Abstract
The excitability of two groups of neurones located in different parts of the sacral spinal cord were examined during micturition in decerebrate adult cats. One group of cells, characterized by their activation by pudendal cutaneous afferents, was located in the dorsal commissure of the first and second sacral spinal segments. The second group, located in the dorsal horn of the first sacral spinal segment, was excited by group II muscle and cutaneous afferents. Micturition was evoked by distension of the urinary bladder or by electrical stimulation of the pontine micturition centre. Tonic firing was induced in the neurones by ejection of DL-homocysteic acid from the recording extracellular micropipette. The instantaneous firing frequency of 11/17 sacral dorsal grey commissure neurones was decreased from 7 to 100 % during micturition, and on average was about half of the prevoid firing frequency. It is hypothesized that these sacral neurones are interposed in polysynaptic excitatory pathways from sacral perineal afferents to sphincter motoneurones and that they are subject to direct postsynaptic inhibition during micturition. One other cell showed no change in firing during micturition, two displayed complex patterns of modulation, while 3/17 of the dorsal grey commissure neurones increased their firing rate 30 to 1000 % during micturition. It is hypothesized that the excited neurones may be part of the inhibitory pathways mediating postsynaptic inhibition of sphincter motoneurones or sacral primary afferent depolarization during micturition. Alternatively, they may be part of the excitatory urethral-bladder reflex circuitry. A small (5–15%) but significant decrease in firing was observed in 4/5 of the group II rostral sacral neurones examined; the firing of a fifth neurone was unchanged. The depression of group II neurones may serve to suppress unwanted hindlimb reflexes that could disrupt micturition.
In the spinal intact adult cat, activation of sacral afferents innervating the urethra, anus, perineum and hindlimb skin evokes reflex contraction of the striated external urethral and anal sphincters (Bishop, 1959; Mackel, 1979; Fedirchuk et al. 1992a). Behaviour (e.g. copulation) and movement that lead to compression of the abdominal cavity and associated increases in bladder pressure may also activate these cutaneous afferents and this continence-promoting reflex. If urine is forced into the urethra, urethral flow receptors and afferents are additionally activated and the continence reflex is reinforced (Barrington, 1914; Kiruluta et al. 1981; Mazieres et al. 1997). Together, cutaneous and urethral afferents activate reflex pathways that ensure continence during rapid changes in bladder pressure when the tonic constriction of smooth muscle in the bladder neck and proximal urethra may not be sufficient to maintain continence. Failure to suppress these excitatory sphincter reflexes during micturition results in bladder sphincter dyssynergia which compromises bladder emptying.
During micturition, excitatory sacral reflexes to the external urethral sphincter (EUS) are suppressed (Shimoda et al. 1992; Fedirchuk et al. 1994). A glycinergic postsynaptic inhibition of EUS motoneurones (Shefchyk et al. 1998) and presynaptic inhibition of pudendal and perineal afferents (Angel et al. 1994; Buss & Shefchyk, 1999) entering the sacral spinal cord have been shown to contribute to suppression of the sphincter reflex. Whether inhibition of interneurones in this polysynaptic pathway also contributes to suppression of this reflex has not been explored. In addition, there appears to be a state-dependent switch in urethral afferent actions that is determined by the state of the bladder (Barrington, 1914; Kiruluta et al. 1981; Mazieres et al. 1997; Shefchyk & Buss, 1998; Geirsson & Fall, 1999).
Two populations of first order sacral interneurones receive inputs from perineal and hindlimb cutaneous afferents and are candidate neurones in the excitatory reflex pathway to sacral sphincter motoneurones. The first population of interest includes neurones within the dorsal grey commissure of the sacral spinal segments. It has been demonstrated anatomically that pudendal primary afferents terminate in this region (Ueyama et al. 1984; Thor et al. 1989) and neurones associated with the control of the striated sphincters have been described using both viral tracing approaches (Nadelhaft & Vera, 1996; de Groat et al. 1996) and electrophysiology (McMahon & Morrisson, 1982a,b; Blok et al. 1998; Coonan et al. 1999; Shefchyk, 2001). Some of these neurones, referred to in the present work as dorsal grey commissure neurones with sensory pudendal afferent input (DGC-SPud neurones), are strongly activated by sensory pudendal afferents (including urethral afferents) and other perineal and hindlimb cutaneous afferents (Coonan et al. 1999), but not by hindlimb muscle afferents. Furthermore, a subset of neurones within the dorsal grey commissure, which have been demonstrated to receive direct projections from the pontine micturition centre (Blok et al. 1997) are glycinergic (Sie et al. 2001) and GABAergic (Blok et al. 1997), and are hypothesized to mediate the inhibition of sphincter motoneurones during micturition (Blok et al. 1998).
The second population of neurones, located in the lateral half of the dorsal horn in the first sacral segment, are activated by group II muscle afferents (referred to from here on as sacral group II neurones) in addition to perineal and hindlimb cutaneous afferents. Although the synaptic connectivity and morphology of these sacral group II neurones have been described in detail (Jankowska & Riddell, 1993, 1994, 1995; Jankowska et al. 1993, 2000), nothing is known about their activity during a motor programmme or behaviour. Based on their inputs, they are hypothesized to be involved in postural adjustments associated with defecation and grooming.
