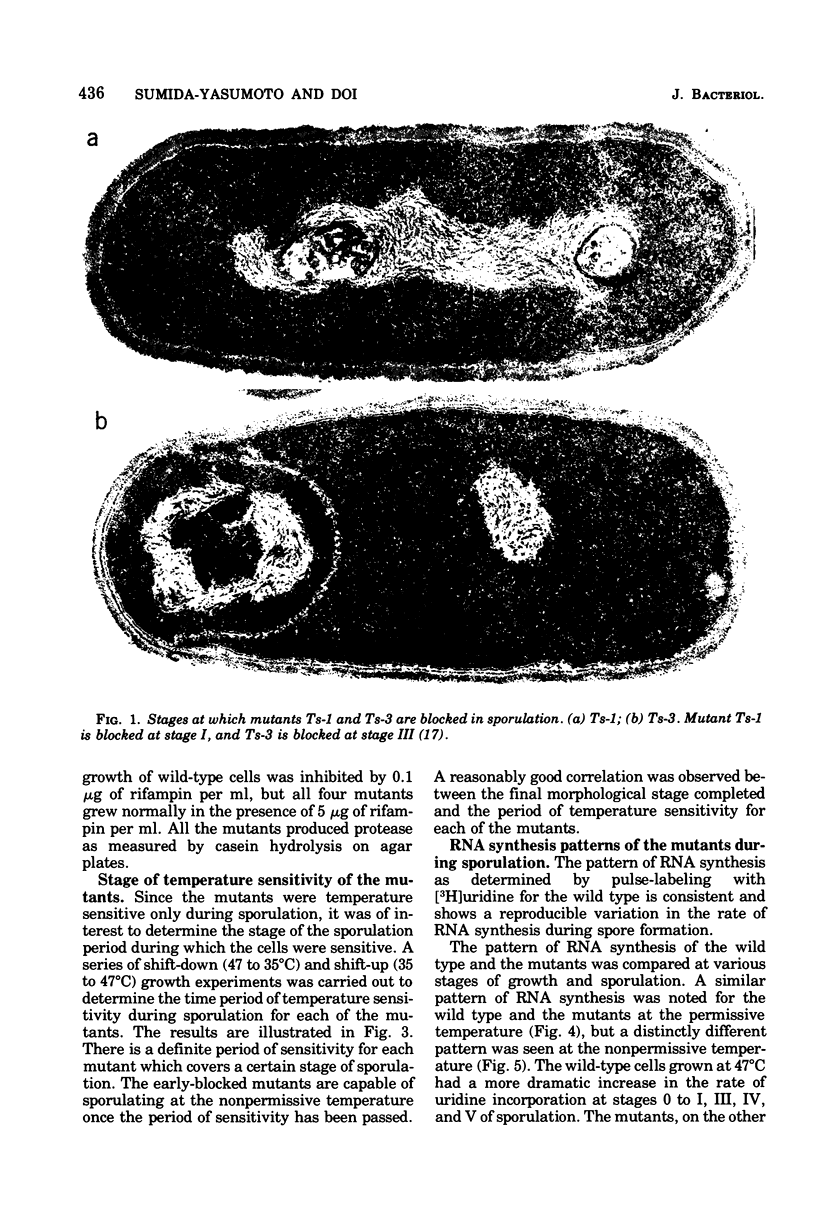
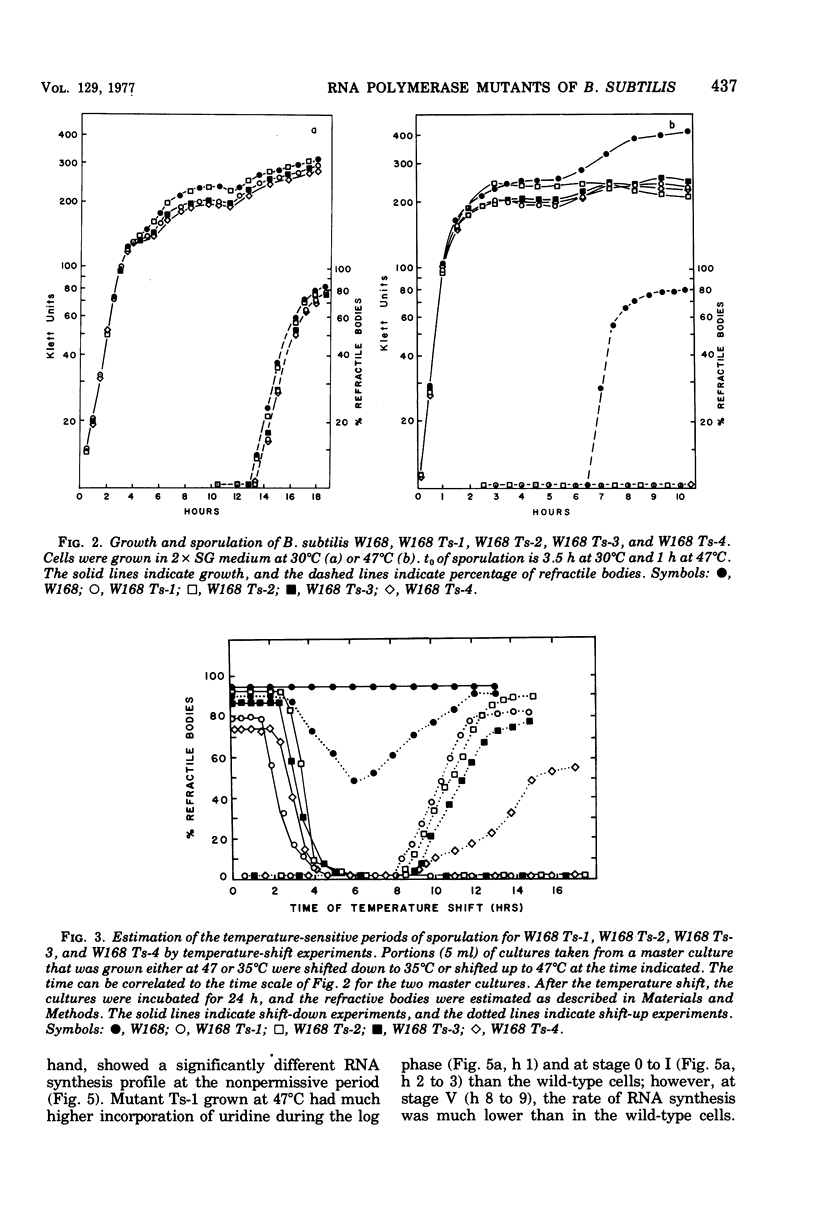
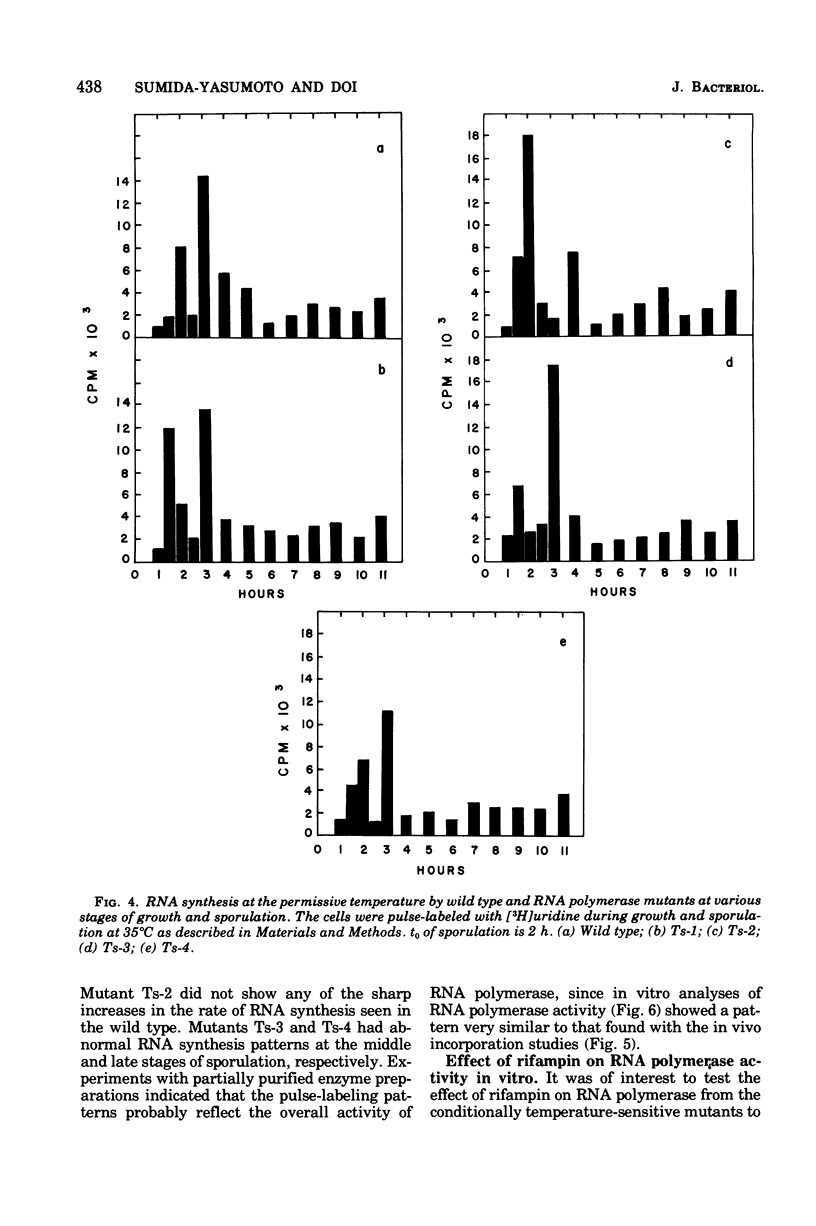
Abstract
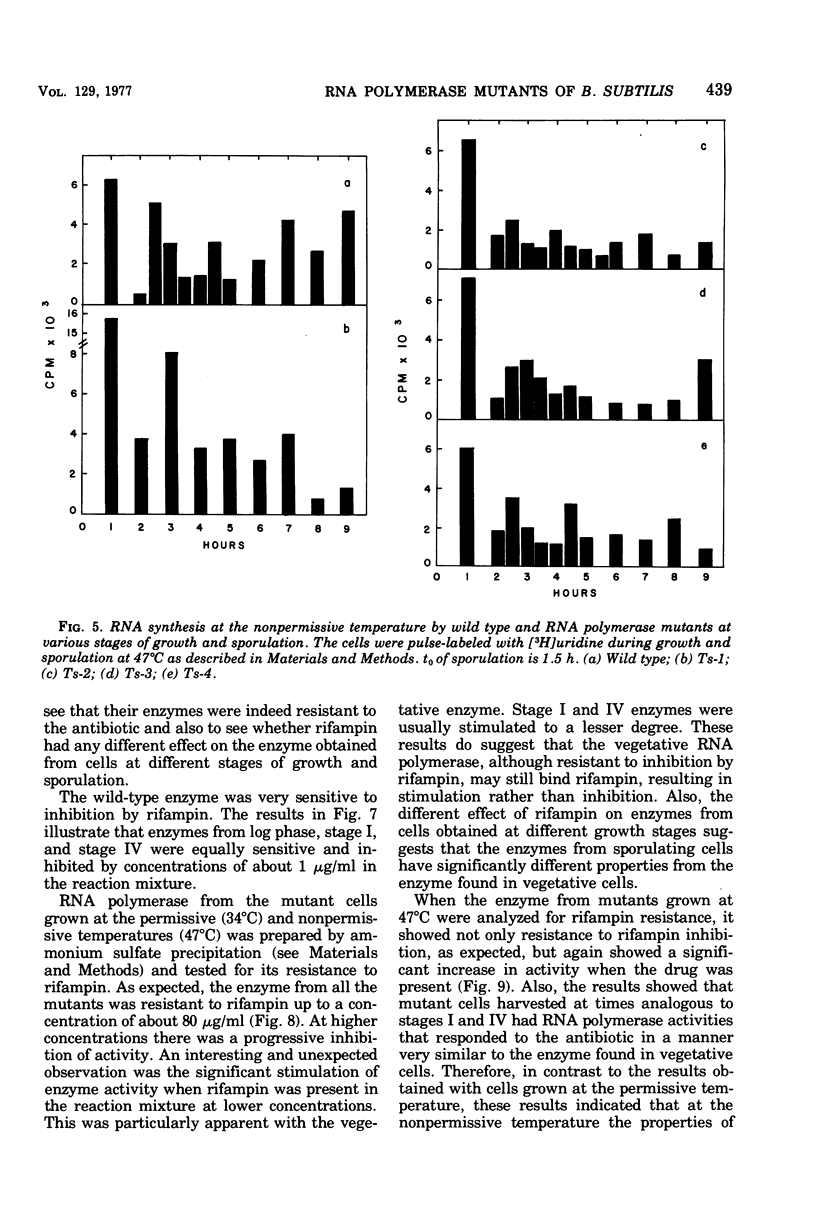
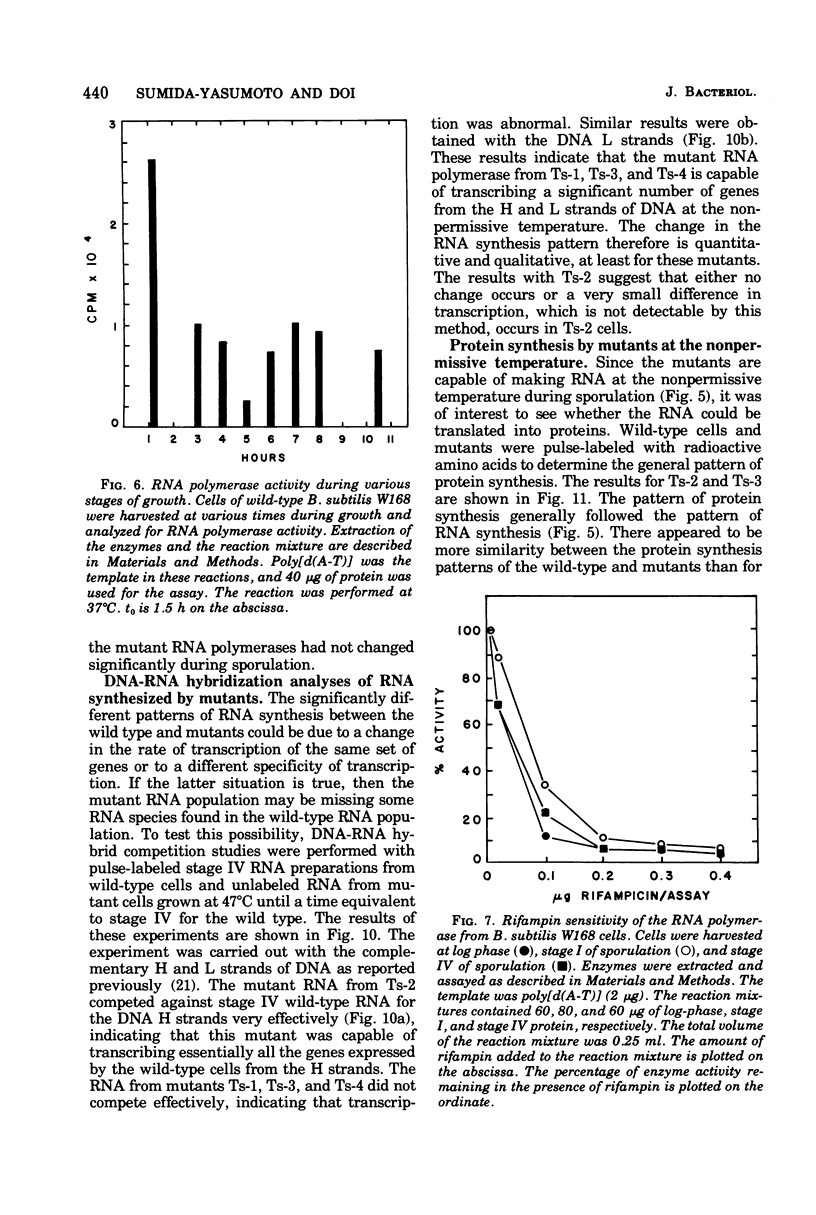
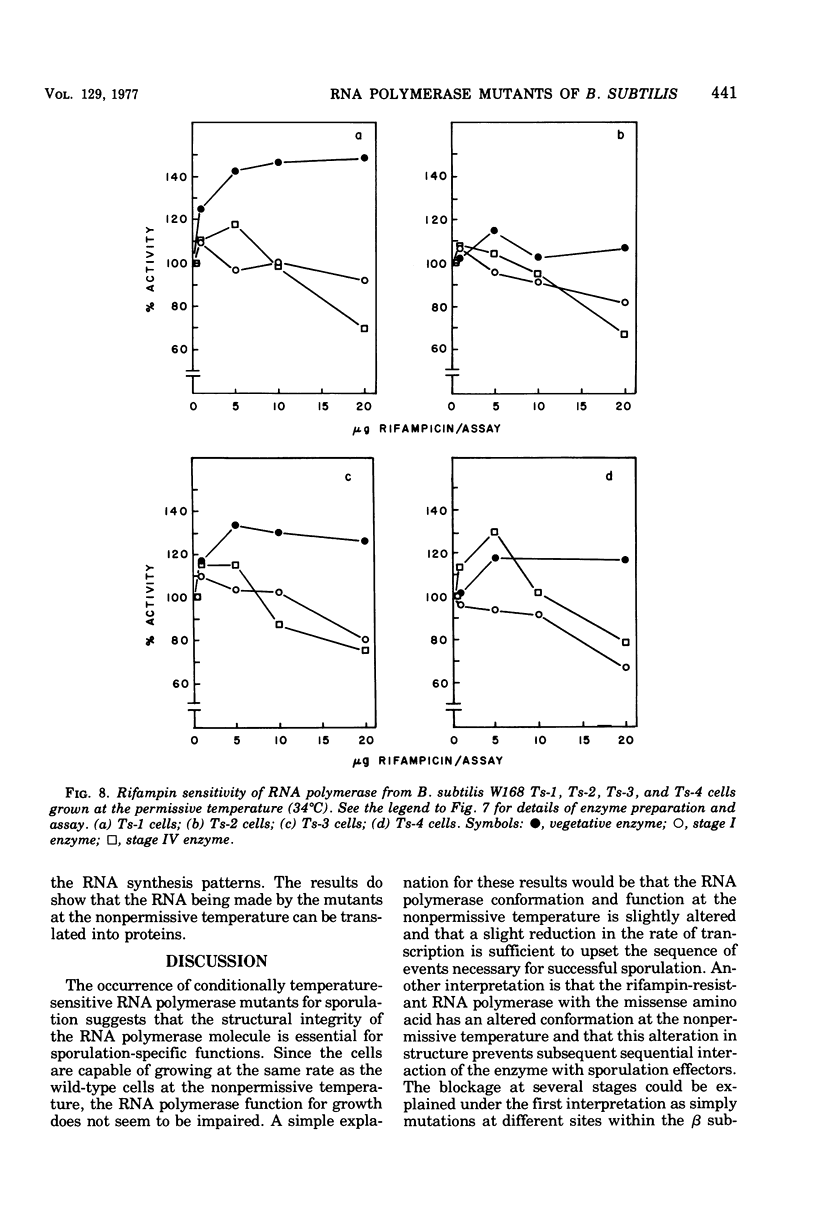
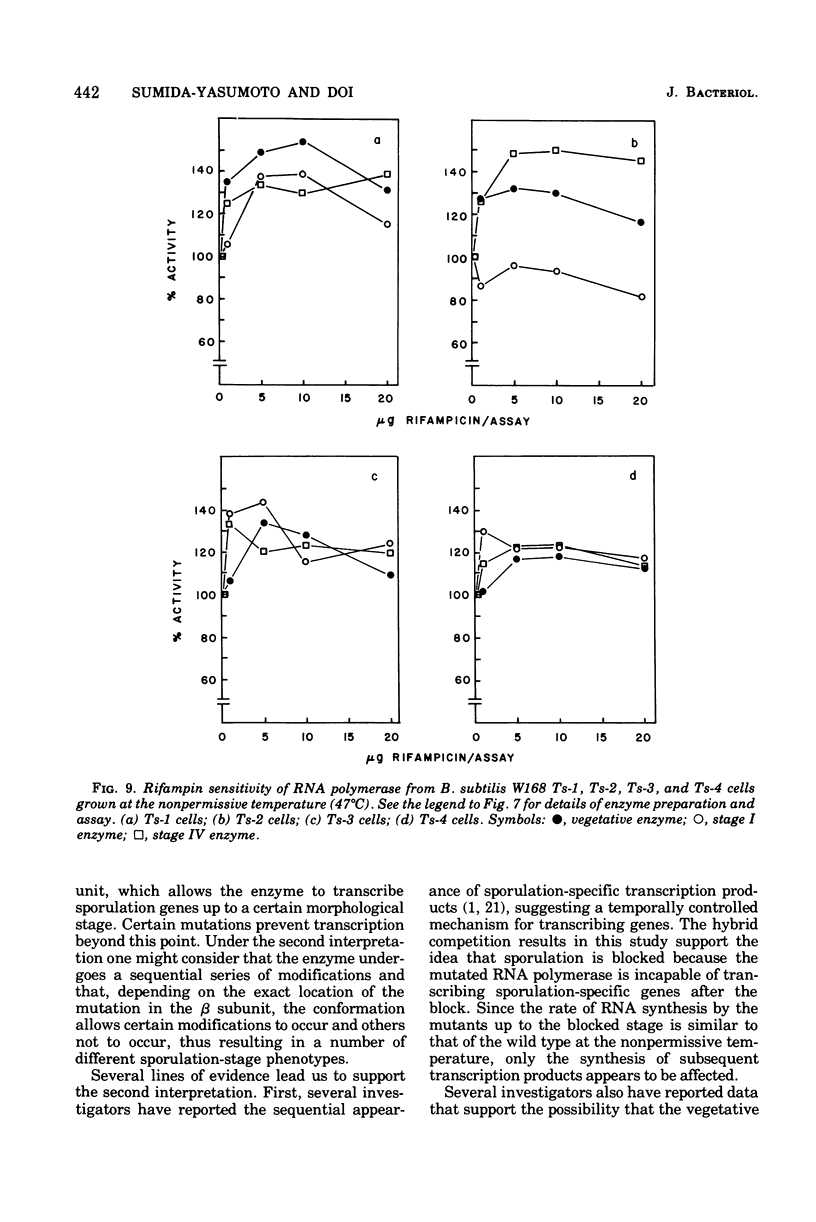
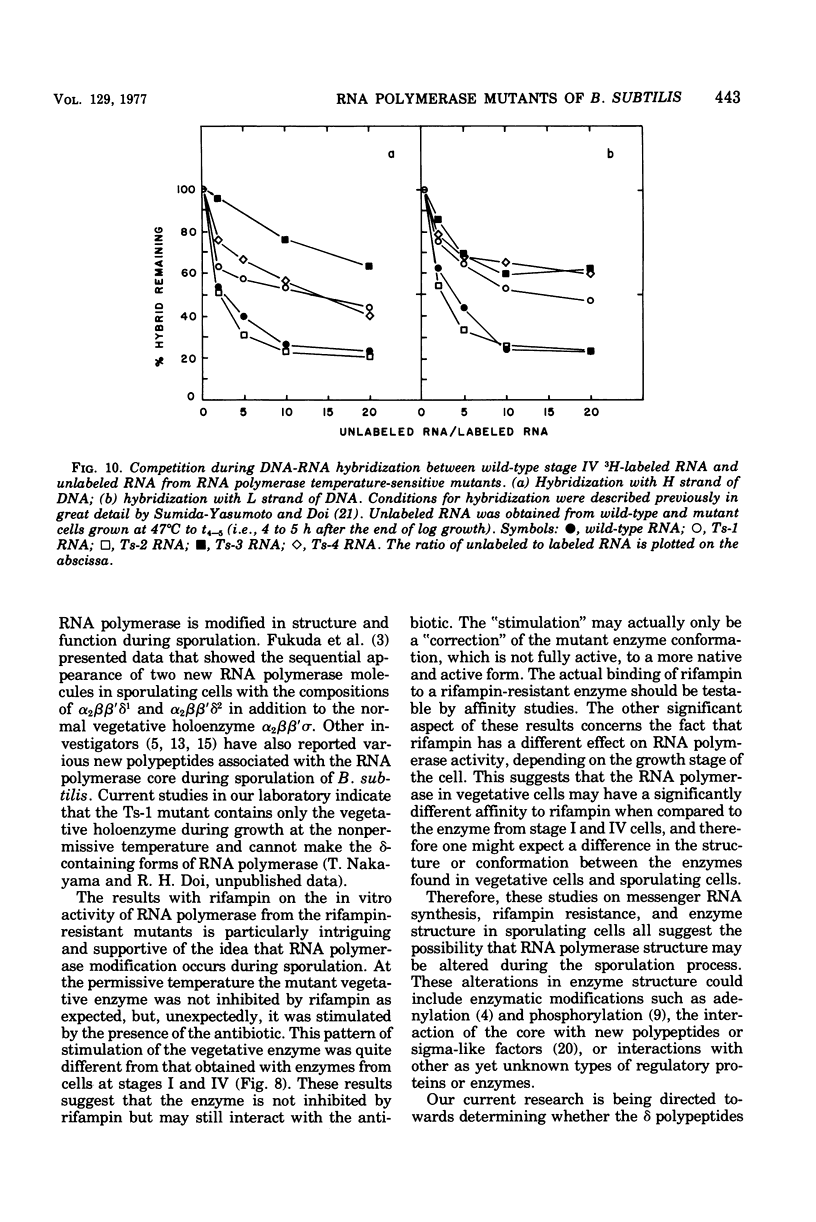
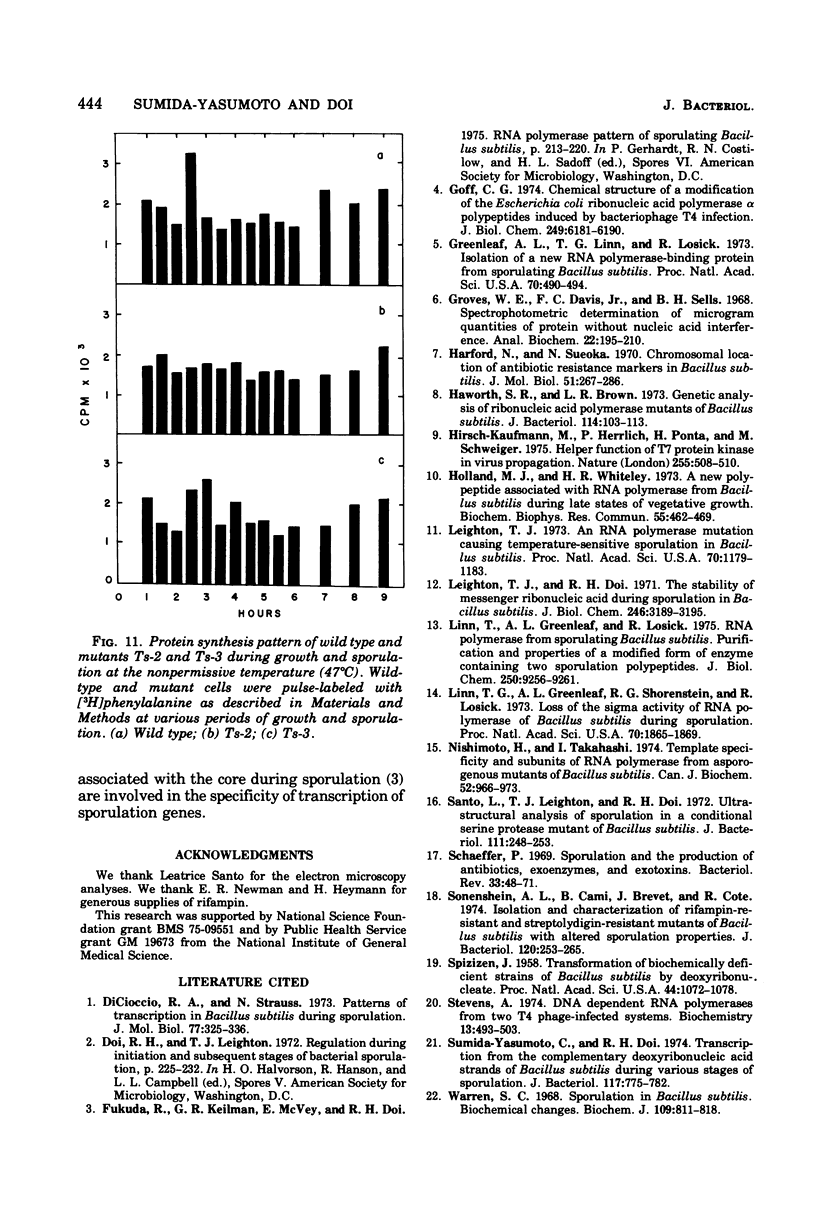
Rifampin-resistant mutants of Bacillus subtilis that are conditionally temperature sensitive during sporulation have been isolated and characterized. The mutants can grow at the same rate as the wild type at the nonpermissive temperature but cannot sporulate. Depending on the mutation, they are blocked at either stage 0 to I, II, II to III, or IV of sporulation. The mutants