Abstract
Using electrophysiological and molecular techniques, we investigated the molecular nature of an inwardly rectifying K+ channel in bovine parotid acinar (BPA) cells and examined its role in setting resting membrane potential. In whole-cell recordings from freshly isolated BPA cells, a predominant current was a K+ current rectified strongly in the inward direction. An inward conductance of the inwardly rectifying K+ (Kir) current was proportional to [K+]o0.57. The selectivity sequence based on permeability ratios was K+ (1.00) > Rb+ (0.63) >> Li+ (0.04) = Na+ (0.02) and the sequence based on conductance ratios was K+ (1.00) >> Rb+ (0.03) = Li+ (0.03) = Na+ (0.02). The current was blocked by extracellular Ba2+ and Cs+ in a voltage- and a concentration-dependent manner, with a Kd at 0 mV of 11.6 µm and 121 mm, respectively. Cell-attached patch measurements identified 27 pS K+ channels as being the most likely to mediate whole-cell Kir currents. Addition of Ba2+ (100 µm) to the bathing solution reversibly depolarized the resting membrane potential in intact unstimulated cells. RT-PCR of RNA from bovine parotid cells revealed transcripts of bovine Kir2.1 (bKir2.1). HEK293 cells stably expressing bKir2.1 cloned from bovine parotid exhibited whole-cell and single channel Kir currents, of which electrophysiological characteristics were quantitatively similar to those of native Kir currents. Immunohistochemical studies showed a bKir2.1 immunoreactivity in BPA cells. Collectively, these results suggest that Kir2.1 may mediate native Kir currents responsible for setting resting membrane potential in BPA cells and might be, at least in part, involved in spontaneous secretion in ruminant parotid glands.
Unlike most other mammals, ruminants secrete large volumes of Na+ and HCO3−-rich and isotonic saliva, roughly equivalent to five to six exchanges of plasma water per day, which not only serve to buffer the acid produced by microbial fermentation in the rumen (Church, 1988), but also play an important role in electrolyte and water homeostasis in ruminants (Silanikove, 1994). A large fraction of this saliva is supplied by the parotid glands. The parotid glands of ruminants also differ strikingly from most other mammalian salivary glands in secreting continuously remarkable quantities of saliva even during resting (Coats et al. 1956; Kay, 1958, 1960; Meot et al. 1997). It has been shown that the parotid glands of unweaned lambs and other juvenile ruminants exhibit little, if any, resting secretion until the ingestion of solid food commences (Kay, 1960), implying developmental regulation of the resting secretion. Previous studies also showed that the resting secretion persists after denervation of the glands or the administration of autonomic blocking agents in adult cattle and sheep (Coats et al. 1956; Coats & Wright, 1957; Kay, 1960). Furthermore, secretion has been demonstrated in isolated perfused sheep parotid glands (Coats et al. 1956), strongly suggesting that it may be attributable to the ‘nature’ of the glands. However, the cellular and molecular mechanisms underlying so-called spontaneous secretion remain largely unknown.
As in other secretory epithelia, the most widely studied model of secretomotor agonist-evoked fluid and electrolyte secretion in salivary glands is based on the secondary active transport of anions, principally Cl− (and/or HCO3−) ions (Cook et al. 1994). In this model, K+ channels play a critical role in primary secretion in salivary glands mainly for two reasons. First, K+ channels in the basolateral membrane of acinar cells keep the apical (as well as the basolateral) cell membrane potential more negative than the Nernst potential for anions such as Cl− (and/or HCO3−), thereby providing a driving force for the sustained electrogenic anion efflux across the apical membrane. Second, the basolateral K+ channels provide a pathway, through which K+ ions pumped into the cells by the Na+-K+-ATPase and/or the Na+-K+-2Cl− cotransporter are recycled. Since most well-studied salivary glands are stimulated to secrete by agonists such as acetylcholine (ACh) that increase cytosolic Ca2+ concentration in acinar cells, the basolateral K+ channels are mediated by Ca2+-activated K+ channels (Cook et al. 1994). Assuming that this model, in terms of the role of K+ channels, is also applicable to spontaneous secretion in the parotid glands of ruminants, it is possible that there are K+ channels that are distinct from Ca2+-activated K+ channels, which allow the ruminant parotid secretory cells to drive fluid and electrolyte secretion even at resting state.
Previous cell-attached patch and whole-cell patch-clamp studies have demonstrated that the basolateral membranes of unstimulated sheep parotid acinar cells contain at least four types of K+ channel, of which two are common enough to be considered as having a role to play in a physiological function (Wegman et al. 1992; Ishikawa et al. 1993; Ishikawa & Cook, 1993). One of these is a tetraethylammonium (TEA)-sensitive, 250 pS voltage- and Ca2+-activated K+ channel (maxi-K+ channel) that underlies an outwardly rectifying K+ current observed in whole-cell patch-clamp studies. The other is a TEA-insensitive 30 pS K+ channel, which mediates an inwardly rectifying whole-cell K+ current. As in other salivary acinar cells, the maxi-K+ channel can be activated by membrane depolarization and by the addition of ACh to the bathing solution in cell-attached configuration. On the contrary, the cell-attached patch study shows that the activity of the inward rectifier, which is the most frequently observed channel type at resting membrane potential, is not affected by the addition of ACh to the bathing solution. Interestingly, the inwardly rectifying K+ currents have not been found in other non-ruminant salivary acinar cells that have been so far studied by the patch-clamp technique (Iwatsuki et al. 1985; Cook et al. 1988; Smith & Gallacher, 1992; Ishikawa & Murakami, 1995; Hayashi et al. 1996; Park et al. 2001). Furthermore, it has been shown that spontaneous secretion in sheep parotid is not blocked by TEA (Wright & Blair-West, 1990; Cook et al. 1992) and that the inward rectifier is absent from the parotid acinar cells of unweaned lambs (Hirono et al. 1994), which do not secrete spontaneously (Kay, 1960). Although it is tempting to hypothesize that inwardly rectifying K+ channels may have a role to play in spontaneous secretion, neither the physiological function nor the molecular basis of the inward rectifier has yet been determined experimentally. Moreover, it is not known whether the inwardly rectifying K+ channels are commonly expressed in other ruminant parotid glands.
In this paper, we addressed these unsolved questions. Using whole-cell and single channel recording techniques we have first demonstrated that unstimulated bovine parotid acinar (BPA) cells contain an inwardly rectifying K+ channel as found in sheep parotid cells. We have then shown that the channels are involved in setting resting membrane potential of intact BPA cells. Finally, using electrophysiological, molecular and immunohistochemical techniques, we have identified Kir2.1 as a molecular candidate of the inwardly rectifying K+ channels. Together, the work forms the first functional and molecular characterization of the inward rectifying K+ channels in ruminant parotid acinar cells. Based on the present and previous studies on bovine and sheep parotid acinar cells, we propose that Kir 2.1 may be, at least in part, involved in setting resting membrane potential during spontaneous secretion, which is a special feature of the parotid glands of ruminants.
METHODS
Patch-clamp experiments
Bovine parotid glands were obtained from a local abattoir (Hokkaido Ebetsu Meat Inspection Center, Ebetsu, Japan). The animals were both males and females. The glands were dissected immediately after slaughter of the animals, put in the standard NaCl-rich solution (solution B in Table 1) on ice and then transported to the laboratory. Isolated acini and acinar cells were prepared as previously described for sheep parotid acinar cells (Ishikawa et al. 1993), with some modifications. In brief, the parotid glands were excised, placed in the standard NaCl-rich solution, trimmed of fat, connective tissues and blood vessels, and cut into small pieces with scissors. The pieces were transferred into 5 ml of the standard bath solution (solution B) without CaCl2 and MgCl2, containing collagenase (1.5 mg ml−1, Worthington Type II, Freehold, NJ, USA), and incubated for 30 min at 37 °C in a shaking water bath. After gentle trituration with a 10 ml pipette, the medium was then replaced with a fresh solution and the tissue incubated once more for 30 min. The tissue was filtered through 148 µm nylon mesh, then centrifuged (40 g, 5 min) and washed twice with the standard bath solution. The filtrate, which contained isolated cells and small clumps of cells, was stored at room temperature (20–23 °C) or at 4 °C until used.
Table 1.