The purpose of the present study was to examine the activity of neurones within these two regions of the sacral spinal cord, the sacral DGC-SPud and the rostral sacral group II interneurones, during micturition to further our understanding of sacral spinal reflex integration and spinal neural control of micturition. It was hypothesized that sacral neurones strongly activated by cutaneous perineal afferents are interposed in polysynaptic pathways to sphincter motoneurones (Fedirchuk et al. 1992a) and that they are inhibited during micturition as a means of suppressing excitatory perineal to pudendal reflex pathways (Fedirchuk et al. 1994). This inhibition would work in concert with the direct postsynaptic inhibition of the motoneurones and the presynaptic inhibitory mechanisms previously demonstrated (reviewed in Shefchyk, 2001). The activity pattern of the sacral group II interneurones during any motor behaviour has not been described and this is the first examination of their activity during sacral motor function.
METHODS
The results reported here were obtained from eight adult male and female decerebrate cats (2.7-4.6 kg) in which stable extracellular intra-spinal recordings were obtained from a total of 22 spinal neurones during micturition. Micturition, characterized by a coordinated bladder contraction and external urethral sphincter (EUS) activity during which time fluid is expelled, was evoked by distension of the bladder in six animals or by electrical stimulation of the pontine micturition centre (PMC) in two animals. The PMC was functionally defined as the site from which a coordinated bladder pressure increase and decreased sphincter activity was produced within seconds of stimulation (see Fedirchuk & Shefchyk, 1991 for details of anatomy).
All procedures were carried out in compliance with the guidelines stipulated by the Canadian Council for Animal Care and the University of Manitoba. Surgery was performed under halothane (1-4 %) carried in a mixture of nitrous oxide (70 %) and oxygen administered by ventilation. The level of anaesthesia was judged to be adequate when pedal withdrawal reflexes were absent and no arterial blood pressure increases were produced by afferent stimulation. A pre-collicular-post-mamillary decerebration was performed under deep anaesthesia and all neural tissue rostral to the transection was removed. Anaesthesia was then discontinued and the animals were subsequently subjected to neuromuscular blockade with pancuronium bromide (Sabex, 0.2-0.3 mg every 30 min) and artificially ventilated (end-tidal CO2 at 3-5 %). The decerebrate, neuromuscularly blocked animals were killed by withdrawal of artificial ventilation with death confirmed by the absence of an arterial pulse pressure. A solution of 100 mM NaHCO3 and glucose (5 %) was infused (5 ml h−1) throughout the experiment and dextran was administered to replace blood loss and maintain blood pressure above 100 mmHg. The following peripheral nerves were dissected on the left side of the animal: the external urethral (EUS) and anal (EAS) sphincter pudendal motor branches; urethral (urSPud) and dorsal penile/clitoral (cutSPud) pudendal sensory branches; superficial perineal (SFP), caudal cutaneous femoral (CCF) and sural (SUR) cutaneous nerves; and the nerves innervating the posterior biceps-semitendinosus (PBST) and triceps surae (GS) hindlimb muscles. The proximal portions of a cut sacral dorsal root filament in which dorsal root potentials (DRPs) were recorded in response to stimulation of the cutSPud and urSPud nerves were used to examine intra-spinal evoked dorsal root potentials.
The cut peripheral nerves and the dorsal root filament were mounted on bipolar Ag-AgCl electrodes. Nerve stimulus strengths are expressed in multiples of the threshold (T) current that produced the first detectable volley at the cord dorsum electrode positioned on the first or second sacral segment. Extracellular intra-spinal recordings were made using single barrelled micropipettes (2-5 MΩ) filled with either 3 M sodium acetate or a solution of 0.5 M DL-homocysteic acid (DL-H, Sigma, St Louis, MO, USA) and 0.5 M NaCl buffered to pH 4 with Tris. The central latency for activation of a neurone from peripheral afferents was determined by measuring the latency from the incoming cutaneous or group I volleys to the onset of the extracellular neurone spike. The DL-H-filled microelectrodes were used to induce a tonic discharge in the neurones. A positive retaining current (< 5 nA) was used to minimize diffusion of DL-H from the micropipette and a negative current (>-5 nA) was used to eject DL-H.
Neurones activated by perineal afferents located within the dorsal grey commissure (DGC-SPud neurones) were located 1100-1700 μm below the surface of the first or second sacral spinal segment near the midline where large extracellular field potentials were evoked by stimulation of the cutSPud afferents. Sacral group II excited neurones were located in the rostral half of the first sacral segment, at a level where a large PBSt-evoked group II afferent component of the cord dorsum was evident and often, group II afferent-evoked extracellular field potentials were evident (Jankowska & Riddell, 1993; 1995). These neurones were recorded at depths of 800-1900 μm below the dorsal surface of the spinal cord and were more lateral than the DGC-SPud neurones. No effort was made to distinguish between interneurones, propriospinal neurones or ascending tract cells in the neurones sampled. To be included in the analysis, the neurone had to have a large negative spike (mean 590 ± 400 μV) that did not vary in amplitude during the recording period, and a rate of firing that could be finely controlled by iontophoresis (±5 nA) of DL-H. A total of 36 voids were evoked in eight animals, including 14 distension-evoked reflexes while recording from 13 units, and 20 PMC-evoked voids during recordings from eight units; one unit was monitored during both a PMC and distension-evoked reflex.