showed an altered pattern of RNA synthesis after the stage at which they were blocked. The effect of rifampin on the activity of enzymes from mutant vegetative cells and sporulating cells was significantly different, suggesting that the RNA polymerase from sporulating cells was different from the RNA polymerase of vegetative cells. These results suggest that the conformation of the RNA polymerase core plays an important role in determining correct transcription during sporulation.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- DiCioccio R. A., Strauss N. Patterns of transcription in Bacillus subtilis during sporulation. J Mol Biol. 1973 Jun 25;77(2):325–336. doi: 10.1016/0022-2836(73)90338-0. [DOI] [PubMed] [Google Scholar]
- Goff C. G. Chemical structure of a modification of the Escherichia coli ribonucleic acid polymerase alpha polypeptides induced by bacteriophage T4 infection. J Biol Chem. 1974 Oct 10;249(19):6181–6190. [PubMed] [Google Scholar]
- Greenleaf A. L., Linn T. G., Losick R. Isolation of a new RNA polymerase-binding protein from sporulating Bacillus subtilis. Proc Natl Acad Sci U S A. 1973 Feb;70(2):490–494. doi: 10.1073/pnas.70.2.490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Groves W. E., Davis F. C., Jr, Sells B. H. Spectrophotometric determination of microgram quantities of protein without nucleic acid interference. Anal Biochem. 1968 Feb;22(2):195–210. doi: 10.1016/0003-2697(68)90307-2. [DOI] [PubMed] [Google Scholar]
