Abstract
Reflexively evoked and eye-related eyelid responses were recorded using the search coil in a magnetic field technique in alert cats. The downward phase of a blink was a large (up to 21 deg), fast (up to 2000 deg s−1) eyelid displacement in the closing direction, with an almost fixed rise time duration (15-20 ms); its maximum velocity was achieved in ≈10 ms. Upward eyelid motion was separated into two phases. The first phase consisted of a fast eyelid displacement, with a short duration (≈30 ms) and a maximum velocity up to 900 deg s−1. The second phase had an exponential-like form, lasting for 200–400 ms, and a maximum velocity ranging between 30 and 250 deg s−1. Maximum blink velocity in the downward direction was linearly related to maximum velocity of the first upward phase. The first phase in the upward direction was never observed if the eyelid stayed closed for a long period (> 50 ms) or moved slowly in the closing direction before it started to open. In these two cases, the upswing motion of the blink reflex contained only the exponential-like movement characteristic of the second upward phase, and maximum velocity in the downward direction was not related to that of the eyelid upward displacement. Mean duration of eyelid downward saccades was ≈130 ms, and their peak velocities ranged between 50 and 440 ms. A physiological model is presented explaining the active and passive forces involved in both reflex and saccadic eyelid responses. A second-order system seems to be appropriate to describe the postulated biomechanical model.
A blink consists of a fast downward displacement of the upper eyelid followed by an upward phase with a slower velocity. The orbicularis oculi is the muscle mainly responsible for reflex eyelid closure (Gordon, 1951; Kugelberg, 1952; Björk & Kugelberg, 1953; Van Allen & Blodi, 1962; Delgado-García et al. 1998). Recently, we have described the firing pattern of orbicularis oculi motoneurones during reflex and conditioned eyelid responses (Trigo et al. 1999a). For reflex responses, orbicularis oculi motoneurones exhibit an initial burst of action potentials that produces the fast eye closure. If the sensory stimulation is strong and long lasting, the initial burst of the motoneurone is usually followed by a short (lasting ≈1 s) tonic firing, able to produce small sags in eyelid position. Between the phasic and tonic phases, there is normally a silent period in motoneurone activity due to a conspicuous hyperpolarization (lasting ≈50 ms) of the resting membrane potential (Trigo et al. 1999a). Indeed, the wavy profile of eyelid position recorded during the late phase of spontaneous and reflex blinks (and also during conditioned responses) is correlated with oscillatory changes in the membrane potential of orbicularis oculi motoneurones. Moreover, these motoneurones tend to fire in doublets, the second spike evoking a larger muscle activation than the preceding one (Trigo et al. 1999a). This reinforcing effect, observed during short interspike intervals, reaches a maximum when the separation between the spikes is ≈10-15 ms; that is, a time period close to the interval between R1 and R2 components of blinks evoked by the electrical stimulation of the supraorbital nerve (Kugelberg, 1952; Gruart et al. 1995).
The levator palpebrae superioris muscle maintains eyelid elevation between blinks. Electromyographic (EMG) records of levator palpebrae superioris muscle, or of the firing activity of its innervating motoneurones, present an increase in the number of active motor units, or in motoneurone firing rate, in linear proportion to the (upward) eyelid position (Björk & Kugelberg, 1953; Holder et al. 1987; Becker & Fuchs, 1988; Fuchs et al. 1992). According to Fuchs et al. (1992), the discharge rate of levator palpebrae superioris motoneurones in monkeys is different during the upswing of a blink to that for upward saccades. During blinks, the reappearance of levator motoneurone activity lagged the movement of the eyelid in the upward direction by ≈20-25 ms. The firing rate pattern of these motoneurones shows a gradual increase with eyelid position, reaching its maximum when the eye is open (see Figs 1 and 6E in Fuchs et al. 1992). In contrast, the time course of motoneuronal activity during upward eyelid saccades is quite different (see Figs 1 and 6B in Fuchs et al. 1992). In this latter case, motoneurones of the levator palpebrae superioris muscle present not a gradual, but a sudden burst of activity followed by a tonic firing related to the final position of the eyelid. The discharge rate of these motoneurones is in agreement with EMG recordings of the levator palpebrae superioris muscle in humans and monkeys (Björk & Kugelberg, 1953; Becker & Fuchs, 1988; Aramideh et al. 1998).
Figure 1. Two different types of air puff-evoked blinks.
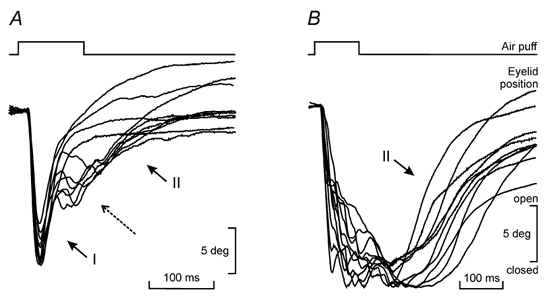
A, examples of blinks evoked by long-lasting (100 ms) strong (3 kg cm−2) air puffs. Blinks were initiated by a fast, downward displacement of the eyelid. The opening of the eyelid was divided in two different phases (I and II). The beginning of the second upward phase (II) was sometimes preceded by a conspicuous oscillation (dashed arrow). B, examples of blinks with a slow velocity or a long duration of the downward phase. In these cases, eyelid opening presented only the second upward phase (II). Note that while in A the change from the downward to the upward direction occurred in a short period of time (≈5 ms), in B there was a larger temporal separation between them (> 50 ms). Eyelid profiles were grouped to a common start to show the variability in amplitude of the evoked responses.