During a void, the DC extracellular recording of unit firing (Dagan 8700 Cell Explorer, 10 kHz low pass), dorsal root filament (3 kHz low pass, 0.1 Hz high pass, × 50 000 amplification), EUS and EAS electroneurograms (ENGs; 3 kHz low pass, 300 Hz high pass, × 20000 amplification) were digitized at 10 kHz. Bladder pressure was digitized at 200 Hz and it was noted when fluid was expelled from the urethra. Analysis of digitized data used software developed at the Winnipeg Spinal Cord Research Centre. Extracellular spikes were discriminated using an amplitude threshold window discriminator after the DC extracellular recording was digitally high pass filtered (100-200 Hz) to remove DC shifts and low pass filtered (800-2000 Hz) to remove high frequency noise. The data were transformed into instantaneous frequency (FI) values which in turn were used to graphically and statistically examine the firing of the neurone. Evoked firing of a neurone was used to spike trigger average 120-5800 responses in the EUS ENG, EAS ENG and the dorsal root filament.
Data collected during each trial were placed into one of five periods within a trial. These included a pre-void period immediately prior to micturition, the evoked micturition reflex and a post-void period. The micturition reflex was subdivided into characteristic parts; the time when the bladder contraction increased the bladder pressure 10 % above baseline, the portion during the peak bladder pressure when fluid was expelled and the period of diminished EUS ENG activity during the bladder contraction. For each of these periods, 90 % of the FI distributions were not normal but rather, were skewed to higher frequencies (skew = 3.4 ± 3.2; mean ± S.D. of mean). Thus, mean values of the distributions poorly described (sample size > 50 in 80 % of the distributions) the data and so median values were used. Median FI values for the three periods during the micturition reflex, as well as the post-void period, were normalized to the median pre-void FI (which was on average 18 ± 17 Hz; mean ± S.D. of mean). This normalization permitted the averaging of data from multiple voids observed in a neurone and averaging among (expressed as median and coefficient of variation of the median) neurones. Non-parametric statistics (Kruskal-Wallis one-way ANOVA on ranks) were used to reveal significant differences (P < 0.05) in FI. It was not appropriate to use ANOVA to compare FI when a large decrease occurred and a neurone stopped firing or fired only a few spikes. In these cases (15 of 144 comparisons), where FI decreased by over an order of magnitude, it was obvious that the unit had stopped firing during micturition and the change was deemed significant. Statistical tests were performed using Sigma Stat software (SPSS Science, Chicago, IL) and equations were provided by Zar (1999).
RESULTS
Sacral neurones in the dorsal grey commissure with sensory pudendal input (DGC-SPud neurones)
In 16/17 DGC-SPud neurones examined, the frequency of DL-H evoked action potentials was significantly modulated during one or more periods (i.e. bladder contraction, EUS inhibition and expulsion periods) of the micturition reflex. Firing changes that extended over several portions of the void were always of the same nature (decrease or increase) although they may vary somewhat in the magnitude throughout the reflex (usually largest during the expulsion phase). The results were similar for both PMC and distension-evoked voiding and the data were pooled.
Decreases in FI during micturition (Table 1) were most common (n = 11), but increases (n = 3) and more complex patterns of modulation (n = 2) were also observed. The firing of only one DGC-SPud neurone was unchanged during micturition. Decreases in FI ranged from 7 % to the nearly complete inhibition shown in Fig. 1 and Fig. 2. In 9/16 cutSPud neurones, FI decreases were observed during all three periods of the micturition reflex. In 2/16 neurones this was not the case; the FI decreased during the bladder contraction and expulsion period in one neurone and during the expulsion period and EUS ENG inhibition in the other. In those cells in which FI decreased during micturition, the FI following the micturition reflex was significantly increased above pre-void values in seven neurones, remained unchanged in one neurone, and was depressed below pre-void levels in three neurones. The modulation of two DGC-SPud neurones was more complex than a simple decrease or increase in FI during micturition. The FI of both neurones decreased during the period of EUS inhibition when the bladder was actively contracting but the FI returned to, or exceeded, pre-void levels prior to the termination of the bladder contraction (Fig. 3). In both neurones, firing returned when EUS ENG activity resumed. However, in one neurone (Fig. 3) where the EUS ENG activity began to diminish ≈40 s prior to the bladder contraction, the firing of the neurone did not decrease until a large micturition reflex bladder contraction occurred. The absence of a close association between neurone FI and EUS ENG activity is also evident in Fig. 2 where neurone firing remained suppressed following the return of activity in the EUS ENG. Compared to pre-void values, post-void FI was higher in one neurone and lower in the other.
Table 1.
Normalized changes in the F1 of sacral neurones during micturition
| Pre-void (%) | EUS inhibition (%) | Bladder contraction (%) | Stream (%) | Post-void (%) | |
|---|---|---|---|---|---|
| DGC-SPud neurones | |||||
| Inhibited (11/17) | 1 ± 90 | 0.48 ± 160 | 0.55 ± 190 | 0.40 ± 430 | 1.3 ± 170 |
| Excited (3/17) | 1 ± 140 | 6.7 ± 140 | 4.3 ± 120 | 4.5 ± 120 | 0.75 ± 1200 |
| Group II neurones | |||||
| Inhibited (4/5) | 1 ± 93 | 0.93 ± 87 | 0.90 ± 93 | 0.88 ± 90 | 0.93 ± 140 |
Median and coefficient of variation of the median values have been normalized to pre–void values and averaged (mean).