Experimental solutions in patch-clamp experiments (all concentrations are mM)
| Whole-cell | Single channel | ||||
|---|---|---|---|---|---|
| Component | Solution A(pipette) | Solution B(bath) | Solution C(bath) | Solution D(bath) | Solution E (pipette) |
| NaCl | — | 145 | — | — | — |
| KCl | 10 | 5 | 150 | 140 | 135 |
| NMDG-Cl | — | — | — | 10 | — |
| TEA-Cl | — | — | 10 | 10 | — |
| K-glutamate | 100 | — | — | — | — |
| CaCl2 | — | 1 | 1 | 1 | — |
| MgCl2 | 1 | 1 | 1 | 1 | 1 |
| EGTA | 10 | — | — | — | 5 |
| NaOH | — | 4.3 | — | — | — |
| KOH | 30 | — | 4.3 | 4.3 | 15 |
All solutions contained Hepes (10 mM) and glucose (10 mM) and were titrated to pH 7.4 with NaOH or KOH. TEA: tetraethylammonium, NMDG: N-methyl-D-glucamine.
The cell preparations described above were plated out onto a coverglass and transferred to a chamber mounted on an Olympus inverted microscope. Current recordings were made using the standard patch-clamp technique (Hamill et al. 1981). Patch-clamp pipettes were pulled from glass capillaries (LG16, Dagan, Minneapolis, MN, USA) using a vertical puller (Model PP-830, Narishige, Tokyo, Japan) so as to have resistances of 1–3 MΩ for whole-cell recordings and 3–10 MΩ for single channel recordings when filled with the standard potassium glutamate-rich solution (solution A in Table 1). The reference electrode was a Ag-AgCl pellet, which was connected to the bathing solution via an agar bridge filled with the standard NaCl-rich bathing solution.
An Axopatch-1D patch-clamp amplifier (Axon Instruments, Union City, CA, USA) was used to measure whole-cell and single channel currents. In whole-cell experiments, the amplifier was driven by pCLAMP6 software (Clampex, Axon Instruments) to allow the delivery of voltage-step protocols with concomitant digitization of the current. The whole-cell currents were filtered at 500 Hz with an internal four-pole Bessel filter, sampled at 2 kHz and stored directly on the computer's hard disk through the Digidata 1200 (Axon Instruments). Subsequent current analysis was performed using programs supplied with pCLAMP 6 software (Clampfit). Current-voltage (I-V) relations were studied using 10 mV voltage pulses, each of 400 ms duration, and voltage pulses were separated by 3 s intervals during which the cell potential was held at either −40 or 0 mV. Steady-state whole-cell currents were measured at 400 ms from the start of each voltage pulse. In experiments where the effects of extracellular Ba2+ were examined, I-V relations were studied using 10 mV voltage pulses, each of 3 s duration, and voltage pulses were separated by a 7 s interval. Steady-state whole-cell currents were measured at 3 s from the start of each voltage pulse. As an alternative to voltage steps, voltage ramps were employed. Typically, the command voltage was varied from −120 to +40 mV over a duration of 800 ms every 10 s as described in the text. The capacitance transient current was compensated in most experiments using the Axopatch-1D amplifier. The cell capacitance was 34.5 ± 0.7 pF (n = 207). The average series resistance (Rs), which was 19.2 ± 0.6 MΩ (n = 233), was not electrically compensated. Therefore, the conductance of currents in the nanoampere range were underestimated as a result of the voltage decrease across the Rs, and the command voltages were not corrected for this, except where external K+ dependence and ion selectivity of the whole-cell conductance were estimated (Fig. 1C, D and F).
Figure 1. Dependence of inwardly rectifying currents on extracellular K+ concentration.
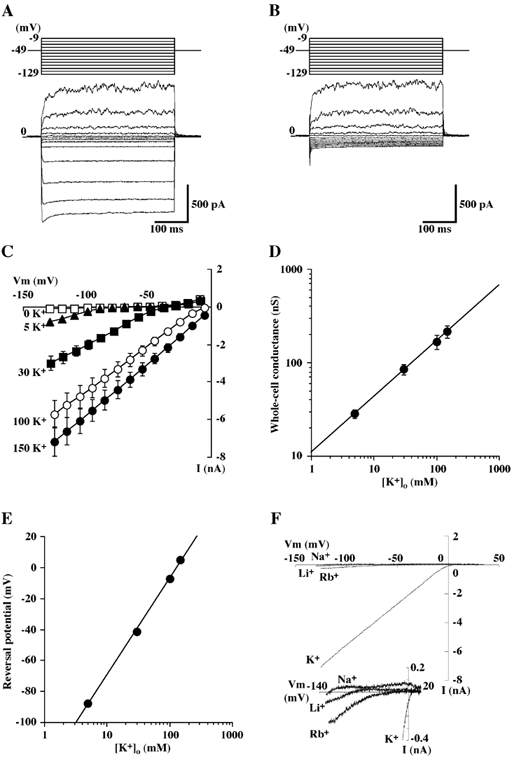
A and B, representative tracings of whole-cell currents obtained from a single bovine parotid acinar (BPA) cells in the presence (A) and the absence (B) of 5 mm K+ (solution B). Hyperpolarizing and depolarizing pulses 400 ms in duration were applied from a holding potential of −49 mV to potentials between −129 and −9 mV in 10 mV intervals. The pipette was filled with a potassium glutamate-rich solution (solution A). C, steady state current-voltage (I-V) relationships of whole-cell currents recorded from BPA cells bathed in different extracellular K+ concentrations. Each point represents the mean ± s.e.m. of 13 experiments. D, log-log plot of the conductance of the inward current as a function of the extracellular K+ concentration; the line is the line of best fit and the data are from C. E, semi-logarithmic plot of the reversal potential of the inward current as a function of extracellular K+ concentration. Each point represents the mean ± s.e.m., but when this was so small as to lie within the symbols, it has been omitted; the line is the line of best fit and the data are from C. F, representative instantaneous I-V relations of whole-cell currents obtained from single BPA cell with Na+, Li+ or Rb+ substituted for K+ (154.3 mm) in the bathing solution (solution C). Ramp command voltages were applied from −125 to +35 mV at a rate of 200 mV s−1. Inset, expanded scale trace for the same experiments.
Single channel currents were recorded in cell-attached patch configuration (Hamill et al. 1981). Single channel currents were filtered at 500 Hz with an internal filter of the instrument, sampled at 2 kHz and stored directly on the computer's hard disk through the Digidata 1200 (Axon Instruments). The data were acquired and analysed with pCLAMP6 software (FETCHEX and FETCHAN, Axon Instruments). The unitary single channel current was measured as the peak-to-peak intervals in the amplitude histogram or estimated as the mean of each current transition.
By convention for cell-attached patches, voltage (-Vp) is displacement of intracellular potential, where Vp is the pipette potential. Therefore, 0 mV indicates that the patch membrane was held at resting membrane potential and +20 mV indicates that it was held at a 20 mV more positive potential than resting membrane potential. Downward single channel currents correspond to K+ flux from the pipette solution to the intracellular side of the membrane.
In both single- and whole-cell experiments, the pipette potential was corrected for the liquid junction potential between the pipette solution and the external solution, and between the external solution and the agar bridge as described by Barry & Lynch (1991) and Neher (1992).
The experimental solutions are described in Table 1. Unless stated otherwise, 10 mm tetraethylammonium chloride (TEA-Cl) was added to the bathing solution in whole-cell experiments to minimize a voltage-dependent, outwardly rectifying current attributable to maxi-K+ channels (Fig. 1A and B). Relative permeabilities for various cations were determined using external solutions (solution C) containing an equimolar amount of the chloride salt of the test cation. N-2-hydroxyethylpiperazine-N‘-ethane-sulfonic acid (Hepes) and EGTA were obtained from Sigma (St Louis, MO, USA), and TEA was obtained from Wako (Osaka, Japan). Other chemicals were of reagent grade.
All experiments were performed at room temperature (20–23 °C). Bath solution changes were accomplished by gravity feed from reservoirs. The results are reported as means ± s.e.m. of independent experiments (n), where n refers to the number of cells patched. Statistical significance was evaluated using Student's two-tailed paired t test or one-way analysis of variance (ANOVA) as appropriate. A value of P < 0.05 was considered significant.
Data fitting
The Km values for external K+ were calculated from the single channel currents induced by varying the concentration (C) of K+ in the pipette solution from 5 to 150 mm. The data were fitted to the Michaelis-Menten equation:
| (1) |
where γmax is the maximal conductance and Km is the half-activation constant using a non-linear fit program.
To analyse titration curves for Ba2+ or Cs+ inhibition of the macroscopic K+ current, the ratio I/Io measured in the presence (I) of the blocker to that in its absence (Io), normalized to the value in the absence of extracellular K+, was described by the following equation:
| (2) |
where Ki is the inhibitory constant of the blocker, A is the concentration of the blocker and n is the pseudo Hill coefficient. In the case of a voltage-dependent block, Ki(V) is the voltage-dependent inhibitory constant, which has been expressed by Woodhull (1973) as a Boltzmann relationship with respect to the voltage:
| (3) |
where Ki(0) is the inhibitory constant at 0 mV, Z is a slope parameter, V is the voltage, and F, R and T have their conventional meanings. Z is equal to the product of the actual valence of the blocking ion and the fraction of the membrane potential (or electrical distance) delta (δ) acting on the ion. To determine the kinetics of Ba2+, the onset of Ba2+ block was fitted with a single exponential.