- Harford N., Sueoka N. Chromosomal location of antibiotic resistance markers in Bacillus subtilis. J Mol Biol. 1970 Jul 28;51(2):267–286. doi: 10.1016/0022-2836(70)90142-7. [DOI] [PubMed] [Google Scholar]
- Haworth S. R., Brown L. R. Genetic analysis of ribonucleic acid polymerase mutants of Bacillus subtilis. J Bacteriol. 1973 Apr;114(1):103–113. doi: 10.1128/jb.114.1.103-113.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirsch-Kauffmann M., Herrlich P., Ponta H., Schweiger M. Helper function of T7 protein kinase in virus propagation. Nature. 1975 Jun 5;255(5508):508–510. doi: 10.1038/255508a0. [DOI] [PubMed] [Google Scholar]
- Holland M. J., Whiteley H. R. A new polypeptide associated with RNA polymerase from Bacillus subtilis during late stages of vegetative growth. Biochem Biophys Res Commun. 1973 Nov 16;55(2):462–469. doi: 10.1016/0006-291x(73)91109-1. [DOI] [PubMed] [Google Scholar]
- Leighton T. J. An RNA polymerase mutation causing temperature-sensitive sporulation in Bacillus subtilis. Proc Natl Acad Sci U S A. 1973 Apr;70(4):1179–1183. doi: 10.1073/pnas.70.4.1179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leighton T. J., Doi R. H. The stability of messenger ribonucleic acid during sporulation in Bacillus subtilis. J Biol Chem. 1971 May 25;246(10):3189–3195. [PubMed] [Google Scholar]
- Linn T. G., Greenleaf A. L., Shorenstein R. G., Losick R. Loss of the sigma activity of RNA polymerase of Bacillus subtilis during sporulation. Proc Natl Acad Sci U S A. 1973 Jun;70(6):1865–1869. doi: 