Abstract
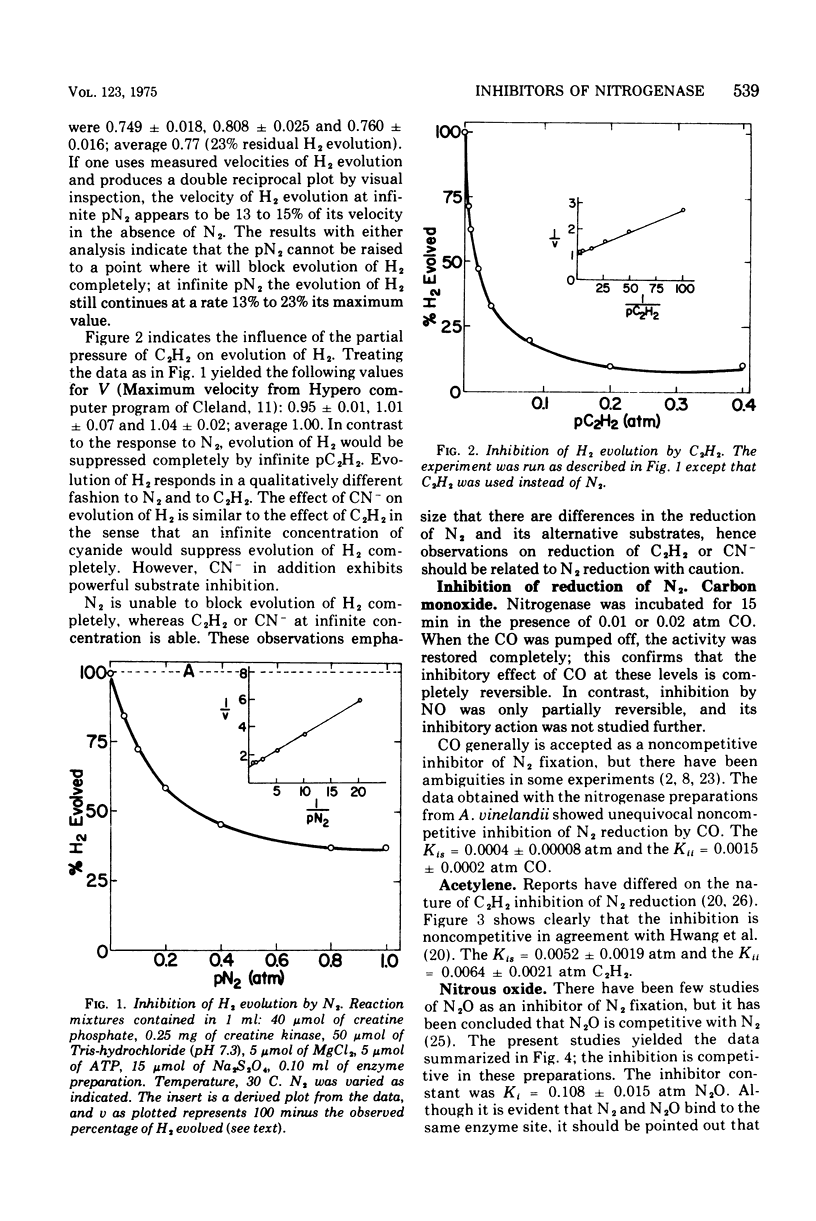
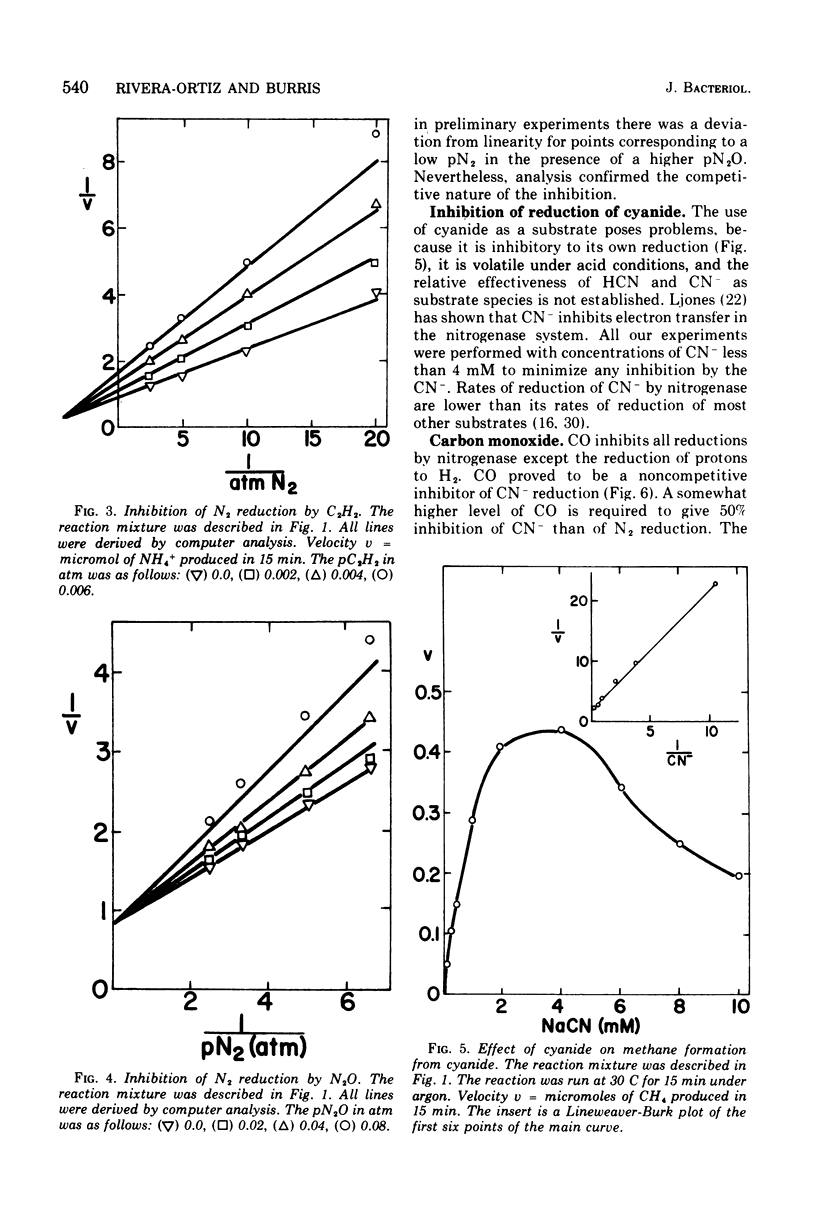
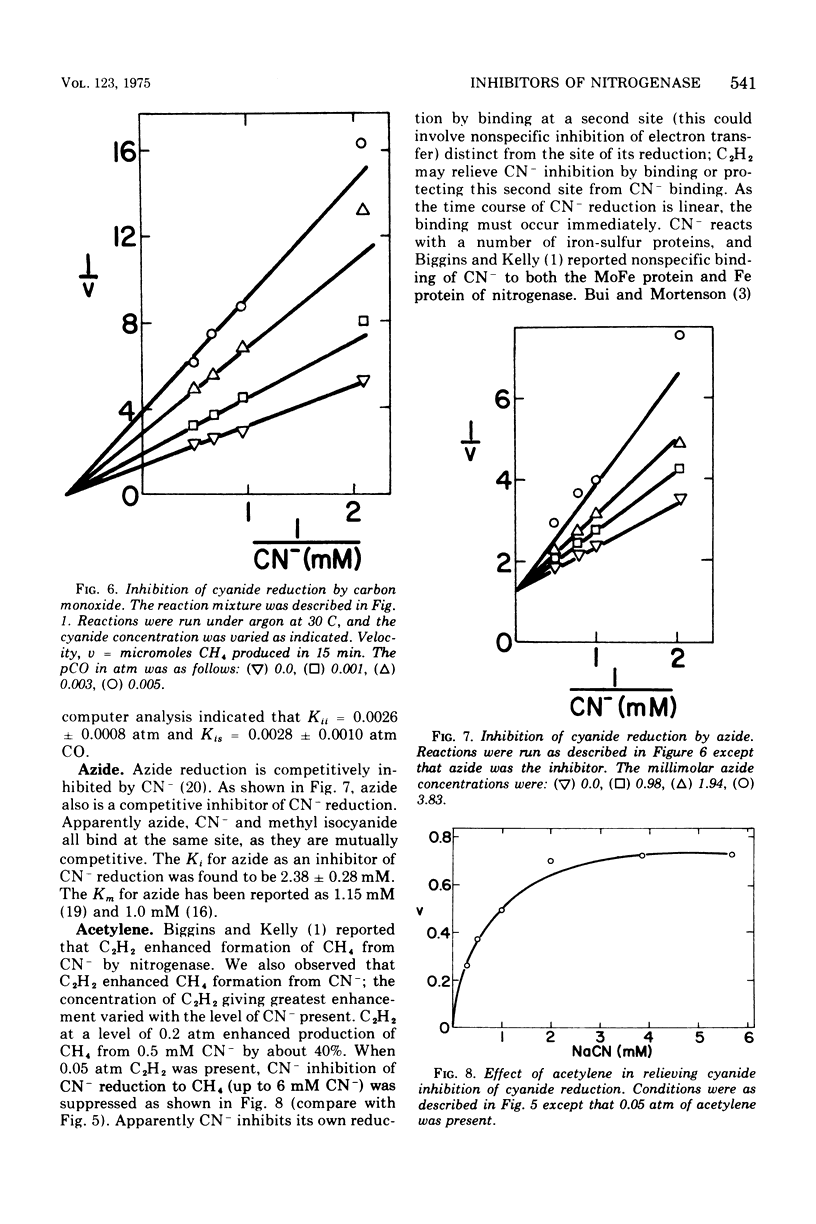
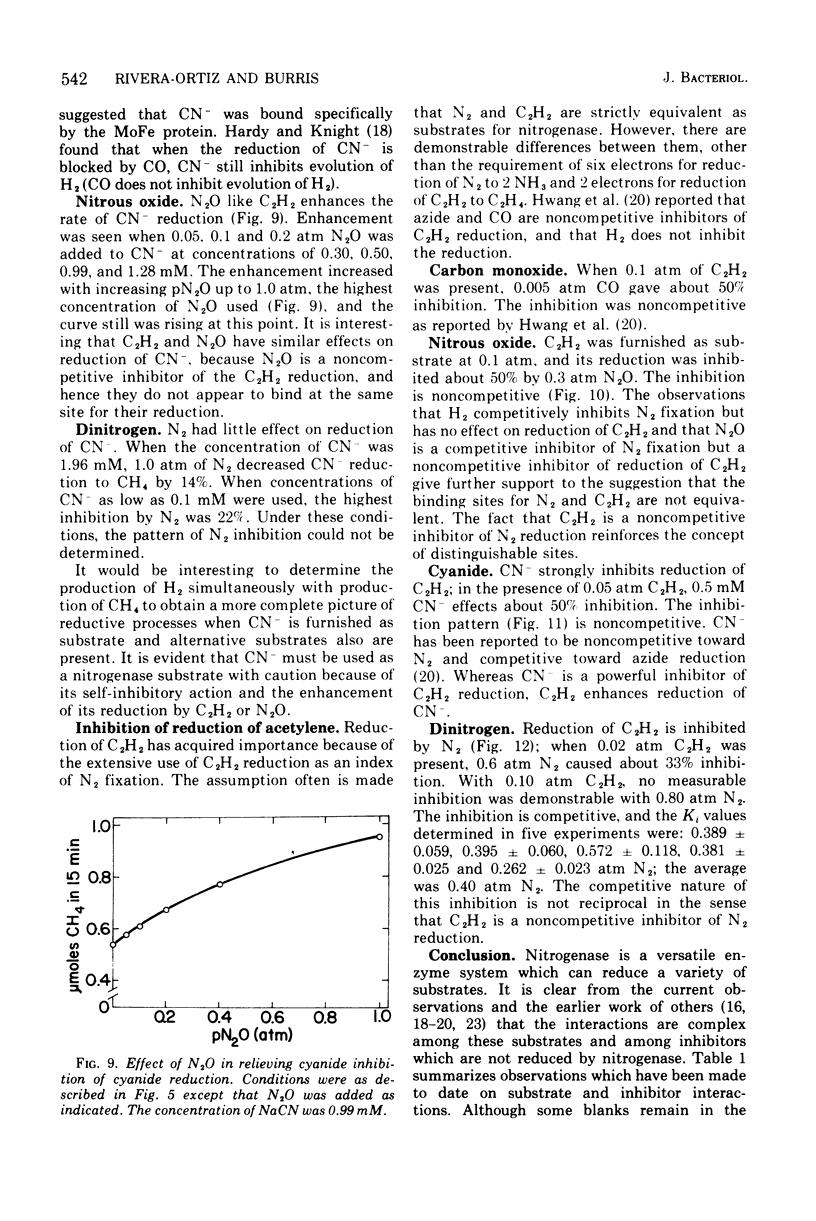
Examination of interactions among various substrates and inhibitors reacting with a partially purified nitrogenase from Azotobacter vinelandii has shown that: nitrous oxide is competitive with N2; carbon monixide and acetylene are noncompetitive with N2; carbon monoxide, cyanide, and nitrous oxide are noncompetitive with acetylene, whereas N2 is competitive with acetylene; carbon monoxide is noncompetitive with cyanide, whereas azide is competitive with cyanide; acetylene and nitrous oxide increase the rate of reduction of cyanide. The results are understandable if nitrogenase serves as an electron sink and substrates and inhibitors bind at multiple modified sites on reduced nitrogenase. It is suggested that substrates such as acetylene may be reduced by a less completely reduced electron sink than is required for the six-electron transfer necessary to reduce N2.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Biggins D. R., Kelly M. Interaction of nitrogenase from Klebsiella pneumoniae with ATP or cyanide. Biochim Biophys Acta. 1970;205(2):288–299. doi: 10.1016/0005-2728(70)90258-6. [DOI] [PubMed] [Google Scholar]