Cloning of Kir 2.1 from bovine parotid cells
Total RNA was extracted from bovine parotid cells prepared as described above using TRIzol reagent (Gibco BRL, Tokyo, Japan) following the manufacturer's instructions. Reverse transcriptase-polymerase chain reaction (RT-PCR) was performed using SuperScript one-step RT-PCR system (Gibco BRL). The specific oligonucleotide primers for amplification of Kir2.1 were as follows:
| (1) |
Samples were cycled first at 50 °C for 30 min and at 94 °C for 2 min followed by 35 cycles of 30 s at 94 °C, 30 s at 50 °C and 1 min at 72 °C. The size of the expected fragment was 636 bp.
First-strand cDNA was also generated from total RNA using SuperScriptII RT (Gibco BRL). To amplify a long fragment (the expected size was 4699 bp) of bovine Kir2.1 (bKir2.1), the PCR reaction was performed with TaKaRa LA Taq (Takara Shuzo Co. Ltd, Japan). The specific oligonucleotide primers were as follows:
| (2) |
Samples were subjected to 35 cycles of 94 °C for 30 s, 69 °C for 30 s and 72 °C for 5 min. The oligonucleotide primers described above were derived from the published sequences of bKir2.1 (Forsyth et al. 1997; GenBank accession No. U95369). As a control, β-actin-cDNA was amplified using the following primers and a 510 bp product was obtained:
PCR products were resolved in 1 % agarose gels with ethidium bromide.
Based on the complete sequence information of bKir2.1, we amplified three overlapping PCR fragments from total mRNA, which was extracted with BioMag mRNA purification kit (Polysciences, Inc., Warrington, PA, USA), by RT-PCR with high fidelity DNA polymerase (Pfu-Turbo, Stratagene, LaJolla, CA, USA) using the following primers:
 |
(3) |
 |
(4) |
 |
(5) |
The fragments were first subcloned into pGEM-T Easy vector (Promega, Tokyo, Japan) and sequenced on both strands, then assembled by triple ligation of fragments EcoRI (in vector)/KpnI (nt 85–601), KpnI/SacI (nt 601–1337), SacI/Sal I (nt 1337–2863) into pCI-neo (Promega) plasmid linearized by EcoRI and Sal I.
HEK293 cells expressing bovine Kir 2.1
HEK293 cells stably expressing bovine Kir2.1 were generated by transfecting cells with a plasmid construct encoding bKir2.1 in the mammalian expression vector pCI-neo using lipofectamine (Life Technologies, Inc., Tokyo, Japan). Mock transfected cells were also prepared using the same vector. G418 (0.8 mg ml−1)-resistant colonies were purified and tested for bKir2.1 expression by whole-cell patch clamp. The cells were then maintained in Dulbecco's modified Eagle's medium (DMEM) supplemented with 10 % fetal bovine serum, penicillin (100 u ml−1)/streptomycin (100 µg ml−1) and 0.2 mg ml−1 G418. The cells were grown at 37 °C in a water-saturated 5 % CO2, 95 % air atmosphere, and passaged 1–2 times weekly. For patch-clamp experiments, the cells were seeded on a coverglass and patched 2–6 days after seeding. For the purpose of the present study, we chose and characterized a clone of the stable cell line, which exhibited a Ba2+-sensitive whole-cell current amplitude similar to that in BPA cells. This was especially important for a quantitative comparison of electrophysiological properties between native and expressed Kir whole-cell currents, because errors introduced by uncompensated series resistance were difficult to avoid due to a large amount of Kir currents in BPA cells. Experimental conditions were exactly the same as those described for the freshly dissociated BPA cells. In whole-cell patch-clamp experiments, the cell capacitance and the average series resistance (Rs) were 18.9 ± 1.0 pF (n = 36) and 9.1 ± 0.7 MΩ (n = 37), respectively.
Immunocytochemistry of bovine Kir2.1
Bovine parotid was dissected into small pieces which were fixed in 3 % paraformaldehyde and 0.1 m phosphate buffer (pH 7.0). After washing with the buffer solution, the specimens were embedded in compound (O.C.T. Compound, Tissue Tek, Torrance, CA, USA) and quick-frozen in liquid nitrogen. Cryosections (10–20 µm) were first labelled with anti-Kir2.1 antibody (developed in rabbit using a highly purified peptide (C)NGVPESTSTDTPPDIDLHN, corresponding to amino acid residues 392–410 of human and 392–410 of bovine Kir2.1 with additional N-terminal cysteine as the immunogen) (Sigma), and then secondary labelled with FITC-conjugated goat affinity-purified antibody to rabbit IgG (Cappel, Aurora, OH, USA). The non-immune serum was used as a primary antibody for the control staining. The specimens were observed in a photomicroscope (BX60; Olympus, Tokyo, Japan) equipped with BX-FLA and photographed by digital cooled CCD camera (SV-16E, Koheisha, Saitama, Japan).
RESULTS
Inwardly rectifying K+ conductance in freshly dissociated BPA cells
The characteristic features of an inward rectifier K+ (Kir) channel are: (1) currents through the channel flow more readily in the inward direction than outward, (2) the non-linear dependence of conductance on external K+ concentration and (3) it is susceptible to blocking of the channel pore by external cations such as Ba2+ and Cs+ (Hille, 1992; Nichols & Lopatin, 1997). In order to examine whether BPA cells express functional Kir channels, we first determined the steady-state whole-cell I-V relation for single BPA cells. When the cells were dialysed with the standard potassium glutamate-rich pipette solution (solution A) in the NaCl-rich bath containing 5 mm K+ (solution B), they exhibited an inwardly rectifying current (Fig. 1A), which was seen at cell potentials more negative than −89 mV, was almost instantaneous in onset (< 10 ms) and was not blocked by the addition of 10 mm TEA to the bathing solution (n = 15) (not shown). The steady-state inward current was due to K+ influx, because it was abolished by replacement of K+ (5 mm) in the bathing solution with equimolar Na+ (Fig. 1B). The slope conductance of the inward current determined from the linear section of the I-V relationships increased as extracellular K+ concentration ([K+]o) was raised and the values in solutions containing 5, 30, 100 and 150 mm K+ were 28.3 ± 2.8, 86.1 ± 10.6, 168.2 ± 29.2 and 219.5 ± 33.3 nS, respectively (n = 13) (Fig. 1C). A plot of the logarithm of the slope conductance as a function of the logarithm of [K+]o revealed a rectilinear relation with a slope of 0.57 ± 0.04 (n = 13) (Fig. 1D). The reversal potentials of the inward current, as estimated by extrapolation from the data lying in the range in which its I-V relation was linear, were −87.9 ± 1.1 mV (5 mm [K+]o), −41.5 ± 0.8 mV (30 mm [K+]o), −7.4 ± 0.8 mV (100 mm [K+]o) and +4.8 ± 1.1 mV (150 mm [K+]o). These values are in good agreement with equilibrium potential for potassium (EK) values in these solutions predicted from the Nernst equation. When the reversal potential was plotted as a function of the logarithm of the extracellular K+ concentration (Fig. 1E), a rectilinear relation was also obtained with a slope of +62.6 ± 1.0 mV per decade. Collectively, these results show that the inwardly rectifying conductance is mediated by K+ channels that activate at membrane potentials below EK and pass inward K+ currents. The inwardly rectifying K+ (Kir) current was observed in 90.1 % (210 out of 233) of the cells tested.
The relative permeabilities for various cations of the whole-cell currents were also examined. Figure 1F shows families of whole-cell Ba2+-sensitive (100 µm) ramp currents recorded from a single cell bathed with either K+, Rb+, Li+ or Na+ as the sole monovalent cation (see below for the Ba2+ block of the currents). The selectivity sequence calculated from relative slope conductances (GX/GK) in the voltage range between −100 and −80 mV or −60 mV was K+ (1.00) >> Rb+ (0.03 ± 0.01) = Li+ (0.03 ± 0.01) = Na+ (0.02 ± 0.01) (n = 10). The reversal potentials were also estimated from the instantaneous I-V relationship of the Ba2+-sensitive current. Assuming that the Ba2+-sensitive current was solely carried by these monovalent cations, we calculated the permeability ratio (PX/PK) from the shift in reversal potential (ΔVrev) when monovalent cation X is substituted for external K+ (Hille, 1992); that is, from:
where R, T and F have their usual thermodynamic meanings. The relative permeability (PX/PK) sequence was K+ (1.00) > Rb+ (0.63 ± 0.15) >> Li+ (0.04 ± 0.01) = Na+ (0.02 ± 0.00) (n = 10).
