Abstract
KCNQ2/3 potassium channel subunits were co-expressed in Chinese hamster ovary (CHO) cells and currents through single channels recorded using cell-attached patches. Channels had a similar slope conductance in the presence (8.04 ± 0.02 pS) and absence (7.6 ± 0.01 pS) of 10 μm retigabine. The mean maximal open probability (Po) for single KCNQ2/3 channels was 0.13 ± 0.02, with a half-maximal Po potential (Vo) of −28.7 ± 1.4 mV for control recordings. Retigabine increased mean maximal Po to 0.38 ± 0.04 and produced a hyperpolarising shift of Vo to −40.1 ± 3.4 mV. Single KCNQ2/3 channels have multiple voltage-dependent kinetic components in their activity (CL-OS-CM-OL-CS; C = closed, O = open, L = long, S = short, M = medium), giving short, medium and long closed times (τCS, τCM, τCL) and short and long open times (τOS and τOL). In the presence of retigabine at 0 mV the combined duration and contributions of the longest closed time τCL decreased tenfold, while the short and long open times increased fourfold and twofold, respectively. Thus, steady-state kinetics were modified to favour the open channel configuration.
KCNQ2 and KCNQ3 subunits form heteromeric potassium channels that underlie slow, subthreshold M-type potassium currents (Wang et al. 1998). KCNQ2 and KCNQ3 K+ channel genes are widely distributed in the nervous system and are related to KCNQ1 (Yang et al. 1997), KCNQ4 (Kubisch et al. 1999) and KCNQ5 (Lerche et al. 2000; Shroeder et al. 2000) subunits. Mutations in KCNQ2 and KCNQ3 subunits resulting in reduced current levels produce a benign form of epilepsy in newborns (Biervert et al. 1998; Charlier et al. 1998; Singh et al. 1998; Lerche et al. 1999). This is consistent with the role that the M-type channels play in dampening neuronal excitability (Brown, 1988; Marrion, 1997).
In a previous publication (Tatulian et al. 2001), we reported how the anti-convulsant drug retigabine (D-23129; N-(2-amino-4-(4-fluorobenzylamino)-phenyl) carbamic acid ethyl ester) (Rostock et al. 1996; Tober et al. 1996) enhances whole cell macroscopic current through expressed heteromeric KCNQ2/3 and homomeric KCNQ2, KCNQ3 and KCNQ4 channels, as well as native M currents recorded in rat SCG neurones. The principal action of retigabine on macroscopic KCNQ currents is a shift in their voltage sensitivity to more hyperpolarised membrane potentials, along with a slowing of current deactivation and acceleration of current activation (Tatulian et al. 2001; see also Main et al. 2000; Rundfeldt & Netzer, 2000; Wickenden et al. 2000; Schroder et al. 2001).
In the present experiments, we have examined the effect of retigabine on the properties of single heteromeric KCNQ2/3 channels expressed in CHO cells, to see if the increase in macroscopic current amplitude caused by retigabine can be attributed to an increase in the number of functional channels, single channel conductance and/or open probability. The results suggest that retigabine enhances macroscopic KCNQ2/3 currents by increasing the channel open probability. This itself can be described by a reduction in the time constant and the contribution of the longest closed state (CL) accompanied with an increase in open times (OS and OL) in the CL-OS-CM-OL-CS scheme of M-channel gating (Selyanko & Brown, 1999).
Methods
The methods of cell culture and transfection, as well as cell-attached patch recording, were as described previously (Selyanko et al. 2001). Briefly, Chinese hamster ovary (CHO) cells were grown in minimum essential medium (α-MEM) supplemented with 10 % fetal calf serum, 1 % l-glutamine and 1 % penicillin- streptomycin with 5 % CO2 at 37 °C. Cells were plated in 35 mm plastic dishes and transfected 1 day later using LipofectAmine Plus (Gibco BRL). KCNQ and CD8 cDNA plasmids, in pCR3.1 vector (Invitrogen, The Netherlands), were co-transfected in a 10:1 ratio. For expression of KCNQ2/3 heteromultimers, equal amounts of KCNQ2 and KCNQ3 cDNA were used. Transfected cells were identified by adding CD8-binding Dynabeads (Dynal, UK) before recording.