- Bui P. T., Mortenson L. E. Mechanism of the enzymic reduction of N2: the binding of adenosine 5'-triphosphate and cyanide to the N2-reducing system. Proc Natl Acad Sci U S A. 1968 Nov;61(3):1021–1027. doi: 10.1073/pnas.61.3.1021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bulen W. A., LeComte J. R. Nitrogenase complex and its components. Methods Enzymol. 1972;24:456–470. doi: 10.1016/0076-6879(72)24091-5. [DOI] [PubMed] [Google Scholar]
- Burris R. H. Nitrogen fixation--assay methods and techniques. Methods Enzymol. 1972;24:415–431. doi: 10.1016/0076-6879(72)24088-5. [DOI] [PubMed] [Google Scholar]
- CLELAND W. W. Computer programmes for processing enzyme kinetic data. Nature. 1963 May 4;198:463–465. doi: 10.1038/198463a0. [DOI] [PubMed] [Google Scholar]
- Chaykin S. Assay of nicotinamide deamidase. Determination of ammonia by the indophenol reaction. Anal Biochem. 1969 Oct 1;31(1):375–382. doi: 10.1016/0003-2697(69)90278-4. [DOI] [PubMed] [Google Scholar]
- Cleland W. W. The statistical analysis of enzyme kinetic data. Adv Enzymol Relat Areas Mol Biol. 1967;29:1–32. doi: 10.1002/9780470122747.ch1. [DOI] [PubMed] [Google Scholar]