10.1073/pnas.70.6.1865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Linn T., Greenleaf A. L., Losick R. RNA polymerase from sporulating Bacillus subtilis. Purification and properties of a modified form of the enzyme containing two sporulation polypeptides. J Biol Chem. 1975 Dec 25;250(24):9256–9261. [PubMed] [Google Scholar]
- Nishimoto H., Takahashi I. Template specificity and subunits of RNA polymerase from asporogenous mutants of Bacillus subtilis. Can J Biochem. 1974 Nov;52(11):966–973. doi: 10.1139/o74-135. [DOI] [PubMed] [Google Scholar]
- Santo L., Leighton T. J., Doi R. H. Ultrastructural analysis of sporulation in a conditional serine protease mutant of Bacillus subtilis. J Bacteriol. 1972 Jul;111(1):248–253. doi: 10.1128/jb.111.1.248-253.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schaeffer P. Sporulation and the production of antibiotics, exoenzymes, and exotonins. Bacteriol Rev. 1969 Mar;33(1):48–71. doi: 10.1128/br.33.1.48-71.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sonenshein A. L., Cami B., Brevet J., Cote R. Isolation and characterization of rifampin-resistant and streptolydigin-resistant mutants of Bacillus subtilis with altered sporulation properties. J Bacteriol. 1974 Oct;120(1):253–265. doi: 10.1128/jb.120.1.253-265.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spizizen J. TRANSFORMATION OF BIOCHEMICALLY DEFICIENT STRAINS OF BACILLUS SUBTILIS BY DEOXYRIBONUCLEATE. Proc Natl Acad Sci U S A. 1958 Oct 15;44(10):1072–1078. doi: 10.1073/pnas.44.10.1072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stevens A. Deoxyribonucleic acid dependent ribonucleic acid polymerases from two T4 phage-infected systems. Biochemistry. 1974 Jan 29;13(3):493–503. doi: 10.1021/bi00700a015. [DOI] [PubMed] [Google Scholar]
- Sumida-Yasumoto C., Doi R. H. Transcription from the complementary deoxyribonucleic acid strands of Bacillus subtilis during various stages of sporulation. J Bacteriol. 1974 Feb;117(2):775–782. doi: 10.1128/jb.117.2.775-782.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warren S. C. Sporulation in Bacillus subtilis. Biochemical changes. Biochem J. 1968 Oct;109(5):811–818. doi: 10.1042/bj1090811. [DOI] [PMC free article] [PubMed] [Google Scholar]