Single channel activity was recorded 2 days after transfection, in order to increase the chances of obtaining functional channels in patches. Cell-attached pipettes (8–10 MΩ) were filled with a ‘low-K+’ solution of the following composition (mm): NaCl 144, KCl 2.5, CaCl2 2, MgCl2 0.5, Hepes 5, glucose 10; pH 7.4 with Tris base. Cells were bath-perfused with a ‘high-K+’ solution in which the KCl concentration was raised from 2.5 to 25 mm to clamp the membrane potential near −30 mV. The methods of recording and analysis were similar to those previously employed for studying M-channels (Selyanko & Brown, 1999). Records were filtered at 1 kHz and sampled at 5.26 kHz.
Data were acquired and analysed using pClamp software (versions 6.0 and 8.0; Axon Instruments, CA, USA). Currents were recorded using an Axopatch 200A patch-clamp amplifier (Axon Instruments). The presence of a single channel in a patch was deduced from the lack of current superimpositions over several minutes of recording, particularly at positive membrane potentials where the activity would be strongly facilitated. When current superimpositions were observed, the number of channels in the patch was estimated from the maximal number of superimposed openings.
Open and closed states of the channel were designated when the current amplitudes were above or below the 50 % level, respectively. Records were filtered at 500 Hz to reduce baseline noise. Channel openings and closings shorter than 1 ms were ignored. Mean single channel current (i) was obtained by fitting open-point amplitude histograms with single Gaussian curves. Distributions of open and closed times were logarithmically binned and fitted with exponential density functions by the method of maximum likelihood (see Colquhoun & Sigworth, 1995) in pSTAT (pClamp).
Where activity was produced by more than one channel, only i and an apparent open probability (Po) were measured. Current levels were estimated by fitting all-points amplitude histograms with multiple Gaussian curves, whereas Po was calculated according to the following equation:
where tj is the time spent at each current level corresponding to j = 0, 1, 2…N; T is the duration of the recording; N is the number of current levels (minimum number of active channels).
Po−Vm relationships were fitted with the following Boltzmann equation:
where Po is the channel open probability and Po,max is the maximum Po attained at strongly depolarised membrane potentials; Vm is the assumed membrane potential (estimated as Vrest − Vpipette, where Vrest = −30 mV and Vo is the half-activation potential, i.e. the potential where Po = 0.5Po,max and k is the slope factor that indicates the potential change required for an e-fold increase in Po. The program Origin (version 5.0, Microcal Software Inc., MA, USA) was used for constructing linear and Boltzmann fits and creating the figures.
cDNAs for human KCNQ2 and rat KCNQ3 were obtained from Dr D. McKinnon. Retigabine was obtained from Glaxo SmithKline (Stevenage, UK); 10 mm stock solutions of retigabine were prepared in distilled water daily and kept on ice and away from sunlight. All other drugs and chemicals were obtained from BDH.
Results
The amplitude of the whole cell current is equal to N × i × Po, where N is the number of functional channels and i and Po are their mean single channel current amplitude and open probability. In order to establish whether the previously reported increase in whole-cell KCNQ2/3 current amplitude following application of retigabine (see Introduction) was related to the difference in the number of functional channels or the difference in their abilities to generate currents, the three aforementioned parameters were investigated using the cell-attached configuration under control conditions and in the presence of 10 μm retigabine. (This concentration was sufficient to produce near-maximum enhancement of the whole cell KCNQ2/3 current and ∼80 % of the maximum voltage shift: Tatulian et al. 2001.)
Currents through single KCNQ2/3 channels were recorded 2 days after transfection. Figure 1A shows the activity of a single KCNQ2/3 channel recorded in the cell-attached configuration at 0 mV under control conditions (top panel), in the presence of 10 μm retigabine (middle panel) and following washout of the drug (bottom panel). Upward deflections represent channel openings from the closed state (dashed line), and downward deflections represent channel closings. Under control conditions the channel spent most of the time in the closed configuration, and channel openings were brief (< 40 ms). When retigabine was added to the bath solution, the channel became predominantly open with only brief closings. These effects of the drug were fully reversible. No openings to a second current level were observed in single channel patch recordings following addition of retigabine, implying that it did not increase the number of functional channels.
Figure 1. Effect of retigabine on the activity of single KCNQ2/3 channels expressed in CHO cells.