Figure 6. Proposed mechanism for downward saccades and blinks.

The dotted lines represent the main structures involved in the movement. For a blink, the active contribution of orbicularis oculi motoneurones (OO Mn) is needed. Abbreviations: APO, aponeurosis of levator palpebrae superioris muscle; ME, margins of the upper eyelid; LPS, levator palpebrae superioris muscle; OO, orbicularis oculi muscle; WL, Whitnall's ligament.
The dynamics of the upward movement of the eyelid during a blink can be separated into two parts: an initial rapid rise, followed by a slower exponential-like displacement (Evinger et al. 1984). According to Evinger and colleagues (Evinger et al. 1991; Evinger, 1995), a burst-step firing pattern in the motoneurones of the levator palpebrae superioris muscle is expected. The burst would account for the first, rapid upward movement of the eyelid, during which maximum velocity is achieved, and the step would produce the second exponential-like motion, during which the eyelid reaches its final position. However, it has been shown that neither levator palpebrae superioris motoneurones nor the EMG activity of the innervated muscle shows such a pattern of activity during blinks (Holder et al. 1987; Becker & Fuchs, 1988; Fuchs et al. 1992; Aramideh et al. 1998). Electrical recordings of levator palpebrae superioris motoneurone and muscle activity show that the burst is absent and there is only a gradual increase in motoneurone and muscle firing (Holder et al. 1987; Becker & Fuchs, 1988; Fuchs et al. 1992; Aramideh et al. 1998).
We present here a physiological mechanism able to explain the rapid raising of the eyelid during reflex eyelid responses. Eyelid kinematics in the downward direction (and how this is related to the upward phase of the blink) is also considered. The values of the physical constants that characterize the spring-like structures participating in the proposed mechanism have been calculated. The results are contrasted with present knowledge of the interaction of the different forces producing reflex blinks and eyelid saccades.
Methods
Animals
The present experiments were carried out on three adult female cats of European and Abyssinian strains. The animals, weighing 2–2.4 kg, were obtained from an authorized supplier (Iffa-Credo, France). Subjects were prepared for the chronic recording of upper eyelid movements with the search coil in a magnetic field technique (Fuchs & Robinson, 1966; Gruart et al. 1995). Experiments followed Spanish guidelines (BOE 67/8509-12, 1988) and European Union Council regulations (86/609/EU) concerning the use of laboratory animals in chronic experiments.
Surgical procedures
Animals were anaesthetized with sodium pentobarbital (sodium pentobarbitone, 35 mg kg−1, i.p.) peceded by an injection of atropine sulphate (0.5 mg kg−1, i.m.) to prevent unwanted vagal reflexes. A five-turn coil (3 mm in diameter) was implanted in the centre of the left upper eyelid under sterile conditions and with the help of an operating microscope. Coils were made of Teflon-coated, seven-strand stainless steel wire (50 μm in diameter). In addition, a three-turn coil made of the same wire was sutured to the sclera of the left eye. Because of their low weights (10 and 34 mg, respectively), the implanted coils did not produce any disturbance in the normal movements of the left eyelid and eye when compared visually with those in the non-implanted side.
Finally, the animals were implanted with a head-holding system for stabilization during the recording sessions. Three bolts were cemented to the skull with the aid of six small screws fastened to the bone. The eyelid and eye coils were soldered to a nine-pin socket attached to the holding system. Further details of this chronic preparation have been published elsewhere (Gruart et al. 1995).
At the end of the experiment the animals were killed using an overdose of anaesthetic (50 mg kg−1 sodium pentobarbitone, i.p.).
General conditions of recording sessions
Recording sessions started 1 week after surgery and lasted a maximum of 1 h day−1. The total number of recording sessions per animal was ≤ 15. The animal was lightly restrained with an elastic bandage and placed on the recording table. The head was immobilized by attaching the holding system to a fixing bar placed inside the magnetic field frame. The animals seemed not to be stressed by the experimental procedures, since cardiac (187 ± 9 beats min−1) and respiratory (60 ± 6 cycles min−1) rates during recording sessions were similar to those obtained with the animals resting on the arms of one of the experimenters (heart rate, 179 ± 9 beats min−1; respiratory rate, 56 ± 7 cycles min−1).
Recording of eyelid and eye movements
Lid movements were recorded using the search coil in a magnetic field technique (Fuchs & Robinson, 1966; Gruart et al. 1995). Eyelid coils were calibrated, with the cat still under the effects of the anaesthetic, by displacing the eyelid manually at known angles and measuring the output voltage of each coil wire. The angles were measured with the aid of a transparent protractor placed sagittally and centred at the external canthus of the eye. The gain of the recording system was adjusted to yield 1 V (10 deg)−1. The animal's head was placed 21 deg nose down with respect to the horizontal plane (Delgado-García et al. 1986). In this position, with the animal looking straight ahead, the lower margin of the upper eyelid was raised by 5–10 deg over the central (zero) position of the eye. Eyelid maximum opening ranged from 35 to 40 deg for the three animals.
