Abstract
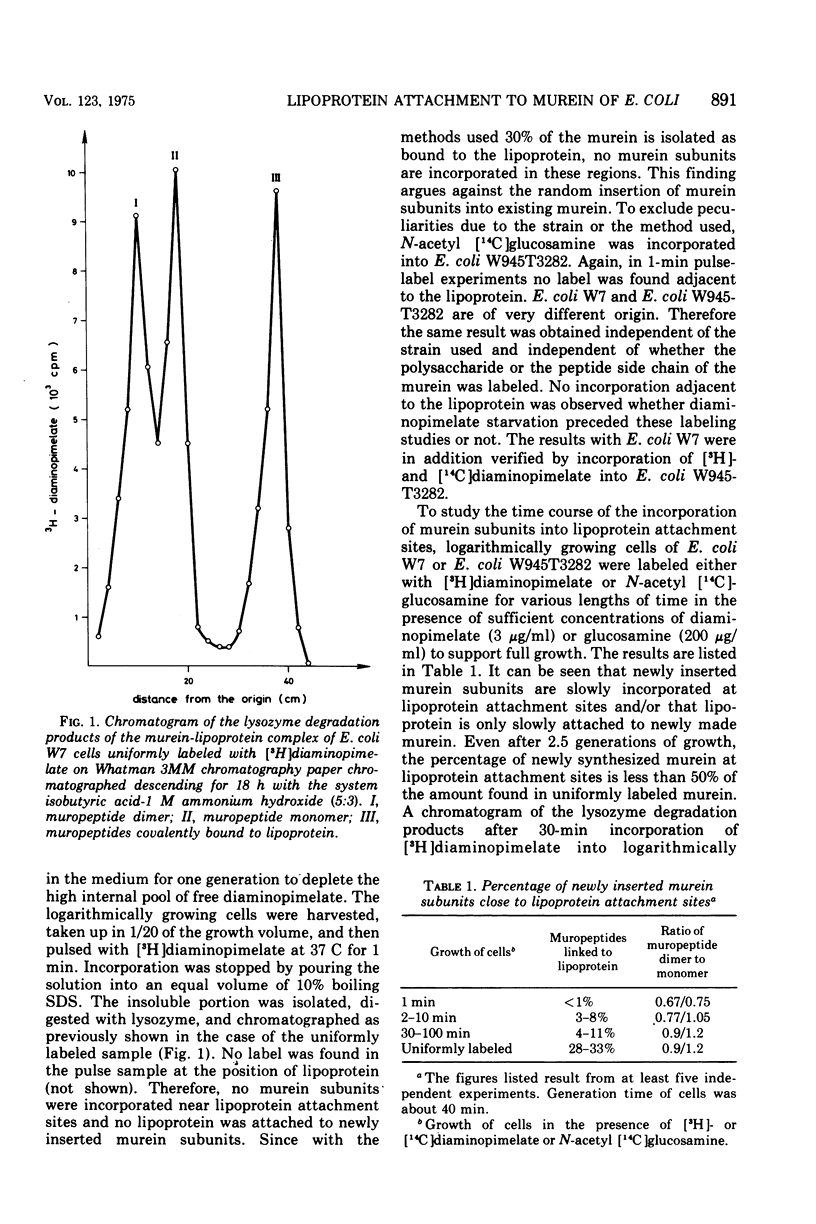
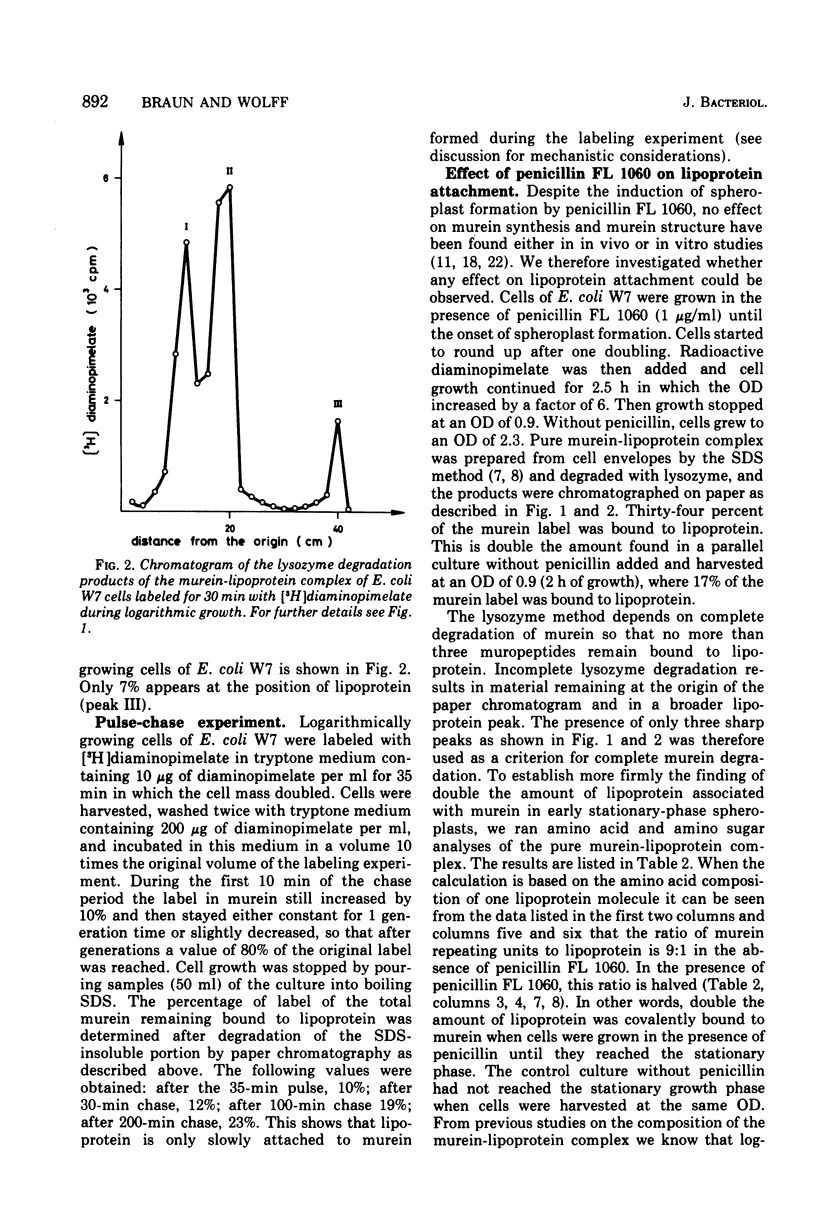
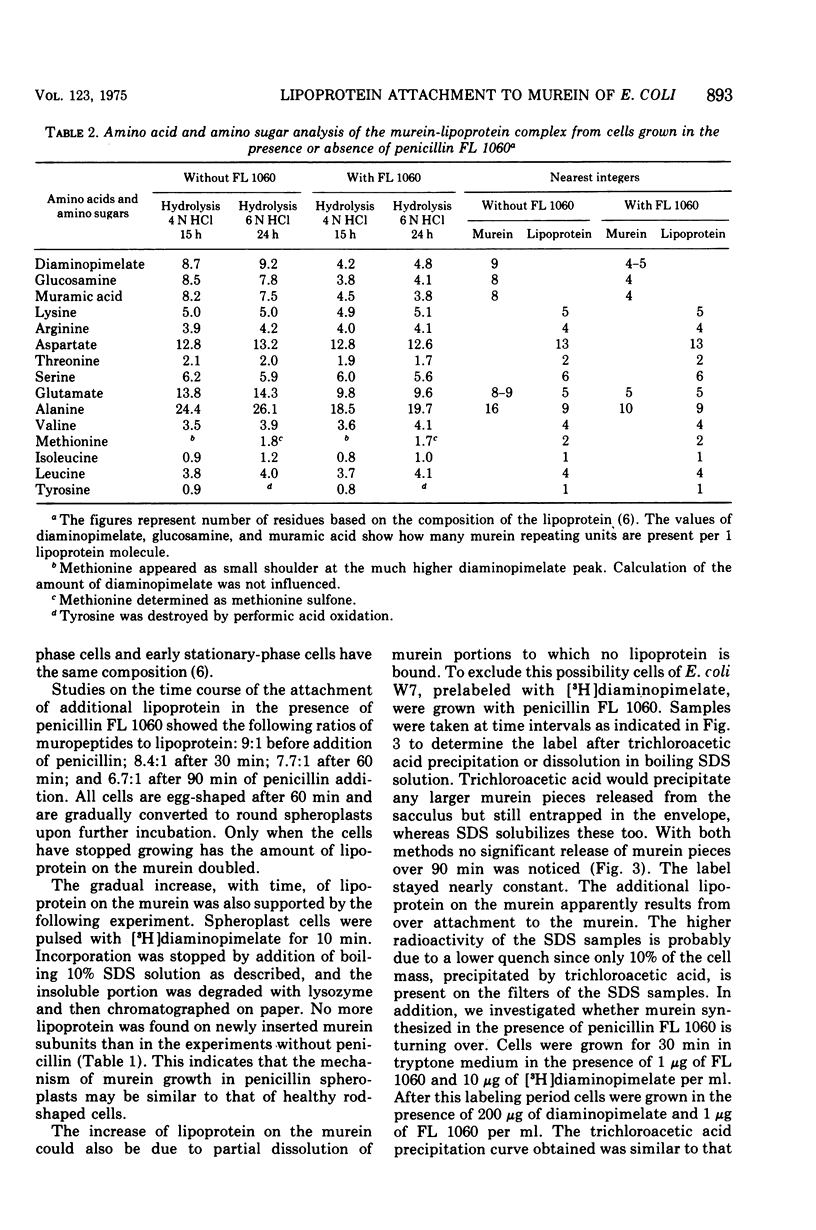
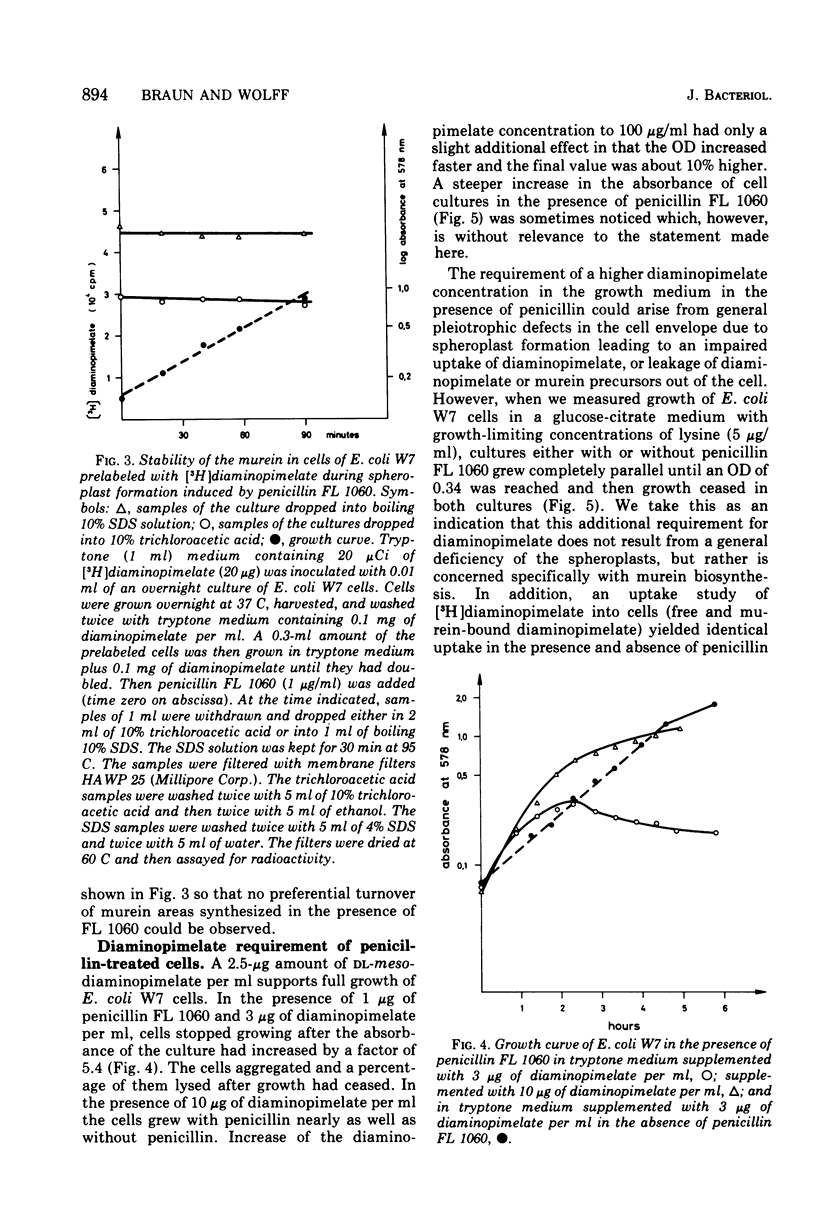
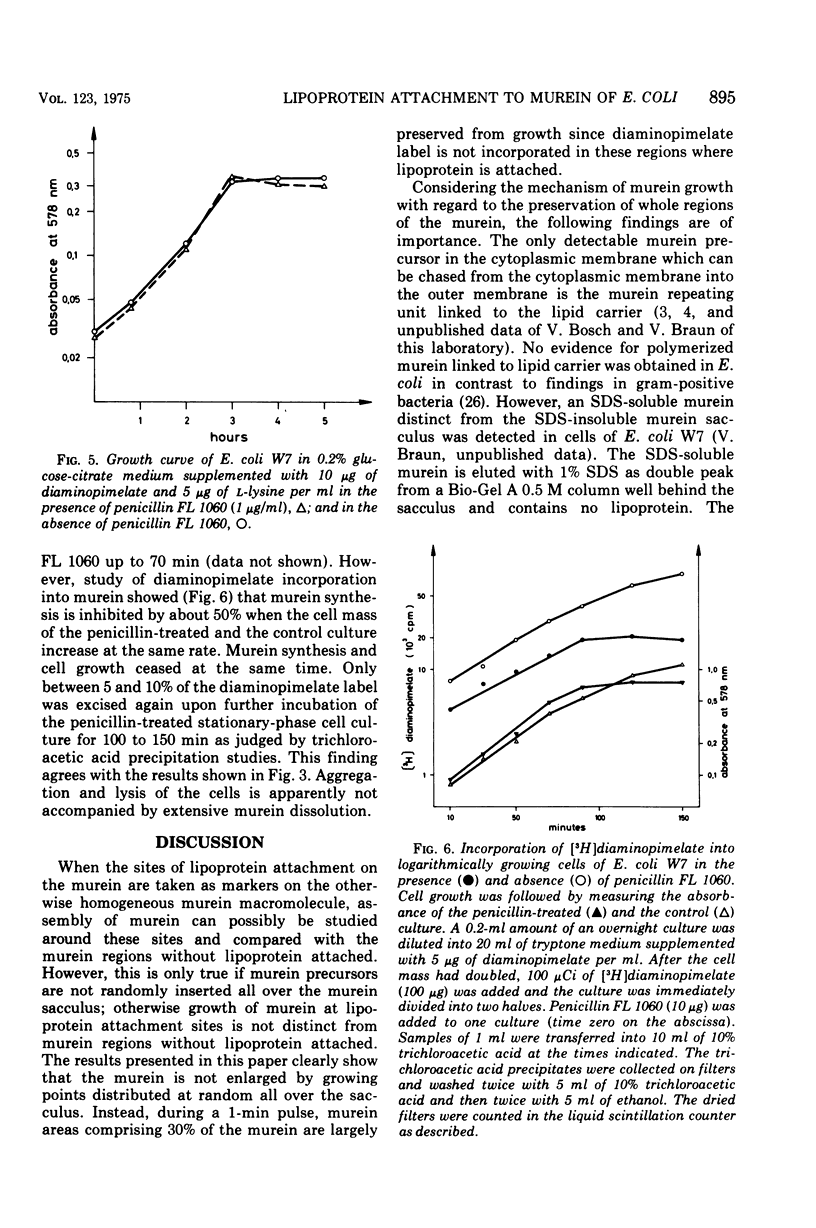
In vivo studies on the attachment of lipoprotein to the murein (peptidoglycan) of Escherichia coli showed that it takes several generations of growth until the amount of lipoprotein on newly made murein is equilibrated. The technique used involves degradation of the sodium dodecyl sulfate-insoluble murein-lipoprotein complex (sacculus, rigid layer) with lysozyme and separation of the labeled products on paper. No lipoprotein was found on murein subunits incorporated during a pulse of [3H]diaminopimelate for 1 min in logarithmically growing cells at 37 C. Even after one doubling of the cell mass, only 4 to 8% of the labeled murein was isolated as bound to lipoprotein. With uniformly labeled murein, 30% remains bound to lipoprotein after lysozyme treatment, corresponding to three murein subunits. Therefore it can be concluded that during pulse labeling either no lipoprotein is incorporated into the newly synthesized murein or no murein subunits are inserted into existing murein around lipoprotein attachment sites. Longer pulse and pulse-chase experiments argue for the latter interpretation. It is therefore