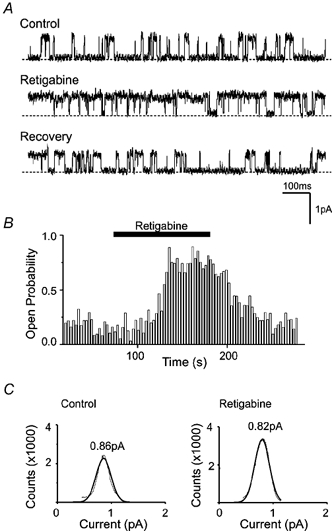
A, steady state activation of a single KCNQ2/3 channel by continuous membrane depolarisation at ∼0 mV (−30 mV patch-potential) recorded in the cell-attached configuration. Records show channel activity under control conditions (top panel), in the presence of 10 μm retigabine in the bath solution (middle panel) and 3 min following washout of the drug (bottom panel). Calibration: 1 pA, 100 ms. B, time course of retigabine action (same channel as A). KCNQ2/3 channel open probability (see Methods) plotted against time (in seconds). Channel open probability began to increase ∼30 s following 10 μm retigabine application. C, open point amplitude histograms measured at 0 mV KCNQ2/3 channel openings from records in A were binned and open point amplitude histograms plotted for control recordings (left panel) and in the presence of 10 μm retigabine (right panel). Smooth lines show Gaussian fits. The mean current components were 0.86 pA for control and 0.82 pA for retigabine-treated channels.
The KCNQ2/3 channel activity began to increase within seconds of retigabine application (10 μm). In Fig. 1B KCNQ2/3 channel open probability (Po) is plotted against time. On application of the drug, channel activity gradually increased, reaching a plateau after ∼60 s of retigabine application.
Current amplitude through a single KCNQ2/3 channel under control conditions at 0 mV was just below 1 pA, and it remained unchanged when retigabine was present in the bath. This is demonstrated in Fig. 1C, where open point amplitude histograms for the activities shown in Figure 1A (top and middle panels) have been plotted. The results were fitted by single Gaussian curves, giving (for this cell) a mean single channel current at 0 mV of 0.86 pA under control conditions and 0.82 pA in the presence of 10 μm retigabine. Thus retigabine did not appear to change the current amplitude through single KCNQ2/3 channels.
To see if this held at other potentials, currents through single KCNQ2/3 channels were measured over the range of −60 to +30 mV (Fig. 2A). Single channel slope conductances obtained from pooled I–Vm relationships were 7.6 ± 0.01 pS for control recordings and 8.04 ± 0.02 pS for recordings in the presence of 10 μ m retigabine (mean ±s.e.m.; n = 8). Thus, KCNQ2/3 channel conductance values were not significantly different in the presence or absence of retigabine.
Figure 2. Effect of retigabine on conductance and open probability of single KCNQ2/3 channels.
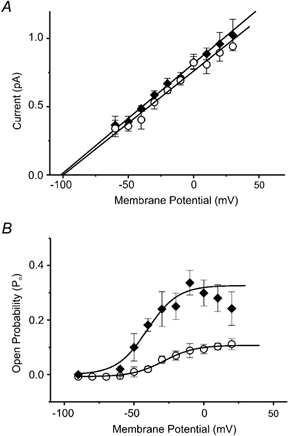
A, single channel currents (mean ± s.e.m.) plotted against patch membrane potential for control conditions (○) and in the presence of 10 μm retigabine (♦). Slope conductances measured between −60 and 0 mV (straight lines) were 7.6 ± 0.01 pS for control recordings (n = 8) and 8.04 ± 0.02 pS in the presence of retigabine (n = 8). B, mean open probabilities (mean ±s.e.m.) for steady-state activities of single KCNQ2/3 channels were estimated as described in Methods and plotted against each patch membrane potential for control recordings (○) and in the presence of 10 μm retigabine (♦). Po−Vm relationships were fitted with Boltzmann equations with maximal Po, half-activation potential (Vo) and slope factor of 0.13 ± 0.02, −28.7 ± 1.4 mV and 11.3 ± 1.12 mV, respectively, for control recordings (n = 8), and 0.38 ± 0.04, −40.2 ± 3.4 mV and 9.7 ± 3.1 mV for retigabine-treated cells (n = 6). (Note: points above 0 mV were not used for these fits.)
Effects of retigabine on the KCNQ2/3 channel open probability were determined by plotting Po−Vm relationships in the absence and presence of 10 μm retigabine (Fig. 2B). Data for different conditions were fitted by a Boltzmann equation (see Methods).