Figure 2A illustrates an example of the effects of Ba2+ on the Kir currents obtained from a single BPA cell. Whole-cell currents were evoked from a holding potential of −5 mV to that between −105 and −5 mV in the absence and the presence of various concentrations of Ba2+ in the bathing solution (solution C). Kir currents were inhibited by external Ba2+ in a concentration- and a voltage-dependent manner (Fig. 2B). The fractional inhibition of Kir currents (expressed as the Ba2+-sensitive current measured at different voltages and concentrations of Ba2+ divided by the Kir current in control) was also plotted against the concentration of Ba2+ (Fig. 2B). The data shown in Fig. 2B were also fitted by eqn (2) (refer to Methods) to determine the apparent dissociation constant (Kd) and Hill coefficient at each potential. The following Kd values and Hill coefficient were obtained: e.g. 0.9 ± 0.2 µm and 1.32 ± 0.06 at −105 mV, 1.0 ± 0.2 µm and 1.26 ± 0.05 at −85 mV, 1.6 ± 0.2 µm and 1.15 ± 0.04 at −65 mV, and 2.5 ± 0.4 µm and 1.11 ± 0.05 at −45 mV, (n = 5). The Kd decreased e-fold per 36.1 ± 2.1 mV hyperpolarization; these data were fitted with eqn (3) (refer to Methods) with a Kd (0) of 11.6 ± 2.3µm and a slope (δ) of 0.36 ± 0.02 (n = 5) (Fig. 2C). Under the present experimental conditions, we could successfully fit the reduction in Kir current by 10µm Ba2+ by a single exponential decay function with time constant (τ) (Fig. 2D). Upon membrane hyperpolarization, Ba2+ block increases at a rate, which depends upon the voltage applied, the time constant (τ) being decreased e-fold per 36.7 ± 0.4 mV (n = 5).
Figure 2. Effects of Ba2+ on the Kir currents.
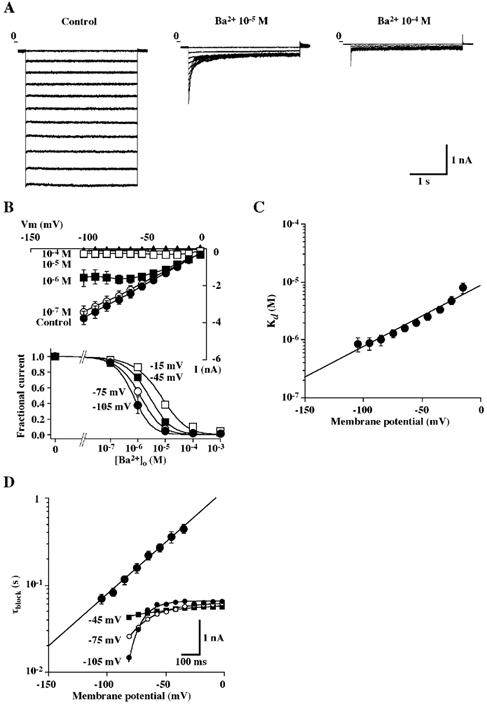
A, representative tracings of whole-cell currents obtained from a single BPA cells in the absence and the presence of Ba2+ (10−5 and 10−4m) in the bathing solution containing 150 mm KCl (solution C). Currents were elicited by voltage steps ranging from −105 to −5 mV in 10 mV increments, each of 3 s duration. Voltage pulses were separated by a 7 s interval and holding potential was −5 mV. B, upper panel, steady-state I-V relationships of whole-cell currents recorded from BPA cells in the presence of 150 mm KCl with varying concentrations of extracellular Ba2+. Steady-state whole-cell currents were measured at 3 s from the start of each voltage pulse. Each point represents the mean ± s.e.m. of 5 experiments. Lower panel, fractional current at steady-state plotted against Ba2+ concentration at a holding potential of −105, −75, −45 or −15 mV. Each point represents the mean ± s.e.m. of 5 experiments. The lines are fits to the Hill equation (eqn (2)). C, Ba2+ concentration dependence of the steady-state block of whole-cell currents at different holding potentials. Each point represents the mean ± s.e.m. of 5 experiments. The line is the fit to eqn (3). D, plot of the blocking time constant as a function of membrane potential. The data points were obtained in the presence of 10−5m Ba2+ and represent means ± s.e.m. of 5 experiments. Inset, representative traces of the currents obtained from a single BPA cell. The currents were elicited by voltage steps to −105, −75 and −45 mV from a holding potential of −5 mV in the presence of 10−5m Ba2+ in the bathing solution. The lines are the fit to a single exponential using a least-squares method.
We also characterized external Cs+ block of Kir currents in BPA cells. Figure 3A shows representative tracings of the effect of different concentrations of Cs+ on currents evoked by 400 ms voltage step from −7 to −117 mV or by 800 ms voltage ramp to membrane potentials between −127 and −7 mV. Inhibition of the current increased with both Cs+ concentration and hyperpolarization (Fig. 3A and B). As the membrane potential became more hyperpolarized, the current inhibition with Cs+ increased, even at the same concentration. When data were fitted by using eqn (2), the following Kd values and Hill coefficient were obtained: e.g. 1.0 ± 0.5 mm and 1.19 ± 0.03 at −117 mV, 2.2 ± 0.7 mm and 1.12 ± 0.03 at −97 mV, 4.3 ± 1.0 mm and 1.08 ± 0.10 at −77 mV, and 8.8 ± 1.3 mm and 0.99 ± 0.08 at −57 mV, (n = 6), respectively. The Kd decreased e-fold per 24.1 ± 3.1 mV with an estimated Kd (0) of 121.3 ± 17.0 mm, and a slope (δ) of 1.15 ± 0.11 (n = 6) (Fig. 3C).
Figure 3. Effects of Cs+ on the Kir currents.
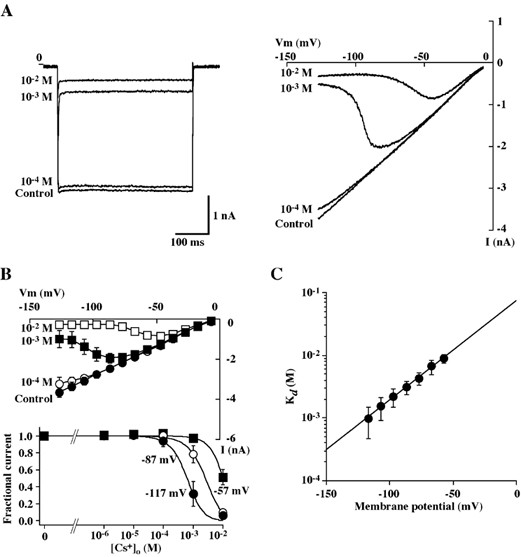
A, representative tracings of whole-cell currents obtained from a single BPA cell in the absence and the presence of Cs+ (10−4, 10−3 and 10−2m) in the bathing solution containing 140 mm KCl (solution D). Currents were elicited by voltage steps from a holding potential of −7 to −117 mV (left panel) or by voltage ramps from −127 to −7 mV at rate of 200 mV s−1 (right panel). B, upper panel, I-V relationships of whole-cell currents recorded from BPA cells in the presence of 140 mm KCl with varying concentrations of extracellular Cs+. Whole-cell currents were elicited by the voltage ramp protocol. Each point represents the mean ± s.e.m. of 5 experiments. Lower panel, fractional current at steady-state plotted against Cs+ concentration at a holding potential of −117, −87 or −57 mV. Each point represents the mean ± s.e.m. of 5 experiments. The lines are fits to the Hill equation (eqn (2)). C, Cs+ concentration dependence of the steady-state block of whole-cell currents at different holding potentials. Each point represents the mean ± s.e.m. of 6 experiments. The line is the fit to eqn (3).