Figure 1. Decrease in the instantaneous firing frequency of a sacral DGC-SPud neurone during distension-evoked micturition.
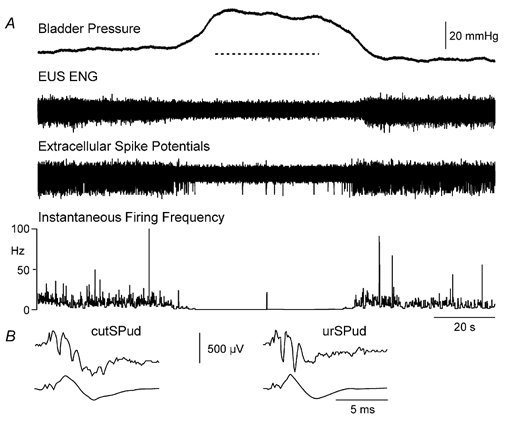
A, micturition was characterized by a bladder contraction that increased the intravesical pressures sufficiently to expel a stream (indicated with dashed line) of fluid from the urethra. During the micturition reflex, the EUS ENG efferent activity decreased. A decrease in evoked firing of the neurone is evident in the extracellular unit recording, however, because the traces are highly compressed, individual spikes are not discernable and firing changes are better illustrated by the transformed instantaneous firing frequency values. B, averages of the extracellular recording (top) and cord dorsum (lower trace) showing the activation by cutSPud and urSPud (stimulation strength 5T) at latencies of 1.4 and 1.3 ms, respectively. The amplitude calibration in B is for the extracellular spike potential in A and B.
Figure 2. Decrease in the instantaneous firing frequency of a DGC-SPud neurone during PMC-evoked micturition.
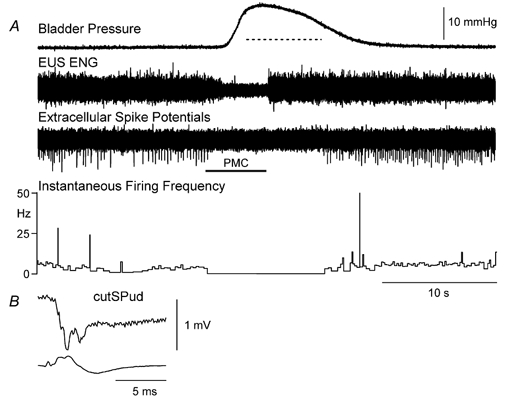
A, the evoked micturition reflex occurred within seconds of the brainstem stimulation (160 μA, 50 Hz, 0.5 ms pulses). In this case, the EUS activity returned at the termination of stimulation even though the reflex bladder contraction and suppression of neurone firing persisted for seconds more. B, this neurone (averaged extracellular record, upper trace) was activated by cutSPud (5T stimulation strength) at a central latency of 2.0 ms; lower trace cord dorsum record. The amplitude calibration in B is for the extracellular spike potential in A and B.
Figure 3. Decrease in a DGC-SPud neurone firing was not linked to changes in EUS activity during distension-evoked micturition.
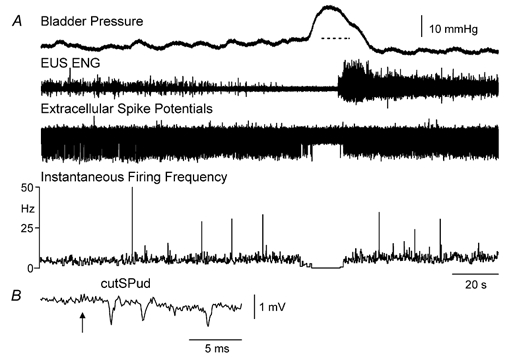
A, during bladder filling, just prior to the micturition reflex, EUS ENG activity began to decrease but the unit firing remained unchanged. At the time of the micturition reflex, EUS activity was completely suppressed as was activity in the neurone. B, the averaged extracellular record of the neurone shows multiple spikes in response to a single stimulus (arrow represents the point of the volley arrival at the cord dorsum) with stimulation of cutSPud at 5T stimulation strengths. The amplitude calibration in B is for the extracellular spike potential in A and B.
Three of the 16 modulated neurones showed an increase in FI ranging from 30-1000 % during micturition (Fig. 4; Table 1). In two of these neurones, FI significantly increased during the bladder contraction, EUS inhibition and expulsion periods, whereas in the other neurone the increase was only significant during the bladder contraction. The FI following micturition remained significantly elevated above pre-void levels in one neurone, was unchanged in another, and was depressed below pre-void levels in the third.
Figure 4. Increased firing in a DGC-SPud neurone during distension-evoked micturition.
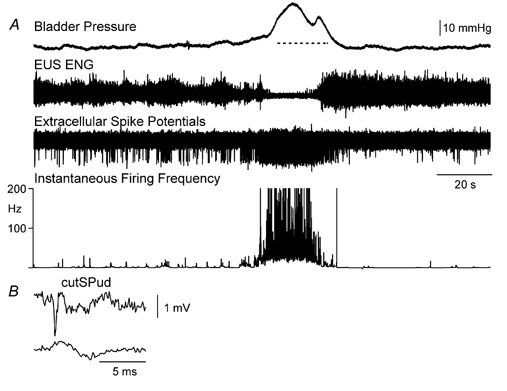
A, during micturition, the neurone was recruited and fired a burst of action potentials starting just before the bladder pressure and EUS ENG changes and ending with the return of EUS activity at the end of the stream. B, this neurone was excited by cutSPud (5T stimulation strength) at a central latency of 1.3 ms. The amplitude calibration in B is for the extracellular spike potential in A and B.
