Abstract
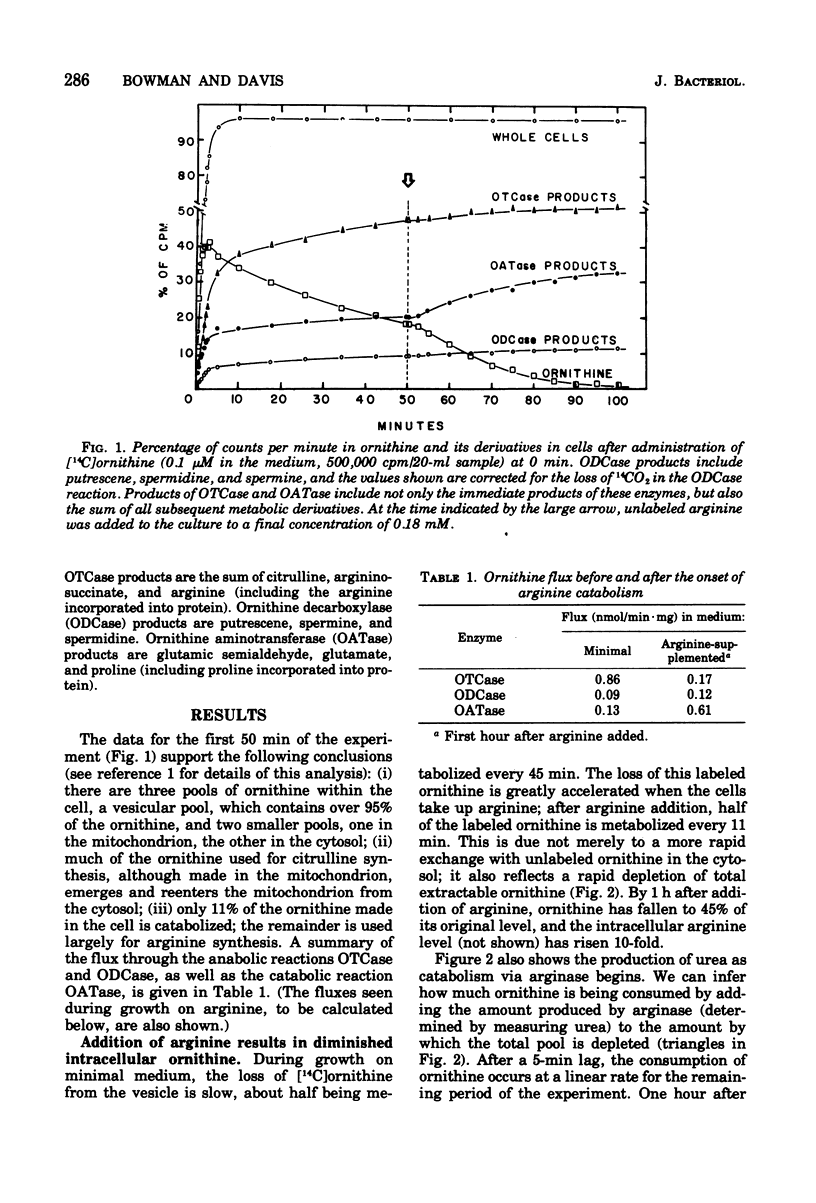
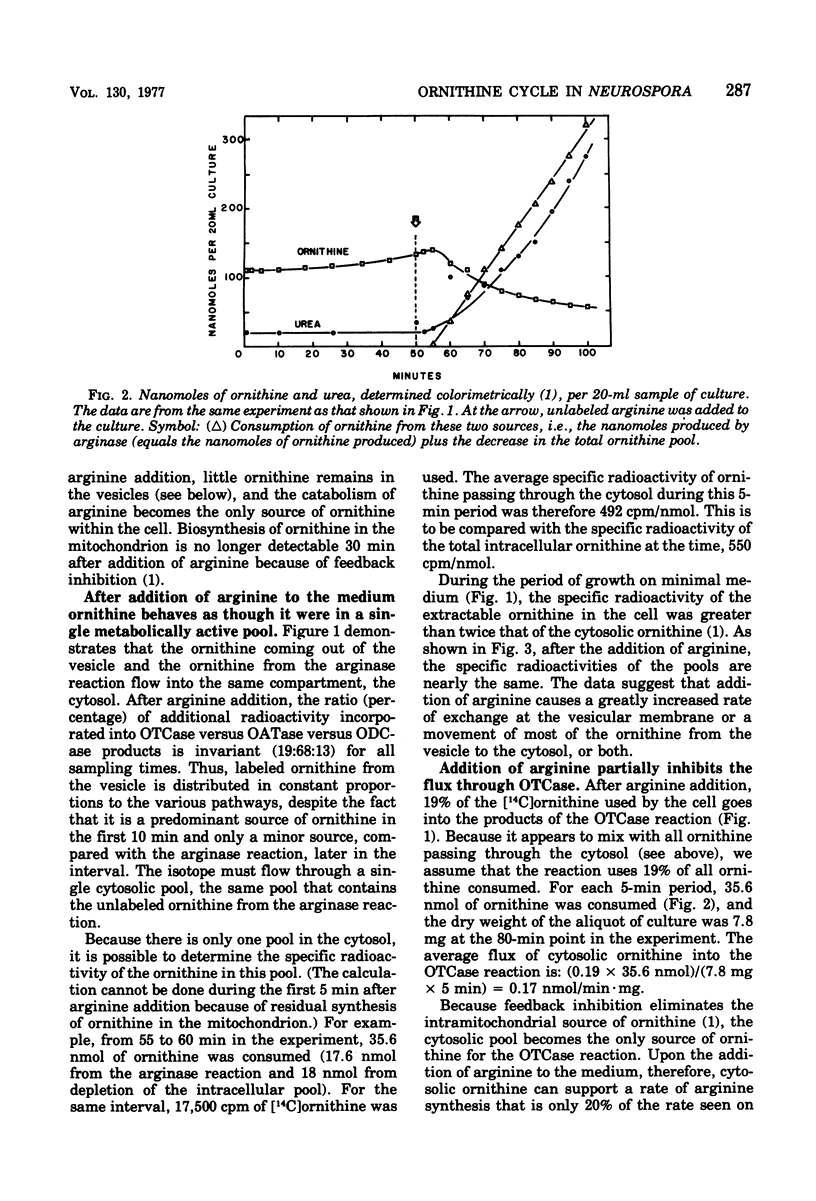
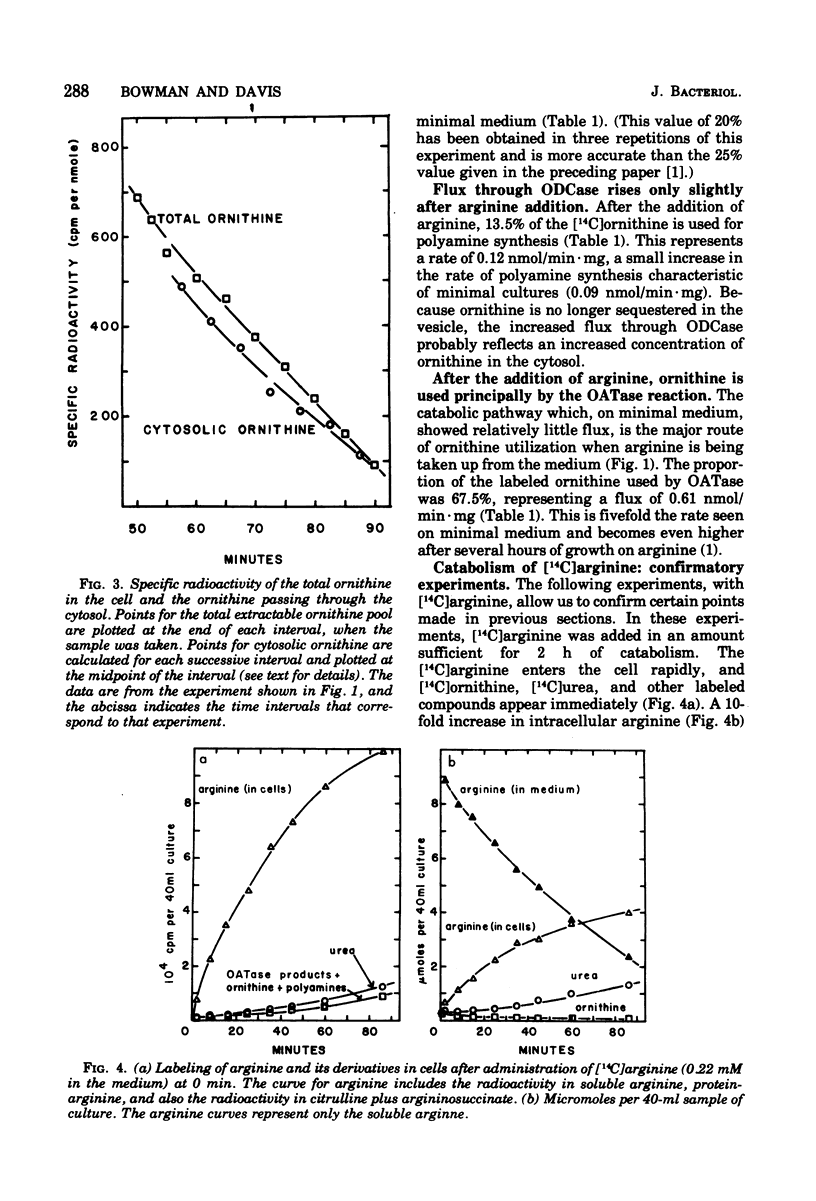
We measured the metabolism of ornithine in Neurospora during the transition from minimal medium to arginine-supplemented medium. Within an hour after arginine supplementation, the amount of intracellular ornithine (95% of which had been stored in vesicles) dropped by 65%, even though the catabolism of arginine produces as much ornithine as had been produced on minimal medium. The arginine level in the cell rose 10-fold. Ornithine flux through the catabolic enzyme ornithine aminotransferase increased fivefold, but flux through the mitochondrial enzyme ornithine transcarbamylase (leading to arginine synthesis) was only 20% of the rate seen on minimal medium. During this transition to arginine catabolism, the enzymes of the arginine pathway operate as an ornithine cycle, but at a restricted rate. We suggest the hypothesis that high levels of arginine may inhibit the movement of ornithine into the vesicles and into the mitochondria.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bowman B. J., Davis R. H. Cellular distribution of ornithine in Neurospora: anabolic and catabolic steady states. J Bacteriol. 