeats without cysteine-rich sequences; FN1–2, FN3–5, and FN6–8, FNIII domains 1–2, 3–5, and 6–8, respectively; FG, fibrinogen-like domain (see ref. 19 for details). (B) Ligand blotting with brevican rCLD. Lysates of bacteria expressing GST fusion proteins representing the following FNIII domains were probed with biotinylated brevican rCLD. Ligand blotting was performed in the presence of 5 mM CaCl2 (Left) or 5 mM EDTA (Right). FN3, domain 3; FN4, domain 4; FN5, domain 5; FN3–4, domain 3–4; FN4–5, domain 4–5; FN3–5, domains 3–5. Arrowheads indicate nonspecific bands reactive to peroxidase-conjugated avidin used in these assays.
To localize the tenascin-R binding site in the brevican ELC chimera, we tested recombinant protein, which consists only of the lectin domain (brevican rCLD). Consistent with the binding profile of the ELC chimera, brevican rCLD bound to FN3–5 fusion protein (Fig. 2B) but not to the other fusion proteins (not shown), showing that the binding is mediated by the C-type lectin domain of brevican.
To define further the brevican binding site on tenascin-R, we examined fusion proteins consisting of a single or paired FNIII domains for binding of brevican rCLD. The brevican rCLD bound to FN4–5 and less strongly to FN3–4, but did not show any significant binding to any of the fusion proteins containing a single FNIII domain (Fig. 2B). Only when higher concentrations of fusion proteins were used, slight, but specific binding of brevican rCLD to FN4 was seen (not shown). These results indicate that the crucial determinant for the binding is present in FNIII domain 4, but that domain 4 alone cannot constitute a high-affinity binding site.
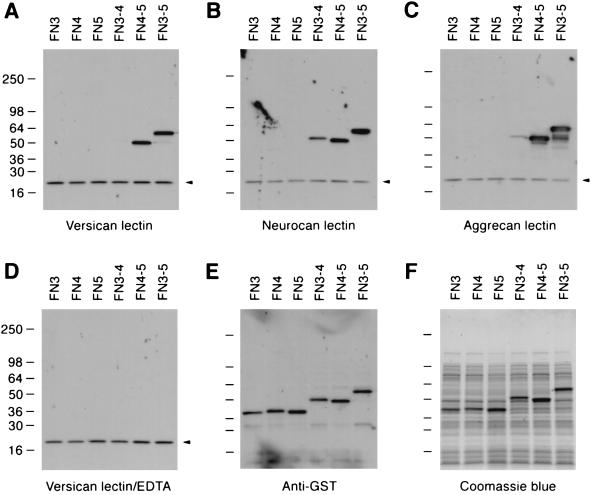
Ligand blotting with the rCLDs of the other lecticans showed that they also bind the FN3–5 fusion protein in an EDTA-sensitive manner (Fig. 3). As was the case with brevican, no binding was observed to the fragments consisting of single type III domains (i.e., FN3, FN4, and FN5 fusion proteins).
Figure 3.
The lectin domains of versican, neurocan, and aggrecan bind bacterial fusion proteins containing FNIII domain 4 of tenascin-R. Lysates of bacteria expressing GST fusion proteins representing different domains of tenascin-R were probed with the biotinylated rCLDs of versican (A and D), neurocan (B), and aggrecan (C) in the presence of 5 mM CaCl2 (A–C) or 5 mM EDTA (D) in ligand blotting. (E) The same blot was probed with anti-GST antiserum. (F) Coomassie brilliant blue staining of the gel loaded with identical amounts of lysates as in A–E. The fusion proteins used in this experiment are the same as described in the legend of Fig. 2. Arrowheads indicate nonspecific bands reactive to peroxidase-conjugated avidin.
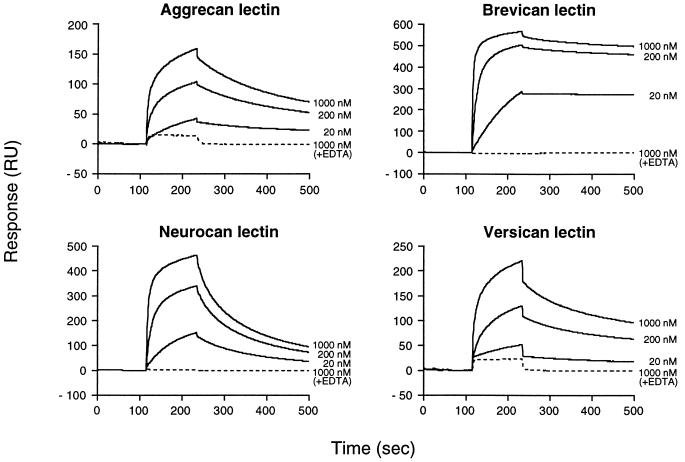
To compare quantitatively the ability of the various lectins to bind the tenascin-R, we determined the affinities of the individual lectin domains to the FN3–5 fragment by surface plasmon resonance analysis. Mono Q-purified rCLDs were coupled to biosensor chips and the purified FN3–5 fragment was injected at different concentrations over the rCLD-coated surfaces. Reversible binding of the FN3–5 fragment to all the rCLDs was observed when a running buffer containing calcium was used (Fig. 4). The interaction was completely obliterated in the presence of EDTA (Fig. 4, broken lines).