The sacral DGC-SPud neurones were activated by cutSPud as well as urSPud (6/17), SFP (6/17) and CCF (3/17) afferents, but not by group I or II muscle afferents (5T PBST or GS). Although always excited by cutSPud afferents, 7/17 of these neurones were activated at shorter latencies by urethral or cutaneous afferents (i.e. urSPud, SFP or CCF). The shortest latencies to activation in an individual neurone from 5T stimulation of cutSPud, urSPud, SFP or CCF nerves was 1.0 ms but could be as long as 2.6 ms (median = 1.7 ms; mean = 1.7 ± 0.4 ms, mean ± S.D.). The central latency to activation was not related to the change in neurone firing during micturition. For instance, the firing of neurones activated at presumably monosynaptic latencies (< 1.3 ms) could decrease or increase during micturition. Whether neurone behaviour was related to a source of afferent input could not be definitively established because of the limited number of observations. The largest sample of neurones, those displaying a decrease in FI during micturition, were activated by one, two, or three cutaneous afferent nerves. Spike-triggered averages from spikes evoked in all 17 DGC-Spud neurones tested did not reveal associated changes in dorsal root filament, EUS or EAS ENG recordings.
Rostral sacral group II neurones
Previously, Jankowska & Riddell (1993; 1994; 1995) described a population of interneurones located in the deep dorsal horn in the rostral sacral spinal cord which were strongly excited by group II muscle afferents as well as pudendal sensory afferents. This population was the target of the present investigation. Cells in this category were activated by stimulation of PBST and/or GS nerves at group II strengths (Fig. 5C). Group II neurones were modulated during micturition but the magnitude of change (5-15 %) was far less than that observed in the more caudal population of DGC-SPud neurones described above. The average decrease in FI was approximately 20 % of that observed in the inhibited DGC-SPud neurones (Table 1). The FI of 4/5 neurones decreased significantly during one or more periods of the micturition reflex. In two neurones the FI significantly decreased during the bladder contraction, EUS ENG inhibition, and expulsion periods. In one neurone decreases were only observed during the bladder contraction and expulsion period and in the other neurone only during the expulsion period. The FI of one neurone significantly increased (4 %) during all periods of the micturition reflex. The post-void FI was depressed below pre-void levels in two neurones, did not significantly change in one neurone, and increased above pre-void levels in two neurones.
Figure 5. Changes in firing in a sacral group II neurone during a distension-evoked void.
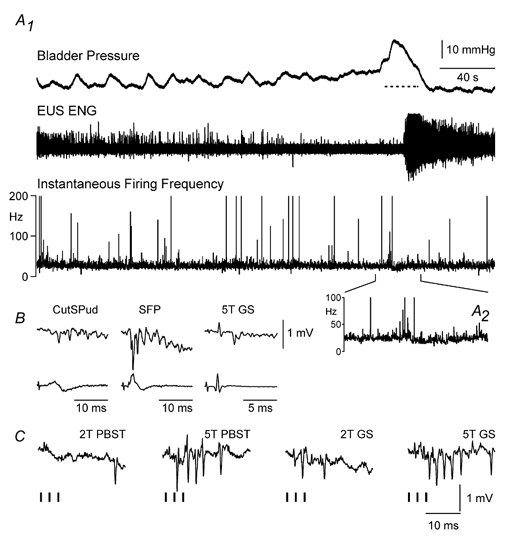
A, the small fluctuation in neurone firing that occurred during the bladder contraction can be seen in A1 and enlarged in A2. These small changes, although statistically significant, were of a much smaller magnitude than the decreases observed in the DGC-SPud neurones. B, averaged records of the interneurone response to single stimuli of cutaneous (CutSPud, SFP; central latencies 3.0 and 1.3 ms, respectively), and GS group II muscle afferents (central latency 2.3 ms). C, single evoked responses show that the neurone was weakly activated by trains of three shocks (indicated by vertical bars below the trace) to PBSt and GS muscle afferents at 2T (no response < 2T) and strongly excited by stimulation which recruited most group II muscle afferents (5T).
Group II neurones were activated by 5T stimulation of PBSt and/or GS nerves at central latencies ranging from 2.2-3.5 ms. All group II neurones were also activated by cutSPud afferents and some were excited by SFP (4/5) or CCF (3/5). Three of five group II neurones were activated at a shorter central latency (1.3-1.5 ms) by cutaneous afferents (SFP) than by group II PBSt or GS muscle afferents (i.e. > 2 ms; Fig. 5). As in the DGC-SPud neurones, group II neurone behaviour during micturition was not related to the central latency from afferent activation. Similar to the neurone that displayed a decrease in FI during micturition (Fig. 5), the neurone that displayed an increase in FI during micturition was activated by group II muscle afferents (latencies of 2.2 ms for GS and 3.3 ms for PBSt) as well as cutSPud, SFP and CCF afferents (at latencies of 3.5, 1.3 and 2.5 ms, respectively). As described above, for the more caudal DGC-SPud neurones, the sample of rostral sacral group II neurones were not likely neurones mediating actions directly on primary afferents nor on sphincter motoneurones since spike-triggered averages of dorsal root filament, EUS or EAS ENG recordings failed to reveal responses associated with the group II neurone spiking.