concluded that incorporation of murein subunits into the growing murein polymer is not at all a random process. Instead, quite large areas of murein, on which lipoprotein is situated, seem to be preserved. Under the influence of penicillin FL 1060 murein synthesis is 50% inhibited. The rate of lipoprotein attachment is less affected so that increasing amounts of lipoprotein become attached during spheroplast formation. By the time the stationary growth phase has been reached, the lipoprotein content of the murein has doubled. Diaminopimelate auxotrophic mutants require, in the presence of penicillin FL 1060, more diaminopimelate for full growth than in the absence of penicillin FL 1060. This finding and the fact that murein synthesis is always inhibited by 50% over a wide range of penicillin concentration (1 to 1,000 mug/ml) point to the inhibition of an enzymatic step of murein synthesis which can be partially bypassed by a second enzyme, less efficient but resistant to penicillin FL 1060.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Braun V., Bosch V., Hantke K., Schaller K. Structure and biosynthesis of functionally defined areas of the Escherichia coli outer membrane. Ann N Y Acad Sci. 1974 May 10;235(0):66–82. doi: 10.1111/j.1749-6632.1974.tb43257.x. [DOI] [PubMed] [Google Scholar]
- Braun V., Bosch V. In vivo biosynthesis of murein-lipoprotein of the outer membrane of E. coli. FEBS Lett. 1973 Aug 15;34(2):302–306. doi: 10.1016/0014-5793(73)80817-8. [DOI] [PubMed] [Google Scholar]
- Braun V., Bosch V. Sequence of the murein-lipoprotein and the attachment site of the lipid. Eur J Biochem. 1972 Jun 23;28(1):51–69. doi: 10.1111/j.1432-1033.1972.tb01883.x. [DOI] [PubMed] [Google Scholar]
- Braun V., Gnirke H., Henning U., Rehn K. Model for the structure of the shape-maintaining layer of the Escherichia coli cell envelope. J Bacteriol. 1973 Jun;114(3):1264–1270. doi: 10.1128/jb.114.3.1264-1270.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braun V., Hantke K. Biochemistry of bacterial cell envelopes. Annu Rev Biochem. 1974;43(0):89–121. doi: 10.1146/annurev.bi.43.070174.000513. [DOI] [PubMed] [Google Scholar]
- Braun V. Molecular organization of the rigid layer and the cell wall of Escherichia coli. J Infect Dis. 1973 Jul;128(Suppl):9–16. doi: 10.1093/infdis/128.supplement_1.s9. [DOI] [PubMed] [Google Scholar]
- Braun V., Rehn K. Chemical characterization, spatial distribution and function of a lipoprotein (murein-lipoprotein) of the E. coli cell wall. The specific effect of trypsin on the membrane structure. Eur J Biochem. 1969 Oct;10(3):426–438. doi: 10.1111/j.1432-1033.1969.tb00707.x. [DOI] [PubMed] [Google Scholar]