Figure 4.
Surface plasmon resonance measurements of the interactions between lectican lectins and tenascin-R. Mono Q-purified tenascin-R FN3–5 fragments at different concentrations were injected over flow cells coupled with individual lectican rCLDs. Solid lines indicate response traces in the presence of 1 mM CaCl2 and broken lines in the presence of 5 mM EDTA. Injection of the tenascin-R fragment started at 115 sec and ended at 235 sec. The response trace average between 75–100 sec was normalized to 0.
As shown in Table 1, the steady-state dissociation constant (KD) of the rCLDs varied by a factor of 30. The differences were almost entirely due to differences in the dissociation rate constant, because the association rate constants (ka) for these interactions were similar. Brevican rCLD was the strongest binder with a Kd at least 10-fold smaller than that of the other lectican rCLDs, and neurocan rCLD was the weakest.
Table 1.
Affinities of the interaction between lectican lectins and tenascin-R
| Lecticans | ka, M−1s−1 | kd, s−1 | KD, M |
|---|---|---|---|
| Brevican | 4.5 × 105 | 0.5 × 10−3 | 1.1 × 10−9 |
| Aggrecan | 2.9 × 105 | 3.5 × 10−3 | 12.2 × 10−9 |
| Versican | 2.2 × 105 | 3.2 × 10−3 | 14.9 × 10−9 |
| Neurocan | 3.3 × 105 | 10.1 × 10−3 | 30.7 × 10−9 |
The purified rCLD of each lectican was coupled to a biosensor chip, and the surface plasmon resonance response to 200 nM purified FN3-5 fragment was measured. Data were fit to dissociation and association models to obtain kd and ka, and KD was calculated from these parameters.
To gain insight into the physiological significance of the brevican–tenascin-R interaction, we examined whether tenascin-R exists as a complex with brevican in brain extracts. Brevican was immunoprecipitated from rat brain extracts with anti-brevican monoclonal antibodies and the precipitated material was probed with brevican ELC chimera for the presence of tenascin-R. As shown in Fig. 5, the brevican ELC chimera detected 160/180 kDa bands of tenascin-R in the precipitated material, and the coprecipitation was inhibited in the presence of EDTA. The identity of the 160/180 kDa bands as tenascin-R was confirmed by probing the precipitated material with anti-tenascin-R monoclonal antibodies followed by peroxidase-conjugated goat anti-mouse IgG. Although, in this case, the secondary anti-mouse antibodies also bound to anti-brevican antibodies used for immunoprecipitation, tenascin-R could be clearly identified (not shown). These results suggest that the brevican–tenascin-R interaction occurs in vivo.
Figure 5.
Tenascin-R coimmunoprecipitates with brevican from adult rat brain extracts. Soluble brain extracts were incubated with anti-brevican monoclonal antibody RB18 (lanes 1 and 2) in the presence of 5 mM CaCl2 (lane 1) or 20 mM EDTA (lane 2). In a control experiment, the incubation was performed without the antibody in the presence of 5 mM CaCl2 (lane 3). The samples were precipitated with protein G-agarose and then probed in ligand blotting assay with brevican ELC chimera.
DISCUSSION
In this paper we demonstrate that the lectin domains of the lectican family proteoglycans bind tenascin-R, and that the binding is a protein–protein interaction with FNIII domains of the tenascin-R. The structure of the C-terminal domain of lecticans is similar to the carbohydrate binding proteins selectins in that both protein families contain a C-type lectin domain and are composed of a similar combination of domains. Furthermore, recombinant lectin domains of versican and aggrecan have been shown to bind simple sugars (8–11). Thus, the protein–protein interaction through the C-type lectin domain of lecticans was unexpected. Yet, there is at least one other example of a C-type lectin domain that mediates a protein–protein interaction; the C-type lectin domain of FcɛRII, a lymphocyte low-affinity receptor for IgE, binds to the Cɛ3 domain of IgE independent of any carbohydrates (25, 26). It may well be that more molecules that are classified as lectins based on primary sequence similarities actually have protein ligands, even if they do bind carbohydrates in vitro.