Eye movements were also recorded with the magnetic search-coil technique. For the calibration of eye movements, the magnetic field frame was rotated ±10 deg about both the horizontal and vertical planes with the cat awake and sitting on the recording table. A zero position of the eye in the orbit was established as the mean position during 60 min of spontaneous eye movements (Delgado-García et al. 1990).
Stimuli evoking eyelid movements
Eyelid blinks were elicited with air puffs presented to the ipsilateral cornea. Stimuli were presented at 30 ± 5 s intervals to avoid inhibitory effects produced by the preceding stimulus (Powers et al. 1997). Air puffs used in this study lasted 100 ms, at an air pressure of 3 kg cm−2. Air puffs were applied through a plastic pipette with a tip diameter of 3 mm. The tip was located 1 cm away from the cornea at a lateral angle of 45 deg with respect to the central direction of gaze. The precise timing of air puff stimuli reaching corneal and periocular skin was determined at the beginning of the recording session with a microphone located at the same side as the eye. The recorded signal was rectified, integrated, and fed into the computer as 1 V rectangular pulses to determine the latency of evoked responses.
Data collection and analysis
Horizontal and vertical eyelid movements, 1 V rectangular pulses corresponding to blink-evoking stimuli, and 1 V trigger pulses were stored digitally on an eight-channel videotape recording system. Data were later transferred for off-line analysis to a computer through a CED 1401-plus converter at 1–4 kHz sampling frequency, with an amplitude resolution of 12 bits. Commercial computer programs (SIGAVG and Spike 2 from CED, Cambridge, UK and Corel Draw 9, Corel Corp., Ottawa, ON, Canada) were adapted to display eyelid position and velocity. The programs allowed the measurement of latency, amplitude, duration, rise time, and peak and mean velocity of evoked eyelid movements and spontaneous saccades (see Delgado-García et al. 1990 and Gruart et al. 1995 for details).
Statistical analyses were carried out using commercially available software (Jandel Scientific, San Rafael, CA, USA and Sigma Stat, SPSS Inc., Chicago, IL, USA) with a statistical significance level of 0.05. Mean values for eyelid movement amplitude, peak velocity, and latency, accompanied by their standard deviation were calculated from ≥ 100 measurements from a minimum of two animals. Regression lines were calculated with ≥ 100 measurements collected from at least two animals.
Results
As illustrated in Fig. 1, a blink consisted of an initial fast downward displacement of the upper eyelid followed by a slower upward phase. The mean amplitude of blinks evoked by the air puff used in this study (100 ms, 3 kg cm−2) was 15.3 ± 2.4 deg (range 11.2-21.7 deg). The downward movement achieved its maximum velocity in 10.6 ± 0.45 ms, reaching values up to 2000 deg s−1. The rise time of the downward phase duration was calculated by the computer program as the time spent by the lid in producing 80 % of its total displacement, as measured from the initial 10 % to the final 90 % of lid movement (see Gruart et al. 1995). Values for the rise time of the downward phase ranged from 15 to 20 ms, regardless of the maximum amplitude of the evoked blink.
A visual analysis of blink profiles indicated that the movement in the upward direction was separated into two phases: an initial rapid motion (I in Fig. 1A) and an exponential-like movement (II in Fig. 1A). Sometimes, the two phases in the upward movement were separated by the presence of oscillations (long dashed arrow in Fig. 1A). In these cases, an exponential-like approach to the final position of the eyelid was always observed following the oscillation (Fig. 1A). Thus, the amplitude of the second, exponential motion depended on the moment that the interruption (oscillations) in the upswing motion of the eyelid was present. This finding was extended to blinks that closed the eyelids slowly (with velocities similar to downward saccades) or for which the eyelids remained, after the downward movement, closed for a long period (> 50 ms). In these two cases, the upward motion of the eyelid did not present the initial rapid phase, but only the exponential-like component (II in Fig. 1B). On occasions, the total amplitude of the upward motion (phases I and II) was larger than the preceding downward movement (Fig. 1A).
The different kinetic properties of the two upward phases of a blink are illustrated in Fig. 2. The latency to maximum velocity for the initial fast upward motion of the eyelid was 11.6 ± 2 ms (histogram in Fig. 2A and velocity trace in Fig. 2B); i.e. a value similar to the latency to peak velocity in the downward direction. The duration of this initial phase was 30.12 ± 3.01 ms (range 25–39 ms; see position trace in Fig. 2B). The second, upward exponential phase had a significantly (P < 0.001) longer duration than the first upward phase, ranging from 200 to 400 ms (Fig. 2C). Its latency to maximum velocity was 98.2 ± 37 ms (range 30–180 ms; histogram in Fig. 2A and velocity trace in Fig. 2C). The latency to peak velocity was independent of blink amplitude for both phases. The shape of eyelid velocity profiles was also different for the two upward components of the blink. The second phase usually had a series of secondary peaks, before or after maximum velocity was achieved, that the first phase did not exhibit.
Figure 2. Representation of the two different phases in the upward movement of the eyelid, during a blink.