- Dilworth M. J. Acetylene reduction by nitrogen-fixing preparations from Clostridium pasteurianum. Biochim Biophys Acta. 1966 Oct 31;127(2):285–294. doi: 10.1016/0304-4165(66)90383-7. [DOI] [PubMed] [Google Scholar]
- Hardy R. W., Holsten R. D., Jackson E. K., Burns R. C. The acetylene-ethylene assay for n(2) fixation: laboratory and field evaluation. Plant Physiol. 1968 Aug;43(8):1185–1207. doi: 10.1104/pp.43.8.1185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hardy R. W., Knight E., Jr ATP-dependent reduction of azide and HCN by N2-fixing enzymes of Azotobacter vinelandii and Clostridium pasteurianum. Biochim Biophys Acta. 1967 May 16;139(1):69–90. doi: 10.1016/0005-2744(67)90114-3. [DOI] [PubMed] [Google Scholar]
- Hwang J. C., Burris R. H. Nitrogenase-catalyzed reactions. Biochim Biophys Acta. 1972 Nov 17;283(2):339–350. doi: 10.1016/0005-2728(72)90250-2. [DOI] [PubMed] [Google Scholar]
- Hwang J. C., Chen C. H., Burris R. H. Inhibition of nitrogenase-catalyzed reductions. Biochim Biophys Acta. 1973 Jan 18;292(1):256–270. doi: 10.1016/0005-2728(73)90270-3. [DOI] [PubMed] [Google Scholar]