- Braun V., Sieglin U. The covalent murein-lipoprotein structure of the Escherichia coli cell wall. The attachment site of the lipoprotein on the murein. Eur J Biochem. 1970 Apr;13(2):336–346. doi: 10.1111/j.1432-1033.1970.tb00936.x. [DOI] [PubMed] [Google Scholar]
- Burdett I. D., Murray R. G. Septum formation in Escherichia coli: characterization of septal structure and the effects of antibiotics on cell division. J Bacteriol. 1974 Jul;119(1):303–324. doi: 10.1128/jb.119.1.303-324.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Formanek H., Formanek S., Wawra H. A three-dimensional atomic model of the murein layer of bacteria. Eur J Biochem. 1974 Jul 15;46(2):279–294. doi: 10.1111/j.1432-1033.1974.tb03620.x. [DOI] [PubMed] [Google Scholar]
- Goodell E. W., Schwarz U. Sphere-rod morphogenesis of Escherichia coli. J Gen Microbiol. 1975 Feb;86(2):201–209. doi: 10.1099/00221287-86-2-201. [DOI] [PubMed] [Google Scholar]
- Henning U., Rehn K., Braun V., Höhn B. Cell envelope and shape of Escherichia coli K12. Properties of a temperature-sensitive rod mutant. Eur J Biochem. 1972 Apr 24;26(4):570–586. doi: 10.1111/j.1432-1033.1972.tb01800.x. [DOI] [PubMed] [Google Scholar]
- Inouye M., Hirashima A., Lee N. Discussion paper: biosynthesis and assembly of a structural lipoprotein in the envelope of Escherichia coli. Ann N Y Acad Sci. 1974 May 10;235(0):83–90. doi: 10.1111/j.1749-6632.1974.tb43258.x. [DOI] [PubMed] [Google Scholar]