A, a blink reflex evoked by a long, strong air puff (100 ms, 3 kg cm−2). The upper trace represents eyelid position, and the lower one eyelid velocity. Note that the upward eyelid movement is separated into a first rapid linear phase (I) followed by an exponential displacement (II) by the occurrence of two downward sags. The histogram at the bottom shows the latency to peak velocity for both phases (11.6 ± 2 and 98.2 ± 37 ms, respectively). B and C, profiles of eyelid velocity for the two components of upward eyelid movement: B shows the initial fast upward phase (I in A), and C the late phase of the upward movement (II in A). The linear (B) and S-shaped (C) traces represent the average (n = 5) eyelid position, while the parabolic-like profiles depict eyelid velocity. Data in C were collected from blinks in which the eyelid stayed closed for a long period, or closed slowly (see Fig. 1B), that is, without a noticeable initial upward component. The inset in C shows the average of 10 such blinks. Calibrations of the bars for the inset in C are 200 ms and 5 deg.
As shown in Fig. 3B, maximum velocity in the downward and upward (phase I) directions maintained a statistically significant (P < 0.001) direct linear relationship. As indicated by the linear analysis, peak velocity in the upward (phase I) direction was about one-third (n = 3 animals) that for the downward movement. It is important to point out that the duration of the eyelid closure for data illustrated in Fig. 3B for phase I was very short (5 ± 1 ms; Fig. 1A). Figure 3B also shows that there was no relationship between maximum velocity in the downward and upward directions when the eyelid remained closed for a long period (> 50 ms), or when the eyelid movement in the closing direction was slow (in a range similar to downward saccades, Fig. 1B and dashed line in Fig. 3A). The duration of eyelid closure for data presented in Fig. 3B for phase II was 102 ± 15 ms (see Fig. 1B). In this latter case, eyelid velocity in the upward direction depended on eyelid movement amplitude (for phases I and II, see Fig. 3A), and not on the velocity achieved during the previous fast downward displacement (Fig. 3C).
Figure 3. Analysis of the relationship between eyelid position and velocity during the two upward phases of reflexively evoked blinks.

A, recordings of eyelid position and velocity during air puff-evoked blinks. Parameters taken to build the relationships shown in B and C are indicated: I, first phase in the upswing of the blink; II, second phase in the upswing. B, relationship between maximum velocity (in deg s−1) of downward and upward eyelid responses for the first (I in A) and second (II in A) upward phases. C, relationship between maximum eyelid velocity (in deg s−1) in the upward direction and blink amplitude (in deg) for both first (I) and second (II) upward phases. Linear equations corresponding to each regression line are indicated.
Figure 3C shows the relationship between amplitude and peak velocity of the eyelid in the upward direction. The linear regressions represent the relationship for the two phases of the upward eyelid motion: (i) during the first fast and linear upswing movement, when maximum velocity was achieved (see I in Figs 1A and 3A); and (ii) during the second, exponential movement (see II in Figs 1 and 3A). The amplitude for the first upward phase was measured from the position of maximum closure of the eyelid to 30 ms later (Fig. 3A). In this manner, the second, slow upward phase was not present. For the second, exponential-like phase, data were collected when the eyelid remained closed for ≥ 50 ms. In this way, the initial rapid movement was avoided (Fig. 1B). As reflected in Fig. 3C, the kinetic properties of these two upward components of a reflex blink were statistically different (Mann-Whitney test, P < 0.0001), the slope of the regression line for the first upward phase being 7.25 times greater than that for the second.
In order to check that maximum eyelid velocity in the downward direction, rather than the size of the eyelid elevation, determines maximum up velocity of the eyelid, the analysis illustrated in Fig. 3B for phase I was repeated for blinks of the same amplitude (15 deg). Indeed, the plot of maximum up velocity against maximum down velocity for 15 deg blinks also presented a linear relationship, and a slope similar to that illustrated in Fig. 3B for blinks of different amplitudes (0.271 for 15 deg blinks against 0.257 for data in Fig. 3B). Accordingly, the maximum velocity of downward motion during a blink seemed to determine the maximum velocity of the first upward phase.
The upward phase of normal blinks presented maximum velocities up to 900 deg s−1, while velocities ranged between 30 and 250 deg s−1 for blinks whose upward rising was slow; i.e. blinks lacking the first upward component (Fig. 3B and C). An attempt was made to analyse blinks of equal amplitude, but with differences in the participation of the first and second upward components (see inset in Fig. 4A, for an example). For this analysis, we computed the difference (continuous trace in Fig. 4A, inset) between the upward phase of blinks of the same downward amplitude (dashed lines in Fig. 4A, inset). The curves illustrated in Fig. 4A represent such differences for five pairs of eyelid responses of the same amplitude. The peak amplitude for these curves ranged between 60 and 85 % of the whole amplitude of the upward eyelid response. A direct linear relationship was also observed between the peak amplitude of the upward movement of the blink (x) and its corresponding maximum calculated difference (y). The linear regression analysis showed that y = 0.75x, r = 0.99 (P < 0.001). The time to reach peak amplitude was 66.8 ± 4.7 ms.
Figure 4. Measurement of the contribution of phases I and II to eyelid upward displacement during a blink and analysis of eyelid movement in the plane of phase.

A, five examples from which were obtained the difference in the eyelid displacement for phases I and II of the upward motion. The inset shows an example of how the represented curves were calculated. The continuous line in the inset (arrow) represents the difference between the upward phases of two blinks of equal downward amplitude (dashed lines). B, representation in the plane of phase (position against velocity) for three normal blinks. C, a representation similar to B but for blinks with a low downward velocity or after which the eyelid stayed closed for a time period > 50 ms.