In control recordings the half-activation potential was −28.7 ± 1.4 mV, the slope factor was 11.3 ± 1.12 mV and maximal Po was 0.13 ± 0.02 (means ±s.e.m. from individual Boltzmann curves fitted to each of 8 cells). By comparison, the Po−Vm relationship for retigabine-treated channels had a more negative half-activation potential (−40.2 ± 3.4 mV), a similar slope value (9.7 ± 3.1 mV) and a higher maximal Po (0.38 ± 0.04) (n = 6). It is interesting to note that the Po−Vm curve appears bell-shaped at more positive potentials, suggesting that mean single channel Po enhancement by retigabine decreases with more positive potentials (possibly due to a secondary blocking action of the drug at these potentials). Values above 0 mV were not used in the calculated Boltzmann fits in Fig. 2B. Notwithstanding, inclusion of points > 0 mV had no significant effect on the derived values for Vo.
Heteromeric KCNQ2/3 potassium channels have been reported to have multiple kinetic components to their activity, suggesting the presence of at least three closed and two open states (Selyanko et al. 2001). Figure 3A exemplifies the distributions of closed and open times obtained from the activity of single KCNQ2/3 channels recorded at 0 mV under control conditions. These distributions were fitted with three and two exponentials, respectively. Thus, for this particular example, the closed state of the KCNQ2/3 channel under control conditions could be described as the sum of three components, with short (τc1), medium (τc2) and long (τc3) time constants (and their relative percentage contributions) being equal to 0.7 ms (55.7 %), 25.8 ms (31.4 %) and 130.7 ms (12.9 %), respectively. The open time distribution in this example could be described as the sum of two components, with short (τo1) and long (τo2) time constants (and their percentage contributions) being equal to 0.5 ms (46 %) and 15.1 ms (54 %). Retigabine (10 μm) had relatively minor effects on the time constants for the short and medium closed-time components, with τo1 = 2.1 ms (41 %) and τo2 = 11.2 ms (45 %), but dramatically reduced the time constant for the long shut time to τc3 = 53.9 ms (13.9 %). The open time constants were also changed by retigabine: both the short and long open time constants were greater, with τo1 = 4.6 ms (67.4 %) and τo2 = 24.4 ms (32.6 %).
Figure 3. Effects of retigabine on the kinetics of KCNQ2/3 channel activity.
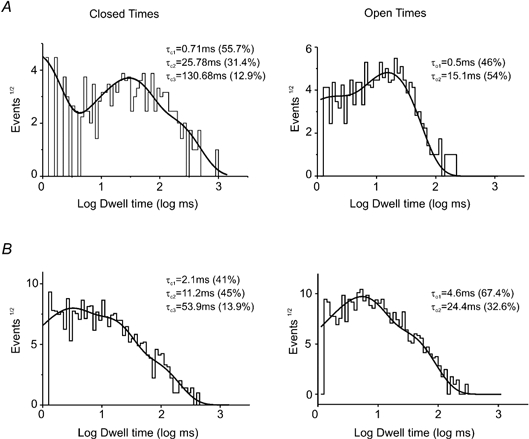
Closed time (left panel) and open time (right panel) distributions for KCNQ2/3 single channel activity recorded at 0 mV are shown for control recordings (A) and in the presence of 10 μm retigabine (B). Closed time distributions were fitted with three exponential density functions and open time distributions with two. Sums of these components are shown as smooth lines, with corresponding kinetic components (and percentage contributions) indicated above each distribution.
Kinetic analyses of steady-state activity at 0 mV were completed for five different single KCNQ2/3 channel records and the results summarised in Fig. 4. It becomes clear that in fact retigabine did not alter the short and medium closed time constants to a great degree but that its principal effect was to shorten the time constant of the long closed time fourfold and to reduce the contribution of this component to the overall distribution of closed states by ∼60 %, thus reducing the total contribution of this state to the total closed time about tenfold. Retigabine also increased both the short and long open time constants, whilst keeping the percentage contributions of these two components very similar to the values obtained during control recordings.
Figure 4. Summary of the effects of retigabine on kinetic components of single KCNQ2/3 channel activity.