DISCUSSION
In the present study, changes in the rate of tonic evoked firing of a neurone during micturition were interpreted as changes in the excitability of the postsynaptic membrane of that neurone. During micturition, the excitability of the majority of DGC-SPud neurones and group II neurones examined in this study decreased, with only a small fraction of the sampled neurones in the DGC-SPud sample displaying an increase in firing during micturition. Since the DL-H-evoked firing of the neurones was set to be below the maximum rates possible for a given cell, we were able to detect both decreases and increases in excitability based on neurone instantaneous firing frequency. Indeed, in 55 % of the neurones, post-void FI was elevated above pre-void levels, potentially contributing to post-void EUS rebound excitation (Buss & Shefchyk, 1999). Furthermore, the pattern of firing change during the micturition reflex was similar for both distension-evoked and brainstem-evoked reflexes, consistent with what has been seen previously when comparisons of events or changes occurring during reflexes evoked by both methods are made (Angel et al. 1994; Fedirchuk et al. 1994; Buss & Shefchyk, 1999).
We hypothesize that the sacral neurones in the dorsal grey commissure that displayed a decrease in excitability during micturition were excitatory neurones interposed in polysynaptic reflex pathways from perineal and pudendal afferents to sphincter motoneurones (Fedirchuk et al. 1992a). Furthermore, we propose that these cells are subject to a direct postsynaptic inhibition during micturition. The decreased firing coincides with the period of maximum suppression of excitatory activation and output from the sphincter motoneurones during the expulsion of urine when the bladder pressure is highest and the sphincter activity lowest. However, the removal of either a tonic excitatory synaptic drive or an intrinsic depolarizing conductance in the neurone that could lead to a decrease in excitability cannot be discounted at this time. Whatever the mechanism, the excitability decrease would act to attenuate transmission through excitatory sacral afferent pathways to EUS motoneurones during voiding.
The neurones examined in the present study were located in a region where the shortest latency field potentials from cutaneous afferents are found, which supports the suggestion that some are probably first-order neurones. The shortest central latency to activation of DGC-SPud neurones was 1.7 ± 0.4 ms (mean ± S.D.). The delay from the incoming volley to the onset of the field is 0.6-0.9 ms for SFP and SPud (Fedirchuk et al. 1992b) and with this minimum delay there is an added delay to reach spike threshold. These latencies are consistent with the neurones being monosynaptically or disynaptically activated from the primary afferents. Central latencies of the sensory pudendal and perineal afferent-evoked EPSPs in EUS motoneurones are 3.8 ± 1 ms, SPud and 2.9 ± 1 ms, SFP (Fedirchuk et al. 1992a), leaving sufficient delay for one or two interneurones to form disynaptic but mainly trisynaptic reflex pathways from the afferents to the motoneurones. Spike-triggered averages did not reveal responses in the EUS or EAS ENGs as would be expected for last-order interneurones projecting to the sphincter motoneurones.
The neurones excited during micturition may be the neurones producing the glycinergic inhibition of EUS motoneurones or the presumed GABAergic neurones responsible for primary afferent depolarization of perineal cutaneous afferents during micturition (Shefchyk, 2001). The GABA and/or glycine-expressing dorsal grey commissure neurones (Blok et al. 1997; Sie et al. 2001), which receive direct inputs from the PMC (Blok et al. 1997) and when stimulated decrease urethral pressure (Blok et al. 1998), could potentially mediate the postsynaptic inhibition of EUS motoneurones or sacral neurones examined in this study. However, because the decreases in excitability observed in some sacral DGC-SPud neurones did not strictly coincide with the inhibition of EUS efferents, it is unlikely that one population of inhibitory interneurones mediates the postsynaptic inhibition of both the motoneurones and the neurones examined in the present study. The DGC-SPud neurones examined in this study were not spontaneously active in the absence of DL-H, which is consistent with a tonic inhibition of these neurones in decerebrate animals but alternatively, they may just not be active at rest. The excitability increase observed in a small fraction of the neurones examined could be due to an increased synaptic drive from the micturition circuitry or to the removal of a tonic inhibition. The excited neurones could be first-order interneurones (central latency from afferent activation 1.5-2.0 ms) which, during continence, are subject to a tonic inhibition that is removed during micturition. For those neurones receiving inputs from urethral afferents, the removal of a tonic inhibition would explain the state-dependent actions of urethral afferents which excite EUS motoneurones during continence but facilitate bladder emptying during micturition. This would permit their activation by urethral afferents and the expression of the urethral-bladder reflex (Barrington, 1914; Kiruluta et al. 1981; Mazieres et al. 1997; Shefchyk & Buss, 1998).
Changes in neurone excitability were most commonly observed during the stream phase of micturition. As urethral afferents are activated during the stream, it is possible that these afferents excite inhibitory inter-neurones that act to depress excitatory pathways to sphincter motoneurones during micturition. In this study, the ipsilateral urSPud nerve was cut, thus reducing urethral afferent actions. The remaining intact urethral afferents may have contributed to the central micturition circuitry-evoked neurone excitability changes observed during the stream period of the bladder contraction.