Figure 4B and C shows eyelid upward displacement in the plane of phase (that is, position against velocity) during blinks of different characteristics. Three eyelid movements of different amplitude in which the high velocity of the downward phase permitted the rapid recovery of the eyelid to its initial position are represented in Fig. 4B. It should be noted that the shape of the downward movement (lower arc) and the upward movement (upper arc) is smooth, and that trajectories are practically closed. In the representation of the upward phase, two different sections can be distinguished: a first rapid transient (with a steep slope) followed by a slower one that returned the system to its equilibrium position. The slope of the first transient presented a direct relationship with velocity of the blink in the downward direction. In this way, higher downward velocities corresponded with higher slopes. This first portion of the upward curve also presented a certain degree of symmetry with the corresponding section in the downward movement of the blink. Blinks with amplitudes similar to those shown in Fig. 4B, but with a slow downward velocity (and consequently with a slow upward velocity), are presented in Fig. 4C. The comparison of this case with the previous one illustrates the fluctuations in the upward phase and the absence of the first rapid transient.
Spontaneous eye saccades made by the animal while scanning the recording room were also recorded in this study. Figure 5 shows a comparison between a spontaneous downward saccade and the downward phase of a blink. The mean duration of downward saccades was 132 ± 56 ms and their peak velocities ranged between 50 and 440 deg s−1. Latency to peak velocity for downward saccades was 55.3 ± 14.0 ms. When the two movements were placed at a common start and presented similar amplitudes (Fig. 5A), the downward saccade always exhibited a much slower (4-10 times) peak velocity (Fig. 5B). The latency to peak velocity was also 4–8 times longer. The maximum difference in the displacement of the eyelid was found when the blink reached its maximum amplitude. In this case, the eyelid displacement during the downward saccade represented 10.1 ± 4.0 % of the amplitude of the blink.
Figure 5. Differences in kinetic properties of downward saccades and reflexively evoked blinks.

A, eyelid displacement during a downward saccade and a blink. In order to make the comparison, the two movements have been set to a common start and are of the same amplitude. The dashed arrow indicates the maximum difference in eyelid position between the two movements in the downward direction. B, velocity profiles for both types of movement illustrated in A. Note the different values for maximum velocities and latencies to peak velocity (arrows).
Discussion
The downward phase of a blink
The kinematics of the downward phase of a blink is the result of a brief activation of orbicularis oculi motoneurones. In fact, this burst of action potentials produces the high velocity (up to 2000 deg s−1) that characterizes the downward motion of the eyelid in cats (Trigo et al. 1999a). Apart from motoneurone firing rate, there are three other factors affecting eyelid velocity (and amplitude) during the downward displacement: (i) the number of active motor units; (ii) their synchronization; and (iii) the enhancing effect on the muscle produced by a second activation of orbicularis oculi motoneurones (Trigo et al. 1999a). In cats, the maximum effect is achieved when a couple of action potentials are separated by ≈10 ms. Apparently, these three factors avoid the need for an extremely high firing rate to accomplish a movement with a high velocity and amplitude.
As shown in Fig. 3B, the upward displacement of the eyelid during a blink is related to the downward motion. The relationship is linear, the peak velocity of the downward phase being ≈3-4 times larger. However, this relationship was present only when the change in direction (from downward to upward) of eyelid movement was fast enough. Figure 3B also shows that there was no correlation between peak velocities for the two movements when eyelids were maintained in the closed position for a long period (> 50 ms), or when eyelids closed slowly. It will be proposed below that the high velocity accomplished during the downward phase is necessary for a rapid opening of the eyelid.
The problem of raising the eyelid during blinks
According to the results presented here, the upward movement of a blink can be separated in two parts with very different kinetic characteristics (Figs 1–3). Figure 3C shows that the slope of the regression line between eyelid amplitude and velocity for the first phase of the upward motion is ≈7 times larger than that for the second, suggesting the participation of two different processes. According to Evinger and colleagues (Evinger et al. 1991; Evinger, 1995), the early upward motion must be the consequence of a burst in the firing pattern of levator palpebrae superioris motoneurones, while a steady firing response should account for the second, exponential motion. However, unitary recordings of these motoneurones, or of the EMG activity of the innervated muscle, do not show such a pattern during actual blinks (Holder et al. 1987; Becker & Fuchs, 1988; Fuchs et al. 1992). Another important fact is that, as Evinger et al. (1991) state, the equilibrium position for the eyelid is eyelid closure. Apparently, then, there is no available force to raise the eyelid rapidly after a reflexively evoked blink.
These apparent inconsistencies can be surmounted considering the following mechanism. The force producing the first fast component in the upward direction is not generated actively, i.e. by a muscle, but is the result of the elastic energy accumulated in the spring-like structures constituting the eyelid (mainly the aponeurosis of the levator palpebrae superioris muscle). There must also be a viscous component to prevent undesirable oscillations when the eyelid reaches its final position after a blink or an eyelid saccade. This viscosity must exert a force that opposes the movement of eyelid structures. During blinks, the additional force provided by the orbicularis oculi muscle to close the eye makes the eyelid move at a high velocity, with the viscous component opposing the movement of the levator palpebrae superioris muscle and Whitnall's ligament. The aponeurosis of the levator palpebrae superioris muscle is a broad and fine band made up of interdigitated thin fibres of connective tissue. This characteristic makes it elastic in all directions. For simplicity, only a vertical component in the elasticity of the aponeurosis will be considered in our model (Fig. 6). Taking this into account, the force exerted by the orbicularis oculi muscle would be employed mainly in stretching the vertical elastic component, providing the elastic potential energy needed for upward eyelid displacement.