Single channel currents responsible for macroscopic Kir currents in BPA cells
To identify single channel currents in BPA cells, which could be responsible for the macroscopic Kir currents, we performed cell-attached single channel recordings in BPA cells. Figure 4A shows typical single channel records and corresponding I-V relations obtained in three independent cell-attached patches with the standard KCl-rich pipette solution (solution E). The most frequently observed channel was the 27 pS channel, which made up almost 50 % of the channels observed (Fig. 4A, left panel). As also shown in Fig. 4A, the 16 pS channel (middle panel) was often observed in the same patches as the 27 pS openings (right panel). The incidence of the four K+ channel types in 107 cell-attached patches obtained from unstimulated BPA cells is also summarized in Table 2. Figure 4B shows the I-V relations of the 27 pS K+ channel in cell-attached patches on BPA cells for pipette solutions in which the K+ concentration was varied between 150 and 5 mm by the substitution of Na+ for K+. The relation between single channel conductance and pipette K+ concentration (Fig. 4C) shows a saturating relation that can be described by the Michaelis-Menten eqn (1) (refer to Methods) with a Km of 23.8 mm and maximum single channel conductance (γmax) of 29.6 pS. A plot of the logarithm of the single channel conductance as a function of the logarithm of pipette K+ concentration (Fig. 4D) reveals a rectilinear relation with a slope of 0.42, close to the value of 0.57 obtained for the whole-cell Kir conductance described above. A plot of the estimated reversal potential as a function of the logarithm of pipette K+ concentration (Fig. 4E) gives a rectilinear relation with a slope of +48.5 mV per decade.
Figure 4. Single channel records from cell-attached patches in BPA cells.
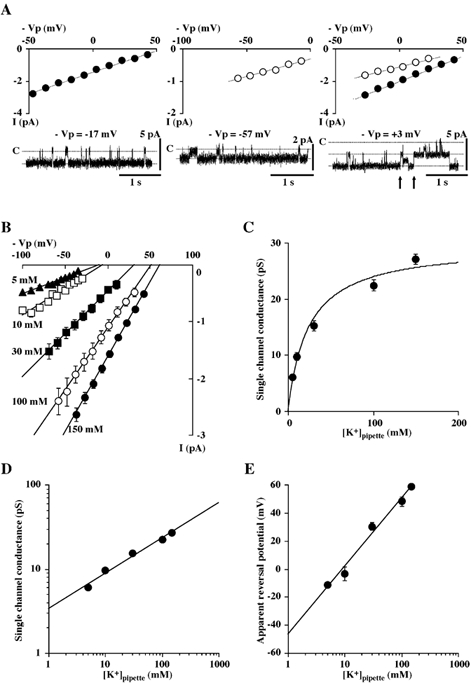
A, representative single channel tracings and corresponding I-V relations of the 27 pS (left and right panels, •) and the 16 pS (middle and right panels, ○) channels in three different cell-attached patches at different voltages (-Vp). In the right panel, a representative single channel tracing and corresponding I-V relations obtained from a patch containing both the 27 pS and the 16 pS channels are also shown. The arrows indicate before and after opening of the 16 pS channels. Voltage (-Vp) is displacement of intracellular potential. Zero millivolts indicates that patch membrane was held at resting membrane potential and +3 mV indicates that it was held at a potential 3 mV more positive than resting membrane potential. Data were obtained from the patch shown. The pipette solution was KCl-rich (solution E). B, single channel I-V relation of the 27 pS channel in cell-attached patches for pipette K+ concentrations of 150 mm (•, n = 14), 100 mm (○, n = 5), 30 mm (▪, n = 5), 10 mm (□, n = 5) and 5 mm (▴, n = 5). A solution containing 5 mm K+ (solution B) was used as a bath solution. The resting potential was most influenced by the equilibrium potential for potassium (EK) and was around −60 mV. C, plot of the conductances of the 27 pS channel in cell-attached patches as a function of pipette K+ concentration. The solid line is a nonlinear least-squares fit of the Michaelis-Menten eqn (1) (see Methods); data are from B. D, log-log plot of the single channel conductance of the 27 pS channel in cell-attached patches as a function of pipette K+ concentration. The solid line is the line of best fit; data are from B. E, semi-logarithmic plot of the apparent reversal potential of the 27 pS K+ channel as a function of pipette K+ concentration in cell-attached patches. The apparent reversal potential of the inward current was estimated by a linear least-squares method and by extrapolation from the data shown in B. The solid line is the line of best fit; data are from B.
Table 2.
Incidence of the four K+ channel types seen in bovine parotid acinar cells
| 27 pS K+ | 16 pS K+ | 60 pS K+ | 160 pS (maxi-K+) | |
|---|---|---|---|---|
| Conductance (pS) | 26.9 ± 0.5(54) | 15.5 ± 0.6(27) | 58.6 ± 5.4(18) | 158.5 ± 6.5(33) |
| Relative incidence (%) | 50.5(107) | 25.2(107) | 16.8(107) | 30.8(107) |
Data were obtained from cell-attached patch experiments using the pipette solution (solution A or E) and the bath solution (solution B). Values are means ± S.E.M.; (n) = number of cells.
Role of the inwardly rectifying K+ channels in setting membrane potential of unstimulated cells
If the Kir channels described above are responsible for spontaneous secretion, they should be, at least, involved in setting membrane potential of intact BPA cells. We next wanted to examine whether the Kir channels would contribute to setting membrane potential of intact unstimulated cells under conditions as close to the physiological situation as possible (i.e. conditions leaving the intracellular environment unaltered). To this end, we measured changes in Vm indicated by the reversal of the single K+ selective channel current (i.e. maxi-K+ channels) in cell-attached patch with the KCl-rich pipette solution (solution E) as described for human T lymphocytes (Verheugen et al. 1995). Given that intracellular K+ concentration in salivary acinar cells ranges between 120 and 150 mm at rest (Poulsen & Oakley, 1979; Izutsu et al. 1987), the theoretical reversal potential for K+ current, with high K+ solution in the pipette, will reverse when pipette potential (Vp) cancels resting membrane potential. Therefore, if Vm in unstimulated BPA cells is determined by Kir channel activity, block of the channels should depolarize Vm. In these experiments, we used Ba2+ rather than Cs+ to block the Kir channels, since Ba2+ was a good blocker of Kir currents at a physiological range of membrane potential as shown in Fig. 2B and Fig. 3B. As expected, addition of Ba2+ (100 µm) to the bathing solution consistently shifted the reversal potential (Vm) toward depolarized membrane potentials and the effect was more pronounced with a higher concentration of Ba2+ (1 mm) (Fig. 5A and B). The mean reversal potential changes induced by 100 µm and 1 mm Ba2+ were −6.3 ± 0.8 mV (n = 5) (P < 0.01) and −16.0 ± 1.8 mV (n = 8) (P < 0.001), respectively. These effects were completely reversible upon washout of the drug. We also examined the effect of addition of TEA (10 mm), a maxi-K+ channel blocker, to the bathing solution and found that it had little, if any, effect on the Vm, so that the mean reversal potential shift was +0.2 ± 1.2 mV (n = 5) (P = 0.88) (Fig. 5C). These results strongly suggest that Ba2+-sensitive Kir channels may be involved in setting resting membrane potential of the intact BPA cells.
Figure 5. Effects of Ba2+ on resting membrane potential of BPA cells.
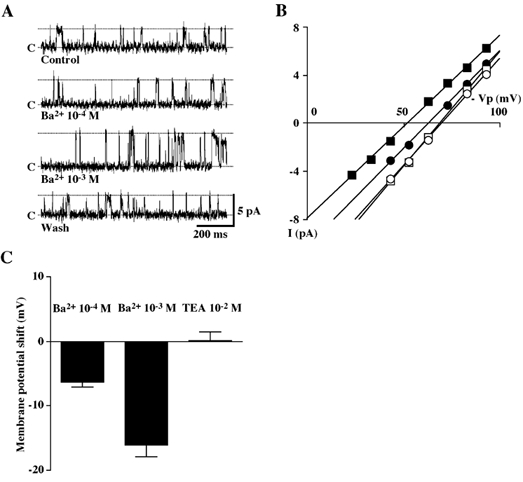
A, representative tracings of single maxi-K+ channel current in a cell-attached patch in the absence or the presence of Ba2+ (100 µm or 1 mm) in the bath solution. Voltage (-Vp) was +83 mV. The patch pipette was filled with the KCl-rich (150 mm K+) solution (solution E) and the bath contained the standard NaCl-rich solution (solution B). B, I-V relations of maxi-K+ channel obtained from the patch shown in A before (○), during (100 µm (•) or 1 mm (▪) addition of Ba2+ to the bathing solution and after washout of Ba2+ (□). C, Ba2+ (100 µm or 1 mm)-induced changes in Vm indicated by the reversal of single maxi-K+ channel current in cell-attached patches. Application of TEA (10 mm) to the bathing solution did not shift the reversal potential.