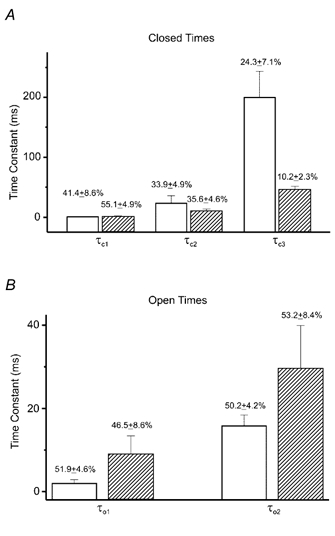
Activities were recorded at 0 mV. τc1, τc2 and τc3 are short, medium and long kinetic components of channel closed times (A), whereas τo1 and τo2 are short and long kinetic components in the distribution of open times (B). Values are means ±s.e.m. with n = 6. □, controls;  , +10 μm retigabine. Values above the columns are corresponding percentage contributions of each component to the total shut or open times.
, +10 μm retigabine. Values above the columns are corresponding percentage contributions of each component to the total shut or open times.
Discussion
The channel currents recorded in these experiments probably represent currents through a single population of heteromeric KCNQ2/3 channels because they had a uniform conductance level, whereas conductance levels of homomeric KCNQ2 and KCNQ3 channels are different (Selyanko et al. 2001). Further, previous measurements of TEA block of macroscopic currents genereated in these cells by co-expression of cDNAs for KCNQ2 and KCNQ3 subunits yielded an IC50 geometrically intermediate between those for homomeric KCNQ2 and KCNQ3 currents (Hadley et al. 2000). Also, we have recently confirmed that the TEA inhibition curve for these KCNQ2/3 currents is indistinguishable from those obtained using a concatenated KCNQ2-3 cDNA construct (Hadley et al. 2003).
Retigabine is a lipophilic compound, and appears to gain access to the KCNQ channels in cell-attached patches even when applied to the bathing solution outside the patch. Although we cannot positively exclude some indirect action, the rapid onset of its effect on in-patch channel activity (within seconds) suggests that it probably acts on the channels themselves, in a similar manner to that observed in whole-cell recording on bath application.
The principal effect of retigabine was to increase single channel open probability, without change in single channel conductance or the number of functional channels in the patch. Thus, no additional channel openings to a second current level were observed in single channel patch recordings, and the conductance remained at ∼8 pS in the absence and presence of retigabine (and similar to that (∼9 pS) previously reported: Selyanko et al. 2001).
The increased activity resulted both from an increase in the maximum open probability, and from a hyperpolarising shift of about 11.5 mV in voltage sensitivity. The hyperpolarising shift accords qualitatively with the effects observed with macroscopic current recording, though somewhat less in magnitude (22 mV: see Tatulian et al. 2001). On the other hand, no increase in maximum current comparable to the increased single channel Po,max could be obtained in previous whole-cell recordings (Tatulian et al. 2001). This we had attributed to a secondary blocking action at positive potentials, which could be observed as a reduction of total current in KCNQ1-expressing cells (the KCNQ1 current being resistant to the enhancing effect of retigabine). The decline in single channel Po at positive potentials in the presence of retigabine may reflect a similar inhibitory action. Possibly this is targeted at a different site in the channel from the enhancing effect, which may be less accessible to retigabine in cell-attached patches than in whole-cell recording.
Kinetic analyses of M- and expressed KCNQ2/3 channel activity conducted in previous studies (Selyanko & Brown, 1999; Selyanko et al. 2001) suggest that this activity can be described by a five-state sequential model, where three closed states are separated by two open states (CL-OS-CM-OL-CS). The longest closed state (CL) in the activity is more than one order of magnitude longer than the other closed states. It was suggested that CL becomes dominant near threshold potentials for M-channel activation and is probably the state in which the channel dwells when it is deactivated. In the presence of retigabine this closed state is dramatically shortened (from ∼200 ms to ∼47 ms at 0 mV), and the percentage contribution of this closed state to total closed times also declines in the presence of the drug (from ∼24 % to ∼10 %). This alone would have the effect of increasing channel Po, particularly at negative potentials, where CL dominates. However, the reduction in CL is also accompanied by an enhancement of both open times; the OS time constant increases from ∼2 to ∼9 ms, and the OM time constant increases from ∼16 to ∼30 ms. Thus, the combined effect of retigabine on open and closed times is to favour the open state, thereby increasing Po.