A mechanism for blinking
Considering all the aforementioned factors, it is possible to distinguish the following events during a blink. (i) The blink is initiated by the activity of the orbicularis oculi muscle. (ii) At a determined latency, the eyelid starts to close. (iii) The eyelid, helped by orbicularis oculi muscle activation, reaches a velocity in its downward motion higher than when the only acting force is the elastic component, i.e. during a downward saccade (Fig. 5). (iv) The blink causes, for a short period of time, the stretching of the vertical elastic component of the eyelid. The vertical elastic component exceeds its own equilibrium position and thus accumulates elastic energy. (v) Simultaneously, during the rapid downward displacement of the eyelid, the levator palpebrae superioris muscle and Whitnall's ligament must be at some point in their return to their equilibrium position (i.e. eyelid closed). Both are unable to follow the movement of the eyelid margins because of the braking action of the viscosity and/or of the time required to overcome their visco-elastic properties. In this way, the change in eyelid elongation is happening, largely, at the expense of the vertical elastic component (Fig. 6). The value of the elasticity constant k must also contribute decisively in the lengthening of the vertical elastic component. Thus, the stiffness must diminish in proportion to the separation from the levator palpebrae superioris muscle, having the lowest value near the margins of the eyelid (sum of springs in series). (vi) The elastic potential energy stored in the vertical springs of the aponeurosis produces the change of direction in the movement of the eyelid and makes it move rapidly in the upward direction. The elastic energy must reach its maximum when the difference in eyelid position between the eyelid saccade (where the movement of the levator palpebrae superioris muscle is the same as that of eyelid margins) and the blink (with the eyelid margins moving faster) is attained (dashed arrow in Fig. 5A). The activity of the orbicularis oculi muscle ceases when the eyelid begins to change direction during the blink, allowing the liberation of the elastic energy accumulated in the vertical component of the aponeurosis.
Figure 6 shows how the disposition of orbicularis oculi muscle fibres (lying over the aponeurosis) and the elastic characteristics of the aponeurosis (broad, fine band of connective tissue) enables the fast displacement of the eyelid margins during the downward phase of a blink. The force exerted by orbicularis oculi fibres then acts directly on the aponeurosis, and not on any other structure. Accordingly, we propose that the first phase in the upswing during a blink is dominated by elastic energy, while the second phase in mainly the result of the action of levator palpebrae superioris motoneurones.
According to the results present here, the existence of an initial rapid opening during a blink needs the previous downward movement to have a velocity significantly higher than that achieved in a saccade. In fact, a rapid return of the eyelid to an opened position was never observed when the eyelid had remained closed for a long period of time (> 50 ms), or when the velocity in the closing direction was slow. In these two latter cases, eyelid velocity in the upward direction depended on the amplitude of the upward eyelid displacement, but not on the velocity achieved during the downward phase. These data suggest that the rapid initial phase of the upward eyelid movement is not the result of the activation of the levator palpebrae superioris muscle. The most parsimonious explanation, according to the proposed mechanism, is that the levator palpebrae superioris muscle and Whitnall's ligament have enough time to follow the displacement of eyelid margins, releasing the stored elastic energy in the vertical component of the aponeurosis, thereby making a fast upward motion of the eyelid impossible.
Further evidence corroborating the proposed mechanism is obtained when eyelid movement is represented in the plane of phase. As shown in Fig. 4B, eyelid movements in which downward velocity was fast enough (lower arc), were characterized by a rapid, smooth upward phase (upper arc). Trajectories of the upward phase presented two transients: the first in charge of opening the eyelid rapidly, and the second in charge of returning the eyelid to its final position. The higher slope of the first transient depended on the velocity achieved in the downward movement of the eyelid. This first transient also presented a certain degree of symmetry with the corresponding portion of downward eyelid displacement. In contrast, upward components of blinks of small amplitude, which maintained the eyes closed for > 50 ms, or which experimentally presented low downward velocities, lacked these characteristics (Fig. 4C). In these latter cases, the upward movement contained many fluctuations, and the first transient was not present. While data shown in Fig. 4B are representative of movements made by elastic structures, those of Fig. 4C seem more appropriate for movements made by muscles (Trigo et al. 1999a; Berdún-Seijo et al. 2001).
Some considerations on the proposed mechanism
In contrast to responses evoked in the skeletal motor system, eyelid movements seem to be generated without the participation of classical proprioceptors (Hosokawa, 1961; Porter et al. 1989; VanderWerf et al. 1997; Trigo et al. 1999b). Although some atypical endings have been described (Hosokawa, 1961; Porter et al. 1989), they are postulated to intervene in the gain regulation of motoneurone output, but not in the stretch reflex (Evinger & Manning, 1988; Trigo et al. 1999b). Levator palpebrae superioris motoneurones, in contrast to motoneurones of the orbicularis oculi muscle, receive input signals related to eye position and velocity (Fuchs et al. 1992). This information, however, has its source in premotor structures that control superior rectus muscle activity, and not in eyelid muscle proprioceptors. Hence, activity in levator palpebrae superioris motoneurones is always coupled to eye movements. However, during blinks, superior rectus response is clearly dissociated from eyelid position and/or velocity (Fuchs et al. 1992). In this situation, levator palpebrae motoneurones remain silent and ‘blind’ to kinetic characteristics of the ongoing blink.