- Koch B., Evans H. J. Reduction of acetylene to ethylene by soybean root nodules. Plant Physiol. 1966 Dec;41(10):1748–1750. doi: 10.1104/pp.41.10.1748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ljones T. Nitrogenase from Clostridium pasteurianum. Changes in optical absorption spectra during electron transfer and effects of ATP, inhibitors and alternative substrates. Biochim Biophys Acta. 1973 Sep 15;321(1):103–113. doi: 10.1016/0005-2744(73)90064-8. [DOI] [PubMed] [Google Scholar]
- Lockshin A., Burris R. H. Inhibitors of nitrogen fixation in extracts from Clostridium pasteurianum. Biochim Biophys Acta. 1965 Nov 15;111(1):1–10. doi: 10.1016/0304-4165(65)90466-6. [DOI] [PubMed] [Google Scholar]
- NEWTON J. W., WILSON P. W., BURRIS R. H. Direct demonstration of ammonia as an intermediate in nitrogen fixation by Azotobacter. J Biol Chem. 1953 Sep;204(1):445–451. [PubMed] [Google Scholar]
- Schöllhorn R., Burris R. H. Acetylene as a competitive inhibitor of N-2 fixation. Proc Natl Acad Sci U S A. 1967 Jul;58(1):213–216. doi: 10.1073/pnas.58.1.213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith B. E., Lowe D. J., Bray R. C. Studies by electron paramagnetic resonance