In some respects – the increased maximum Po, the hyperpolarising shift in the Po−Vm curve, and the marked reduction in the long closed times – the effect of retigabine resembles the effects observed following excision of M-channel patches into a Ca2+-free solution (Selyanko & Brown, 1999). Under such conditions, addition of intracellular Ca2+ reduces M-channel activity (Selyanko & Brown, 1996). Recent evidence suggests that the inhibitory effect of Ca2+ ions on either expressed KCNQ channels or native M-channels is mediated by calmodulin (Gamper & Shapiro, 2002). One possibility, therefore, is that retigabine might modulate Ca2+-calmodulin-KCNQ channel interaction. We have considered this possibility but think it unlikely for two reasons. First, the increase in open times produced by retigabine appeared not to be a prominent component of the effect of reducing Ca2+ (Selyanko & Brown, 1999). Second, retigabine still enhanced whole-cell M-currents in sympathetic neurones patched with open-tip electrodes containing an EGTA-buffered Ca2+-free solution (L. Tatulian & D. A. Brown, unpublished experiments). Hence, the molecular mechanism of retigabine action remains unclear. Nevertheless, the present data should assist in the eventual resolution of this question.
References
- Biervert C, Schroeder BC, Kubisch C, Berkovic SF, Propping P, Jentsch T, Steinlein OK. A potassium channel mutation in neonatal human epilepsy. Science. 1998;279:403–406. doi: 10.1126/science.279.5349.403. [DOI] [PubMed] [Google Scholar]
- Brown D. M-currents: an update. Trends Neurosci. 1988;11:294–299. doi: 10.1016/0166-2236(88)90089-6. [DOI] [PubMed] [Google Scholar]
- Charlier C, Singh NA, Ryan SG, Lewis TB, Reus BE, Leach RJ, Leppert M. A pore mutation in a novel KQT-like potassium channel gene in an idiopathic epilepsy family. Nature Genetics. 1998;18:53–55. doi: 10.1038/ng0198-53. [DOI] [PubMed] [Google Scholar]
- Colquhoun D, Sigworth FJ. Fitting and statistical analysis of single channel records. In: Sakmann B, Neher E, editors. Single Channel Recording. 2. New York: Plenum; 1995. pp. 483–587. [Google Scholar]
- Gamper N, Shapiro MS. Calmodulin mediates Ca2+ modulation of KCNQ2/3 K+ channels. Soc Neurosci Abstr. 2002;217.2 [Google Scholar]
- Hadley JK, Noda M, Selyanko AA, Wood IC, Abogadie FC, Brown DA. Differential tetraethylammonium sensitivity of KCNQ1-4 potassium channels. Br J Pharmacol. 2000;129:413–415. doi: 10.1038/sj.bjp.0703086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hadley JK, Passmore GM, Tatulian L, Al-Qatari M, Ye F, Wickenden AD, Brown DA. Stoichiometry of expressed KCNQ2/KCNQ3 channels and subunit composition of native ganglionic M-currents deduced from block by tetraethylammonium (TEA) J Neurosci. 2003 doi: 10.1523/JNEUROSCI.23-12-05012.2003. (in the Press) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kubisch C, Schroeder BC, Friedrich T, Lutjohann B, El-Amraoui A, Marlin S, Petit C, Jentsch TJ. KCNQ4, a novel potassium channel expressed in sensory outer hair cells, is mutated in dominant deafness. Cell. 1999;96:437–446. doi: 10.1016/s0092-8674(00)80556-5. [DOI] [PubMed] [Google Scholar]
- Lerche C, Scherer CR, Seebohm G, Derst C, Wei AD, Busch AE, Steinmeyer K. Molecular cloning and functional expression of KCNQ5, a potassium channel subunit that may contribute to neuronal M-current diversity. J Biol Chem. 2000;275:22395–22400. doi: 10.1074/jbc.M002378200. [DOI] [PubMed] [Google Scholar]
- Lerche H, Biervert C, Alekov AK, Schleithoff L, Lindner M, Klinger W, Bretschneider F, Mitrovic N, Jurkat-Rott K, Bode H, Lehmann-Horn F, Steinlein OK. A reduced K+ current due to a novel mutation in KCNQ2 causes neonatal convulsions. Ann Neurol. 1999;46:305–312. doi: 10.1002/1531-8249(199909)46:3<305::aid-ana5>3.0.co;2-5. [DOI] [PubMed] [Google Scholar]