The absence of classical proprioceptors makes it difficult to explain the linear relationship found between maximum velocity in the downward and upward directions during blinks (see the analysis for phase I in Fig. 3B, and Trigo et al. 1999b). Another contradictory situation is when the activation of a single orbicularis oculi motoneurone is considered (Trigo et al. 1999a). As already stated, the second spike in the doublet has an enhanced effect on the velocity and amplitude of the downward phase of the blink. According to present knowledge, for the occurrence of such a situation the activity of levator palpebrae superioris motoneurones should be highly coordinated with that of orbicularis oculi motoneurones. Such coordination would demand a precise circuitry, still unknown, capable of evaluating the interspike interval of even a single orbicularis oculi motoneurone just to produce a corresponding relaxation and activation in levator palpebrae superioris motoneurones. Similarly, small oscillations found in the late phase of the blink, or during eyelid-conditioned responses, must be the consequence of the stretching of the aponeurosis. The only other possibility is the repetitive relaxation and activation of the levator palpebrae superioris muscle to avoid stretching the aponeurosis. In contrast to the mechanism described in the previous section, the two opposing forces acting to stretch the vertical elastic component are produced by the simultaneous, but antagonistic, action of the levator palpebrae superioris and orbicularis oculi muscles, because in these situations the levator palpebrae muscle is presumably not relaxed. On the other hand, the mechanism proposed here allows the existence of such events without the presence of proprioceptors, or of an (unknown) internal circuitry. Accordingly, the synchronous and high firing rate of the orbicularis oculi motoneurones produces the rapid descent of the eyelid. Depending on the evoked eyelid velocity, the vertical elastic component of the aponeurosis is correspondingly lengthened, regulating the velocity of the subsequent upswing.
An attempt was made to describe the proposed physiological model at a mathematical level. As a first step, we determined the contribution of the elastic force to the upward movement of the eyelid during blinks, measured as the difference between normal blinks and those that presented slow upward phases (accomplished solely by the action of the levator palpebrae muscle, see Fig. 4A). This difference ranged between 60 and 85 % of the total amplitude, indicating the large contribution of elastic structures.
The next step was to calculate the physical constants describing the proposed biomechanical model. Representations in the plane of phase of position against velocity suggested a second-order system for blinks:
where θ is the position in degrees, I is inertia, c is damping, and k is elasticity. Dividing by I:
Substituting to get standardized parameters:
where δ is the damping ratio and Ωn is the natural frequency. Applying Laplace's transform to solve the differential equation, we get the following characteristic equations:
where s is Laplace's frequency operator, or
the solution of which is:
In this case,
so expressing the two solutions as a function of k and c:
Besides the plane of phase, movements of the eyelid carried out by the isolated action of the elastic potential energy were also taken into account to solve this second-order system. In this sense, tiny blinks elicited by the action of a single motoneurone of the orbicularis oculi muscle can be considered to be of such kind (Trigo et al. 1999a). The two solutions were similar for eyelid movements < 4 deg, and a critically damped model is postulated:
where A1 is the amplitude of the movement,
and
 |
Eyelid movements > 4 deg (up to 6 deg) presented two different solutions, and an over-damped model is postulated:
where A = 1.66 ± 0.5, B = −6.73 ± 0.8, τa = 0.0046 ± 0.0015 s and τb = 0.0236 ± 0.0013 s. As stated above,
so the two solutions depend on k. If k ≈c2/4I, s is unique and equal to -c/2I. Lower values of k result in two different values of s. Thus, k must diminish with the amplitude of the blink. Note also that the strategy followed by the mechanical system for blinks > 4 deg is to achieve an initial rapid recovery of the eyelid (τ with lower value = τa, corresponding with the steep beginning of the upper arc in the plane of phase), continued by the second transient, which carries the eyelid close to its initial opened position.
Acknowledgments
This work was supported by grants PM98-0011 and BFI2002-00936 from the Spanish DGICYT, and 00/032-00 from La Caixa Foundation. The authors acknowledge the editorial help of Mr R. Churchill.