on the catalytic mechanism of nitrogenase of Klebsiella pneumoniae. Biochem J. 1973 Oct;135(2):331–341. doi: 10.1042/bj1350331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strandberg G. W., Wilson P. W. Molecular H2 and the PN2 function of azotobacter. Proc Natl Acad Sci U S A. 1967 Oct;58(4):1404–1409. doi: 10.1073/pnas.58.4.1404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tso M. Y., Burris R. H. The binding of ATP and ADP by nitrogenase components from Clostridium pasteurianum. Biochim Biophys Acta. 1973 Jun 6;309(2):263–270. doi: 10.1016/0005-2744(73)90024-7. [DOI] [PubMed] [Google Scholar]
- Wong P. P., Burris R. H. Nature of oxygen inhibition of nitrogenase from Azotobacter vinelandii. Proc Natl Acad Sci U S A. 1972 Mar;69(3):672–675. doi: 10.1073/pnas.69.3.672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zumft W. G., Mortenson L. E., Palmer G. Electron-paramagnetic-resonance studies on nitrogenase. Investigation of the oxidation-reduction behaviour of azoferredoxin and molybdoferredoxin with potentiometric and rapid-freeze techniques. Eur J Biochem. 1974 Aug 1;46(3):525–535. doi: 10.1111/j.1432-1033.1974.tb03646.x. [DOI] [PubMed] [Google Scholar]