- Inouye M., Shaw J., Shen C. The assembly of a structural lipoprotein in the envelope of Escherichia coli. J Biol Chem. 1972 Dec 25;247(24):8154–8159. [PubMed] [Google Scholar]
- Kohiyama M., Cousin D., Ryter A., Jacob F. Mutants thermosensibles d'Escherichia coli K 12. I. Isolement et caractérisation rapide. Ann Inst Pasteur (Paris) 1966 Apr;110(4):465–486. [PubMed] [Google Scholar]
- Lund F., Tybring L. 6 -amidinopenicillanic acids--a new group of antibiotics. Nat New Biol. 1972 Apr 5;236(66):135–137. doi: 10.1038/newbio236135a0. [DOI] [PubMed] [Google Scholar]
- Matsuhashi S., Kamiryo T., Blumberg P. M., Linnett P., Willoughby E., Strominger J. L. Mechanism of action and development of resistance to a new amidino penicillin. J Bacteriol. 1974 Feb;117(2):578–587. doi: 10.1128/jb.117.2.578-587.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melchior N. H., Blom J., Tybring L., Birch-Andersen A. Light and electron microscopy of the early response of Escherichia coli to a 6beta-amidinopenicillanic acid (FL 1060). Acta Pathol Microbiol Scand B Microbiol Immunol. 1973 Aug;81(4):393–407. doi: 10.1111/j.1699-0463.1973.tb02222.x. [DOI] [PubMed] [Google Scholar]