DGC-SPud neurones underwent larger excitability changes than group II neurones. The small but significant decrease in firing in 4/5 rostral sacral neurones was observed in neurones di- and polysynaptically activated by group II afferents. They are probably not last-order neurones projecting to the sphincter motoneurones since spike-triggered averaging failed to reveal responses in the EUS or EAS ENGs and previous work has shown that group II proprioceptive information from hindlimb muscles (e.g. PBST and GS) do not have strong actions on sacral sphincter motoneurones (Fedirchuk et al. 1992a). The small decrease in the firing of group II neurones may not reflect a specific postsynaptic inhibition but rather the removal of a tonic excitatory drive from primary afferents by presynaptic inhibition (Buss & Shefchyk, 1999). The inhibition of group II interneurones during micturition may function to reduce the gain of spinal pathways related to postural adjustments during micturition, in a similar manner to decreases in muscle afferent transmission observed during locomotion (Perreault et al. 1999).
Based on the observations presented, it is hypothesized that sacral afferent reflex pathways to sphincter motoneurones are not only suppressed through direct motoneurone inhibition and presynaptic inhibition of afferents as previously described (Shefchyk, 2001), but that sacral neurones interposed in excitatory sacral reflex pathways may also receive direct inhibition from the central micturition circuitry and/or urethral afferents activated during micturition. Similar to that which has been described during locomotion (Perreault et al. 1999), there appear to be central mechanisms recruited to modulate sensory feedback during micturition, perhaps reflecting a basic principle essential for controlling unwanted reflexes during motor function. As for the population of neurones that displayed an enhanced excitability during micturition, these may be the inhibitory neurones just discussed, or they may be neurones interposed in pathways to the parasympathetic bladder preganglionic neurones. These possibilities remain the subject of future experiments.
Acknowledgments
Shannon Deschamps, Sharon McCartney and Maria Setterbom provided technical assistance and Drs B. Fedirchuk and D. McCrea provided helpful discussion. This work was supported by grants from the Canadian Institutes of Health Research and the National Institutes of Health (R. R. B.'s salary).
REFERENCES
- Angel MJ, Fyda D, McCrea DA, Shefchyk SJ. Primary afferent depolarization of cat pudendal afferents during micturition and segmental afferent stimulation. J Physiol. 1994;479:451–461. doi: 10.1113/jphysiol.1994.sp020309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barrington FJF. The nervous mechanism of micturition. Q J Exp Physiol. 1914;8:33–71. [Google Scholar]
- Bishop B. Reflex activity of external anal sphincter of cat. J Neurophysiol. 1959;22:679–692. doi: 10.1152/jn.1959.22.6.679. [DOI] [PubMed] [Google Scholar]
- Blok BF, De Weerd H, Holstege G. The pontine micturition center projects to sacral cord GABA immunoreactive neurons in the cat. Neurosci Lett. 1997;233:109–112. doi: 10.1016/s0304-3940(97)00644-7. [DOI] [PubMed] [Google Scholar]
- Blok BF, Van Maarseveen JT, Holstege G. Electrical stimulation of the sacral dorsal gray commissure evokes relaxation of the external urethral sphincter in the cat. Neurosci Lett. 1998;249:68–70. doi: 10.1016/s0304-3940(98)00382-6. [DOI] [PubMed] [Google Scholar]
- Buss RR, Shefchyk SJ. Excitability changes in sacral afferents innervating the urethra, perineum and hindlimb skin of the cat during micturition. J Physiol. 1999;514:593–607. doi: 10.1111/j.1469-7793.1999.593ae.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coonan EM, Downie JW, Du HJ. Sacral spinal cord neurons responsive to bladder pelvic and perineal inputs in cats. Neurosci Lett. 1999;260:137–40. doi: 10.1016/s0304-3940(98)00970-7. [DOI] [PubMed] [Google Scholar]
- de Groat WC, Vizzard MA, Araki I, Roppolo J. Spinal interneurons and preganglionic neurons in sacral autonomic reflex pathways. Prog Brain Res. 1996;107:97–111. doi: 10.1016/s0079-6123(08)61860-9. [DOI] [PubMed] [Google Scholar]
- Fedirchuk B, Downie JW, Shefchyk SJ. Reduction of perineal evoked excitatory postsynaptic potentials in cat lumbar and sacral motoneurons during micturition. J Neurosci. 1994;14:6153–6159. doi: 10.1523/JNEUROSCI.14-10-06153.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fedirchuk B, Hochman S, Shefchyk SJ. An intracellular study of perineal and hindlimb afferent inputs onto sphincter motoneurons in the decerebrate cat. Exp Brain Res. 1992a;89:511–516. doi: 10.1007/BF00229875. [DOI] [PubMed] [Google Scholar]
- Fedirchuk B, Shefchyk SJ. Effects of electrical stimulation of the thoracic spinal cord on bladder and external urethral sphincter activity in the decerebrate cat. Exp Brain Res. 1991;84:635–642. doi: 10.1007/BF00230976. [DOI] [PubMed] [Google Scholar]