This study demonstrates that all lectican lectins bind tenascin-R fragments containing FNIII domain 4. The binding to the fragment consisting only of domain 4 is weak, and strong binding requires at least one neighboring domain (domain 3 or 5) in addition to domain 4. Thus, although these data suggest that the crucial determinant is present in domain 4, the binding site could not be dissected further. It is possible that a dual interaction in which two FNIII domains interact with two different sites on the brevican lectin is required for a high-affinity interaction. Alternatively, only one FNIII domain (FN4) may be directly involved in interaction, but requires another FNIII domain for the correct conformation of the binding site. In the case of FcɛRII–IgE binding, the presence of Cɛ4 domain of IgE is necessary for the FcɛRII-binding site in Cɛ3 to assume the correct conformation for the interaction (25).
Although we showed that all lecticans can specifically bind tenascin-R in vitro, this may not be the case in vivo. In fact, we found that the neurocan lectin domain, which showed the lowest affinity to tenascin-R fragments in surface plasmon resonance analysis, does not bind in ligand blotting assay to natural tenascin-R present in brain extracts (not shown). On the other hand, our results suggest that tenascin-R is a physiological ligand of brevican. First, the brevican lectin domain showed highest affinity for tenascin-R among the four lecticans (Table 1). The difference in affinity is quite significant; KD of brevican lectin is 12-fold lower than that of aggrecan lectin, the next strongest binder, and 30-fold lower than that of neurocan lectin. Second, temporal expression patterns of tenascin-R and brevican are similar; the expression of both tenascin-R and brevican begins late during development and both are expressed in adult rat brain (13, 27). Third, we found tenascin-R to coprecipitate with brevican from rat brain extracts. These results suggest that the binding of brevican and tenascin-R we demonstrated here reflects an in vivo interaction between these proteins.
Given that both tenascin-R and chondroitin sulfate proteoglycans have been found to inhibit neural cell adhesion and neurite outgrowth, the brevican–tenascin-R complex could constitute a particularly repulsive substrate. Such a substrate may provide a highly effective barrier against axonal extension and synapse formation. Alternatively, the lecticans could modulate the repulsive activity of tenascin-R. The repulsive activity has been mapped to the FNIII domain 3–5 of tenascin-R (19), apparently overlapping with the lectican binding region. The binding of lecticans could, by steric hindrance, neutralize the repulsive activity of tenascin-R. Thus, future experiments testing how lecticans affect the repulsive activity of tenascin-R (or vice versa) would provide crucial information for understanding the physiological significance of these interactions. Another issue to be studied is the physiological significance of the reported binding of versican and aggrecan lectins to simple sugars and glycosaminoglycans in vitro (8–11). Although this study has clearly ruled out carbohydrates as binding sites on tenascin-R, there remains a possibility that lectican lectins may also have physiological carbohydrate ligands, as reflected by their binding to these simple carbohydrates. Such putative carbohydrate binding may modulate the protein–protein interactions of lecticans and tenascin-R.
Acknowledgments
We thank Dr. Minoru Fukuda for providing Ig-chimera of l-selectin, Dr. Zhi-Cheng Xiao for constructs for tenascin-R fusion proteins, and Drs. Fukuda and Bill Stallcup for critical reading of the manuscript. This work was supported by Grants CA28896 (to E.R.), NS32717 (to Y.Y.), and Cancer Center Support Grant CA30199 from the National Cancer Institute and the National Institute for Neurological Disorders and Stroke, National Institutes of Health, Department of Health and Human Services. A.A. was supported by a fellowship from the Swedish Cancer Research Fund.