1977 Apr;130(1):274–284. doi: 10.1128/jb.130.1.274-284.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cybis J. J., Davis R. H. Acetylglutamate kinase: a feedback-sensitive enzyme of arginine biosynthesis in Neurospora. Biochem Biophys Res Commun. 1974 Sep 23;60(2):629–634. doi: 10.1016/0006-291x(74)90287-3. [DOI] [PubMed] [Google Scholar]
- Cybis J., Davis R. H. Organization and control in the arginine biosynthetic pathway of Neurospora. J Bacteriol. 1975 Jul;123(1):196–202. doi: 10.1128/jb.123.1.196-202.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karlin J. N., Bowman B. J., Davis R. H. Compartmental behavior of ornithine in Neurospora crassa. J Biol Chem. 1976 Jul 10;251(13):3948–3955. [PubMed] [Google Scholar]
- Messenguy F., Penninckx M., Wiame J. M. Interaction between arginase and ornithine carbamoyltransferase in Saccharomyces cerevisiae. The regulatory site for ornithine. Eur J Biochem. 1971 Sep 24;22(2):277–286. doi: 10.1111/j.1432-1033.1971.tb01542.x. [DOI] [PubMed] [Google Scholar]
- Subramanian K. N., Weiss R. L., Davis R. H. Use of external, biosynthetic, and organellar arginine by Neurospora. J Bacteriol. 1973 Jul;115(1):284–290. doi: 10.1128/jb.115.1.284-290.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thwaites W. M. A mutation reducing feedback regulation by arginine in suppressed pyr-3 mutants in Neurospora. Genetics. 1967 Apr;55(4):769–781. doi: 10.1093/genetics/55.4.769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thwaites W. M., Pendyala L. Regulation of amino acid assimilation in a strain of Neurospora crassa lacking basic amino acid transport activity. Biochim Biophys Acta. 1969 Dec 30;192(3):455–461. doi: 10.1016/0304-4165(69)90394-8. [DOI] [PubMed] [Google Scholar]
- Weiss R. L., Davis R. H. Intracellular localization of enzymes of arginine metabolism in Neurospora. J Biol Chem. 1973 Aug 10;248(15):5403–5408. [PubMed] [Google Scholar]
- Weiss R. L. Intracellular localization of ornithine and arginine pools in Neurospora. J Biol Chem. 1973 Aug 10;248(15):5409–5413. [PubMed] [Google Scholar]
- Williams L. G., Bernhardt S. A., Davis R. H. Evidence for two discrete carbamyl phosphate pools in Neurospora. J Biol Chem. 1971 Feb 25;246(4):973–978. [PubMed] [Google Scholar]