References
- Aramideh M, Ongerboer De Visser BW, Vander Werf F, Bour LJ, Brands JWM, Spielman JD. The inhibitory response of levator palpebrae muscle to evoked blink reflexes. In: Valls-Solé J, Tolosa E, editors. Brainstem Reflexes and Functions. S. A., Madrid: Litofinter; 1998. pp. 89–98. [Google Scholar]
- Becker W, Fuchs AF. Eyelid-eye coordination during vertical gaze changes in man and monkey. J Neurophysiol. 1988;60:1227–1252. doi: 10.1152/jn.1988.60.4.1227. [DOI] [PubMed] [Google Scholar]
- Berdún-Seijo JI, Roa LM, Trigo JA. Análisis del parpadeo mediante el plano de estados. Proceedings of CASEIB. Madrid: 2001. pp. 77–80. [Google Scholar]
- Björk A, Kugelberg E. The electrical activity of the muscles of the eye and eyelid in various positions and during movement. Electroencephalogr Clin Neurophysiol. 1953;5:595–602. doi: 10.1016/0013-4694(53)90037-6. [DOI] [PubMed] [Google Scholar]
- Delgado-García JM, Del Pozo F, Baker R. Behavior of neurons in the abducens nucleus of the alert cat - I. Motoneurons. Neuroscience. 1986;17:929–952. doi: 10.1016/0306-4522(86)90072-2. [DOI] [PubMed] [Google Scholar]
- Delgado-García JM, Evinger C, Escudero M, Baker R. Behavior of accessory abducens and abducens motoneurons during eye retraction and rotation in the alert cat. J Neurophysiol. 1990;64:413–422. doi: 10.1152/jn.1990.64.2.413. [DOI] [PubMed] [Google Scholar]
- Delgado-García JM, Gruart A, Trigo JA, Morcuende S. Neuronal organization and functional properties of the eyelid motor system. In: Valls-Solé J, Tolosa E, editors. Brainstem Reflexes and Functions. S. A., Madrid: Litofinter; 1998. pp. 25–35. [Google Scholar]
- Evinger C. A brain stem reflex in the blink of an eye. News Physiol Sci. 1995;10:147–153. [Google Scholar]
- Evinger C, Manning KA. A model system for motor learning: adaptive gain control of the blink reflex. Exp Brain Res. 1988;70:527–538. doi: 10.1007/BF00247600. [DOI] [PubMed] [Google Scholar]
- Evinger C, Manning KA, Sibony PA. Eyelid movements. Mechanisms and normal data. Invest Ophthalmol Vis Sci. 1991;32:387–400. [PubMed] [Google Scholar]
- Evinger C, Shaw MD, Peck CK, Manning KA, Baker R. Blinking and associated eye movements in humans, guinea pigs and rabbits. J Neurophysiol. 1984;52:323–339. doi: 10.1152/jn.1984.52.2.323. [DOI] [PubMed] [Google Scholar]
- Fuchs AF, Becker W, Ling L, Langer TP, Kaneko CRS. Discharge patterns of levator palpebrae superioris motoneurons during vertical lid and eye movements in the monkey. J Neurophysiol. 1992;68:233–243. doi: 10.1152/jn.1992.68.1.233. [DOI] [PubMed] [Google Scholar]
- Fuchs AF, Robinson DA. A method for measuring horizontal and vertical eye movements chronically in the monkey. J Appl Physiol. 1966;21:1068–1070. doi: 10.1152/jappl.1966.21.3.1068. [DOI] [PubMed] [Google Scholar]
- Gordon G. Observations upon the movements of the eyelids. Br J Ophthalmol. 1951;35:339–351. doi: 10.1136/bjo.35.6.339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gruart A, Blázquez P, Delgado-García JM. Kinematics of spontaneous, reflex and conditioned eyelid movements in the alert cat. J Neurophysiol. 1995;74:226–247. doi: 10.1152/jn.1995.74.1.226. [DOI] [PubMed] [Google Scholar]
- Holder DS, Scott A, Hannaford B, Stark L. High resolution electromyogram of the human eyeblink. Electromyogr Clin Neurophysiol. 1987;27:481–488. [PubMed] [Google Scholar]
- Hosokawa H. Proprioceptive innervation of striated muscles in the territory of cranial nerves. Tex Rep Biol Med. 1961;19:405–464. [PubMed] [Google Scholar]
- Kugelberg E. Facial reflexes. Brain. 1952;75:385–396. doi: 10.1093/brain/75.3.385. [DOI] [PubMed] [Google Scholar]
- Porter JD, Burns LA, May PJ. Morphological substrate for eyelid movements: innervation and structure of primate levator palpebrae superioris and orbicularis oculi muscles. J Comp Neurol. 1989;287:64–81. doi: 10.1002/cne.902870106. [DOI] [PubMed] [Google Scholar]
- Powers AS, Schicatano EJ, Basso MA, Evinger C. To blink or not to blink: inhibition and facilitation of reflex blinks. Exp Brain Res. 1997;113:283–290. doi: 10.1007/BF02450326. [DOI] [PubMed] [Google Scholar]
- Trigo JA, Gruart A, Delgado-García JM. Discharge profiles of abducens, accessory abducens and orbicularis oculi motoneurons during reflex and conditioned responses. J Neurophysiol. 1999a;81:1666–1684. doi: 10.1152/jn.1999.81.4.1666. [DOI] [PubMed] [Google Scholar]
- Trigo JA, Gruart A, Delgado-García JM. Role of proprioception in the control of eyelid position during reflex and conditioned blink responses in the alert behaving cat. Neuroscience. 1999b;90:1515–1528. doi: 10.1016/s0306-4522(98)00539-9. [DOI] [PubMed] [Google Scholar]
- Van Allen MW, Blodi FC. Electromyographic study of reciprocal innervation in blinking. Neurology. 1962;12:371–377. doi: 10.1212/wnl.12.5.371. [DOI] [PubMed] [Google Scholar]
- VanderWerf F, Aramideh M, Ongerboer De Visser BW, Baljet B, Speelman JD, Otto JA. A retrograde double fluorescent tracing study of the levator palpebrae superioris muscle in the cynomolgus monkey. Exp Brain Res. 1997;113:174–179. doi: 10.1007/BF02454155. [DOI] [PubMed] [Google Scholar]