ABBREVIATIONS
- FNIII
fibronectin type III
- GST
glutathione S-transferase
- rCLD
recombinant C-type lectin domain
- EGF
epidermal growth factor
References
- 1.Ruoslahti E. Glycobiology. 1996;6:489–492. doi: 10.1093/glycob/6.5.489. [DOI] [PubMed] [Google Scholar]
- 2.Doege K, Sasaki M, Horigan E, Hassell J R, Yamada Y. J Biol Chem. 1987;262:17757–17767. [PubMed] [Google Scholar]
- 3.Zimmermann D R, Ruoslahti E. EMBO J. 1989;8:2975–2981. doi: 10.1002/j.1460-2075.1989.tb08447.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rauch U, Karthikeyan L, Maurel P, Margolis R U, Margolis R K. J Biol Chem. 1992;267:19536–19547. [PubMed] [Google Scholar]
- 5.Yamada H, Watanabe K, Shimonaka M, Yamaguchi Y. J Biol Chem. 1994;269:10119–10126. [PubMed] [Google Scholar]
- 6.Nelson R M, Venot A, Bevilacqua M P, Lindhardt R J, Stamenkovic I. Annu Rev Cell Dev Biol. 1995;11:601–631. doi: 10.1146/annurev.cb.11.110195.003125. [DOI] [PubMed] [Google Scholar]
- 7.Bevilacqua M P, Nelson R M. J Clin Invest. 1993;91:379–387. doi: 10.1172/JCI116210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Halberg D F, Proulx G, Doege K, Yamada Y, Drickamer K. J Biol Chem. 1988;263:9486–9490. [PubMed] [Google Scholar]
- 9.Saleque S, Ruiz N, Drickamer K. Glycobiology. 1993;3:185–190. doi: 10.1093/glycob/3.2.185. [DOI] [PubMed] [Google Scholar]
- 10.Ujita M, Shinomura T, Ito K, Kitagawa Y, Kimata K. J Biol Chem. 1994;269:27603–27609. [PubMed] [Google Scholar]
- 11.Aspberg A, Binkert C, Ruoslahti E. Proc Natl Acad Sci USA. 1995;92:10590–10594. doi: 10.1073/pnas.92.23.10590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Pesheva P, Spiess E, Schachner M. J Cell Biol. 1989;109:1765–1778. doi: 10.1083/jcb.109.4.1765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yamaguchi Y. Perspect Dev Neurobiol. 1996;3:307–317. [PubMed] [Google Scholar]
- 14.Yamada H, Watanabe K, Shimonaka M, Yamasaki M, Yamaguchi Y. Biochem Biophys Res Commun. 1995;216:957–963. doi: 10.1006/bbrc.1995.2713. [DOI] [PubMed] [Google Scholar]
- 15.Aruffo A, Kolanus W, Walz G, Fredman P, Seed B. Cell. 1991;67:35–44. doi: 10.1016/0092-8674(91)90570-o. [DOI] [PubMed] [Google Scholar]
- 16.Stallcup W B, Cohn M. Exp Cell Res. 1976;98:285–297. doi: 10.1016/0014-4827(76)90440-7. [DOI] [PubMed] [Google Scholar]
- 17.Yasugi E, Yokoyama Y, Seyama Y, Kano K, Hayashi Y, Oshima M. Biochem Biophys Res Commun. 1995;216:848–853. doi: 10.1006/bbrc.1995.2699. [DOI] [PubMed] [Google Scholar]
- 18.Pouyani T, Seed B. Cell. 1995;83:333–343. doi: 10.1016/0092-8674(95)90174-4. [DOI] [PubMed] [Google Scholar]
- 19.Xiao Z, Taylor J, Montag D, Rougon G, Schachner M. Eur J Neurosci. 1996;8:766–782. doi: 10.1111/j.1460-9568.1996.tb01262.x. [DOI] [PubMed] [Google Scholar]
- 20.Gill S C, von Hippel P H. Anal Biochem. 1989;182:319–326. doi: 10.1016/0003-2697(89)90602-7. [DOI] [PubMed] [Google Scholar]
- 21.Sueyoshi S, Tsuboi S, Sawada-Hirai R, Dang U N, Lowe J B, Fukuda M. J Biol Chem. 1994;269:32342–32350. [PubMed] [Google Scholar]
- 22.Kruse J, Keilhauer G, Faissner A, Timple R, Schachner M. Nature (London) 1985;316:146–148. doi: 10.1038/316146a0. [DOI] [PubMed] [Google Scholar]
- 23.Imai Y, Lasky L A, Rosen S D. Nature (London) 1993;361:555–557. doi: 10.1038/361555a0. [DOI] [PubMed] [Google Scholar]
- 24.Sako D, Comess K M, Barone K M, Camphausen R T, Cumming D A, Shaw G D. Cell. 1995;83:323–331. doi: 10.1016/0092-8674(95)90173-6. [DOI] [PubMed] [Google Scholar]
- 25.Vercelli D, Helm B, Marsh P, Padlan E, Geha R, Gould H. Nature (London) 1989;338:649–651. doi: 10.1038/338649a0. [DOI] [PubMed] [Google Scholar]
- 26.Bettler B, Maier R, Rüegg D, Hoftetter H. Proc Natl Acad Sci USA. 1989;86:7118–7122. doi: 10.1073/pnas.86.18.7118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wintergerst E S, Fuss B, Bartsch U. Eur J Neurosci. 1993;5:299–310. doi: 10.1111/j.1460-9568.1993.tb00497.x. [DOI] [PubMed] [Google Scholar]
- 28.Yamada, H., Fredette, B., Shitara, K., Hagihara, K., Miura, R., Ranscht, B., Stallcup, W. B. & Yamaguchi, Y. (1997) J. Neurosci., in press. [DOI] [PMC free article] [PubMed]