- Fedirchuk B, Song L, Downie JD, Shefchyk SJ. Spinal distribution of extracellular field potentials generated by electrical stimulation of pudendal and perineal afferents in the cat. Exp Brain Res. 1992b;89:517–520. doi: 10.1007/BF00229876. [DOI] [PubMed] [Google Scholar]
- Geirsson G, Fall M. Reflex interaction between the proximal urethra and the bladder. Scand J Urol Nephrol. 1999;33:24–26. doi: 10.1080/003655999750016221. [DOI] [PubMed] [Google Scholar]
- Jankowska E, Bichler E, HammaR I. Areas of operation of interneurons mediating presynaptic inhibition in sacral spinal segments. Exp Brain Res. 2000;133:402–406. doi: 10.1007/s002210000429. [DOI] [PubMed] [Google Scholar]
- Jankowska E, Riddell JS. A relay for input from group II muscle afferents in sacral segments of the cat spinal cord. J Physiol. 1993;465:561–580. doi: 10.1113/jphysiol.1993.sp019693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jankowska E, Riddell JS. Interneurones in pathways from group II muscle afferents in sacral segments of the feline spinal cord. J Physiol. 1994;475:455–468. doi: 10.1113/jphysiol.1994.sp020085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jankowska E, Riddell JS. Interneurones mediating presynaptic inhibition of group II muscle afferents in the cat spinal cord. J Physiol. 1995;483:461–471. doi: 10.1113/jphysiol.1995.sp020597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jankowska E, Riddell JS, Szabo-Lackberg Z, Hammar I. Morphology of interneurones in pathways from group II muscle afferents in sacral segments of the cat spinal cord. J Comp Neurol. 1993;337:518–528. doi: 10.1002/cne.903370312. [DOI] [PubMed] [Google Scholar]
- Kiruluta HG, Downie JW, Awad SA. The continence mechanisms: the effect of bladder filling on the urethra. Invest Urol. 1981;18:460–465. [PubMed] [Google Scholar]
- Mackel R. Segmental and descending control of the external urethral and anal sphincters in the cat. J Physiol. 1979;294:105–122. doi: 10.1113/jphysiol.1979.sp012918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McMahon SB, Morrison JFB. Spinal neurones with long projections activated from the abdominal viscera of the cat. J Physiol. 1982a;322:1–20. doi: 10.1113/jphysiol.1982.sp014018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McMahon SB, Morrison JFB. Two group of spinal interneurones that respond to stimulation of the abdominal viscera of the cat. J Physiol. 1982b;322:21–34. doi: 10.1113/jphysiol.1982.sp014019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mazieres L, Jiang C, Lindstrom S. Bladder parasympathetic response to electrical stimulation of urethral afferents in the cat. Neurourol Urodyn. 1997;16:471–472. [Google Scholar]
- Nadelhaft I, Vera PL. Neurons in the rat brain and spinal cord labeled after pseudorabies virus injected into the external urethral sphincter. J Comp Neurol. 1996;375:502–17. doi: 10.1002/(SICI)1096-9861(19961118)375:3<502::AID-CNE11>3.0.CO;2-N. [DOI] [PubMed] [Google Scholar]
- Perreault M-C, Shefchyk SJ, Jimenez I, McCrea D. Depression of monosynaptic field potentials evoked by muscle and cutaneous afferents during fictive locomotion in the cat. J Physiol. 1999;521:691–703. doi: 10.1111/j.1469-7793.1999.00691.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shefchyk SJ. Sacral spinal interneurones and the control of urinary bladder and urethral striated sphincter muscle function. J Physiol. 2001;533:57–63. doi: 10.1111/j.1469-7793.2001.0057b.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shefchyk SJ, Buss RR. Urethral pudendal afferent-evoked bladder and sphincter reflexes in decerebrate and acute spinal cats. Neurosci Lett. 1998;244:137–140. doi: 10.1016/s0304-3940(98)00155-4. [DOI] [PubMed] [Google Scholar]
- Shefchyk SJ, Epsey MJ, Carr P, Nance D, Sawchuk M, Buss R. Evidence for a strychnine-sensitive mechanism and glycine receptors involved in the control of urethral sphincter activity during micturition in the cat. Exp Brain Res. 1998;119:297–306. doi: 10.1007/s002210050345. [DOI] [PubMed] [Google Scholar]
- Shimoda N, Takakusaki K, Nishizawa O, Tsuchida S, Mori S. The changes in the activity of pudendal motoneurons in relation to reflex micturition evoked in decerebrate cats. Neurosci Lett. 1992;135:175–178. doi: 10.1016/0304-3940(92)90430-f. [DOI] [PubMed] [Google Scholar]
- Sie JA, Blok BF, De Weerd H, Holstege G. Ultrastructural evidence for direct projections from the pontine micturition center to glycine-immunoreactive neurons in the sacral dorsal gray commissure in the cat. J Comp Neurol. 2001;429:631–637. doi: 10.1002/1096-9861(20010122)429:4<631::aid-cne9>3.0.co;2-m. [DOI] [PubMed] [Google Scholar]
- Thor KB, Morgan C, Nadelhaft I, Houston M, De Groat WC. Organization of afferent and efferent pathways in the pudendal nerve of the female cat. J Comp Neurol. 1989;288:263–279. doi: 10.1002/cne.902880206. [DOI] [PubMed] [Google Scholar]
- Ueyama T, Mizuno N, Nomura S, Konishi A, Itoh K, Arakawa H. Central distribution of afferent and efferent components of the pudendal nerve in cat. J Comp Neurol. 1984;222:38–46. doi: 10.1002/cne.902220104. [DOI] [PubMed] [Google Scholar]
- Zar JH. Biostatistical Analysis. Upper Saddle River, NJ, USA: Prentice Hall; 1999. [Google Scholar]