Molecular identification of Kir2.1 from bovine parotid cells
The electrophysiological properties of macroscopic Kir currents and the 27 pS K+ channels in unstimulated BPA cells led us to hypothesize that the Kir2 subfamily, in particular Kir2.1 originally cloned from mouse macrophage (Kubo et al. 1993) could be responsible for the native Kir currents. To test this hypothesis, we performed RT-PCR analysis using specific primers of described bKir2.1 (U95369) and confirmed that bovine parotid cells indeed expressed transcripts of Kir2.1 (Fig. 6). We subsequently amplified three overlapping PCR fragments using the specific primers with high fidelity DNA polymerase (Pfu-Turbo) and found that there was only one difference in the nucleotide sequence of the coding region between the bovine parotid Kir2.1and the described bKir2.1 (U95369), but it did not result in amino acid change or translation frame shift, indicating that the bovine parotid Kir2.1 clone also encodes a 427 amino acid protein.
Figure 6. RT-PCR analysis of bKir2.1 channel.

A ethidium bromide-stained agarose gel showing RT-PCR products generated from total RNA isolated from freshly dissociated bovine parotid acinar cells using Kir2.1-specific primers. Size markers (1 kb DNA ladder, Life Technologies, Inc.) of representative bands are also indicated. Arrows show the RT-PCR products for bKir2.1 at 4699 and 636 bp and for β-actin at 510 bp.
We also confirmed that basic electrophysiological properties of bKir2.1 stably expressed in HEK293 cells, which agree with the known properties of heterologously expressed Kir2.1 channels, are quantitatively similar to those of native Kir channels in BPA cells (Table 3).
Table 3.
Comparison of the electrophysiological properties of native bovine and sheep parotid Kir currents and bKir2.1 expressed in HEK293 cells
| bKir2.1 current | Native Kir current of bovine parotid acinar cells | Native Kir current of sheep parotid acinar cells | |
|---|---|---|---|
| Whole-cell current | |||
| Type of rectification | Strong | Strong | Strong |
| Inward conductance (G) ([K+]o= 5 mM) | 11 nS | 28 nS | 24 nS |
| [K+]o dependency of G | [K+]o0.52 | [K+]o0.57 | [K+]o0.51 |
| Conductance sequence | K+≫ Rb+= Na+ | K+≫ Rb+= Li+= Na+ | K+≫ Na+ |
| Permeability sequence | * | K+ > Rb+≫ Li+= Na+ | K+≫ Na+ |
| Ba2+block | Kd(0) = 8 μM, δ= 0.36 | Kd(0) = 12 μM, δ= 0.36 | K1/2= 1 μM (at −140 mV) |
| Cs+block | Kd(0) = 116 mM, δ= 1.40 | Kd(0) = 121 mM, δ= 1.15 | K1/2= 100 μM (at −140 mV) |
| Single channel current | |||
| Inward conductance (γ) | 27 and 16 pS | 27 pS(γmax= 29.6 pS, Km= 23.8 mM) | 29 pS (γmax= 33.4 pS, Km= 36.8 mM) |
| [K+]o dependency of γ | — | [K+]o0.42 | [K+]o0.55 |
The selectivity sequence based on permeability (PX/PK) ratio could not be estimated properly because Ba2+-sensitive inward and outward currents were very small when Rb+ or Na+ was in the bath and such a small Ba2+-sensitive current was also observed in mock-transfected cells under the same experimental conditions (data not shown).
Data for bKir2.1 currents were obtained from 5–28 experiments. Values are means.
Immunohistochemistry
We finally carried out immunohistochemistry using anti-Kir2.1 antibody to confirm the localization of Kir2.1 protein in BPA cells. In the control experiments where the non-immune serum was used as a primary antibody, no labelling for Kir2.1 was detected in the control specimens of the parotid gland (Fig. 7A). In the experiments with a Kir2.1 antibody, clear labelling for Kir2.1 was observed in the cells of acini and interstitial segments (Fig. 7B and C). The labelling was observed most intensely in the cells of the interstitial segments, but not detected in the cells of the striated ducts at all (Fig. 7D). As for both interstitial segments and acinar cells, the labelling was observed in the entire surroundings of the cells. Although the surroundings of most acinar cells reacted intensely, a small number of acinar cells, which reacted only faintly to the antibody, were also observed.
Figure 7. Immunofluorescence labelling for Kir2.1 in bovine parotid gland.
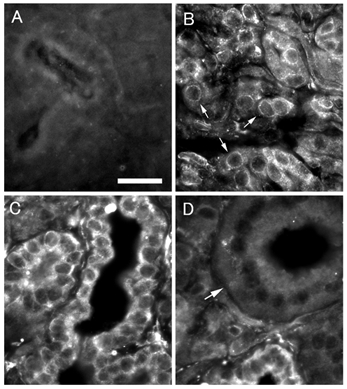
A, control: no labelling is observed. Bar indicates 20 µm. B, acini: peripheries of acinar cells (arrows) are labelled. C, interstitial segment: labelling is observed clearly in entire periphery of interstitial segment cells. D, striated duct: striated duct (arrow) where no labelling is observed.
DISCUSSION
Possible involvement of Kir2.1 in the inward rectifier K+ channels in BPA cells
In the present study, we have shown for the first time that BPA cells exhibit an inward rectifier K+ (Kir) channel and provided several lines of indirect evidence that the native Kir channel may be composed of Kir2.1 subunits. First, RT-PCR study indicated that transcripts for Kir2.1 are expressed in bovine parotid cells (Fig. 6). Second, immunohistochemical analysis showed the immunoreactivity of bKir2.1 in BPA cells (Fig. 7). Finally, electrophysiological properties of the native Kir whole-cell currents were remarkably and quantitatively similar to those of bKir2.1 cloned from BPA cells under the same experimental conditions (Table 3). Shared properties of the bovine parotid Kir channels and the cloned bKir2.1 were also confirmed at single channel level. Cell-attached single channel recordings showed the mean single channel conductance of 27 pS as the major conductance for both the native Kir channels and the bKir2.1 stably expressed in HEK293 cells (Table 3).
Interestingly, we also found that both BPA cells and HEK293 cells expressing bKir2.1 exhibited a single channel conductance of 16 pS. A previous study showed that tsA-201 cells expressing the Kir2.1 cloned from chick lens epithelium displayed subconductance levels similar to those seen for naturally occurring inward rectifier in chick lens epithelium (Rae & Shepard, 1998). More recently, it has been demonstrated that mouse Kir2.1 expressed in Xenopus oocytes and in HEK293 cells exhibit the unitary conductances ranging from 2 to 33 pS (Picones et al. 2001). This study also showed that channels of all amplitudes share several biophysical properties, including inward rectification, voltage sensitivity of open probability, sensitivity of open probability to external divalent cations and shape of the open channel I-V relation, suggesting that the channels of different conductances may be related to Kir2.1 proteins. Further single channel studies are needed to characterize in detail the biophysical properties of both the 27 pS and the 16 pS channels in native BPA cells.
The electrophysiological properties of the native and bKir2.1 currents differed from those of expressed other mammalian Kir2.1 in some respects. The sensitivity of the native bovine parotid Kir and the expressed bKir2.1 currents to the block by Cs+ seems to be lower than those reported for mouse and rat Kir2.1 expressed in murine erythroleukaemia cells and in Xenopus oocytes, where the Kd values for Cs+ at −100 mV range between 70 and 90µm (Abrams et al.1996; Bradley et al. 1999). The inconsistency could be in part due to errors introduced by the uncompensated series resistance (i.e. the voltage decrease across the uncompensated Rs) in the present whole-cell experiments, which would affect the voltage-dependent blocking properties of Cs+, because the Kir-mediated whole-cell currents (in high-K+ solution) were extremely large in both native BPA and HEK293 cells expressing bKir2.1 (see also Methods). A similar explanation may also apply to the reason that the blocking properties of external Ba2+ determined in the present study were slightly different from those for other mammalian Kir2.1 expressed in Xenopus oocytes (Shieh et al. 1998; Bradley et al. 1999; Alagem et al. 2001).
It should be stressed that the present study cannot rule out the contribution of other Kir subunits including Kir2.x subunits to the native Kir channels in BPA cells. It has been demonstrated that Kir2.1 can form functional hetero-oligomers with Kir2.2, Kir2.3, Kir2.4, Kir4.1 and Kir5.1 and that channels consisting of different subunits may alter the electrophysiological characteristics as distinct from those of homomeric channels composed of either subunit (Fakler et al. 1996; Derst et al. 2001; Preisig-Müller et al. 2002; Schram et al. 2002). Furthermore, it has been shown that changes in subunit arrangement can affect electrophysiological properties, as demonstrated for Kir4.1 and Kir5.1 (Pessia et al. 1996), the finding adding an additional complication. Therefore, electrophysiological data may have a limitation in identification of Kir subunit composing a native Kir channel. Future study is indeed necessary to establish whether such a heterotetramerization might also occur in BPA cells, as well as to establish what functional relevance it may have.