- Main MJ, Cryan JE, Dupere JRB, Cox B, Clare JJ, Burbidge SA. Modulation of KCNQ2/3 potassium channels by the novel anticonvulsant retigabine. Mol Pharmacol. 2000;58:253–262. doi: 10.1124/mol.58.2.253. [DOI] [PubMed] [Google Scholar]
- Marrion NV. Control of M-current. Ann Rev Physiol. 1997;59:483–504. doi: 10.1146/annurev.physiol.59.1.483. [DOI] [PubMed] [Google Scholar]
- Rostock A, Tober C, Rundfeldt C, Barsch R, Engel J, Polymeropoulos EE, Kutscher B, Loscher W, Homack D, White HS, Wolf HH. D-23129, a new anticonvulsant with a broad spectrum activity in animal models of epileptic seizures. Epilepsy Res. 1996;23:211–223. doi: 10.1016/0920-1211(95)00101-8. [DOI] [PubMed] [Google Scholar]
- Rundfeldt C, Netzer R. The novel anticonvulsant retigabine activates M-currents in Chinese hamster ovary-cells transfected with human KCNQ2/3 subunits. Neurosci Lett. 2000;282:73–76. doi: 10.1016/s0304-3940(00)00866-1. [DOI] [PubMed] [Google Scholar]
- Schroder RL, Jesperson T, Christopherson P, Strobaek D, Jensen BS, Oleson SP. KCNQ4 channel activation by BMS-204352 and retigabine. Neuropharmacology. 2001;40:888–898. doi: 10.1016/s0028-3908(01)00029-6. [DOI] [PubMed] [Google Scholar]
- Schroeder BC, Hechenberger M, Weinreich F, Kubisch C, Jentsch TJ. KCNQ5, a novel potassium channel broadly expressed in brain, mediates M-type currents. J Biol Chem. 2000;31:24089–24096. doi: 10.1074/jbc.M003245200. [DOI] [PubMed] [Google Scholar]
- Selyanko AA, Brown DA. Intracellular calcium directly inhibits potassium M channels in excised membrane patches from rat sympathetic neurons. Neuron. 1996;16:151–162. doi: 10.1016/s0896-6273(00)80032-x. [DOI] [PubMed] [Google Scholar]
- Selyanko AA, Brown DA. M-channel gating and simulation. Biophys J. 1999;77:701–713. doi: 10.1016/S0006-3495(99)76925-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Selyanko AA, Hadley JK, Brown DA. Properties of single M-type KCNQ2/KCNQ3 potassium channels expressed in mammalian cells. J Physiol. 2001;534:15–24. doi: 10.1111/j.1469-7793.2001.00015.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh NA, Charlier C, Stauffer D, Dupont BR, Leach RJ, Melis R, Ronen GM, Bjerre I, Quattlebaum T, Murphy JV, McHarg ML, Gagnon D, Rosales T, Peiffer A, Anderson VE, Leppert M. A novel potassium channel gene KCNQ2, is mutated in an inherited epilepsy of newborns. Nature Genetics. 1998;18:25–29. doi: 10.1038/ng0198-25. [DOI] [PubMed] [Google Scholar]
- Tatulian L, Delmas P, Abogadie FC, Brown DA. Activation of expressed KCNQ potassium currents and native neuronal M-type potassium currents by the anti-convulsant drug retigabine. J Neurosci. 2001;21:5535–5545. doi: 10.1523/JNEUROSCI.21-15-05535.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tober C, Rostock A, Bartsch R. D-23129: a potent anticonvulsant in the amygdala kindling model of complex partial seizures. Eur J Pharmacol. 1996;303:163–169. doi: 10.1016/0014-2999(96)00073-8. [DOI] [PubMed] [Google Scholar]
- Wang H-S, Pan Z, Shi W, Brown BS, Wymore R, Cohen IS, Dixon JE, McKinnon D. KCNQ2 and KCNQ3 potassium channel subunits: molecular correlates of the M-channel. Science. 1998;282:1890–1893. doi: 10.1126/science.282.5395.1890. [DOI] [PubMed] [Google Scholar]
- Wickenden AD, Yu W, Zou A, Jegla T, Wagoner PK. Retigabine, a novel anti-convulsant, enhances activation of KCNQ2/3 potassium channels. Mol Pharmacol. 2000;58:591–600. doi: 10.1124/mol.58.3.591. [DOI] [PubMed] [Google Scholar]
- Yang W-P, Levesque PC, Little WA, Condor ML, Shalaby FY, Blanae MA. KvLQT1, a voltage-gated potassium channel responsible for human arrhythmias. Proc Natl Acad Sci U S A. 1997;94:4017–4021. doi: 10.1073/pnas.94.8.4017. [DOI] [PMC free article] [PubMed] [Google Scholar]


