Abstract
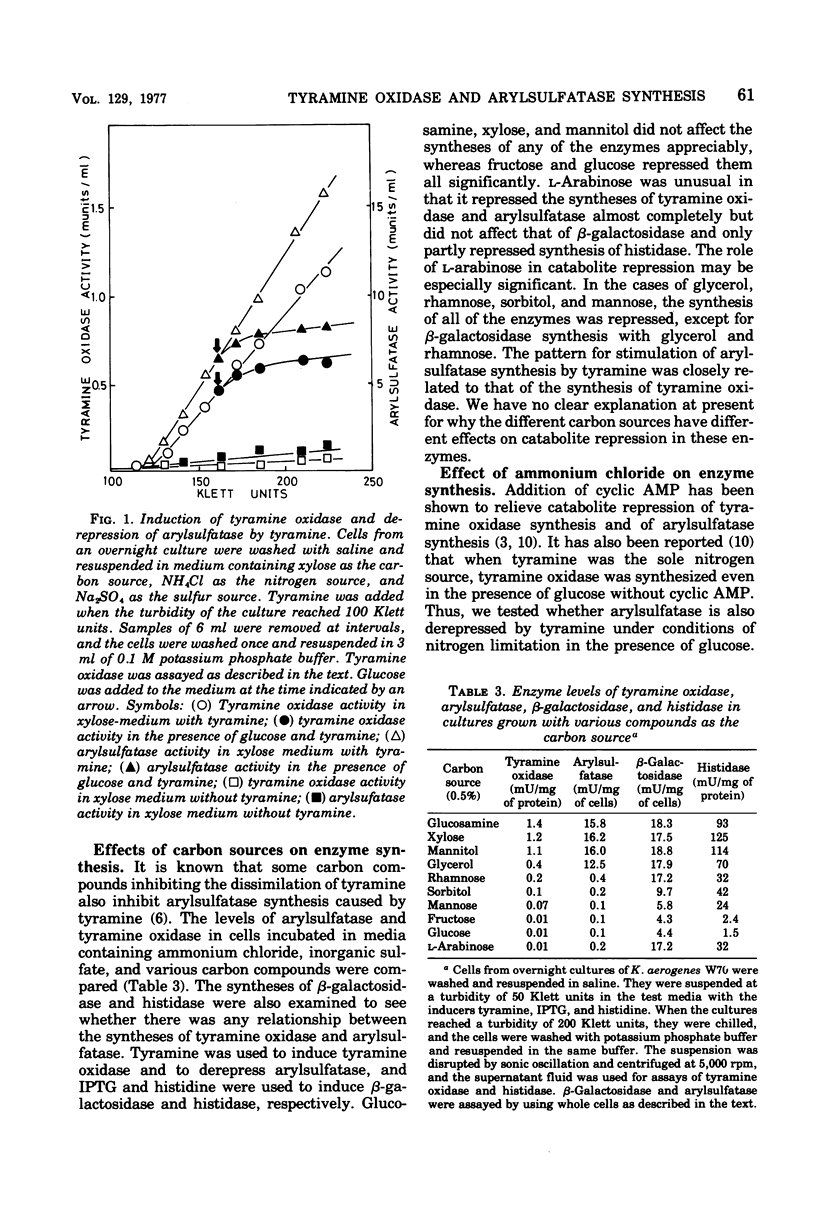
The participation of tyramine oxidase in the regulation of arylsulfatase synthesis in Klebsiella aerogenes was studied. Arylsulfatase was synthesized when this organism was grown with methionine or taurine as the sulfur source (nonrepressing conditions) and was repressed by inorganic sulfate or cysteine; this repression was relieved by tyramine and related compounds (derepressing conditions). Under nonrepressing conditions, arylsulfatase synthesis was not regulated by tyramine oxidase synthesis. However, derepression of arylsulfatase and induction of tyramine oxidase synthesis by tyramine were both antagonized by glucose and other carbohydrate compounds. The derepressed synthesis of arylsulfatase, like that of tyramine oxidase, was released from catabolite repression by use of tyramine as the sole source of nitrogen. A mutant strain that exhibits constitutive synthesis of glutamine synthetase and high levels of histidase when grown in glucose-ammonium medium was subject to the catabolite repression of both tyramine oxidase and arylsulfatase syntheses. Mutants in which repression of arylsulfatase could not be relieved by tyramine could not utilize tyramine as the sole source of nitrogen and were defective in the gene for tyramine oxidase.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adachi T., Murooka Y., Harada T. Derepression of arylsulfatase synthesis in Aerobacter aerogenes by tyramine. J Bacteriol. 1973 Oct;116(1):19–24. doi: 10.1128/jb.116.1.19-24.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Adachi T., Murooka Y., Harada T. Regulation of arylsulfatase synthesis by sulfur compounds in Klebsiella aerogenes. J Bacteriol. 1975 Jan;121(1):29–35. doi: 10.1128/jb.121.1.29-35.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Adachi T., Okamura H., Murooka Y., Harada T. Catabolite repression and derepression of arylsulfatase synthesis in Klebsiella aerogenes. J Bacteriol. 1974 Nov;120(2):880–885. doi: 10.1128/jb.120.2.880-885.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harada T., Spencer B. Repression and induction of arylsulphatase synthesis in Aerobacter aerogenes. Biochem J. 1964 Nov;93(2):373–378. doi: 10.1042/bj0930373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Okamura H., Murooka Y., Harada T. Regulation of tyramine oxidase synthesis in Klebsiella aerogenes. J Bacteriol. 1976 Jul;127(1):24–31. doi: 10.1128/jb.127.1.24-31.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prival M. J., Brenchley J. E., Magasanik B. Glutamine synthetase and the regulation of histidase formation in Klebsiella aerogenes. J Biol Chem. 1973 Jun 25;248(12):4334–4344. [PubMed] [Google Scholar]
- Prival M. J., Magasanik B. Resistance to catabolite repression of histidase and proline oxidase during nitrogen-limited growth of Klebsiella aerogenes. J Biol Chem. 1971 Oct 25;246(20):6288–6296. [PubMed] [Google Scholar]
- Smith G. R., Halpern Y. S., Magasanik B. Genetic and metabolic control of enzymes responsible for histidine degradation in Salmonella typhimurium. 4-imidazolone-5-propionate amidohydrolase and N-formimino-L-glutamate formiminohydrolase. J Biol Chem. 1971 May 25;246(10):3320–3329. [PubMed] [Google Scholar]