Physiological implications of the Kir channels in ruminant parotid glands
In the present study, we have provided strong evidence for the existence of functional Kir channels in BPA cells as also found in sheep parotid acinar cells (Wegman et al. 1992; Ishikawa & Cook, 1993; Ishikawa et al. 1993). Given the fact that electrophysiological characteristics of native Kir currents in both bovine and sheep parotid acinar cells are remarkably similar (Table 3), Kir2.1 could be expressed commonly in the parotid glands of ruminants.
The present whole-cell voltage-clamp and single channel recording experiments allow estimation of active channel numbers per cell with an assumption of open probability of the channel in freshly dissociated BPA cells. Taking the values of whole-cell Kir conductance of 220 nS (Fig. 1C and D) and a single channel conductance of 27 pS (Fig. 4B and C) with 150 mm [K+]o into account, with an assumption of open probability of 1, one can calculate the minimum number of active channels to be 8150 per cell. Thus, given a large number of functional Kir channels in unstimulated cells of bovine parotid glands, in spite of their property of anomalous rectification, the channels may allow efficient voltage clamping of the membrane potential to drive anion efflux across the apical membrane with minimum loss of K+ from the cell and hence to support resting secretion. Consistent with this view, we were able to demonstrate that addition of Ba2+ (0.1 and 1 mm) to the bathing solution reversibly depolarized membrane potential in a dose-dependent manner (Fig. 5). It is well known that Ba2+, especially at millimolar concentrations, can block not only Kir channels but also other types of K+ channels including maxi-K+ channels in various cell types as well as in sheep parotid acinar cells (Ishikawa & Cook, 1993). Since extracellular TEA, a blocker of maxi-K+ channels failed to depolarize resting membrane potential in intact unstimulated cells (Fig. 5C), maxi-K+ channels are highly unlikely to participate in setting resting membrane potential. At present, however, we cannot completely preclude the possibility that other unidentified Ba2+-sensitive K+ channels might also contribute to the resting membrane potential in BPA cells.
The other possible role of the Kir channel in BPA cells might be to buffer a rise in extracellular K+, since salivary glands in many species including ruminant parotids have been shown to lose K+ due to the activation of Ca2+-activated K+ channels to their surroundings when they are stimulated to secrete (Cook et al. 1994). In other well-studied salivary glands, the Na+-K+-2Cl− cotransporter serves as a basolateral K+ uptake mechanism during stimulated secretion, whereas in sheep and bovine parotid, the Na+-K+-2Cl− cotransporter appears to play no role in the stimulated secretion (Wright & Blair-West, 1990; Lee & Turner, 1993; Poronnik et al. 1995). The inwardly rectifying property of the Kir channel thus might be suited for fast uptake of accumulated K+ ions with a combination of Na+-K+-ATPase into ruminant parotid acinar cells, the K+ influx depending on the relation between the membrane potential and the concentration gradient of K+ across the basolateral membrane. Such a K+ buffering action of Kir channels has been proposed in brain astrocytes (Higashi et al. 2001), in retinal Müller cells (Ishii et al. 1997) and also in retinal pigmented epithelial cells (Kusaka et al. 2001).
In conclusion, the present study suggests that the molecular nature of the channel responsible for generating inwardly rectifying K+ currents in ruminant parotid acinar cells may be composed of Kir2.1 subunits and that the native Kir channels may be involved in setting resting membrane potential. This work is a first step toward gaining mechanistic insight into the roles played by Kir channels in ruminant parotid secretion. With a molecular target now focused, further studies can be carried out to examine the physiological role of Kir2.1, in association with other channels and transporters, in ruminant parotid secretion, and to determine whether the Kir2.1 expression is developmentally regulated in the parotid glands of ruminants.
Acknowledgments
This was supported by Grants-in-Aid from the Japan Society for the Promotion of Science and by the grants from the Ito Foundation and Northern Advancement Center for Science & Technology Foundation, and in part by Grants-in-Aid from Japan Society for Scientific Research from the Ministry of Education, Culture, Sports, and Science of Japan. M. Hayashi was supported by Research Fellowships of the Japan Society for the Promotion of Science for Young Scientists.
REFERENCES
- Abrams CJ, Davies NW, Shelton PA, Stanfield PR. The role of a single aspartate residue in ionic selectivity and block of a murine inward rectifier K+ channel Kir2. 1. J Physiol. 1996;493:643–649. doi: 10.1113/jphysiol.1996.sp021411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alagem N, Dvir M, Reuveny E. Mechanism of Ba2+ block of a mouse inwardly rectifying K+ channel: differential contribution by two discrete residues. J Physiol. 2001;534:381–393. doi: 10.1111/j.1469-7793.2001.00381.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barry PH, Lynch JW. Liquid junction potentials and small cell effects in patch-clamp analysis. J Membr Biol. 1991;121:101–117. doi: 10.1007/BF01870526. [DOI] [PubMed] [Google Scholar]
- Bradley KK, Jaggar JH, Bonev AD, Heppner TJ, Flynn ER, Nelson MT, Horowitz B. Kir2. 1 encodes the inward rectifier potassium channel in rat arterial smooth muscle cells. J Physiol. 1999;515:639–651. doi: 10.1111/j.1469-7793.1999.639ab.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Church DC. Salivary function and production. In: Church DC, editor. The Ruminant Animal. Digestive Physiology and Nutrition. Englewood Cliffs, NJ, USA: Prentice Hall; 1988. pp. 117–124. [Google Scholar]
- Coats DA, Denton DA, Goding JR, Wright RD. Secretion by the parotid gland of the sheep. J Physiol. 1956;131:13–31. doi: 10.1113/jphysiol.1956.sp005441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coats DA, Wright RD. Secretion by the parotid gland of the sheep: the relationship between salivary flow and composition. J Physiol. 1957;135:611–622. doi: 10.1113/jphysiol.1957.sp005734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cook DI, Gard GB, Champion M, Young JA. Patch-clamp studies of the electrolyte secretory mechanism of rat mandibular gland cells stimulated with acetylcholine or isoproterenol. In: Thorn NA, Treiman M, Petersen OH, Thaysen JH, editors. Molecular Mechanisms in Secretion. Copenhagen: Munksgaard; 1988. pp. 131–151. [Google Scholar]
- Cook DI, Van Lennep EW, Roberts ML, Young JA. Secretion by the major salivary glands. In: Johnson LR, Alpers DH, Christensen J, Jacobson ED, Walsh JH, editors. Physiology of the Gastrointestinal Tract. New York: Raven Press; 1994. pp. 1061–1117. [Google Scholar]
- Cook DI, Wegman EA, Ishikawa T, Poronnik P, Allen DG, Young JA. Tetraethylammonium blocks muscarinically evoked secretion in the sheep parotid gland by a mechanism additional to its blockade of BK channels. Pflugers Arch. 1992;420:167–171. doi: 10.1007/BF00374986. [DOI] [PubMed] [Google Scholar]
- Derst C, Karschin C, Wischmeyer E, Hirsch JR, Preisig-Müller R, Rajan S, Engel H, Grzeschik K, Daut J, Karschin A. Genetic and functional linkage of Kir5. 1 and Kir2.1 channel subunits. FEBS Lett. 2001;491:305–311. doi: 10.1016/s0014-5793(01)02202-5. [DOI] [PubMed] [Google Scholar]
- Fakler B, Bond CT, Adelman JP, Ruppersberg JP. Heterooligomeric assembly of inward-rectifier K+ channels from subunits of different subfamilies: Kir2. 1(IRK1) and Kir4.1 (BIR10) Pflugers Arch. 1996;433:77–83. doi: 10.1007/s004240050251. [DOI] [PubMed] [Google Scholar]
- Forsyth SE, Hoger A, Hoger JH. Molecular cloning and expression of a bovine endothelial inward rectifier potassium channel. FEBS Lett. 1997;409:277–282. doi: 10.1016/s0014-5793(97)00514-0. [DOI] [PubMed] [Google Scholar]