- Normark S., Boman H. G., Bloom G. D. Cell division in a chain-forming envA mutant of Escherichia coli K12. Fine structure of division sites and effects of EDTA, lysozyme and ampicillin. Acta Pathol Microbiol Scand B Microbiol Immunol. 1971;79(5):651–664. doi: 10.1111/j.1699-0463.1971.tb00093.x. [DOI] [PubMed] [Google Scholar]
- Osborn M. J., Gander J. E., Parisi E., Carson J. Mechanism of assembly of the outer membrane of Salmonella typhimurium. Isolation and characterization of cytoplasmic and outer membrane. J Biol Chem. 1972 Jun 25;247(12):3962–3972. [PubMed] [Google Scholar]
- Park J. T., Burman L. FL-1060: a new penicillin with a unique mode of action. Biochem Biophys Res Commun. 1973 Apr 16;51(4):863–868. doi: 10.1016/0006-291x(73)90006-5. [DOI] [PubMed] [Google Scholar]
- Ryter A., Hirota Y., Schwarz U. Process of cellular division in Escherichia coli growth pattern of E. coli murein. J Mol Biol. 1973 Jun 25;78(1):185–195. doi: 10.1016/0022-2836(73)90437-3. [DOI] [PubMed] [Google Scholar]
- VOGEL H. J., BONNER D. M. Acetylornithinase of Escherichia coli: partial purification and some properties. J Biol Chem. 1956 Jan;218(1):97–106. [PubMed] [Google Scholar]
- Ward J. B., Perkins H. R. The direction of glycan synthesis in a bacterial peptidoglycan. Biochem J. 1973 Dec;135(4):721–728. doi: 10.1042/bj1350721. [DOI] [PMC free article] [PubMed] [Google Scholar]