- Hamill OP, Marty A, Neher E, Sakmann B, Sigworth FJ. Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pflugers Arch. 1981;391:85–100. doi: 10.1007/BF00656997. [DOI] [PubMed] [Google Scholar]
- Hayashi T, Poronnik P, Young JA, Cook DI. The ACh-evoked, Ca2+-activated whole-cell K+ current in mouse mandibular secretory cells. Whole-cell and fluorescence studies. J Membr Biol. 1996;152:253–259. doi: 10.1007/s002329900103. [DOI] [PubMed] [Google Scholar]
- Higashi K, Fujita A, Inanobe A, Tanemoto M, Doi K, Kubo T, Kurachi Y. An inwardly rectifying K+ channel, Kir4. 1, expressed in astrocytes surrounds synapses and blood vessels in brain. Am J Physiol Cell Physiol. 2001;281:C922–931. doi: 10.1152/ajpcell.2001.281.3.C922. [DOI] [PubMed] [Google Scholar]
- Hille B. Ionic Channels of Excitable Membranes. 2. Sunderland, MA, USA: Sinauer Associates; 1992. [Google Scholar]
- Hirono C, Steward MC, Sangster N, Poronnik P, Young JA, Cook DI. Developmental regulation and maintenance of inwardly and outwardly rectifying K+ currents in sheep parotid cells. Am J Physiol. 1994;267:G947–953. doi: 10.1152/ajpgi.1994.267.5.G947. [DOI] [PubMed] [Google Scholar]
- Ishii M, Horio Y, Tada Y, Hibino H, Inanobe A, Ito M, Yamada M, Gotow T, Uchiyama Y, Kurachi Y. Expression and clustered distribution of an inwardly rectifying potassium channel, KAB-2/Kir4. 1, on mammalian retinal Müller cell membrane: their regulation by insulin and laminin signals. J Neurosci. 1997;17:7725–7735. doi: 10.1523/JNEUROSCI.17-20-07725.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishikawa T, Cook DI. Effects of K+ channel blockers on inwardly and outwardly rectifying whole-cell K+ currents in sheep parotid secretory cells. J Membr Biol. 1993;133:29–41. doi: 10.1007/BF00231875. [DOI] [PubMed] [Google Scholar]
- Ishikawa T, Murakami M. Tetraethylammonium-insensitive, Ca2+-activated whole-cell K+ currents in rat submandibular acinar cells. Pflugers Arch. 1995;429:748–750. doi: 10.1007/BF00373998. [DOI] [PubMed] [Google Scholar]
- Ishikawa T, Wegman EA, Cook DI. An inwardly rectifying potassium channel in the basolateral membrane of sheep parotid secretory cells. J Membr Biol. 1993;131:193–202. doi: 10.1007/BF02260108. [DOI] [PubMed] [Google Scholar]
- Iwatsuki N, Maruyama Y, Matsumoto O, Nishiyama A. Activation of Ca2+-dependent Cl− and K+ conductances in rat and mouse parotid acinar cells. Jpn J Physiol. 1985;35:933–944. doi: 10.2170/jjphysiol.35.933. [DOI] [PubMed] [Google Scholar]
- Izutsu KT, Johnson DE, Goddard M. Intracellular elemental concentrations in resting and secreting rat parotid glands. J Dent Res. 1987;66:537–540. doi: 10.1177/00220345870660022501. [DOI] [PubMed] [Google Scholar]
- Kay RNB. Continuous and reflex secretion by the parotid gland in ruminants. J Physiol. 1958;144:463–475. doi: 10.1113/jphysiol.1958.sp006113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kay RNB. The rate of flow and composition of various salivary secretions in sheep and calves. J Physiol. 1960;150:515–537. doi: 10.1113/jphysiol.1960.sp006402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kubo Y, Baldwin TJ, Jan YN, Jan LY. Primary structure and functional expression of a mouse inward rectifier potassium channel. Nature. 1993;362:127–133. doi: 10.1038/362127a0. [DOI] [PubMed] [Google Scholar]
- Kusaka S, Horio Y, Fujita A, Matsushita K, Inanobe A, Gotow T, Uchiyama Y, Tano Y, Kurachi Y. Expression and polarized distribution of an inwardly rectifying K+ channel, Kir4. 1, in rat retinal pigment epithelium. J Physiol. 1999;520:373–381. doi: 10.1111/j.1469-7793.1999.00373.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee SI, Turner RJ. Secretagogue-induced 86Rb+ efflux from bovine parotid is HCO3− dependent. Am J Physiol. 1993;264:R162–168. doi: 10.1152/ajpregu.1993.264.1.R162. [DOI] [PubMed] [Google Scholar]
- Meot F, Cirio A, Boivin R. Parotid secretion daily patterns and measurement with ultrasonic flow probes in conscious sheep. Exp Physiol. 1997;82:905–923. doi: 10.1113/expphysiol.1997.sp004072. [DOI] [PubMed] [Google Scholar]
- Neher E. Correction for iquid junction potentials in patch clamp experiments. In: Rudy B, Iverson L, editors. Methods in Enzymology. San Diego, CA, USA: Academic Press; 1992. pp. 123–131. [DOI] [PubMed] [Google Scholar]
- Nichols CG, Lopatin AN. Inward rectifier potassium channels. Annu Rev Physiol. 1997;59:171–191. doi: 10.1146/annurev.physiol.59.1.171. [DOI] [PubMed] [Google Scholar]
- Park K, Case RM, Brown PD. Identification and regulation of K+ and Cl− channels in human parotid acinar cells. Arch Oral Biol. 2001;46:801–810. doi: 10.1016/s0003-9969(01)00047-4. [DOI] [PubMed] [Google Scholar]
- Pessia M, Tucker SJ, Lee K, Bond CT, Adelman JP. Subunit positional effects revealed by novel heteromeric inwardly rectifying K+ channels. EMBO J. 1996;15:2980–2987. [PMC free article] [PubMed] [Google Scholar]
- Picones A, Keung E, Timpe LC. Unitary conductance variation in Kir2. 1 and in cardiac inward rectifier potassium channels. Biophys J. 2001;81:2035–2049. doi: 10.1016/S0006-3495(01)75853-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poronnik P, Schumann SY, Cook DI. HCO3−-dependent ACh-activated Na+ influx in sheep parotid secretory endpieces. Pflugers Arch. 1995;429:852–858. doi: 10.1007/BF00374810. [DOI] [PubMed] [Google Scholar]
- Poulsen JH, Oakley B., II Intracellular potassium ion activity in resting and stimulated mouse pancreas and submandibular gland. Proc R Soc Lond B Biol Sci. 1979;204:99–104. doi: 10.1098/rspb.1979.0015. [DOI] [PubMed] [Google Scholar]
- Preisig-Müller R, Schlichthörl G, Goerge T, Heinen S, Brüggemann A, Rajan S, Derst C, Veh RW, Daut J. Heteromerization of Kir2. x potassium channels contributes to the phenotype of Andersen's syndrome. Proc Natl Acad Sci U S A. 2002;99:7774–7779. doi: 10.1073/pnas.102609499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rae JL, Shepard AR. Inwardly rectifying potassium channels in lens epithelium are from the IRK1 (Kir 2. 1) family. Exp Eye Res. 1998;66:347–359. doi: 10.1006/exer.1997.0432. [DOI] [PubMed] [Google Scholar]
- Schram G, Melnyk P, Pourrier M, Wang Z, Nattel S. Kir2. 4 and Kir2.1 K+ channel subunits co-assemble: a potential new contributor to inward rectifier current heterogeneity. J Physiol. 2002;544:337–349. doi: 10.1113/jphysiol.2002.026047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shieh RC, Chang JC, Arreola J. Interaction of Ba2+ with the pores of the cloned inward rectifier K+ channels Kir2. 1 expressed in Xenopus oocytes. Biophys J. 1998;75:2313–2322. doi: 10.1016/S0006-3495(98)77675-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silanikove N. The struggle to maintain hydration and osmoregulation in animals experiencing severe dehydration and rapid rehydration: the story of ruminants. Exp Physiol. 1994;79:281–300. doi: 10.1113/expphysiol.1994.sp003764. [DOI] [PubMed] [Google Scholar]
- Smith PM, Gallacher DV. Acetylcholine- and caffeine-evoked repetitive transient Ca2+-activated K+ and C1− currents in mouse submandibular cells. J Physiol. 1992;449:109–120. doi: 10.1113/jphysiol.1992.sp019077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verheugen JA, Vijverberg HP, Oortgiesen M, Cahalan MD. Voltage-gated and Ca2+-activated K+ channels in intact human T lymphocytes. Noninvasive measurements of membrane currents, membrane potential, and intracellular calcium. J Gen Physiol. 1995;105:765–794. doi: 10.1085/jgp.105.6.765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wegman EA, Ishikawa T, Young JA, Cook DI. Cation channels in basolateral membranes of sheep parotid secretory cells. Am J Physiol. 1992;263:G786–794. doi: 10.1152/ajpgi.1992.263.5.G786. [DOI] [PubMed] [Google Scholar]
- Woodhull AM. Ionic blockage of sodium channels in nerve. J Gen Physiol. 1973;61:687–708. doi: 10.1085/jgp.61.6.687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wright RD, Blair-West JR. The effects of K+ channel blockers on ovine parotid secretion depend on the mode of stimulation. Exp Physiol. 1990;75:339–348. doi: 10.1113/expphysiol.1990.sp003408. [DOI] [PubMed] [Google Scholar]


